Abstract
The ability of antibody (Ab) to modulate HSV pathogenesis is well recognized but the mechanisms by which HSV-specific IgG antibodies protect against genital HSV-2 disease are not well understood. The requirement for Ab interactions with Fcγ receptors (FcγR) in protection was examined using a murine model of genital HSV-2 infection. IgG antibodies isolated from the serum of HSV-immune mice protected normal mice against HSV-2 disease when administered prior to genital HSV-2 inoculation. However, protection was significantly diminished in recipient mice lacking the gamma chain subunit utilized in FcγRI, FcγRIII, FcγRIV and FcepsilonRI receptors and in normal mice depleted of Gr-1+ immune cell populations known to express FcγR, suggesting protection was largely mediated by an FcγR-dependent mechanism. To test whether neutralizing Ab might provide superior protection, a highly neutralizing HSV glycoprotein D (gD)- specific monoclonal antibody (mAb) was utilized. Similar to results with HSV-specific polyclonal IgG, administration of the gD-specific mAb did not prevent initial infection of the genital tract but resulted in lower virus loads in the vaginal epithelium and provided significant protection against disease and acute infection of the sensory ganglia; however, this protection was independent of host FcγR expression and was manifest in mice depleted of Gr-1+ immune cells. Together, these data demonstrate that substantial Ab-mediated protection against genital HSV-2 disease could be achieved by either FcγR-dependent or -independent mechanisms. These studies suggest that HSV vaccines might need to elicit multiple, diverse antibody effector mechanisms to achieve optimal protection.
Keywords: herpes simplex virus type 2, Fc gamma receptors, genital mucosa, sensory neurons, antibodies
1. Introduction
The role of Ab in protection against HSV-2 is not clear. In humans, the presence of HSV-specific Ab in HSV-infected mothers has been suggested to decrease the risk of acquisition of HSV-2 by newborns (Brown et al., 1991). However, immunization with candidate HSV-2 vaccines comprised of recombinant HSV-2 glycoproteins elicited high titers of neutralizing Ab but provided, at best, a modest level of protection (Corey et al., 1999; Stanberry et al., 2002). Although sterilizing immunity was not achieved with these candidate vaccines, efficacy related to limiting the establishment of latency, virus reactivation and virus shedding during recurrences has not been fully determined. Recent analysis of serum Ab elicited by a candidate HSV vaccine demonstrated low levels of Ab capable of mediating Ab-dependent cellular cytotoxicity (ADCC), suggesting that mechanisms beyond virus neutralization may be required for the Ab-mediated component of protection elicited by HSV vaccines (Kohl et al., 2000).
Ab elicited by natural infection or immunization has been shown clearly to modulate HSV pathogenesis in animal models (McDermott et al., 1984; Stanberry et al., 1986; Kuklin et al., 1998; Bourne et al., 2003). Numerous studies have demonstrated also that passive administration of HSV-specific polyclonal Ab or mAb can interrupt the normal disease course (Oakes and Lausch, 1981; McKendall, 1985; Schneweis et al., 1988; Bourne et al., 2002). However, the relevant mechanisms of Ab-mediated protection are not well understood and may be influenced by antigen specificity, Ab isotype or the site of HSV challenge. In the current study, we have transferred HSV-specific IgG antibodies prior to genital HSV-2 challenge to examine the mechanism of protection conferred by HSV-specific antibody against HSV-2 exposure. We examined the efficacy of virus-neutralizing mAb and involvement of Ab-FcγR interactions in a model of protection against genital HSV-2 infection and disease. The results have implications for the development of effective HSV vaccines as well as other prophylactic treatments to prevent transmission of HSV-2.
2. Materials and Methods
2.1 Virus
HSV-2 strain 186 was obtained originally from Dr. Lawrence Stanberry (University of Texas Medical Branch, Galveston, TX).
2.2 Mice
C57BL/6J (B6) and B6.129S7-Rag1tm1Mom (Rag 1−/−) mice genetically deficient in adaptive immune cells but possessing an intact innate immune system including macrophages, dendritic cells and NK cells (Mombaerts et al., 1992) were purchased from the Jackson Laboratory (Bar Harbor, ME). B cell-deficient µMT mice (Kitamura et al., 1991) were used as recipient mice deficient in endogenous antibody production and were maintained as a breeding colony. Swiss Webster mice were obtained from Harlan Sprague Dawley (Indianapolis, IN). B6.129P2-Fcεr1gtm1RavN12 mice (FcγR−/−) deficient in the gamma chain subunit utilized in FcγRI, FcγRIII and FcepsilonRI receptors (Ravetch and Bolland, 2001) and FcγRIV (Nimmerjahn et al., 2005) were obtained from Taconic Farms (Germantown, NY).
2.3 Virus inoculation and titration
Mice treated one week previously with medroxyprogesterone acetate (SICOR Pharmaceuticals, Inc., Irvine, CA) were inoculated intravaginally (ivag) as described previously (Dobbs et al., 2005) without pre-swabbing prior to inoculation. Pretreatment with medroxyprogesterone was required for successful vaginal infection of mice, most likely due to the induction of the viral entry receptor, nectin 1, on vaginal epithelial cells (Linehan et al., 2004). Inoculated mice exhibiting severe disease symptoms were euthanized. Infectious virus in vaginal swab samples as well as in the lumbosacral ganglia and adjacent section of spinal cord was quantified in tissue homogenates as described previously (Milligan et al., 1998).
2.4 Quantification of HSV-specific T cells
IFN-γ -secreting lymphocytes were quantified by ELISPOT analysis 10 days after challenge with HSV-2 strain 186, as described previously (Milligan and Bernstein, 1995). Lymphocytes were incubated with mitomycin C (Sigma-Aldrich, St. Louis, MO)-treated splenocytes pulsed with UV-irradiated HSV virions or an immunogenic peptide from HSV glycoprotein B (gB498–505).
