Abstract
TCR engagement on adherent human effector memory (EM) CD4+ T cells by TNF-treated human umbilical vein endothelial cells (HUVEC) under flow induces formation of a transendothelial protrusion (TEP) by the T cell but fails to induce transendothelial migration (TEM). In contrast, TCR engagement of the same T cell populations by TNF-treated human dermal microvascular cells (HDMEC) not only induces TEP formation, but triggers TEM at or near the interendothelial cell junctions via a process in which TEP formation appears to be the first step. Transduction of adhesion molecules in unactivated HDMEC and use of blocking antibodies as conducted with TNF-activated HDMEC indicate that ICAM-1 plays a nonredundant role in TCR-driven TEP formation and TEM, and that TCR-driven TEM is also dependent upon fractalkine. TEP formation, dependence on ICAM-1 and dependence on fractalkine distinguish TCR-induced TEM from IP-10 induced TEM. These in vitro observations suggest that presentation of antigen by human microvascular EC to circulating CD4+ EM T cells may function to initiate recall responses in peripheral tissues.
Keywords: Endothelial cells, T cells, T cell receptors, chemokines, superantigens
Introduction
In uninflamed peripheral tissues of humans, microvascular endothelial cells (EC) display both class I and II MHC molecules at substantially higher levels than any other cell type with the possible exception of resident dendritic cells (1-3). The only well established function of MHC molecules is to present bound peptides in a form that may be recognized by T cell receptors for antigens (TCRs). Human EC also display costimulators and adhesion molecules that preferentially engage memory T cells and, in vitro, selectively activate proliferation and cytokine synthesis by memory but not naïve T cells (4-8). These observations suggest that human EC are positioned to present peptide antigens to circulating memory T cells.
The in vivo functions and consequences of antigen presentation by human EC is unknown. EC could present self peptides in association with self MHC molecules to provide low-affinity signals to the TCRs of circulating memory cells that sustain memory cell viability. Alternatively, EC could present non-self-derived (e.g., microbial) peptides or, in the case of transplantation, non-self MHC molecules to promote local recruitment and/or activation of antigen-specific memory T cell populations, initiating protective recall responses or allograft rejection, respectively. These hypotheses are not mutually exclusive. Studies in mice have provided in vitro and in vivo evidence that antigen presentation by EC does increase the recruitment of antigen-specific CD8+ T cells into tissue (9-11). A role played by EC in recruiting antigen specific CD4+ T cells in vivo, however, cannot be addressed in mice since murine EC constitutively express only class I, but not class II MHC molecules in situ (12, 13). Moreover, even when induced to express class II MHC molecules in vitro, mouse EC, unlike human EC, do not activate T cell effector responses (14, 15). Recent studies using a human-SCID mouse chimera model show that human effector memory (EM) CD4+ T cells can recognize and destroy EC in skin grafts from donors allogeneic to the T cells, consistent with the hypothesis that human EC directly present class II MHC molecules to circulating T cells in vivo (7, 8). However, studies of human TCR-driven antigen recruitment in vitro, assessed as transendothelial migration (TEM) of T cells across cultured EC monolayers, have been contradictory, with some showing inhibition of migration (16, 17) and others stimulation (16, 18). Much of this variation may be attributed to the analysis of T cells and of EC of different origins and states of activation. An additional important variable has been the presence or absence of physiological shear stress, the force imparted through viscous drag of flowing blood, in these in vitro models. T cells, unlike neutrophils or monocytes, require venular levels of shear stress (∼1 dyne/cm2) to initiate rapid (in minutes) TEM in response to chemokines (19, 20). Using a simple parallel plate flow chamber, we have shown that TEM across a monolayer of cultured HUVEC by effector memory (EM) CD4+ T cells in response to the inflammatory chemokine IP-10 (CXCL10) occurs within 20 minutes only when shear stress is applied (20). Importantly, our studies have utilized primary CD4+ T cells that were neither activated nor activated and expanded in culture as T cell lines and/or clones. We also noted that the expression of EC adhesion molecules, especially ICAM-1 or VCAM-1, whether induced by TNF treatment or by retroviral transduction, was necessary and sufficient for T cells to be captured by the EC; in the absence of EC adhesion molecules, T cells failed to bind to the EC monolayer under flow and could not, therefore, transmigrate.
Another potential variable is the source of EC. HUVEC are derived from a large vessel whereas most TEM occurs in the microvasculature, especially in the post-capillary vessels. An alternative model are EC isolated and cultured from human dermal microvessels (i.e., HDMEC). HDMEC differ from HUVEC in their kinetics of expression of adhesion molecules for leukocytes (21), in their inducible expression of chemokine, especially fractalkine (CX3CL1) (22), and in their ability to support transmigration of activated T cells through the EC body (23).
Unlike chemokine responses, the analysis of TCR signaling by human EC presents specific technical challenges. The frequency of antigen-specific T cells in the circulation is very low, even for alloantigens. We have recently developed two approaches to address this problem by engineering HUVEC to express molecules capable of engaging the TCRs of polyclonal T cell populations. Unexpectedly, TCR engagement in either model blocked the rapid TEM of freshly isolated EM CD4+ T cells across HUVEC monolayers in response to IP-10 under conditions of venular shear stress, and that this block of TEM persisted for up to one hour (17). Examination of the T cells revealed that, despite their migratory arrest on the EC apical surface, EM CD4+ T cells whose TCR had been engaged both activated NFAT and AP-1 and formed a cytoplasmic protrusion that extended across the EC monolayer. However, the formation of this transendothelial protrusion (TEP) was not followed by TEM. Here we report that TEP formation is the first step in a mechanism of TEM that is TCR-dependent when HDMEC are used instead of HUVEC. Furthermore, this response is restricted to EM CD4+ T cells, is significantly augmented by shear stress, and depends upon ICAM-1 and fractalkine.
