Abstract
SOCS-1, a member of the suppressor of cytokine signaling (SOCS) family, was identified in a genetic screen for inhibitors of interleukin 6 signal transduction. SOCS-1 transcription is induced by cytokines, and the protein binds and inhibits Janus kinases and reduces cytokine-stimulated tyrosine phosphorylation of signal transducers and activators of transcription 3 and the gp130 component of the interleukin 6 receptor. Thus, SOCS-1 forms part of a feedback loop that modulates signal transduction from cytokine receptors. To examine the role of SOCS-1 in vivo, we have used gene targeting to generate mice lacking this protein. SOCS-1−/− mice exhibited stunted growth and died before weaning with fatty degeneration of the liver and monocytic infiltration of several organs. In addition, the thymus of SOCS-1−/− mice was reduced markedly in size, and there was a progressive loss of maturing B lymphocytes in the bone marrow, spleen, and peripheral blood. Thus, SOCS-1 is required for in vivo regulation of multiple cell types and is indispensable for normal postnatal growth and survival.
Cytokines induce a variety of biological responses by binding to specific cell surface receptors and activating cytoplasmic signal transduction pathways (1). Many of the proteins that mediate these signals from the cell surface to the nucleus, such as Janus kinases and signal transducers and activators of transcription (STATs), have been well characterized (2, 3). There is clearly a need for tight control of these signal transduction pathways, but little is known about how these signals are turned off. The adverse consequences of an imbalance between positive and negative signals is evident in the motheaten mouse, which lacks the phosphatase SHP-1. These mice fail to regulate cytokine responses appropriately, resulting in hyperproliferation and accumulation of cells in several hematopoietic lineages and the development of systemic autoimmune disease (4).
A new family of proteins that regulates responses to cytokines has been identified recently. Suppressor of cytokine signaling1 (SOCS-1) was cloned in a functional screen for molecules that inhibit interleukin (IL) 6-induced macrophage differentiation of M1 cells (5). SOCS-1 (also known as Janus kinase binding protein and STAT-induced STAT inhibitor) was isolated independently by its ability to interact with Janus kinase 2 in a two-hybrid screen (6) and on the basis of antigenic similarity with the SH2 domain of STAT3 (7). The SOCS family now comprises at least eight members, SOCS-1 to SOCS-7 and CIS (cytokine-inducible SH2-containing protein) (8, 9).
The SOCS proteins appear to act in a negative feedback loop to suppress signal transduction from cytokine receptors. SOCS gene expression is induced by cytokines both in vitro and in vivo, and, once produced, they act directly on components of the cytokine signaling pathways to switch them off (5–7, 10). The SOCS proteins suppress cytokine signaling in at least two distinct ways: SOCS-1 inhibits the signal by interacting with Janus kinases and inhibiting their catalytic activity (6, 7) whereas CIS binds directly to the cytokine receptor, competing with STATs for access to docking sites on the activated receptor (9).
Little is understood about specificity within the SOCS family of proteins. Constitutive over-expression of SOCS-1 inhibits cellular responses in vitro to a variety of cytokines, including IL-6, leukemia inhibitory factor, interferon-γ, and thrombopoietin (5–7, 11), but the primary targets and biological consequences of SOCS-1 action in vivo are unknown. To address these questions, we have generated mice that lack the SOCS-1 protein. These animals exhibited stunted growth and abnormalities in a diverse range of organs and died before weaning, indicating an essential role of this negative regulator in postnatal growth and survival.
MATERIALS AND METHODS
Generation of Targeted Embryonic Stem (ES) Cells and SOCS-1−/− Mice.