2.5 Detection of Gr-1+ cells by flow cytometric analysis
Vaginal leukocytes were collected by lavage, and lumbosacral ganglia and adjacent spinal cord sections were dissected from normal mice on day 2 following HSV-2 inoculation. Tissue was dispersed through a stainless steel screen, suspended in 30% Percoll (Sigma-Aldrich) and layered over a 70% Percoll cushion. After centrifugation, the leukocyte-rich fraction was collected from the interface. Cells were stained with FITC-anti-Gr-1 (Serotec, Raleigh, NC) and PE-conjugated F4/80 (Caltag/Invitrogen, Carlsbad, CA) or isotype control antibodies. Data were acquired on a Becton Dickson FACS Canto flow cytometer at the University of Texas Medical Branch Flow Cytometry Core Facility and analyzed using FlowJo Software (Treestar Inc., Ashland, OR).
2.6 In vivo depletion of Gr-1+ cells
The RB6.8C5 mAb was obtained by ammonium sulfate precipitation of serum-free culture supernatants. Mice were treated with sterile RB6.8C5 mAb or control IgG mAb, as described previously (Milligan, 1999; Milligan et al., 2001). Treatment with RB6.8C5 mAb resulted in depletion of greater than 90% Gr-1+ granulocytes from the vaginal lumen of HSV-2 inoculated mice.
2.7 Generation of HSV-2 gD-specific hybridomas
BALB/c mice were immunized with 50µg rgD2 (Protein Express, Cincinnati, OH) in Imject Alum (Pierce Chemical Co., Rockford, IL), then re-immunized 6 weeks later with 50µg rgD2. Two days later, splenocytes were fused to SP2/0 cells. Hybridomas were screened for secretion of gD-specific IgG on gD2-coated ELISA plates, as described previously for serum Ab (Milligan et al., 2001). Hybridomas of interest were cloned by limiting dilution.
2.8 Preparation of HSV-specific Ab
Polyclonal Ab was prepared by ammonium sulfate precipitation of sera from mice immunized twice in a 4 week period with 20µg rgD2 and challenged ivag with HSV-2 strain 186 four weeks after the second immunization. Monoclonal Ab was prepared as described previously (Milligan, 1999) by ammonium sulfate precipitation of serum-free culture supernatants. Quantification of Ab was performed by ELISA on rgD2-coated plates (for mAb) or HSV antigen-coated plates for serum IgG, Ab as described previously (Milligan et al., 2001). Mice received a single i.p. injection of HSV-specific Ab 2 days prior to ivag HSV-2 challenge to allow distribution and equilibration of Ab to all tissues prior to virus inoculation. Virus neutralization assays were performed as described previously (Milligan et al., 1998) in the presence or absence of complement (Accurate Chemical and Scientific, Westbury, NY).
2.9 Statistics
Survival curves were compared by Logrank test. Incidence of infection and disease were analyzed by Fisher’s exact test. HSV-specific T cell quantification and HSV-2 titers were analyzed by Student’s t test or ANOVA, with the Bonferoni correction for multiple groups as appropriate. All analyses were performed using GraphPad Prism Version 3.0 for Windows (GraphPad Prism Software, San Diego, CA). P values less than 0.05 were considered to indicate statistical significance.
3. Results
3.1 FcγR-dependent protection following passive transfer of HSV-specific antibodies
To examine the mechanisms of Ab-mediated protection, antibodies were precipitated from the serum of HSV-immune mice, characterized and tested for protective efficacy. The ratio of HSV-specific IgG1, IgG2b and IgG2a subclasses in this Ab preparation was 3.2 : 5.2 : 1.0, respectively, with 50% neutralization concentrations of 696 and 980ng/ml in the presence and absence of complement, respectively. This difference did not reach significance (p= 0.53, Student’s t test).
To examine protection provided by the polyclonal HSV glycoprotein-specific Ab, groups of 13–15 B6 or FcγR−/− mice received 50µg polyclonal anti-HSV Ab 2 days prior to ivag HSV-2 challenge. This dose of Ab resulted in a concentration of HSV-specific serum IgG in the range commonly detected following genital inoculation with attenuated HSV-2 (Milligan et al, 2004). B6 mice receiving polyclonal Ab were significantly protected compared to PBS-treated B6 control animals and FcγR −/− Ab recipients (p< 0.0001 and p< 0.008, respectively, Logrank test; Fig. 1A). Administration of polyclonal Ab to B6 mice did not prevent infection, but did significantly decrease the incidence of disease symptoms, including mortality, compared to PBS-treated B6 mice and FcγR−/− Ab recipients (p < 0.0003 and p < 0.028, respectively, Fisher’s exact test; Table 1). There was no significant difference in expression of disease symptoms of HSV-2 infection, excluding mortality, between B6 and FcγR−/− HSV-specific antibody recipients, suggesting cutaneous manifestations of disease may have been less susceptible to FcγR-dependent antibody-mediated protection. Administration of polyclonal Ab to B6 mice resulted in significantly lower HSV-2 titers in the vaginal epithelium on day 2 after inoculation compared to PBS –treated controls (Fig. 1B; p < 0.001, ANOVA) or FcγR−/− mice (p< 0.05, ANOVA). Infectious virus titers in PBS-treated control mice and FcγR−/− Ab recipients were not significantly different (p > 0.05).
Fig. 1.