Materials and Methods
Cells and reagents
Human umbilical vein EC (HUVEC) were isolated and cultured as described (20). All HUVEC were used at no later than subculture five; all cells in a given experiment were used at the same level of subculture. CIITA HUVEC were generated by retroviral transduction as described (17). Human dermal microvascular endothelial cells (HDMEC) were isolated and cultured as described (24). FcγRII-HDMEC and CIITA-HDMEC were generated and characterized in an analogous manner. CIITA HDMEC co-transduced with E-selectin, ICAM-1, and VCAM-1 were likewise generated and characterized as described for HUVECs (20). For many experiments, EC were incubated in the presence of 10 ng/ml recombinant human TNF (TNFα, R&D Systems) for 20−26 hours prior to the flow assay. For the costimulator blocking experiments, EC were incubated in the presence of blocking antibodies to 41BBL, ICOSL, LFA-3, OX40L, or control IgG as described (17) for 30 minutes prior to the flow assay. Similarly, 10 μg/ml goat anti-fractalkine, goat anti E-selectin, mouse anti-ICAM-1, and mouse anti-VCAM-1 (all from R&D Systems) were used to block fractalkine, E-selectin, ICAM-1, and VCAM-1, respectively.
CD4+ T cells were isolated by positive selection with magnetic beads and released with Detachabead (Dynal) from PBMCs prepared by Ficoll gradient of blood collected from healthy donors. Naïve (CD4+CD45RO−) T cells were isolated by depletion of CD45RO+ cells from CD4+ T cells using anti-CD45RO mAb (eBiosciences) and pan-mouse IgG beads (Dynal). Memory (CD4+CD45RA−) T cells were isolated by depletion of CD45RA+ cells from CD4+ T cells using anti-CD45RA mAb (eBiosciences) and pan-mouse IgG beads. EM cells were prepared by depleting memory cells with anti-CCR7 mAb (R&D Systems) and pan-mouse IgG beads. Alternatively, CD4+CD45RA− T cells were stained with FITC-conjugated anti-CCR7, and CCR7high and CCR7low cells were isolated by FACS to obtain EM and CM CD4+ T cells, respectively. T cells were cultured in RPMI 1640 medium supplemented with 10% fetal bovine serum, 2 mM glutamine, penicillin/streptomycin, and nonessential amino acids overnight prior to assays. For pharmacological inhibitor studies, EM CD4+ T cells were treated with 20 μM Ly294002 30 min, 100 ng/ml pertussis toxin overnight, or 100 ng/ml cholera toxin one hour prior to transmigration assays.
TEM assays
CIITA HUVEC, FcγRII HDMEC, CIITA HDMEC and adhesion molecule co-transductants, grown to confluence on 35 mm fibronectin-coated coverglasses, were incubated with 3 μg/ml OKT3 mAb (eBioscience) (for FcγRII transductants) or 100 ng/ml TSST-1 (Toxin Technologies) (for CIITA transductants) 30 minutes prior to the flow assay, washed twice with RPMI/10% FBS, overlaid or not with chemokine (3 μg/ml IP-10, R&D Systems) in RPMI/10% FBS for 5 minutes, washed two more times, and assembled with a parallel plate flow chamber apparatus (Glycotech) using the 0.01 inch height, 5 mm wide slit gasket provided by the manufacturer. On a 37°C heating surface, CD4+ CD45RO− (naïve), CD4+CD45RA−CCR7high (CM) or CD4+CD45RA−CCR7low (EM) T cells (106 cells/500 μl) suspended in the same medium were loaded onto the EC monolayer at 0.75 dyne/cm2 for 2 minutes, followed by medium only at 0 or 1 dyne/cm2 for 5, 15, 30, 45 or 60 minutes. Samples were then fixed with 3.7% formaldehyde in PBS, stained with antibodies as appropriate [anti-Vβ2 mAb (Immunotech) followed by Alexafluor 594-or 488-conjugated donkey anti-mouse IgG (Molecular Probes); FITC-conjugated anti CD3 (Immunotech); anti-NFAT-1 mAb (BD Bioscences) or anti-CD31 mAb (DAKO) pre-complexed with Alexafluor 488- or 647-conjugated anti-mouse IgG Fab (Molecular Probes); goat anti-VE-cadherin followed by Alexafluor 647-conjugated chicken anti-goat IgG], mounted on slides using mounting medium containing DAPI (Molecular Probes), and examined by microscopy. A FITC filter was used to detect FITC or Alexafluor 488-stained cells, a TRITC filter was used to detect Alexafluor 594-stained cells, a Cy5 filter was used to detect Alexafluor-647-stained cells, and a DAPI filter used to detect nuclei. Phase contrast optics were used to determine whether CD4+ T cells were either on top or underneath the HUVEC monolayer. The percentage of transmigrated CD4+ T cells and cells with a transendothelial protrusion were counted per group of 20; five to ten groups were analyzed for each sample.
For time lapse experiments, pictures were taken once a minute using a 10× objective and TRITC filter of a field containing 0.14 μM PKH26 (Sigma) labeled EM CD4+ T cells on FcγRII HDMEC preloaded with control IgG or OKT3, in the flow chamber. At the end (60 min), samples were fixed and mounted in DAPI-containing medium on a cover slide, the field that was captured localized, and Z-stack images were captured with a 63× objective and deconvoluted (Openlab).
Statistics
For experiments in which more than two groups were compared, statistical significance was determined by one-way ANOVA using a 95% confidence interval and the Tukey post-test (Prism 4.0 for Macintosh). Statistical error is expressed as s.e.m. For experiments in which two groups were compared, a t-test was used.
