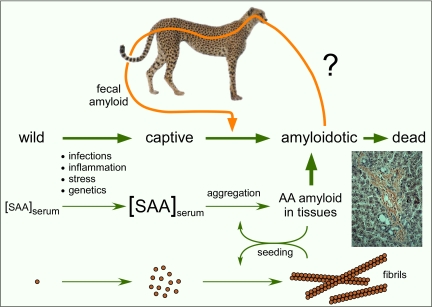
The misfolding and aggregation of proteins is often an accident waiting to happen. Consequently, organisms have developed sophisticated chaperone and quality-control systems to limit abnormal protein interactions and the accumulation of toxic aggregates (1). However, sometimes these systems can be overwhelmed, and diseases, namely protein misfolding diseases, can result. One such disease, amyloid protein A (AA) amyloidosis, is wreaking havoc in the captive cheetah population, complicating efforts to rescue this endangered species from extinction (2, 3). One key to managing this fatal disease in cheetahs is to understand why it is so prevalent. Most cases of AA amyloidosis in mammals appear to occur spontaneously, usually as a result of chronic inflammation or genetic peculiarities that predispose the organism to the deposition of serum amyloid A (SAA) protein in fibrillar deposits called amyloid (Fig. 1). In this issue of PNAS, Zhang et al. (4) report that AA amyloid is excreted in the feces of cheetahs with AA amyloidosis and that this fecal amyloid can in turn promote a similar disease in mice. These results suggest that cheetah AA amyloidosis may not be simply a spontaneous disease, but also a natural prion-like, transmissible protein misfolding disease.
Fig. 1.
Diagram of AA amyloid formation and the potential prion-like transmission of AA amyloidosis by fecal shedding and oral uptake of the amyloid. The photo shows an example of Congo red-stained AA amyloid fibril deposits in hamster liver tissue (courtesy of John Coe, Rocky Mountain Laboratories, National Institute of Allergy and Infectious Diseases).
Prions are protein-based infectious agents or elements of inheritance that, unlike conventional pathogens, lack agent-specific nucleic acid genomes (5, 6). Prions have been described in both mammals (e.g., bovine spongiform encephalopathy and Creutzfeldt–Jakob disease) and fungi ([URE3], [PSI+], and [Het-s]). Replication of prions requires a self-propagating modification of an otherwise non-prion host protein. Usually the mechanism involves the recruitment of the normal form of the protein into a growing amyloid-like prion aggregate. In many cases the presence of prions is a disease state, but some prions play normal physiological roles (7). Although amyloid-like protein aggregation is typical of many important protein misfolding diseases, including Alzheimer's disease and type 2 diabetes, most of these diseases are not known to be naturally transmissible or heritable because of transfer of the amyloid. However, experimental inoculations of amyloid preparations can enhance amyloidosis in naïve mice that are strongly primed for the development of amyloidosis (8, 9). This suggests that there is potential for amyloidoses, in general, and AA amyloidosis specifically, to be transmissible and, hence, prion-like. For such transmissions to be significant in the real world, there must be practical routes of transmission and the potential for inducing disease in natural, rather than artificially primed, hosts.
The shedding of AA amyloid into the feces of cheetahs suggests a potential route of transmission (Fig. 1) (4). Although the fecal amyloid can promote amyloidosis on i.v. inoculation into mice, this is only true in mice that were primed for amyloidosis by injections of an inflammatory chemical (silver nitrate) that dramatically boosts serum SAA levels. The silver nitrate treatment alone causes spontaneous amyloidosis in these mice, albeit at a slower pace than when the mice are inoculated with exogenous amyloid. Thus, it remains to be determined whether fecal amyloid can actually initiate, rather than enhance, amyloidosis in either mice or cheetahs and, if so, by what route of inoculation. One possible mode of entry would be oral because AA amyloid can be active in primed mice when administered orally as well as intravenously (9, 10). Another potential route would be direct inoculation of fecal amyloid into the blood stream through cuts or abrasions. It should be noted that Zhang et al. (4) inoculated mice with highly concentrated preparations of fecal amyloid. Hence, it is unknown whether amyloid concentrations in feces would allow transmission to naïve recipients by any peripheral route.