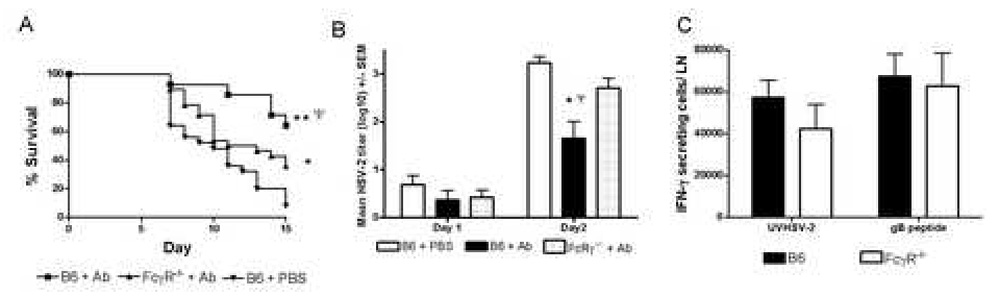
Protection afforded by HSV-specific polyclonal Ab requires host cell expression of FcγR. A, Survival curves of B6 or FcγR−/− polyclonal Ab recipients (n=13–16) following ivag challenge with 5×103 PFU HSV-2 strain 186. Results shown were pooled from 2 experiments. Survival curves are marked to denote statistical significance as follows: *p< 0.0001, **p < 0.006 compared to PBS-treated B6 mice, and ψp < 0.008 compared to FcγR −/− Ab recipients by Logrank test. B, HSV-2 titers in the vaginae of Ab-treated or control-treated mice (n=8) on day 1 and 2 post ivag HSV-2 inoculation. Day 2 titers for B6 Ab recipients are significantly different compared to PBS-treated B6 mice (p < 0.0001, Logrank test) and FcγR−/− Ab recipients (p < 0.05, Logrank test). C, Quantification of IFN-γ-producing cells from the iliac lymph nodes of B6 or FcγR−/− mice challenged 6 days previously with 5×103 PFU HSV-2 strain 186. Results shown represent the mean response of 6 mice to UV-killed HSV-2 or the CD8 epitope from HSV glycoprotein B (gB498–505). Less than 10 IFN-γ-secreting cells/LN were detected in naïve mice.
Table I.
Passive transfer of polyclonal anti-HSV-2 antibody to B6 or FcγR−/− mice
| Incidence a |
||||
|---|---|---|---|---|
| HSV-2 Shedding b | Disease Symptoms c | Infection d | ||
| Mouse strain | Transfer e | |||
| B6 | PBS | 25 \ 25 | 25 \ 25 | 25 \ 25 |
| B6 | Polyclonal IgG | 28 \ 28 | 17 \ 28 f | 28 \ 28 |
| FcγR −/− | PBS | 15 \ 15 | 15 \ 15 | 15 \ 15 |
| FcγR −/− | Polyclonal IgG | 28 \ 28 | 25 \ 28 g | 28 \ 28 |
Results are expressed as the number of mice exhibiting the indicated outcome per total number of mice. Results are pooled from 2 experiments of identical design.
Mice were scored as positive for shedding if infectious virus was detected in the vagina on day 1 or 2 post challenge.
Disease symptoms included: erythema, perivaginal hair loss, swelling, and mortality.
Mice were considered infected if they shed virus from the vagina on day 1 or 2 post inoculation or developed disease symptoms.
Mice received either 50µg of polyclonal anti-HSV-2 antibody or PBS as a control 2 days prior to ivag inoculation of 5×103 PFU HSV-2 strain 186.
p<0.0003 compared to PBS-treated control mice, 2-tailed Fisher’s exact test.
p< 0.028 compared to Ab-treated B6 mice, 2-tailed Fisher’s exact test.
The number of HSV-specific T cells detected in the iliac lymph node did not differ significantly between B6 and FcγR −/− Ab recipients (Fig. 1C; p > 0.05, Student’s t test) suggesting that the diminished protection in FcγR −/− Ab recipients reflected diminished Ab-dependent protection rather than a decreased HSV-specific cell-mediated response.
3.2 FcγR-independent protection conferred by a highly neutralizing gD2-specific mAb
The possibility that a highly neutralizing Ab capable of interfering with essential virus-binding steps may provide a different level or manifestation of protection, such as sterilizing immunity, was considered. We tested the efficacy of an HSV glycoprotein D-specific IgG2a mAb (10E4.2G2a) that was approximately 50-fold more efficient at virus neutralization than the previously tested polyclonal IgG preparation. Transfer of 50µg mAb resulted in significant protection against mortality compared to PBS-treated controls (p < 0.0001, Logrank test) and to 1.0µg mAb recipients (p = 0.016, Logrank test; Fig. 2A). To test whether the protection observed in mAb recipients reflected only Ab-mediated mechanisms in the complete absence of T cells, we tested the ability of 10E4.2G2a mAb to protect Rag 1−/− mice. Groups of 15 mice received 50µg 10E4.2G2a mAb or PBS 2 days prior to ivag challenge with HSV-2. Thirteen of 15 PBS-treated controls succumbed to infection by day 15 post-challenge (Fig. 2B). By contrast, Rag 1−/− mAb recipients were protected to a significantly higher level compared to control-treated mice (p <0.001, Logrank test), demonstrating the ability of this mAb to protect in the absence of adaptive immune cells.
Fig. 2.