Results
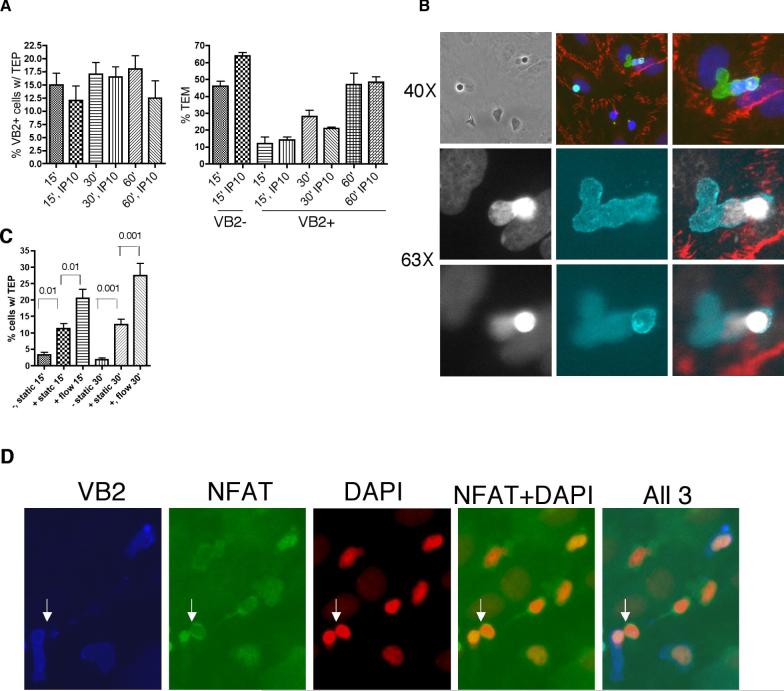
We recently reported that recognition of TSST-1 bound to HLA-DR, expressed by CIITA-transduced HUVEC pretreated with TNF to induce adhesion molecules, prevented rapid (15 min) shear-stress-dependent transendothelial migration (TEM) of Vβ2TCR+ effector memory (EM) CD4+ T cells in response to IP-10 without affecting IP-10-dependent TEM of Vβ2TCR- EM CD4+ T cells in the same chamber (17). The inhibition of the response to IP-10 was still evident at 60 min, but we noted that, in increasing numbers, Vβ2TCR+ cells underwent a morphological change during this time interval. While Vβ2TCR- cells, whether on the EC surface, transmigrating across the EC monolayer, or spread out beneath the EC, typically lay flat with a spread out nucleus that occupied most of the intracellular area, an increasing number of Vβ2TCR+ cells rounded up on the EC surface and extended a nuclear-free protrusion (Figure 1). In deconvoluted Z-stack images, these protrusions are seen to extend across the HUVEC monolayer and for this reason we refer to these structures as transendothelial protrusions (TEPs). Furthermore, TEPs are only formed by EM as opposed to central memory (CM) and naïve CD4+ T cells and their formation is dependent on the application of venular levels of shear stress (Figure 1C).
Figure 1.

TCR engagement induces transendothelial protrusions in effector memory CD4+ T cells on cultured HUVEC. A. Phase contrast (PC), nuclear staining with DAPI, and immunofluorescence staining of Vβ2TCR+ (upper and middle panels) and Vβ2TCR- EM CD4+ T cell captured by a TNF-treated CIITA-transduced HUVEC monolayer preloaded with TSST-1under conditions of venular flow. Upper and lower panels taken with a 40× objective. Middle panels show deconvoluted images of a series of 1 μm Z-stack of pictures taken with a 63× objective of Vβ2TCR staining. Note that the TEP extends through the HUVEC monolayer. Lower panels show a Vβ2TCR- cell. Note the spread morphology and close proximity of the nucleus to the leading edge. B. Left graph: Flow TEM assay was performed with EM CD4+ cells for 30 and 60 minutes at 1 dyne/cm2 on TNF-treated CIITA HUVECs preloaded with TSST-1, fixed, and stained for Vβ2TCR and CD3 (to detect protrusions in Vβ2TCR- cells). Mean and s.e.m. of the number of cells showing a TEP in 5 sets of 20 cells. P<0.01 for comparison of Vβ2TCR– vs Vβ2TCR+ at 60’. One representative experiment of three is shown. Right graph: Flow TEM assay was performed with EM CD4+ cells for 60 minutes at 1 dyne/cm2 on TNF-treated CIITA HUVECs preloaded or not with TSST-1, fixed and stained for Vβ2TCR. Mean and s.e.m. of the number of cells showing a TEP in ten sets of 20 cells. P<0.0001. C. CD4+ cells were depleted of CD45RO+ positive cells (naïve) or depleted of CD45RA+ cells and then stained with CCR7-FITC and FACS-sorted for CCR7 high (central memory, CM) and CCR7 low (EM) and used in a one hour flow TEM assay or static TEM assay with TNF-treated CIITA HUVECs preloaded with TSST-1 and stained for Vβ2TCR. Mean and s.e.m. of number of Vβ2TCR+ cells showing a TEP in 10 sets of 20 cells. P<0.001 for comparison of EM, flow versus all other groups; P>0.05 for all other comparisons.
We hypothesized that the extension of a TEP is the first step in chemokine-independent, TCR-initiated TEM. However, little if any TEM by TCR-activated EM CD4+ T cells is observed across HUVEC monolayers (17). As noted in the introduction, large vessel EC, like HUVEC, may be less capable than microvascular EC to support TEM. We therefore transduced cultured HDMEC with FcRγII or CIITA to examine the formation of TEP and stimulation of TEM with this EC type. TNF-treated HDMEC, compared to HUVEC, show increased spontaneous (exogenous chemokine-independent) TEM of EM CD4+ T cells under conditions of shear stress, but still showed a further increment in response to exogenous IP-10. Both the spontaneous and the IP-10-induced increase in TEM is inhibited by TSST-1 in Vβ2TCR+ but not Vβ2TCR- CD4+ EM T cells (Figure 2A). In contrast to the HUVEC model, the TEM of Vβ2TCR+ cells across HDMEC monolayers increases over time, with approximately 50 % of Vβ2TCR+ cells transmigrating after 60 minutes (Figure 2A). As in our CIITA HUVEC model, only Vβ2TCR+ cells responding to TSST-1 presented by CIITA HDMEC form TEPs. Without exception, we have observed that TEPs form only in T cells attached near inter-EC junctions, as detected by VE-cadherin, DAPI and Vβ2TCR three color staining (Figure 2B). On HDMEC but not HUVEC monolayers, some of the T cell nuclei are observed within the TEP, and analysis of these images by deconvolution reveal that such T cells appear to be in the process of moving their cell body across the EC monolayer, either close to or through the inter-EC junctions (Figure 2B). On HDMEC, TCR-dependent TEP formation of EM CD4+ T cells is enhanced by shear stress, although some TEPs do form in the absence of flow (Figure 2C). As with HUVEC, NFAT translocation to the nuclei of Vβ2TCR+, but not Vβ2TCR-, EM CD4+ T cells is seen by 15’ (not shown), and is still evident in almost every cell stimulated by TSST-1 (77/78 for Vβ2TCR+, 3/79 for Vβ2TCR-) at 60’, even in Vβ2TCR+ EM CD4 cells that had transmigrated as well as those with a TEP (Figure 2D), suggesting that these T cells that undergo TEM in response to TCR signaling initiate a transcriptional program different from those T cells that transmigrate without engaging their TCR e.g., in response to IP-10.