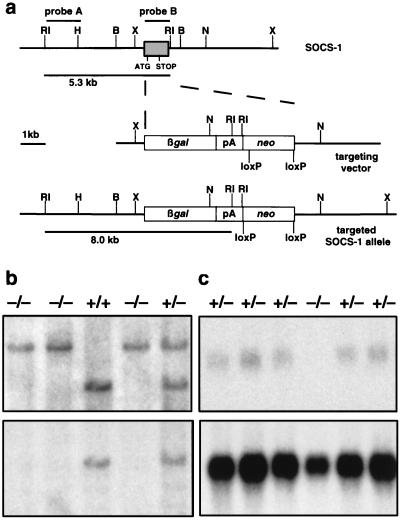
A 5′ fragment of the murine SOCS-1 gene extending ≈2.5 kilobases (kb) from the protein initiation ATG was generated by PCR. This fragment was ligated directly upstream of the initiation codon of β-galactosidase via a BamHI site in the plasmid pβgalpAloxneo, which also contains a PGKneo cassette flanked by loxP sites. A 3.2-kb BamHI-EcoRV fragment from the genomic SOCS-1 locus was blunted and ligated into a blunted XhoI site 3′ to the PGKneo cassette. This construct was linearized and electroporated into 129/Sv-derived W9.5 ES cells (12). Clones surviving selection in 175 μg/ml G418 were screened for those in which the targeting vector had recombined with an endogenous SOCS-1 allele by using Southern blots of EcoRI-digested genomic DNA probed with a 1.5-kb EcoRI-HindIII genomic SOCS-1 fragment (Fig. 1, probe A). A targeted ES cell clone was injected into C57BL/6 blastocysts to generate chimeric mice. Male chimeras were mated with C57BL/6 females to yield SOCS-1 heterozygotes, which were interbred to produce wild-type (SOCS-1+/+), heterozygous (SOCS-1+/−), and homozygous mutant (SOCS-1−/−) mice. The genotypes of offspring were determined by Southern blot analysis of genomic DNA extracted from tail biopsies as described above. The deletion of SOCS-1 coding sequence and subsequent inability to produce SOCS-1 mRNA in mutant mice was confirmed in nucleic acid blots, which were performed as described (13). Northern blots were probed with probe B (Fig. 1) from the SOCS-1 coding region and then with a 1.2-kb PstI chicken glyceraldehyde-3-phosphate dehydrogenase fragment.
Figure 1.
Disruption of the SOCS-1 locus by homologous recombination. (a) The murine SOCS-1 genomic locus is shown (B, BamHI; H, HindIII; RI, EcoRI; X, XhoI), with the exon containing the coding region as a shaded box. In the targeting vector, the entire SOCS-1 coding region was replaced by a β-gal-PGKneo cassette in which the β-galactosidase coding region was fused to the SOCS-1 initiation codon. (b) Southern blot of EcoRI-digested genomic DNA extracted from the tails of mice derived from a cross between heterozygous (SOCS-1+/−) mice. The blot initially was hybridized with probe A (upper panel), which distinguishes between endogenous (5.3 kb) and mutant SOCS-1 (8.0 kb) alleles, and then was hybridized with a coding region probe (probe B, lower panel) to confirm the loss of genetic material in SOCS-1−/− mice. (c) Northern blot showing lack of SOCS-1 expression in the thymus of SOCS-1−/− mice. In the upper panel, the blot was hybridized with a coding region probe, which detects the 1.4-kb SOCS-1 transcript (1), and the integrity of the RNA was confirmed by hybridization with glyceraldehyde-3-phosphate dehydrogenase (1.4-kb transcript, lower panel).
Flow Cytometry.
Single cell suspensions of bone marrow cells, splenocytes, and thymocytes from 9- to 16-day-old SOCS-1−/− mice and normal littermates were prepared, and the erythrocytes were lysed as described (14). The cells were stained with rat mAbs for specific cell surface markers and were analyzed by flow cytometry as described (15). To analyze B-lymphoid differentiation in vitro, B220-positive cells from the bone marrow of 11-day-old SOCS-1−/− mice and normal littermates were sorted into individual wells of 96-well plates seeded with irradiated (30 Gy) IL-7-expressing NIH 3T3 fibroblasts. After 20 days of culture, 1 × 106 cells were washed, were cultured for 48 hours in the absence of NIH 3T3 cells and IL-7, and then were analyzed by flow cytometry for surface IgM expression as described (14). Fluorescence-activated cell sorter-Gal assays were performed on cells previously stained with antibodies for cell surface markers. Cells were analyzed on a FACStar Plus (Becton Dickinson).
Hematological and Histological Analysis.