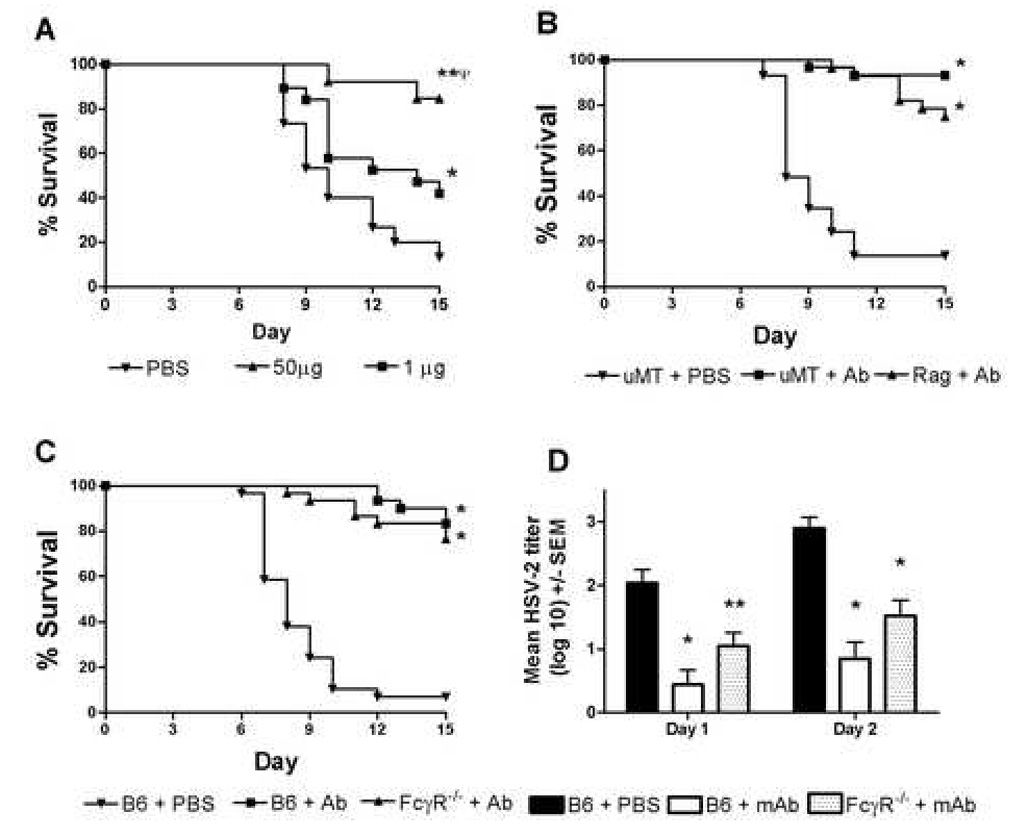
Survival of mice following passive transfer of gD-specific mAb. A, Groups of 10–20 mice received PBS, or either 50µg or 1.0µg 10E4.2G2a, prior to ivag inoculation with 5×103 PFU HSV-2. Survival curves are marked to denote statistical significance as follows: **p< 0.0001 compared to PBS-treated B6; ψp < 0.016 compared to FcγR−/− Ab recipients; and *p < 0.04 compared to PBS-treated B6 mice by Logrank test. Groups of 15µMT and Rag 1 −/− mice (B) or B6 and FcγR−/− mice (C) were given 50µg 10E4.2G2a or PBS 2 days before ivag inoculation with 5×103 PFU HSV-2. Results shown are from representative experiments of two performed. Survival curves significantly different than PBS controls are marked with an asterisk (p<0.001, Logrank test). D, HSV-2 titers in the genital tracts from groups of 15 B6 mAb recipients, FcγR−/− mAb recipients or B6 PBS-treated controls were quantified on the indicated days after HSV-2 inoculation. Marked values are significantly different compared to PBS-treated controls (*p < 0.001; **p < 0.01, ANOVA).
To determine whether expression of FcγR was required for protection mediated by the 10E4.2G2a mAb, groups of 15 B6 or FcγR−/− mice received 50µg 10E4.2G2a mAb or PBS 2 days prior to ivag inoculation with HSV-2. As shown in Fig. 2C, mAb-treated recipients were significantly protected compared to PBS-treated controls (p< 0.001, Logrank test). Unlike polyclonal IgG recipients (Fig1A), there was no significant difference in the survival curves of FcγR−/− and B6 mAb recipients (p=0.21, Logrank test). As shown in Fig. 2D, significantly less infectious HSV-2 was detected in the vaginae of B6 and FcγR−/− mAb recipients on days 1–2 post inoculation compared to PBS-treated controls (p< 0.001 and p< 0.01, respectively, ANOVA). Although HSV-2 titers in FcγR−/− mAb recipients were slightly higher than in B6 mAb recipients, the difference did not reach significance.
3.3 Depletion of Gr-1+, FcγR-expressing cells diminishes protection by polyclonal HSV-specific IgG antibodies
Granulocytes and monocyte/macrophages expressing the Gr-1 surface marker are involved in resistance to HSV infection in naïve and HSV-immune animals (Tumpey et al., 1996; Gangappa et al., 1997; Milligan 1999; Milligan et al., 2001). These cells also represent major cell types which express Fcγ receptors (Ravetch and Bolland, 2001) and may be involved in Ab-mediated protection. Populations of Gr-1+, F4/80− granulocytes and Gr-1+, F4/80+ monocyte/macrophages were readily detected in genital tracts and spinal cords following genital inoculation of HSV-2 (Fig. 3). As described previously (Milligan, 1999), a population of Gr-1+ granulocytes was detected in the vaginal lumen of uninfected, medroxyprogesterone-treated mice (Fig. 3A). An additional population of Gr-1+, F4/80+ cells was detected in the vaginal lumen 2 days after HSV-2 inoculation (Fig. 3B). Gr-1+, F4/80+ monocyte/macrophages and Gr-1+, F480− granulocytes were detected also in the spinal cords of uninfected mice (Fig. 3C). Additionally, an F4/80+, Gr-1− macrophage population was detected at this site 2 days after virus inoculation (Fig. 3D).