Figure 2.
TCR-dependent TEP formation and TEM of EM CD4+ cells on cultured HDMEC. A. CIITA/TSST-1 HDMEC model. EM CD4+ T cell on TNF-treated CIITA HDMECs preloaded with TSST-1, overlaid or not with IP-10, and subjected to 15, 30, or 60 minutes flow. Left panel: mean and s.e.m. of Vβ2TCR+ cells with a TEP. No significant increase is observed between 15’ and 60’ and no effect of IP-10 is noted. Right panel: mean and s.e.m. of transmigrated cells. P<0.05 between Vβ2TCR- cells, with and without IP-10. P<0.001 between Vβ2TCR- and Vβ2TCR+ cells at 15’. P<0.001 between Vβ2TCR+ cells at 15’ compared to 60”. Note that there is a significant level of spontaneous TEM across HDMEC monolayers that is augmented by IP-10 and that TCR signals inhibit both spontaneous and IP-10-dependent TEM at 15’. However, TCR-stimulated cells do undergo TEM by 60’. B. TEP formation and TEM occurs at or near EC cell junctions. EM CD4+ cells on TNF-treated CIITA HDMECs preloaded with TSST-1 after 30 minutes flow, fixed and stained for Vβ2TCR (green), VE-cadherin (red), and DAPI (blue). Upper panels taken with a 40X objective show phase contrast and merged fluorescent staining; the top right panel is an enlarged view of the Vβ2TCR+ cell with a TEP. Lower panels show deconvoluted images (layer 8 and 20) from a series of 0.2 μm Z-stack of pictures taken with a 63X objective of Vβ2TCR staining (green) and DAPI (white) and merge of Vβ2TCR, DAPI, and VE-cadherin (red). Note that the TEP and DNA in the TEP are in focus coincident with HDMEC nuclei, but out of focus on the apical surface where the T cell body still resides. C. Shear stress augments TEP formation. EM CD4+ T cells were used in TEM assay for 15 or 30 minutes at 0 (static) or 1 dyne/cm2 (flow) on TNF-treated CIITA HDMEC pre-loaded or not with TSST-1. Numbers above brackets indicate p values between groups. D. Sustained NFAT nuclear translocation in transmigrated antigen-specific EM CD4+ T cells. EM CD4+ T cells on TNF-treated CIITA HDMEC preloaded with TSST-1 were fixed and stained for Vβ2TCR (blue), NFAT1 (green), and nuclei (DAPI, red) after 60’ flow. Arrow indicates a Vβ2TCR+ cell with a TEP and NFAT in the nucleus next to a Vβ2TCR- cell with NFAT excluded from the nucleus. Other transmigrated Vβ2TCR+ cells with NFAT nuclear colocalization, in contrast to the transmigrated Vβ2TCR- cells, are also evident, for example, in the upper portion. In 10 separate fields examined, NFAT translocation to the nucleus was observed in 77/78 and 3/79 of Vβ2TCR+ and Vβ2TCR- cells, respectively.
To confirm that this phenomenon was not confined to superantigen-delivered TCR signal, we also analyzed these parameters in HDMECs presenting TCR-activating antibody on the cell surface, i.e., on FcγRII-transduced HDMEC pre-loaded with the TCR activating mAb OKT3. TEM of TCR-engaged EM CD4+ cells occurs even faster with FcγRII-transduced HDMEC pre-loaded with the TCR activating mAb OKT3, with approximately 50% and 70% transmigrating after 30 and 60 minutes, respectively (P<0.001 for all comparisons, Figure 3A). Despite the kinetic differences, the similarity in the response in the two systems supports the hypothesis that TCR-mediated signals are responsible both for TEP formation and for TCR-mediated TEM of EM CD4+ T cells. Time lapse imaging of EM CD4+ T cells on FcγRII-transduced HDMEC pre-loaded with the TCR activating mAb OKT3 confirmed that TEP formation precedes TEM by the same T cells (Figure 3B).
Figure 3.
Photomicroscopic analyses of TCR formation and TEM in the FcγRII HDMEC/OKT3 model. A. Quantification of TEP formation and TEM of EM CD4+ cells at the indicated times under venous flow conditions on TNF-treated, FcγRII HDMEC preloaded with OKT3 mAb. Samples were fixed and stained with anti-CD3-FITC and DAPI and analyzed as described in Materials and Methods. Mean and s.e.m. of % of cells with a TEP and % TEM (left and right graph, respectively) from 10 groups of 20 cells each. P<0.001 for comparison of all TEM groups. B. Time lapse fluorescence and fluorescence deconvolution photomicroscopic analysis of TEP formation. PKH26-labeled EM CD4+ T cells on FcγRII HDMEC preloaded with OKT3 mAb, under flow.