The peripheral blood white cell and platelet counts were determined manually by using hemocytometers. Single-cell suspensions from femoral bone marrow, spleen, and liver were prepared by standard techniques, and manual leukocyte differential counts of peripheral blood, bone marrow, liver, and spleen were performed on smears or cytocentrifuge preparations stained with May–Grunwald–Giemsa. Tissue sections were prepared by standard techniques, were stained with hematoxylin and eosin, and were examined by light microscopy.
RESULTS
Production of Mice Lacking SOCS-1.
A targeting construct was assembled to enable homologous recombination to replace the entire SOCS-1 coding region with β-galactosidase in ES cells (Fig. 1a). An ES cell clone bearing a disrupted SOCS-1 allele was used to generate chimeric mice, from which animals lacking a functional SOCS-1 gene were bred. Southern blot analysis of tail biopsies from offspring of heterozygous parents (Fig. 1b) revealed that SOCS-1−/− mice were born in similar numbers to their wild-type littermates (30:88:32 for SOCS-1+/+:SOCS-1+/−:SOCS-1−/−), suggesting that SOCS-1 was not essential for normal embryonic development. Northern blots of RNA extracted from the thymus of SOCS-1−/− mice confirmed that SOCS-1 mRNA was not transcribed from the disrupted allele (Fig. 1c).
Postnatal Lethality and Histopathology of SOCS-1−/− Mice.
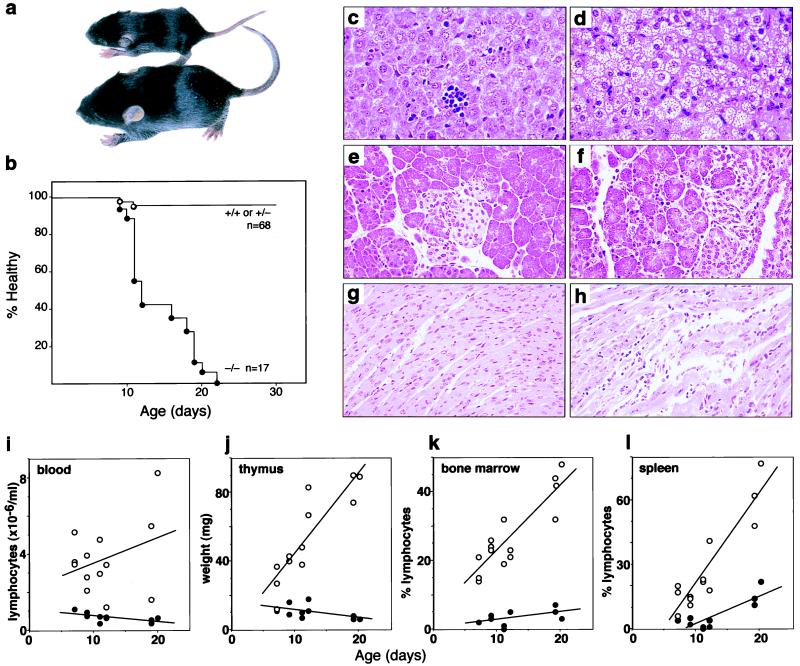
At birth, SOCS-1-deficient mice were indistinguishable from their normal littermates (birth weights: SOCS-1+/+, 1.90 ± 0.29 g; SOCS-1+/−, 1.89 ± 0.29 g; and SOCS-1−/−, 1.94 ± 0.10 g, n = 2–8). However, within 10 days the SOCS-1−/− mice were significantly smaller (body weight at days 9–12: SOCS-1+/+ or +/−, 6.27 ± 1.58 g; and SOCS-1−/−, 4.06 ± 1.12 g; n = 9–11) and became ill and died before they reached 3 weeks of age (Fig. 2 a and b). Histological examination of the organs of SOCS-1−/− mice at 7–20 days of age revealed major abnormalities in the liver. Parenchymal cells commonly contained an accumulation of lipid-containing vacuoles, resulting in cells with expanded cytoplasm and shrunken nuclei (Fig. 2 c and d). The fatty degeneration either involved local areas not related to portal vessels or was generalized throughout the whole organ. In the latter case, which represented half of the SOCS-1−/− livers examined, areas of subcapsular necrosis were present. Unlike livers from normal neonatal mice, which showed focal aggregates of erythroid and, less commonly, granulocytic cells, the SOCS-1−/− livers exhibited both focal and generalized infiltration, predominantly by immature and mature monocytic and granulocytic cells (Fig. 2 c and d). Less frequently, megakaryocytes and eosinophils were present and were more numerous in SOCS-1-deficient mice than in their heterozygous or normal littermates. Abnormalities were also evident in three other organs of SOCS-1−/− mice. In seven of eight mice examined, the pancreas was infiltrated by monocytes invading septal regions between acini, associated with some reduction in acinar size (Fig. 2 e and f). In the majority of SOCS-1−/− mice, infiltration of the heart by monocytes and less numerous granulocytes also was observed (Fig. 2 g and h), as was increased cellularity of alveolar walls in the lung, often associated with macrophage cuffing around major vessels (data not shown). Other than the overall reduction in organ size, paralleling the stunted growth of these animals, no abnormalities were observed in skeletal muscle, gut, bladder, kidney, gonads, or brain of SOCS-1−/− mice.
