Abstract
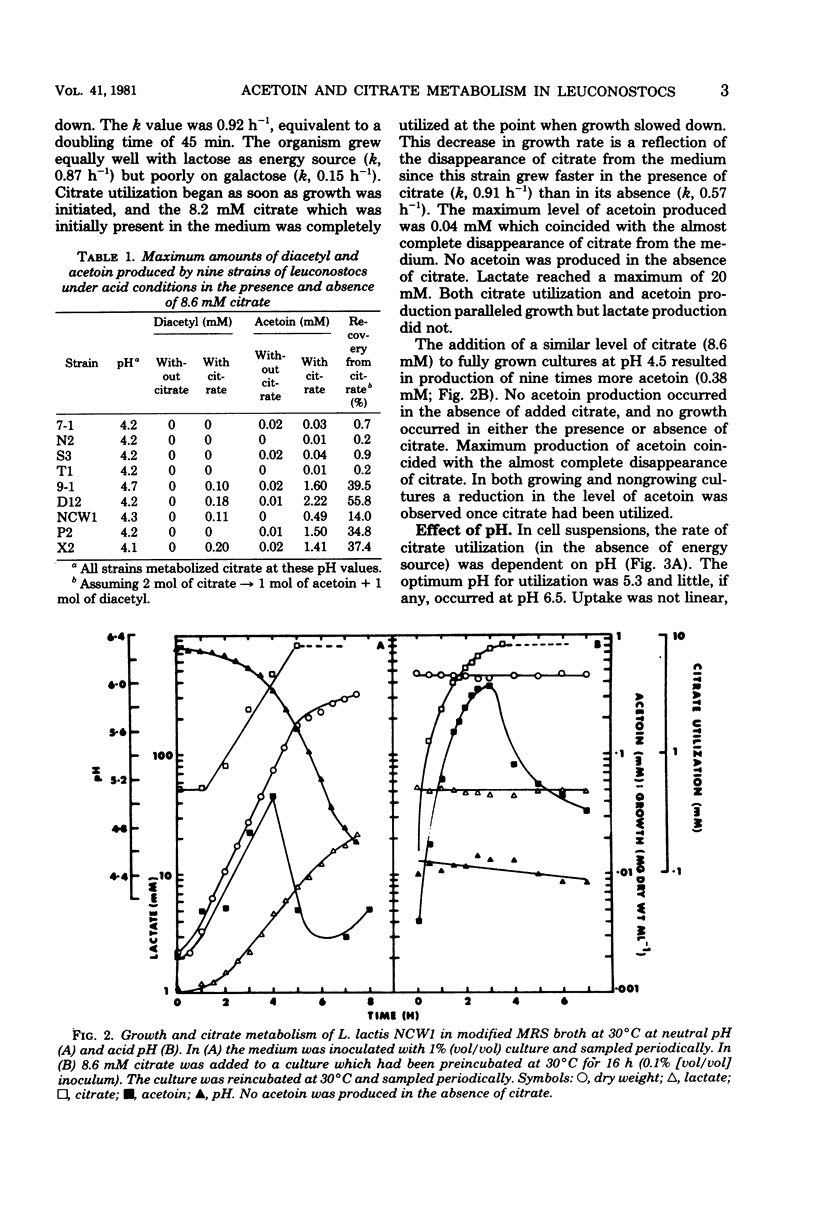
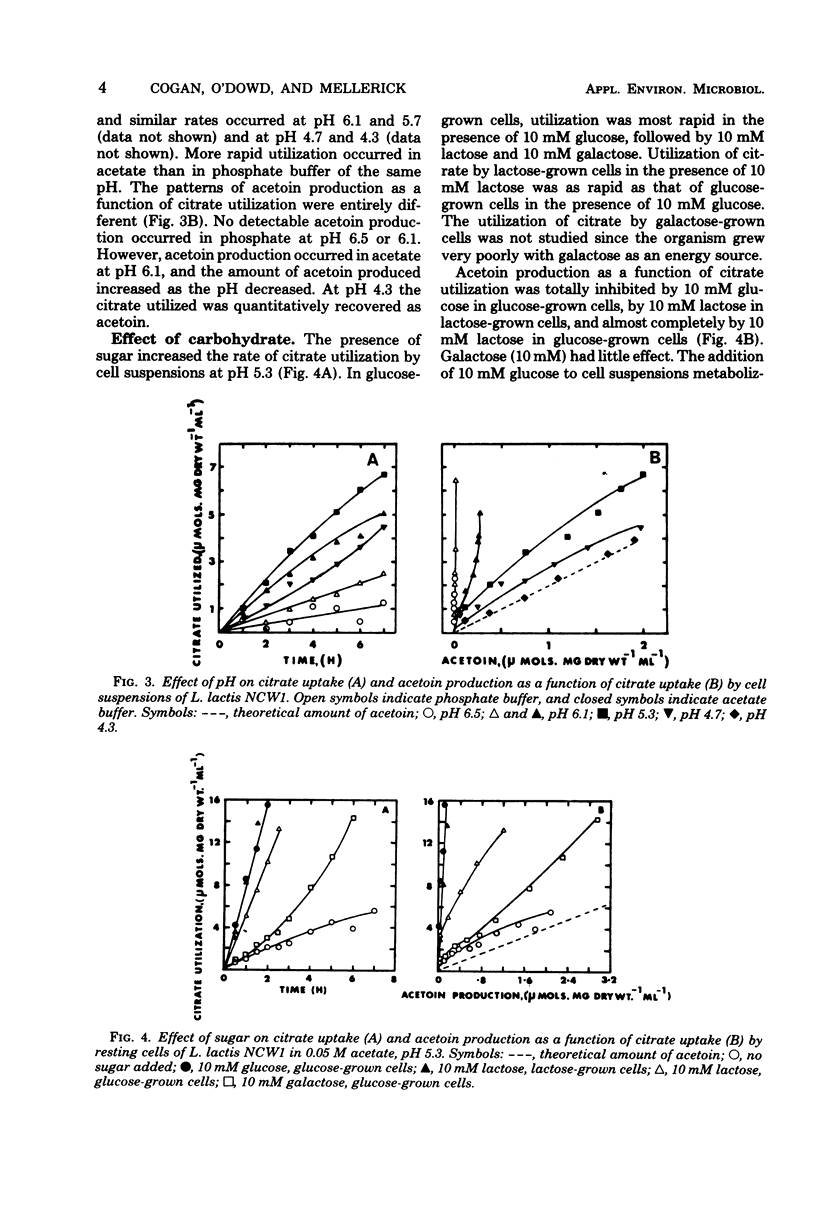
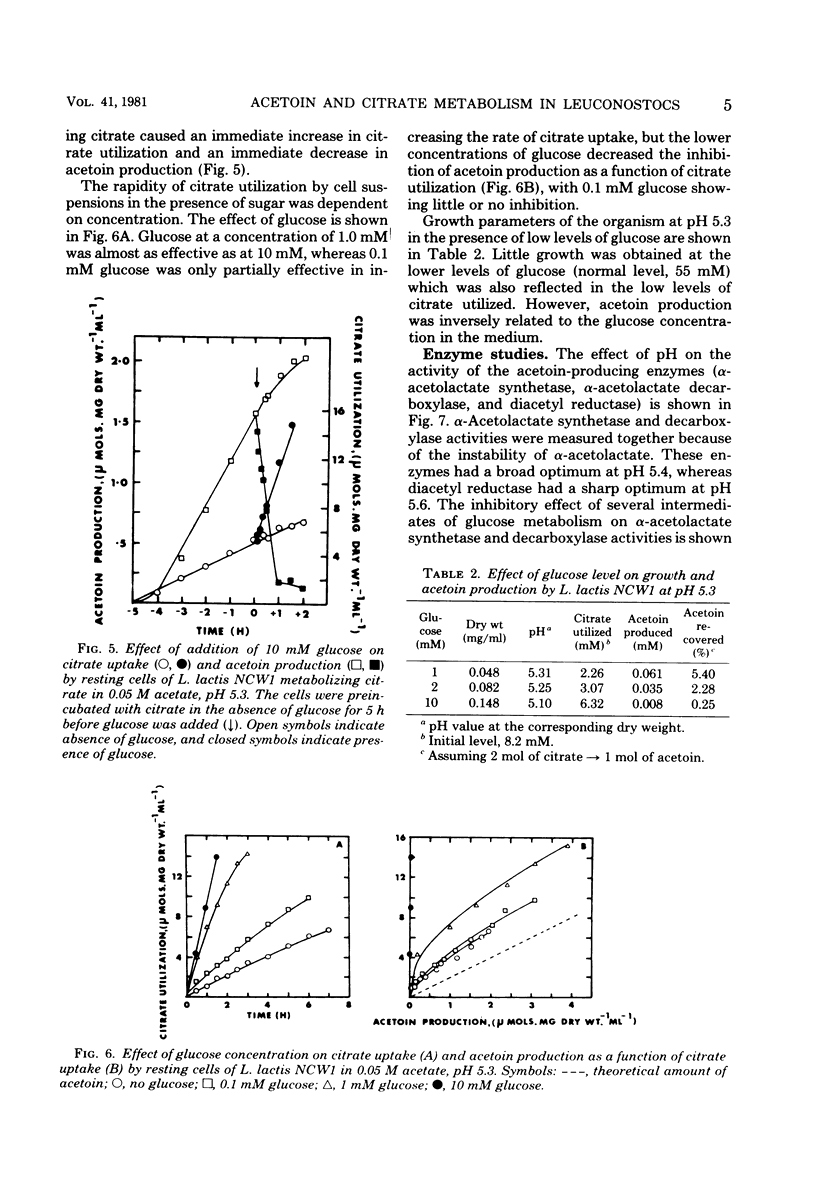
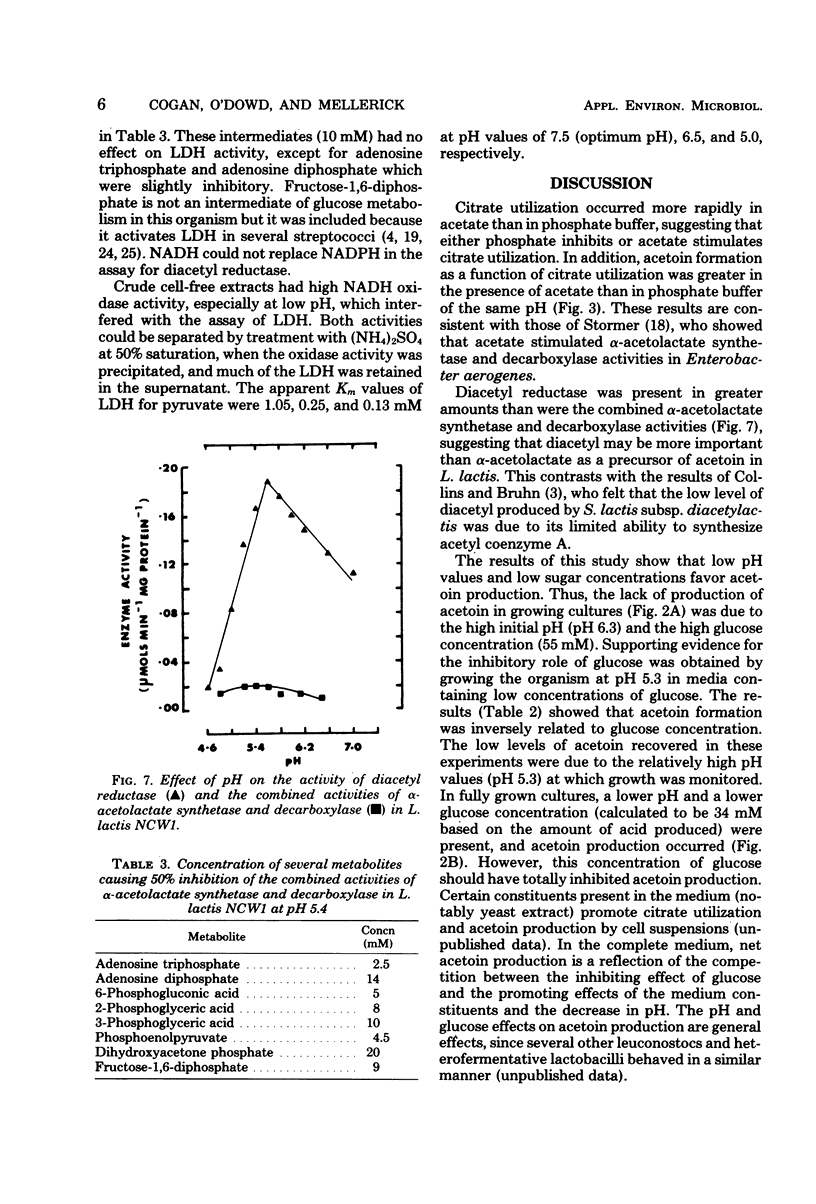
The relationship between acetoin production and citrate utilization in Leuconostoc lactis NCW1 was studied. In a complex medium the organism utilized citrate at neutral pH (initial pH, 6.3) and at acid pH (initial pH, 4.5) but produced nine times more acetoin at the latter pH. In resting cells the utilization of citrate was optimum at pH 5.3. Production of acetoin as a function of citrate utilization increased as the pH decreased, and at pH 4.3 all of the citrate utilized was recovered as acetoin. Glucose (10 mM) and lactose (10 mM) markedly stimulated citrate utilization but totally inhibited acetoin production in glucose- and lactose-grown cells. Addition of glucose to cells actively metabolizing citrate caused an immediate increase in citrate uptake and a reduction in the level of acetoin. The apparent Km values of lactic dehydrogenase for pyruvate were 1.05, 0.25, and 0.15 mM at pH 7.5, 6.5, and 5.0, respectively. Several heterofermentation intermediates inhibited α-acetolactate synthetase and decarboxylase activities. The implications of these results in regulating acetoin formatin are discussed.