If fecal amyloid can be transmitted to other captive cheetahs, what makes these animals so susceptible to AA amyloidosis? The fact that mice can be primed for AA amyloidosis by inflammatory stimuli raises the possibility that inflammation is also important in cheetahs. Indeed, inflammatory diseases are prominent in captive cheetahs with AA amyloidosis, and a number of precipitating factors, including chronic infections, diet, and stress, have been identified (2, 3). Other possibilities include genetic predispositions of cheetahs to AA amyloidosis because of their SAA sequence or expression level. Interestingly, a gene polymorphism has been identified in captive cheetahs that flanks the SAA1 gene and strongly affects its transcriptional induction in response to inflammation (11). Expression of other proteins can also profoundly enhance susceptibility of animals to AA amyloidosis, as shown by modulation of pentraxin levels in hamsters (12). The genetic homogeneity of captive cheetahs may enhance these susceptibility problems (11, 13), but does not appear to be the sole issue (3).
The very factors that might make cheetahs susceptible to exogenous AA amyloid “infections” should also potentiate spontaneous AA amyloidosis in these animals. There is precedent for this in the spontaneous amyloidosis that occurs in silver nitrate-primed mice. By analogy, it remains possible that the high incidence of AA amyloidosis in cheetahs is caused by spontaneous disease exacerbated by the inflammatory stimuli, stresses, and inbreeding of captivity rather than exposure to fecal amyloid. Further studies will be required to resolve these questions.
AA amyloidosis susceptibility issues may have serious implications for cheetah conservation efforts. If the objective is to rescue the wild cheetah population by releasing cheetahs bred in captivity, then it will be important to know the impact of releasing amyloidotic animals into the wild. Will AA amyloidosis continue to progress and affect the survival of released cheetahs? Can the disease be spread to wild cheetahs? One encouraging observation is that, relative to captive cheetahs, wild Namibian cheetahs are remarkably free of disease, including inflammatory diseases such as AA amyloidosis and gastritis (3). Perhaps lower chronic levels of inflammation, and hence serum SAA levels, make them less susceptible than captive cheetahs to AA amyloid shed by other animals.
Although major questions remain about the etiology of AA amyloidosis in captive cheetahs, it may be wise to take measures to limit exposure of cheetahs to potential sources of amyloid “infectivity.” The demonstration that cheetah AA amyloid is active in mice indicates that there is cross-species promiscuity in its amyloid-inducing capacity. This promiscuity might also work in reverse, rendering cheetahs susceptible to AA-amyloid-laden tissues of other species that might be fed to them. Interestingly, foie gras was recently shown to contain AA amyloid that could accelerate amyloidosis when fed to mice (9). Thus, consideration of a variety of potential sources of exposure for cheetahs seems warranted. Furthermore, if chronic inflammation enhances disease susceptibility, then anti-inflammatory therapies may be helpful.
Relative to captive cheetahs, wild Namibian cheetahs are remarkably free of disease.
Is AA amyloidosis in cheetahs a prion disease? The answer depends on whether AA amyloidosis in captive cheetahs is caused by spontaneous disease or transmission of amyloid between animals. Environmental influences on AA amyloidosis epidemiology could be due to the presence of either “infectious” amyloid, a prion-like etiology, or to factors that enhance the incidence of spontaneous disease, i.e., a non-prion etiology. Even if transfer of AA amyloid between cheetahs enhances AA amyloidosis, the question would remain as to whether the transferred amyloid initiates the disease de novo or merely accelerates ongoing disease. The latter scenario would place AA amyloidosis into a gray area with respect to the basic prion concept. In this instance, prion transmission would affect the kinetics of the disease without actually initiating it.