Figure 2.
Phenotypic abnormalities in SOCS-1−/− mice. (a) SOCS-1−/− and littermate mice at 10 days of age showing retarded growth of the SOCS-1-deficient mouse. (b) Rapid onset of morbidity and mortality in SOCS-1−/− mice. The proportion of mice remaining disease-free (percent healthy) with age is shown. All of the SOCS-1−/− mice died within 3 weeks of birth. Normal histological appearance of the liver (c), pancreas (e), and heart (g) of littermate mice is in contrast to the fatty degeneration of the liver (d) and monocytic infiltration of the pancreas (f) and heart (h) of SOCS-1−/− mice. The progressive deficit with increasing age of SOCS-1−/− mice (•) in peripheral blood lymphocytes (i), thymic weight (j), and lymphocyte composition of the bone marrow (k) and spleen (l) is shown in comparison with values from littermate SOCS-1+/+ or SOCS-1+/− controls (○).
Lymphocyte Deficiencies in SOCS-1−/− Mice.
In comparison with wild-type littermates, the peripheral blood of SOCS-1−/− mice revealed a consistent and severe reduction in lymphocyte levels, a moderate reduction in eosinophil and platelet numbers, a consistently lower hematocrit, and a variable elevation of blood granulocytes (Table 1 and Fig. 2i). The smaller body weight of SOCS-1-deficient mice was associated with a femoral marrow cell count that was not that of littermate mice, and cytological analysis revealed a consistent deficit of lymphocytes in this population (Fig. 2k). Although spleen weight appeared to be normal in SOCS-1−/− mice, this organ also exhibited a relative deficit in lymphocytes, which became more evident with increasing age (Fig. 2l). Histological analysis of the spleen showed that, in SOCS-1−/− mice, lymphoid follicles either were completely absent or were composed of immature cells, often mixed with monocytes and granulocytes. Most SOCS-1-deficient spleens showed expanded areas of red pulp with nucleated erythroid cells as the dominant population. The most striking hemopoietic abnormality in SOCS-1−/− mice was the small size of the thymus (Fig. 2j). Unlike normal neonatal mice, the number of cortical lymphocytes failed to increase with age, and, instead, the cortex became progressively depleted of lymphoid cells. The lymph nodes showed a similar deficit in lymphoid follicle formation, and, although Peyer’s patches were present, these often contained few lymphocytes.
Table 1.
Hematological profile of peripheral blood in SOCS-1−/− mice
| Genotype
|
||
|---|---|---|
| SOCS-1+/− or +/+ | SOCS-1−/− | |
| Platelets, ×10−6/ml | 737 ± 22 | 435 ± 251 |
| Hematocrit, % | 37 ± 3 | 28 ± 3 |
| White cell count, ×10−6/ml | 5.55 ± 2.46 | 4.08 ± 2.64 |
| Neutrophils | 0.95 ± 0.64 | 2.42 ± 1.93 |
| Lymphocytes | 3.63 ± 1.85 | 0.73 ± 0.36 |
| Monocytes | 0.73 ± 0.36 | 0.94 ± 1.22 |
| Eosinophils | 0.16 ± 0.24 | 0.01 ± 0.02 |
Mean ± SD of data from 10 (SOCS-1−/−) and 14 (SOCS-1+/+ or +/−) mice.