Pictures were taken once every minute using a 10× objective and TRITC filter (upper panels). Shown are pictures corresponding to 6−45 minutes presented from left to right in each row. At the end of the flow assay, the sample was mounted on a slide in DAPI-containing mounting medium, and a series of Z-stack images were taken using a 63× objective and a DAPI filter at 0.3 μm intervals and deconvoluted. Lower panel shows two slices, 13 slices apart, from the deconvoluted images. The arrow points to the cell that had formed a TEP in the upper panels. Note that the EC nuclei and the nucleus of the T cell exhibiting a TEP are in the same plane of focus, whereas the nucleus of the other T cell in the same field that had not formed a TEP remains in a different plane, corresponding to the apical surface of the EC, as determined by phase contrast optics (not shown).
Since TEP formation and delayed TEM is TCR-driven, we investigated whether EC co-stimulatory molecules important for memory T cell activation, namely 41BBL, ICOSL, OX40L, and LFA-3, may contribute to this process by using blocking antibodies (7). No significant inhibitory effect is seen, either with individual antibodies or all four combined (data not shown).
To define the role of EC adhesion molecules in the processes of TEP formation and TCR-dependent TEM of EM CD4 cells, we measured these parameters using HDMEC co-transduced to express CIITA and either E-selectin, ICAM-1, VCAM-1, or all three (EIV), pre-loaded with TSST-1, and either overlaid or not with IP-10. TEM significantly increases between 15 and 60 minutes for Vβ2TCR+ cells on EIV, ICAM-1 and VCAM-1 transductants (P<0.001 in each case); ICAM-1 is more effective at supporting TEM of Vβ2TCR+ cells than either E-selectin or VCAM-1, which is reflected in the earlier appearance of TEPs on ICAM-1 transductants (Figure 4B, C). We also assessed the cells that were not TCR-engaged, i.e., the Vβ2TCR- cells. ICAM-1 and VCAM-1, but not E-selectin, supports rapid IP-10-dependent TEM (Figure 4C). Blocking antibodies to ICAM-1, but not E-selectin or VCAM-1, inhibited TCR-driven TEP formation and TEM on TNF-treated EC, but did not affect chemokine-driven TEM (Figure 6), suggesting that ICAM-1 is specifically required for TCR-driven TEP formation and TEM, but is redundant with VCAM-1 for chemokine-driven TEM of EM CD4+ T cells.
Figure 4.

TEM and TEP formation in flow assays using TSST-1 preloaded CIITA HDMECs co- transduced with E-selectin, ICAM-1, VCAM-1, or all three (EIV). A. FACS analysis of transductants. B. % of Vβ2TCR+ cells (Vβ2+, IP10, 15 and 60’) with a TEP. C. % TEM of EM CD4+ cells used in 15’ and 60’ flow assay with CIITA co-transductants preloaded with TSST-1 and overlaid or not with IP-10, fixed and stained for Vβ2TCR and nuclei (DAPI). Left graph shows Vβ2TCR- cells with or without IP-10 at 15 minutes. Right graph shows Vβ2TCR+ cells (with IP-10) at 15 and 60 minutes. Mean and s.e.m. from data combined from two separate experiments are shown. P<0.001 between 15 and 60 minutes for Vβ2TCR+ cells on EIV, ICAM-1 and VCAM-1 transductants.
Figure 6.
Both anti-ICAM-1 and anti-fractalkine antibodies block TCR-driven TEM, but only anti-ICAM-1 antibody blocks TEP formation. EM CD4+ T cells used in TEM assay on TNF-treated CIITA HDMEC preloaded with TSST-1 and incubated with IgG, anti-E-selectin, anti-ICAM-1, anti-VCAM-1 or anti-fractalkine blocking antibody. Shown are the mean and s.e.m. of % TEM (left graph; P<0.001 (**) between Vβ2TCR+ cells at 60’ on HDMEC treated with IgG or with anti-ICAM-1 or anti-fraktalkine Ab) and % TEP of Vβ2TCR+ cells (right graph; P<0.05 (*) between cells at 60’ on HDMEC treated or not with anti-ICAM-1 or anti-fractalkine Ab).
Chemokine-driven TEM of T cells across HUVEC has been shown to be dependent on heterotrimeric G protein (of the Gαi class, blocked by pertussis toxin), but independent of PI-3 kinase (blocked by LY294002) signals (19). We investigated whether these agents would affect TCR-dependent TEM. Pertussis toxin inhibits rapid TEM of EM CD4+ T cells across HDMEC, whereas Ly294002 has no effect. Pertussis toxin also blocks both TCR-dependent TEP formation and TEM whereas LY294002 does not (Figure 5). The inhibitory effect of pertussis toxin is not mimicked by cholera toxin (data not shown), a control experiment to rule out an indirect inhibitory effect due to elevation of cAMP levels, a common effect of both toxins. These results suggest that the process of TCR-driven TEM also involves a Gαi signaling pathway.
Figure 5.

Analysis of signals that contribute to antigen-driven TEM. A. Antigen-driven TEM is pertussis toxin sensitive. EM CD4+ T cells were treated with vehicle, LY294002 (LY), or pertussis toxin (ptx) prior to flow assays on TNF-treated CIITA HDMEC preloaded with TSST-1. Shown are the mean and s.e.m. of % TEM (left graph; P<0.001 for comparison of Vβ2TCR- cells ptx, 15’ compared to vehicle and LY 15’; P<0.001 for comparison of Vβ2TCR+ cells ptx, 60’ compared to vehicle and LY 60’) and % TEP of Vβ2TCR+ cells (right graph; P<0.001 for comparison of vehicle to ptx at 15’; P<0.05 for comparison of vehicle to ptx at 60’; P>0.05 for comparisons of vehicle vs LY at both 15’ and 60’).
One attractive candidate EC ligand/EM CD4+ T cell counter-ligand interaction that may involve GαI signaling is fractalkine (CX3CL1)/fractalkine receptor (CX3CR1). Fractalkine is a unique chemokine possessing a long mucin stalk and a transmembrane region that serves as both an activator and adhesion molecule for leukocytes (25). To investigate the role of fractalkine in TCR-dependent TEP formation and TEM, we used fractalkine blocking antibody in our CIITA HDMEC model. The number of TEPs increased over time while the number of TCR-driven transmigrated cells was inhibited, indicating that fractalkine antibody blocks TCR-driven TEM at a point after TEP formation (Figure 6).