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Chuang L. F., Collins E. B. Biosynthesis of diacetyl in bacteria and yeast. J Bacteriol. 1968 Jun;95(6):2083–2089. doi: 10.1128/jb.95.6.2083-2089.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cogan T. M. Citrate utilization in milk by Leuconostoc cremoris and Streptococcus diacetilactis. J Dairy Res. 1975 Feb;42(1):139–146. doi: 10.1017/s0022029900015168. [DOI] [PubMed] [Google Scholar]
- Collins E. B., Bruhn J. C. Roles of acetate and pyruvate in the metabolism of Streptococcus diacetilactis. J Bacteriol. 1970 Sep;103(3):541–546. doi: 10.1128/jb.103.3.541-546.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crow V. L., Pritchard G. G. Fructose 1,6-diphosphate-activated L-lactate dehydrogenase from Streptococcus lactis: kinetic properties and factors affecting activation. J Bacteriol. 1977 Jul;131(1):82–91. doi: 10.1128/jb.131.1.82-91.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DAY H. S. Planning and equipping a disease diagnostic control laboratory. Lab Anim Care. 1963 Jun;2:431–441. [PubMed] [Google Scholar]
- Drinan D. F., Robin S., Cogan T. M. Citric acid metabolism in hetero- and homofermentative lactic acid bacteria. Appl Environ Microbiol. 1976 Apr;31(4):481–486. doi: 10.1128/aem.31.4.481-486.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gordon G. L., Doelle H. W. Molecular aspects for the metabolic regulation of the nicotinamide adenine dinucleotide-dependent D(-)-lactate dehydrogenase from Leuconostoc. Microbios. 1974 Mar-Apr;9(36):199–215. [PubMed] [Google Scholar]
- HARVEY R. J., COLLINS E. B. Role of citritase in acetoin formation by Streptococcus diacetilactis and Leuconostoc citrovorum. J Bacteriol. 1961 Dec;82:954–959. doi: 10.1128/jb.82.6.954-959.1961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johansen L., Bryn K., Stormer F. C. Physiological and biochemical role of the butanediol pathway in Aerobacter (Enterobacter) aerogenes. J Bacteriol. 1975 Sep;123(3):1124–1130. doi: 10.1128/jb.123.3.1124-1130.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Maloney P. C. Membrane H+ conductance of Streptococcus lactis. J Bacteriol. 1979 Oct;140(1):197–205. doi: 10.1128/jb.140.1.197-205.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Brien R. W. Induction of citrate lyase in Enterobacter cloacae grown under aerated conditions and its effect on citrate metabolism. J Bacteriol. 1975 Dec;124(3):1084–1088. doi: 10.1128/jb.124.3.1084-1088.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Speckman R. A., Collins E. B. Diacetyl biosynthesis in Streptococcus diacetilactis and Leuconostoc citrovorum. J Bacteriol. 