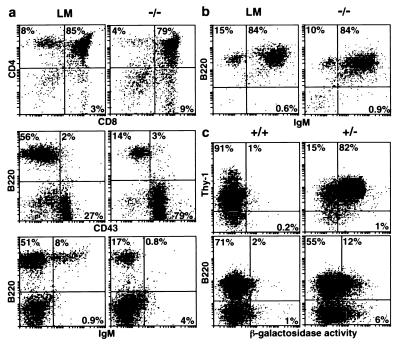
The reduction in thymic cellularity in SOCS-1−/− mice was not associated with selective loss of a specific subset of thymocytes. Fluorescence-activated cell sorting revealed normal ratios of T cells expressing the CD4 and/or CD8 cell surface markers (Fig. 3a). In contrast, selective depletion of maturing B-lymphoid cells was evident in the bone marrow and spleen of SOCS-1−/− mice at 9–16 days of age (Fig. 3a). Although pro-B cells (B220+ CD43+) comprised only a few percent of the littermate marrow population, their relative frequency in SOCS-1-deficient mice did not appear to be reduced. However, as exemplified in Fig. 3a, pre-B and B cells (B220+ CD43−), which comprised nearly half of the bone marrow cells in littermate mice (46 ± 20%, n = 4), were depleted significantly in the marrow of SOCS-1−/− mice (8 ± 6%, n = 5). The number of mature B cells expressing surface Ig in the bone marrow was reduced to a similar extent (SOCS-1−/−, 1 ± 0.5%, n = 4; littermate, 7 ± 3%, n = 3). Similarly, the proportion of B-lymphoid cells in the spleens of SOCS-1−/− mice also was reduced (data not shown).
Figure 3.
Lymphocyte profiles in SOCS-1−/− mice. (a) Fluorescence-activated cell sorter analysis showing unaltered distribution of thymocytes expressing the CD4 and CD8 cell surface markers and the deficiency in pre-B and B (B220+CD43−) as well as mature B cells (B220+IgM+) in the bone marrow of a representative SOCS-1−/− mouse. Profiles of the equivalent populations from age-matched littermate mice (LM) are shown for comparison. (b) Analysis of cultures of bone marrow cells from SOCS-1−/− and littermate (LM) mice showing similar in vitro capacity for B cell (B220+IgM+) maturation. (c) Double-staining with β-galactosidase and Thy-1 (thymus) or B220 (spleen) from heterozygous SOCS-1+/− and wild-type mice demonstrating transcriptional activity of the SOCS-1 locus in thymocytes and B-lymphoid cells.
Despite their abnormally low number, SOCS-1−/− B-lymphocytes were not intrinsically incapable of maturation. B220+ cells were sorted from the bone marrow of littermate and SOCS-1−/− mice and were seeded onto IL-7-producing fibroblasts. Consistent with the lymphoid deficiencies observed in SOCS-1-deficient mice, the mutant population contained significantly fewer precursors capable of proliferation in this assay (SOCS-1−/−, one colony-forming cell in 45 B220+ cells; SOCS-1+/+ or +/−, one in four to nine B220+ cells). Pre-B cells that did develop from SOCS-1−/− marrow were replated in the absence of fibroblasts or added cytokine. Under these conditions, the development of mature B cells (B220+ IgM+) was as efficient as in cultures derived from littermates (Fig. 3b).
Transcription of the SOCS-1 Locus in Thymocytes and B-Lymphoid Cells.
In the targeted SOCS-1 allele, the SOCS-1 coding sequence was replaced by the β-gal gene (Fig. 1). β-galactosidase activity was, therefore, examined as a marker of SOCS-1 expression in phenotypically normal SOCS-1+/− mice. Cell sorting studies combining analysis of the Thy-1 cell surface marker with β-galactosidase activity suggested that >80% of thymocytes normally express SOCS-1 (Fig. 3c). Consistent with the deficit in SOCS-1−/− mice, expression of β-galactosidase was evident in all CD4 and CD8 thymic subsets (data not shown). β-galactosidase activity was also evident in a proportion of pre-B (B220+ IgM−) and mature B (B220+ IgM+) cells from the spleen (Fig. 3c and data not shown) or the bone marrow (data not shown) of SOCS-1+/− mice. As expected, β-galactosidase activity was negligible in control wild-type lymphoid populations (Fig. 3c). These data confirm that SOCS-1 normally is expressed in the T and B lymphoid lineages.