Fig. 3.
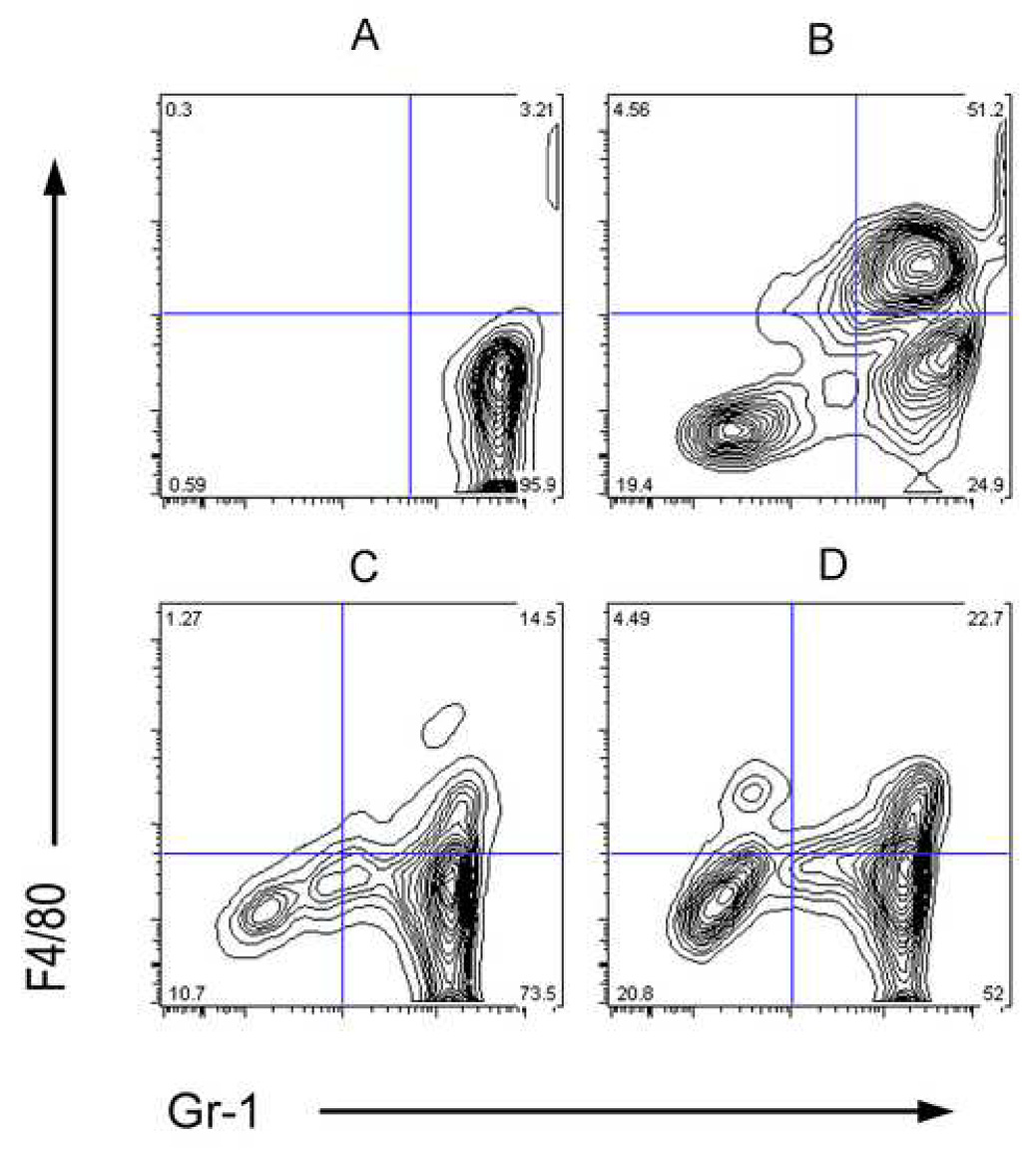
Presence of Gr-1+ cells in the vagina, sensory ganglia and spinal cords of HSV-2 inoculated mice. Vaginal lavages, sensory ganglia and spinal cords were harvested and pooled from 5 uninfected or HSV-2 inoculated mice on day 2 post-inoculation. Leukocytes were isolated and stained for expression of Gr-1 or F4/80, as described in Materials and Methods. A, Vaginal lavage cells from uninfected mice. B, Vaginal lavage cells from mice inoculated 2 days previously with 5×103 PFU HSV-2. C, Spinal cord cells from uninfected mice. D, Spinal cord cells obtained on day 2 post-HSV-2 inoculation. Results are representative of at least two experiments performed.
HSV-2 spreads and replicates to high titer in the sensory ganglia and spinal cords of naïve mice (Milligan et al., 2001). We have shown previously that the kinetics and magnitude of virus replication in the sensory ganglia and spinal cords of HSV-2 –infected naïve mice was not affected by depletion of Gr-1+ cells (Milligan et al., 2001). To test whether Gr-1+ cells were involved in protection resulting from polyclonal Ab administration, HSV-specific Ab recipients were depleted of Gr-1+ cells by administration of specific Ab (RB6.8C5) and the presence and titer of infectious virus in the sensory ganglia and spinal cords was determined. The incidence of infectious HSV-2 in neuronal tissues of control-treated, polyclonal Ab recipients (3 of 11) on day 8 after virus inoculation was significantly less than that of Gr-1-depleted, Ab recipients (7 of 11; p < 0.05, Fisher’s exact test) and Gr-1-depleted PBS recipients (6 of 6; p < 0.05, Fisher’s exact test; Table 2, Experiment A). Higher titers of virus were detected in neuronal tissue of Gr-1-depleted compared to control-treated polyclonal Ab recipients, although the difference did not reach significance. These results suggest that depletion of a large population of FcγR-expressing cells in HSV-specific Ab recipients resulted in a greater incidence of neuronal infection and provide further evidence of the importance of Ab-FcγR interactions in Ab-mediated protection. In contrast, infectious HSV-2 was detected in the sensory ganglia and spinal cords of only 1 of 14 Gr-1-depleted mAb recipients and 0 of 14 control-treated mAb recipients, suggesting protection afforded by administration of the highly neutralizing mAb was independent of the action of Gr-1+ cells (Table 2, Experiment B).