1968 Jan;95(1):174–180. doi: 10.1128/jb.95.1.174-180.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stormer F. C. Evidence for regulation of Aerobacter aerogenes pH 6 acetolactate-forming enzyme by acetate ion. Biochem Biophys Res Commun. 1977 Feb 7;74(3):898–902. doi: 10.1016/0006-291x(77)91603-5. [DOI] [PubMed] [Google Scholar]
- Thomas T. D. Regulation of lactose fermentation in group N streptococci. Appl Environ Microbiol. 1976 Oct;32(4):474–478. doi: 10.1128/aem.32.4.474-478.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson J. In vivo regulation of glycolysis and characterization of sugar: phosphotransferase systems in Streptococcus lactis. J Bacteriol. 1978 Nov;136(2):465–476. doi: 10.1128/jb.136.2.465-476.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WOLIN M. J. FRUCTOSE-1,6-DIPHOSPHATE REQUIREMENT OF STREPTOCOCCAL LACTIC DEHYDROGENASES. Science. 1964 Nov 6;146(3645):775–777. doi: 10.1126/science.146.3645.775. [DOI] [PubMed] [Google Scholar]
- Yamada T., Carlsson J. Regulation of lactate dehydrogenase and change of fermentation products in streptococci. J Bacteriol. 1975 Oct;124(1):55–61. doi: 10.1128/jb.124.1.55-61.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]