DISCUSSION
Through the generation of mice bearing a targeted disruption of the gene encoding SOCS-1, we have shown that this protein is indispensable for normal postnatal development. Although apparently normal at birth, SOCS-1−/− mice exhibited stunted growth and died within the first 3 weeks of life. Major abnormalities were evident in the liver, with fatty degeneration of parenchymal cells, which may be sufficient to account for the mortality of SOCS-1−/− mice. Deficiencies also were observed in the lymphoid organs, with dramatically reduced cellularity of the thymus and a severe progressive depletion of pre-B and B cells. Extensive focal and generalized hematopoietic infiltration was also evident in the livers of SOCS-1−/− mice, and the pancreas, lungs, and heart of these animals often exhibited a less severe monocytic infiltration (Fig. 2).
The complexity of the phenotypic abnormalities in SOCS-1−/− mice implies that SOCS-1 is indispensable for the normal in vivo regulation of multiple cell types. Although it is feasible that some of the abnormalities observed in SOCS-1−/− mice may be the indirect consequences of a primary defect in one organ, such as the liver, it is striking that the cells that are lost or damaged in SOCS-1−/− mice are among those that normally express this gene (T and B lymphocytes; Fig. 3) or in which expression is induced by cytokine stimulation (liver; ref. 5). Thus, the lymphocyte deficits, as well as the degeneration of liver parenchymal cells, seem likely to be a direct consequence of the lack of SOCS-1 in these cells. Although the diverse abnormalities that characterize mice lacking SOCS-1 may reflect a common action in divergent cell types, our data do not exclude the possibility that SOCS-1 may mediate differing and independent effects in different organs.
Previous studies in vitro have established that SOCS-1 can act to inhibit cytokine signaling (5), suggesting that abnormalities in mice lacking this protein may result from inappropriate responses to cytokine stimulation. Indeed, STAT1 activation, which normally occurs in response to agents such as IL-6 or interferon γ, appears to occur constitutively in the livers of SOCS-1-deficient mice (data not shown). As overexpression of interferon γ is known to cause liver damage and B-lymphocyte depletion (16, 17), the disease that arises in SOCS-1−/− mice may be the result of dysregulation of signals from such a cytokine. Alternatively, activation of STATs also has been linked to the induction of apoptosis (18, 19), raising the possibility that SOCS-1−/− mice may be unable to regulate apoptotic signals appropriately. To explore these possibilities, we currently are crossing the SOCS-1−/− mice to mice lacking interferon γ or regulators of apoptosis. An additional possibility is raised by the monocyte infiltration observed in multiple organs of SOCS-1−/− mice (Fig. 2). Preliminary data indicate that SOCS-1-deficient granulocyte-macrophage progenitor cells are hyper-responsive to proliferative stimulation by granulocyte–macrophage colony-stimulating factor. If this is associated with the functional activation of monocytes and granulocytes, infiltrating hemopoietic cells also might contribute to tissue damage in SOCS-1−/− mice.
Acknowledgments
We thank Elizabeth Viney, Naomi Sprigg, Steven Rakar, Jason Corbin, Sandra Mifsud, and Ladina DiRago for excellent technical assistance, Dr. Andreas Strasser for antibodies and advice, and Dr. Antonius Rolink for IL-7-expressing NIH 3T3 cells. This work was supported by the Australian National Health and Medical Research Council, the Anti-Cancer Council of Victoria, an Australian Government Cooperative Research Centres Program Grant, the National Institutes of Health (Grant CA22556), the J. D. and L. Harris Trust, and Australian Medical Research and Development. Operations Pty Ltd. R.S. was supported by an Australian Research Council Postdoctoral Fellowship.