Table II.
Effect of depletion of Gr-1+ cells on protection mediated by passive transfer of HSV-specific polyclonal antibodies or monoclonal anti-gD2 10E4.2G2a antibodies
| Titer (PFU/ mg tissue) | ||||
|---|---|---|---|---|
| Depletiona | HSV-specific Ab Transferredb | Incidencec | Lumbosacral ganglia | Spinal Cord |
| A | ||||
| Anti- Gr-1+ | Polyclonal Ab | 7 \ 11 | 754 +/− 502 | 616 +/− 162 |
| Control IgG | Polyclonal Ab | 3 \ 11 d | 130 +/− 129 | 155 +/− 152 |
| Anti- Gr-1+ | None (PBS) | 6 \ 6e | 1087 +/− 518 | 903 +/− 249 |
| B | ||||
| Anti- Gr-1+ | 10E4.2G2a | 1 \ 14d | 49 +/− 49d | 45 +/− 45 d |
| Control IgG | 10E4.2G2a | 0 \ 14 d | 0d | 0 d |
| Anti- Gr-1+ | None (PBS) | 12 \ 12 | 3475 +/− 723 | 1159 +/− 231 |
Rag 1 −/− mice were depleted of Gr-1+ cells by in vivo treatment with RB6.8C5 antibody or treated with an isotype control antibody of irrelevant specificity.
Mice received either 50µg HSV-specific polyclonal Ab (A), 50µg 10E4.2IgG2a antibody (B), or PBS as a control 2 days prior to ivag inoculation of 5×103 PFU HSV-2 strain 186. Lumbosacral ganglia and spinal cords were sampled on day 8 post inoculation.
Results are expressed as the number of samples containing infectious virus per total number of samples. Results were pooled from 2 experiments of identical design.
p<0.05 compared to PBS-treated Gr-1 depleted mice, 2-tailed Fisher’s exact test.
Four PBS treated, anti-Gr-1 treated mice died prior to tissue sampling and were not included in the analysis.
4. Discussion
The isotype, antigen specificity and affinity of Ab are shaped by the nature of the immunogen or pathogen. In turn, these characteristics determine the effector functions mediated by the Ab. Protection mediated by pathogen-specific Ab may result from the contribution of several effector functions and these may be modulated by immune escape strategies incorporated by the pathogen. Historically, much attention has focused on neutralizing antibody in which the antibody bound to infectious virus prohibits protein interactions involved in binding and internalization steps by either direct recognition of important proteins or steric hindrance of their interaction with the target cell (Fig. 4A). Additionally, antibody may aid in protection by recognition of viral-encoded proteins on the surface of infected cells, resulting in fixation of complement and lysis of the infected cell (Fig. 4B). Alternatively, the Fc region of the IgG antibody bound to cell-surface expressed viral proteins may, in turn, be bound by the FcγR of granulocytes, monocytes, macrophages or NK cells, resulting in destruction of the virus-infected cell by ADCC (Fig 4C). While virus neutralization may occur irrespective of the IgG subclass expressed, the latter two mechanisms are executed by specific IgG subclasses. The results of the current studies suggest that the protection conferred by passive transfer of an HSV-specific polyclonal IgG preparation was mediated primarily, but not exclusively, by mechanisms involving Ab-FcγR interactions. The presence of a relatively high proportion of HSV-specific IgG2b and IgG2a Ab in the HSV-immune serum preparation was consistent with efficient FcγR-Ab interactions. Residual protection observed in the absence of FcγR expression (Fig. 1A) suggested that neutralizing activity contributed also to resistance to HSV-2 infection. In this regard, the HSV-specific IgG1 component of polyclonal serum Ab, while not expected to contribute markedly in protection via Ab–FcγR interactions, might still have provided protection via complement-independent virus neutralization.
Fig. 4.