Discussion
We have described the formation of TEPs, novel cell morphological structures restricted to EM CD4+ T cells interacting with antigen presenting ECs whose formation are augmented under conditions of blood flow. The HDMEC model indicates that this structure is, in fact, formed as the first step in antigen-specific TEM of EM CD4+ T cells across microvascular ECs. TEPs clearly differ from lamellipods, pseudopods, and even the very recently described invasive podosome of activated human lymphocytes undergoing transcellular diapedesis (23). The distinctions with this latter structure are worth noting. First, invasive podosomes (IP) are confined to T cells undergoing transcellular (through the EC), as opposed to paracellular (around the EC; between ECs) diapedesis, whereas TEPs appear to extend through EC junctions. Second, TEP formation depends on TCR signals whereas IP are formed in response to chemokines. Third, TEPs are formed by EM freshly isolated from peripheral blood whereas IP are only formed by activated human T cells cultured for several days with IL-2. Fourth, TEM associated with IP formation occur with the same kinetics as TEM without IP whereas antigen-dependent TEP formation and eventual TEM is somewhat delayed in comparison to rapid chemokine-driven TEM. Fifth, to initiate transcellular IP, T cells must be spread out over the EC surface, forming so-called podoprints that precede the large pore; TEP formation is preceded by cells rounding up into a compact ball on the EC surface, at cellular junctions. Finally, IP formation is unaffected by shear stress, in contrast to TEP formation, which is augmented by it.
NFAT translocation to the nuclei of antigen-specific Vβ2TCR+ EM CD4 cells that had transmigrated suggests that these T cells may initiate a transcriptional program different from those T cells that transmigrate without engaging their TCR. This has important implications in that TCR-activated CD4+ EM T cells could produce cytokines, e.g., IFN-γ and TNF, which would induce inflammatory activation of the EC and thereby upregulate adhesion molecules and chemokines on the EC (26). In other words, early TCR-dependent recruitment of EM CD4+ T cells could trigger subsequent TCR-independent recruitment of additional EM CD4+ T cells. This may be relevant to the eventual outcome of an immune response as recent data from our laboratory has shown that so-called bystander T cells, which have not received a TCR signal, and have likely been recruited by chemokines such as IP-10, may enhance the response of antigen-activated T cells through release of low levels of NO (27). In contrast to bystander T cells, TCR activated T cells suppress iNOS expression, allowing these cells to avoid inhibition evident at higher levels of NO. These in vivo experiments, like our in vitro model, support the idea that chemokines and TCR signals produce distinct patterns of gene expression in infiltrating T cells.
Analysis of the roles of EC adhesion molecules, both by transduction with E-selectin, ICAM-1, VCAM-1, or all three, and by blocking antibodies, indicates that ICAM-1 plays a nonredundant role in TCR-driven TEP formation and TEM, but can be substituted by other adhesion molecules (probably VCAM-1, as we previously showed for transmigration across HUVEC monolayers) in inflammatory chemokine-dependent TEM. The key point is that TCR-driven TEM uses different EC molecules than does IP-10- driven TEM. A second important difference involves the requirement for fractalkine, which also may serve as an adhesion molecule in addition to its role as a chemokine (28-30).
Blocking antibodies to the EC costimulatory molecules 41BBL, ICOSL, OX40L, and LFA-3 failed to affect the TCR-mediated block of rapid TEM in response to IP-10 on HUVEC (17), as well as TEP formation and TEM of EM CD4+ T cells on HDMEC. These molecules have been shown to affect cytokine production and proliferation (7), implying that TCR signals in TEP may be co-stimulator-independent. However, fractalkine has been shown to be a co-stimulatory molecule for CX3CR1+ CD4+ T cell activation by non-professional APCs (31), and here we show that fractalkine contributes to TCR-dependent TEM of EM CD4+ T cells.
A role for fractalkine in TCR-dependent TEM of EM CD4+ T cells is consistent with several additional observations. First, CX3CR1 is expressed on EM, but not CM or naïve CD4+ T cells, which may partly explain why only this subtype under goes TCR-driven TEM (28, 32, 33). Interestingly, monocytes present a similar scenario, in which a small subset (5−10%), namely CD16+, express CX3CR1 and respond to fractalkine (34). Second, TNF induces more fractalkine in microvascular EC as compared to macrovascular EC (HUVEC), which may underlie the differences between TCR-driven TEM on HDMEC and HUVEC (22). Our findings also suggest that fractalkine can initiate responses of EM CD4+ T cells not induced by other (soluble) chemokines present on the EC surface and that fractalkine responses, unlike those initiated by other chemokines, are not sensitive to inhibition by TCR signals. The basis for this difference is unclear. The PTX-inhibited step in both types of TEM is likely to be downstream of chemokine receptors since these molecules are known to signal via PTX-sensitive trimeric G proteins that utilize Gαi. The absence of an effect of a PI-3 kinase inhibitor distinguishes this process from that recently described for mouse T cells (35). Further analysis of the signaling events that distinguish TCR-driven from IP-10 driven TEM is a promising future direction for research. Defining the molecular underpinnings regulating antigen-specific TEP formation and delayed TEM of EM CD4+ T cells may identify unique therapeutic targets for T cell-mediated inflammatory reactions such as acute allograft rejection.
Acknowledgments
We thank William Sessa for use of the immunofluorescence microscope, Nicholas Torpey for the CIITA retrovirus producing cells, Martin Kluger for the use of the flow chamber apparatus, and Louise Benson, Gwendoline Davis, and Lisa Gras for excellent assistance in cell culture.
Abbreviations
- TEM
Transendothelial migration
- TEP
transendothelial protrusion
- EM
effector memory
- CM
central memory
- HUVEC
human umbilical vein endothelial cells
- HDMEC
human dermal microvascular endothelial cells
Footnotes
Grant Support: This work is funded by National Institute of Health grants P01-HL070295 and HL051014.