ABBREVIATIONS
- SOCS
suppressor of cytokine signaling
- STAT
signal transducers and activators of transcription
- IL
interleukin
- kb
kilobase
- ES
embryonic stem
References
- 1.Nicola N A. Guidebook to Cytokines and Their Receptors. Oxford: Oxford Univ. Press; 1994. pp. 1–261. [Google Scholar]
- 2.Darnell J E., Jr Science. 1997;277:1630–1636. doi: 10.1126/science.277.5332.1630. [DOI] [PubMed] [Google Scholar]
- 3.Pelligrini S, Dusanter-Fourt I. Eur J Biochem. 1997;248:615–633. doi: 10.1111/j.1432-1033.1997.00615.x. [DOI] [PubMed] [Google Scholar]
- 4.Shultz L D, Rajan T V, Greiner D L. Trends Biotechnol. 1997;15:302–307. doi: 10.1016/S0167-7799(97)01060-3. [DOI] [PubMed] [Google Scholar]
- 5.Starr R, Willson T A, Viney E M, Murray L J L, Rayner J R, Jenkins B J, Gonda T J, Alexander W S, Metcalf D, Nicola N A, et al. Nature (London) 1997;387:917–921. doi: 10.1038/43206. [DOI] [PubMed] [Google Scholar]
- 6.Endo T A, Masuhara M, Yokouchi M, Suzuki R, Sakamoto H, Mitsui K, Matsumoto A, Tanimura S, Ohtsubo M, Misawa H, et al. Nature (London) 1997;387:921–924. doi: 10.1038/43213. [DOI] [PubMed] [Google Scholar]
- 7.Naka T, Narazaki M, Hirata M, Matsumoto T, Minamoto S, Aono A, Nishimoto N, Kajita T, Taga T, Yoshizaki K, et al. Nature (London) 1997;387:924–929. doi: 10.1038/43219. [DOI] [PubMed] [Google Scholar]
- 8.Hilton D J, Richardson R T, Alexander W S, Viney E M, Willson T A, Sprigg N S, Starr R, Nicholson S E, Metcalf D, Nicola N A. Proc Nat Acad Sci USA. 1998;95:114–119. doi: 10.1073/pnas.95.1.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Yoshimura A, Ohkubo T, Kiguchi T, Jenkins N A, Gilbert D J, Copeland N G, Hara T, Miyajima A. EMBO J. 1985;14:2816–2826. doi: 10.1002/j.1460-2075.1995.tb07281.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Matsumoto A, Masuhara M, Mitsui K, Yokouchi M, Ohtsubo M, Misawa H, Miyajima A, Yoshimura A. Blood. 1997;89:3148–3154. [PubMed] [Google Scholar]
- 11.Sakamoto H, Yasukawa H, Masuhara M, Tanimura S, Sasaki A, Yuge K, Ohtsubo M, Ohtsuka A, Fujita T, Ohta T, et al. Blood. 1998;92:1668–1676. [PubMed] [Google Scholar]
- 12.Szabo P, Mann J R. Development (Cambridge, UK) 1994;120:1651–1660. doi: 10.1242/dev.120.6.1651. [DOI] [PubMed] [Google Scholar]
- 13.Alexander W S, Metcalf D, Dunn A R. EMBO J. 1995;14:5569–5578. doi: 10.1002/j.1460-2075.1995.tb00244.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Elefanty A G, Begley C G, Metcalf D, Barnett L, Kontgen F, Robb L. Proc Natl Acad Sci USA. 1998;95:11876–11881. doi: 10.1073/pnas.95.20.11897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Strasser A, Harris A W, Cory S. Cell. 1991;67:889–899. doi: 10.1016/0092-8674(91)90362-3. [DOI] [PubMed] [Google Scholar]
- 16.Toyonaga T, Hino O, Sugai S, Wakasugi S, Abe K, Shichiri M, Yamamura K. Proc Natl Acad Sci USA. 1994;91:614–618. doi: 10.1073/pnas.91.2.614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Young H A, Klinman D M, Reynolds D A, Grzegorzewski K J, Nii A, Ward J M, Winkler-Pickett R T, Ortaldo J R, Kenny J J, Komschlies K L. Blood. 1997;89:583–595. [PubMed] [Google Scholar]
- 18.Schindler C. Trends Cell Biol. 1998;8:97–98. doi: 10.1016/s0962-8924(98)01233-1. [DOI] [PubMed] [Google Scholar]
- 19.Chin Y E, Kitagawa M, Kuida K, Flavell R A, Fu X Y. Mol Cell Biol. 1997;17:5328–5337. doi: 10.1128/mcb.17.9.5328. [DOI] [PMC free article] [PubMed] [Google Scholar]