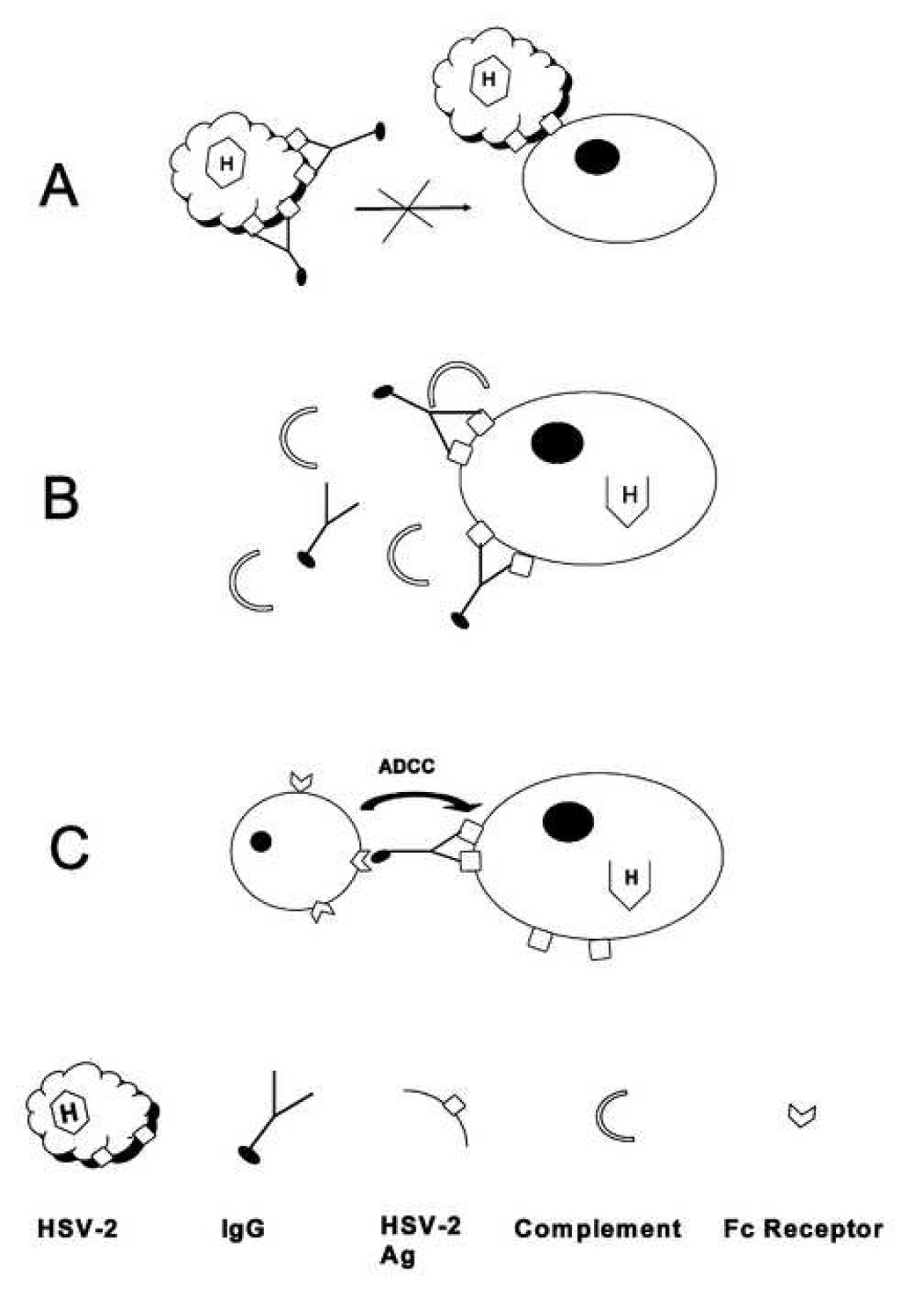
Mechanisms of antibody-mediated protection against HSV-2. A, Virus neutralization. B, Complement-dependent lysis of HSV-infected cells. C, ADCC.
It has been suggested that the poor efficacy of a candidate HSV vaccine may partially have been the result of failure to elicit high levels of Ab capable of mediating ADCC (Kohl et al., 2000). ADCC has been implicated as important for control of HSV infections of newborns and cutaneous HSV-1 infections (Mester et al., 1991; Kohl, 2000), yet the role of ADCC in protection against genital HSV-2 infection has not been examined. In the current study, protection conferred by administration of polyclonal anti-HSV Ab was greatly diminished in FcγR−/− mice, suggesting that anti-viral mechanisms involving Ab-FcγR interactions, such as those involved in ADCC, were responsible for the majority of the Ab-mediated protection. The presence of FcγR-expressing cells in both the genital and neuronal tissues suggests the potential for operation of FcγRdependent protective mechanisms at both sites of HSV infection. The decreased resistance to acute infection of the sensory ganglia and spinal cords in polyclonal anti-HSV Ab recipients depleted of ADCC effector cells, such as Gr-1+ granulocytes and monocytes, is consistent also with an important role for ADCC in Ab-mediated protection against genital HSV-2 exposure.
Although it had been hoped that highly neutralizing Ab might provide sterilizing immunity at the genital epithelium, Ab resulting from natural infection or immunization with experimental vaccines has not consistently provided sterilizing immunity against subsequent vaginal challenge with HSV (Kuklin et al., 1998; Milligan et al., 1998; Bourne et al., 2003). While neither polyclonal Ab acting primarily via FcγR-dependent mechanisms nor highly neutralizing mAb completely prevented infection, passive administration of either Ab preparation resulted in lower initial virus loads in the genital tract early in infection. This result suggests that this FcγR-dependent mechanism is active very early after HSV-2 exposure and is consistent with the detection of FcγR-expressing cells capable of mediating ADCC in the genital tract within the first 2 days after HSV-2 challenge. Ab has been shown to interact with HSV at several sites including the vaginal epithelium, sensory nerve endings, nerve axons, and on the surface of infected neurons in the sensory ganglia (McDermott et al., 1984; Eis Hubinger et al., 1993; Mikloska et al., 1999; Sanna et al., 1999). The detection of populations of granulocytes and monocyte/macrophages in both genital and neuronal sites of HSV infection suggests the FcγR-dependent mechanism may have been active at either or both sites. Our results also are consistent with the concept that Ab acting by either FcγR-dependent or-independent mechanisms may potentially limit HSV-2 spread to sensory neurons, ultimately resulting in lower latent virus loads in sensory ganglia (Morrison et al., 2001; Dalai et al., 2002; Bourne et al., 2002). Taken together, the results of these studies suggest that FcγR-dependent and -independent mechanisms confer similar manifestations of protection against genital HSV-2 challenge. The predominance of a given mechanism may vary in response to different antigenic proteins or routes of inoculation.