References
- 1.Hayry P, von Willebrand E, Andersson LC. Expression of HLA-ABC and -DR locus antigens on human kidney, endothelial, tubular and glomerular cells. Scand J Immunol. 1980;11:303–310. doi: 10.1111/j.1365-3083.1980.tb00238.x. [DOI] [PubMed] [Google Scholar]
- 2.Natali PG, De Martino C, Quaranta V, Nicotra MR, Frezza F, Pellegrino MA, Ferrone S. Expression of Ia-like antigens in normal human nonlymphoid tissues. Transplantation. 1981;31:75–78. doi: 10.1097/00007890-198101000-00017. [DOI] [PubMed] [Google Scholar]
- 3.Hancock WW, Kraft N, Atkins RC. The immunohistochemical demonstration of major histocompatibility antigens in the human kidney using monoclonal antibodies. Pathology. 1982;14:409–414. doi: 10.3109/00313028209092120. [DOI] [PubMed] [Google Scholar]
- 4.Epperson DE, Pober JS. Antigen-presenting function of human endothelial cells. Direct activation of resting CD8 T cells. J Immunol. 1994;153:5402–5412. [PubMed] [Google Scholar]
- 5.Dengler TJ, Pober JS. Human vascular endothelial cells stimulate memory but not naive CD8+ T cells to differentiate into CTL retaining an early activation phenotype. J Immunol. 2000;164:5146–5155. doi: 10.4049/jimmunol.164.10.5146. [DOI] [PubMed] [Google Scholar]
- 6.Khayyamian S, Hutloff A, Buchner K, Grafe M, Henn V, Kroczek RA, Mages HW. ICOS-ligand, expressed on human endothelial cells, costimulates Th1 and Th2 cytokine secretion by memory CD4+ T cells. Proc Natl Acad Sci U S A. 2002;99:6198–6203. doi: 10.1073/pnas.092576699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Shiao SL, McNiff JM, Pober JS. Memory T cells and their costimulators in human allograft injury. J Immunol. 2005;175:4886–4896. doi: 10.4049/jimmunol.175.8.4886. [DOI] [PubMed] [Google Scholar]
- 8.Shiao SL, Kirkiles-Smith NC, Shepherd BR, McNiff JM, Carr EJ, Pober JS. Human Effector Memory CD4+ T Cells Directly Recognize Allogeneic Endothelial Cells In Vitro and In Vivo. J Immunol. 2007;179:4397–4404. doi: 10.4049/jimmunol.179.7.4397. [DOI] [PubMed] [Google Scholar]
- 9.Savinov AY, Wong FS, Stonebraker AC, Chervonsky AV. Presentation of antigen by endothelial cells and chemoattraction are required for homing of insulin-specific CD8+ T cells. J Exp Med. 2003;197:643–656. doi: 10.1084/jem.20021378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Marelli-Berg FM, James MJ, Dangerfield J, Dyson J, Millrain M, Scott D, Simpson E, Nourshargh S, Lechler RI. Cognate recognition of the endothelium induces HY-specific CD8+ T-lymphocyte transendothelial migration (diapedesis) in vivo. Blood. 2004;103:3111–3116. doi: 10.1182/blood-2003-08-2717. [DOI] [PubMed] [Google Scholar]
- 11.Galea I, Bernardes-Silva M, Forse PA, van Rooijen N, Liblau RS, Perry VH. An antigen-specific pathway for CD8 T cells across the blood-brain barrier. J Exp Med. 2007;204:2023–2030. doi: 10.1084/jem.20070064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Choo JK, Seebach JD, Nickeleit V, Shimizu A, Lei H, Sachs DH, Madsen JC. Species differences in the expression of major histocompatibility complex class II antigens on coronary artery endothelium: implications for cell-mediated xenoreactivity. Transplantation. 1997;64:1315–1322. doi: 10.1097/00007890-199711150-00014. [DOI] [PubMed] [Google Scholar]
- 13.Takei Y, Sims TN, Urmson J, Halloran PF. Central role for interferon-gamma receptor in the regulation of renal MHC expression. J Am Soc Nephrol. 2000;11:250–261. doi: 10.1681/ASN.V112250. [DOI] [PubMed] [Google Scholar]
- 14.Kreisel D, Krasinskas AM, Krupnick AS, Gelman AE, Balsara KR, Popma SH, Riha M, Rosengard AM, Turka LA, Rosengard BR. Vascular endothelium does not activate CD4+ direct allorecognition in graft rejection. J Immunol. 2004;173:3027–3034. doi: 10.4049/jimmunol.173.5.3027. [DOI] [PubMed] [Google Scholar]
- 15.Savage CO, Hughes CC, McIntyre BW, Picard JK, Pober JS. Human CD4+ T cells proliferate to HLA-DR+ allogeneic vascular endothelium. Identification of accessory interactions. Transplantation. 1993;56:128–134. doi: 10.1097/00007890-199307000-00024. [DOI] [PubMed] [Google Scholar]
- 16.Tay SS, McCormack A, Lawson C, Rose ML. IFN-gamma reverses the stop signal allowing migration of antigen-specific T cells into inflammatory sites. Journal of Immunology. 2003;170:3315–3322. doi: 10.4049/jimmunol.170.6.3315. [DOI] [PubMed] [Google Scholar]
- 17.Manes TD, Shiao SL, Dengler TJ, Pober JS. TCR signaling antagonizes rapid IP-10-mediated transendothelial migration of effector memory CD4+ T cells. J Immunol. 2007;178:3237–3243. doi: 10.4049/jimmunol.178.5.3237. [DOI] [PubMed] [Google Scholar]
- 18.Marelli-Berg FM, Frasca L, Weng L, Lombardi G, Lechler RI. Antigen recognition influences transendothelial migration of CD4+ T cells. J Immunol. 1999;162:696–703. [PubMed] [Google Scholar]