Regardless of prion semantics, there could be practical consequences of such kinetic phenomena in both animals and humans. For instance, recent studies have shown that injection of β-amyloid can enhance Alzheimer's-like amyloidosis in transgenic mice (14). This raises the possibility that inadvertent transfer of β-amyloid from one person to another could accelerate the neurodegenerative process to the point where it becomes Alzheimer's disease as opposed to normal aging. In this example, as well as in cheetah AA amyloidosis and many other protein misfolding diseases, the basic problem is likely the outpacing of an organism's protein quality control mechanisms. This may sometimes be more a problem of the rate, rather than of the instigation, of protein misfolding.
Acknowledgments.
This work was supported by the intramural program of the National Institute of Allergy and Infectious Diseases, National Institutes of Health.
Footnotes
The authors declare no conflict of interest.
See companion article on page 7263.
References
- 1.Balch WE, Morimoto RI, Dillin A, Kelly JW. Adapting proteostasis for disease intervention. Science. 2008;319:916–919. doi: 10.1126/science.1141448. [DOI] [PubMed] [Google Scholar]
- 2.Papendick RE, Munson L, O'Brien TD, Johnson KH. Systemic AA amyloidosis in captive cheetahs (Acinonyx jubatus) Vet Pathol. 1997;34:549–556. doi: 10.1177/030098589703400602. [DOI] [PubMed] [Google Scholar]
- 3.Munson L, et al. Extrinsic factors significantly affect patterns of disease in free-ranging and captive cheetah (Acinonyx jubatus) populations. J Wildl Dis. 2005;41:542–548. doi: 10.7589/0090-3558-41.3.542. [DOI] [PubMed] [Google Scholar]
- 4.Zhang B, et al. Fecal transmission of AA amyloidosis in the cheetah contributes to high incidence of disease. Proc Natl Acad Sci USA. 2008;105:7263–7268. doi: 10.1073/pnas.0800367105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Prusiner SB. Prions. Proc Natl Acad Sci USA. 1998;95:13363–13383. doi: 10.1073/pnas.95.23.13363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Wickner RB, et al. Prion genetics: New rules for a new kind of gene. Annu Rev Genet. 2004;38:681–707. doi: 10.1146/annurev.genet.38.072902.092200. [DOI] [PubMed] [Google Scholar]
- 7.Coustou V, Deleu C, Saupe S, Begueret J. The protein product of the het-s heterokaryon incompatibility gene of the fungus Podospora anserina behaves as a prion analog. Proc Natl Acad Sci USA. 1997;94:9773–9778. doi: 10.1073/pnas.94.18.9773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kisilevsky R, Boudreau L. Kinetics of amyloid deposition. I. The effects of amyloid-enhancing factor and splenectomy. Lab Invest. 1983;48:53–59. [PubMed] [Google Scholar]
- 9.Solomon A, et al. Amyloidogenic potential of foie gras. Proc Natl Acad Sci USA. 2007;104:10998–11001. doi: 10.1073/pnas.0700848104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lundmark K, et al. Transmissibility of systemic amyloidosis by a prion-like mechanism. Proc Natl Acad Sci USA. 2002;99:6979–6984. doi: 10.1073/pnas.092205999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zhang B, et al. Characterization of the cheetah serum amyloid A1 gene: Critical role and functional polymorphism of a cis-acting element. J Hered. 2008 Mar 28; doi: 10.1093/jhered/esn015. [DOI] [PubMed] [Google Scholar]
- 12.Coe JE, Ross MJ. Amyloidosis and female protein in the Syrian hamster: Concurrent regulation by sex hormones. J Exp Med. 1990;171:1257–1267. doi: 10.1084/jem.171.4.1257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.O'Brien SJ, et al. Genetic basis for species vulnerability in the cheetah. Science. 1985;227:1428–1434. doi: 10.1126/science.2983425. [DOI] [PubMed] [Google Scholar]
- 14.Meyer-Luehmann M, et al. Exogenous induction of cerebral beta-amyloidogenesis is governed by agent and host. Science. 2006;313:1781–1784. doi: 10.1126/science.1131864. [DOI] [PubMed] [Google Scholar]