- 19.Cinamon G, Shinder V, Alon R. Shear forces promote lymphocyte migration across vascular endothelium bearing apical chemokines. Nat Immunol. 2001;2:515–522. doi: 10.1038/88710. [DOI] [PubMed] [Google Scholar]
- 20.Manes TD, Pober JS, Kluger MS. Endothelial cell-T lymphocyte interactions: iP-10 stimulates rapid transendothelial migration of human effector but not central memory CD4+ T cells. Requirements for shear stress and adhesion molecules. Transplantation. 2006;82:S9–14. doi: 10.1097/01.tp.0000231356.57576.82. [DOI] [PubMed] [Google Scholar]
- 21.Aird WC. Phenotypic heterogeneity of the endothelium: I. Structure, function, and mechanisms. Circ Res. 2007;100:158–173. doi: 10.1161/01.RES.0000255691.76142.4a. [DOI] [PubMed] [Google Scholar]
- 22.Beck G, Ludwig F, Schulte J, van Ackern K, van der Woude FJ, Yard BA. Fractalkine is not a major chemoattractant for the migration of neutrophils across microvascular endothelium. Scand J Immunol. 2003;58:180–187. doi: 10.1046/j.1365-3083.2003.01298.x. [DOI] [PubMed] [Google Scholar]
- 23.Carman CV, Sage PT, Sciuto TE, de la Fuente MA, Geha RS, Ochs HD, Dvorak HF, Dvorak AM, Springer TA. Transcellular diapedesis is initiated by invasive podosomes. Immunity. 2007;26:784–797. doi: 10.1016/j.immuni.2007.04.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Clark PR, Manes TD, Pober JS, Kluger MS. Increased ICAM-1 expression causes endothelial cell leakiness, cytoskeletal reorganization and junctional alterations. J Invest Dermatol. 2007;127:762–774. doi: 10.1038/sj.jid.5700670. [DOI] [PubMed] [Google Scholar]
- 25.Bazan JF, Bacon KB, Hardiman G, Wang W, Soo K, Rossi D, Greaves DR, Zlotnik A, Schall TJ. A new class of membrane-bound chemokine with a CX3C motif. Nature. 1997;385:640–644. doi: 10.1038/385640a0. [DOI] [PubMed] [Google Scholar]
- 26.Choi J, Enis DR, Koh KP, Shiao SL, Pober JS. T lymphocyteendothelial cell interactions. Annu Rev Immunol. 2004;22:683–709. doi: 10.1146/annurev.immunol.22.012703.104639. [DOI] [PubMed] [Google Scholar]
- 27.Choy JC, Wang Y, Tellides G, Pober JS. Induction of inducible NO synthase in bystander human T cells increases allogeneic responses in the vasculature. Proc Natl Acad Sci U S A. 2007 doi: 10.1073/pnas.0607731104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Imai T, Hieshima K, Haskell C, Baba M, Nagira M, Nishimura M, Kakizaki M, Takagi S, Nomiyama H, Schall TJ, Yoshie O. Identification and molecular characterization of fractalkine receptor CX3CR1, which mediates both leukocyte migration and adhesion. Cell. 1997;91:521–530. doi: 10.1016/s0092-8674(00)80438-9. [DOI] [PubMed] [Google Scholar]
- 29.Fong AM, Robinson LA, Steeber DA, Tedder TF, Yoshie O, Imai T, Patel DD. Fractalkine and CX3CR1 mediate a novel mechanism of leukocyte capture, firm adhesion, and activation under physiologic flow. J Exp Med. 1998;188:1413–1419. doi: 10.1084/jem.188.8.1413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Haskell CA, Cleary MD, Charo IF. Molecular uncoupling of fractalkine-mediated cell adhesion and signal transduction. Rapid flow arrest of CX3CR1-expressing cells is independent of G-protein activation. J Biol Chem. 1999;274:10053–10058. doi: 10.1074/jbc.274.15.10053. [DOI] [PubMed] [Google Scholar]
- 31.Sawai H, Park YW, Roberson J, Imai T, Goronzy JJ, Weyand CM. T cell costimulation by fractalkine-expressing synoviocytes in rheumatoid arthritis. Arthritis Rheum. 2005;52:1392–1401. doi: 10.1002/art.21140. [DOI] [PubMed] [Google Scholar]
- 32.Foussat A, Coulomb-L'Hermine A, Gosling J, Krzysiek R, Durand-Gasselin I, Schall T, Balian A, Richard Y, Galanaud P, Emilie D. Fractalkine receptor expression by T lymphocyte subpopulations and in vivo production of fractalkine in human. Eur J Immunol. 2000;30:87–97. doi: 10.1002/1521-4141(200001)30:1<87::AID-IMMU87>3.0.CO;2-7. [DOI] [PubMed] [Google Scholar]
- 33.Chtanova T, Newton R, Liu SM, Weininger L, Young TR, Silva DG, Bertoni F, Rinaldi A, Chappaz S, Sallusto F, Rolph MS, Mackay CR. Identification of T cell-restricted genes, and signatures for different T cell responses, using a comprehensive collection of microarray datasets. J Immunol. 2005;175:7837–7847. doi: 10.4049/jimmunol.175.12.7837. [DOI] [PubMed] [Google Scholar]
- 34.Ancuta P, Rao R, Moses A, Mehle A, Shaw SK, Luscinskas FW, Gabuzda D. Fractalkine preferentially mediates arrest and migration of CD16+ monocytes. J Exp Med. 2003;197:1701–1707. doi: 10.1084/jem.20022156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Jarmin SJ, David R, Ma L, Chai JG, Dewchand H, Takesono A, Ridley AJ, Okkenhaug K, Marelli-Berg FM. T cell receptor-induced phosphoinositide-3-kinase p110delta activity is required for T cell localization to antigenic tissue in mice. J Clin Invest. 2008;118:1154–1164. doi: 10.1172/JCI33267. [DOI] [PMC free article] [PubMed] [Google Scholar]