Loss of protection was not complete in Gr-1-depleted polyclonal Ab recipients, suggesting that other effector cells important for FcγR-dependent protective functions such as natural killer (NK) cells were not depleted by anti-Gr-1 treatment, that virus neutralizing Ab provided some protection or that alternative FcγR-dependent mechanisms were involved in resisting infection of neuronal tissue by HSV-2. In addition to granulocytes, monocytes and NK cells, FcγR1 has been detected on sensory neurons and cross-linking of FcγR1 bound Ab with specific antigen has been shown to result in intracellular signaling events and release of neutotransmitters (Andoh and Kuraishi, 2004). Any protective role for neuron-expressed FcγR during HSV infection is currently unknown.
HSV has evolved mechanisms to inhibit Ab function in humans, thereby enhancing virus pathogenicity. HSV encoded glycoprotein E and glycoprotein I complexes are incorporated in both virion and infected cell membranes and may serve as an Fc receptor capable of binding human IgG Ab and inhibiting Ab effector functions (Budt et al., 2004). However, the extent to which this putative immune evasion mechanism impacts pathogenesis in humans is not well understood. Elegant studies have shown a near-ablation of antibody-mediated protection in mice receiving human anti-HSV antibody and infected with wild-type virus compared to virus with non-functional Fc receptor (Nagashunmugam et al., 1998). However, other studies in which human anti-HSV antibodies were passively transferred to mice have clearly shown significant protection following infection with wild-type HSV strains (Erlich and Mills, 1986; Sanna et a., 1996; LeBlanc et al., 1999; Dalai et al., 2002). These differences in experimental outcome may reflect the use of different HSV strains or routes of infection. Recently, it has been shown that HSV-encoded Fc receptor preferentially binds certain human IgG subclasses (Armour et al., 2002), suggesting some antibody-mediated protective mechanisms may remain intact in the presence of functional HSV Fc receptor. Additional studies are needed to understand fully the in vivo effects of HSV-2-encoded Fc receptors on HSV-2 pathogenesis as well as vaccine-elicited Ab protection.
In summary, the results of this study suggest that both FcγR-dependent and FcγR-independent mechanisms are capable of providing some level of protection against genital HSV-2 challenge. The main benefits manifest by both mechanisms appear to be limiting the initial infection of the genital tract, decreasing the infectious virus titer in sensory ganglia and spinal cords, and prevention of disease symptoms. These results suggest that, in addition to neutralizing Ab, effective HSV-2 vaccines may need to elicit Ab expressing Fc regions compatible with interaction with FcγR-expressing immune cells to provide the Ab-mediated component for optimal protection against genital HSV-2 infection.
Acknowledgements
The authors thank Dr. Lawrence R. Stanberry and Dr. Premkumar Christadoss for critical reading of the manuscript. This work was supported by National Institutes of Health grants AI42815 and AI054444.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Andoh T, Kuraishi Y. Direct action of immunoglobulin G on primary sensory neurons through Fc gamma receptor I. FASEB J. 2004;18:182–184. doi: 10.1096/fj.02-1169fje. [DOI] [PubMed] [Google Scholar]
- Armour KL, Atherton A, Williamson LM, Clark MR. The contrasting IgG-binding interactions of human and herpes simplex virus Fc receptors. Biochem. Soc. Trans. 2002;30:495–500. doi: 10.1042/bst0300495. [DOI] [PubMed] [Google Scholar]
- Bourne N, Bravo FJ, Francotte M, Bernstein DI, Myers MG, Slaoui M, Stanberry LR. Herpes simplex virus (HSV) type 2 glycoprotein D subunit vaccines afford guinea pigs comparable protection against genital HSV-1 or HSV-2 disease. J. Infect. Dis. 2003;187:542–549. doi: 10.1086/374002. [DOI] [PubMed] [Google Scholar]
- Bourne N, Pyles RB, Bernstein DI, Stanberry LR. Modification of primary and recurrent genital herpes in guinea pigs by passive immunization. J. Gen. Virol. 2002;83:2797–2801. doi: 10.1099/0022-1317-83-11-2797. [DOI] [PubMed] [Google Scholar]
- Brown Z, Benedetti J, Ashley R, Burchett S, Selke S, Berry S, Vontver LA, Corey L. Neonatal herpes virus infection in relation to asymptomatic maternal infection at the time of delivery. N. Engl. J. Med. 1991;324:1247–1252. doi: 10.1056/NEJM199105023241804. [DOI] [PubMed] [Google Scholar]
- Budt M, Reinhard H, Bigl A, Hengel H. Herpesviral Fcγ receptors: culprits attenuating antiviral IgG? Int. Immunopharmacol. 2004;4:1135–1148. doi: 10.1016/j.intimp.2004.05.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corey L, Langenberg AGM, Ashley R, Sekulovich RE, Izu AE, Douglas JM, Handsfield H, Warren T, Marr L, Tyring S, DiCarlo R, Adimora AA, Leone P, Dekker CL, Burke RL, Leong WP, Straus SE. Two double-blind, placebo-controlled trials of a vaccine containing recombinant gD2 and gB2 antigens in MF59 adjuvant for the prevention of genital HSV-2 acquisition. J. Am. Med. Assoc. 1999;282:331–340. doi: 10.1001/jama.282.4.331. [DOI] [PubMed] [Google Scholar]
- Dalai SK, Pesnicak L, Miller GF, Straus SE. Prophylactic and therapeutic effects of human immunoglobulin on the pathobiology of HSV-1 infection, latency, and reactivation in mice. J. Neurovirol. 2002;8:35–44. doi: 10.1080/135502802317247794. [DOI] [PubMed] [Google Scholar]
- Dobbs ME, Strasser JE, Chu C-F, Chalk C, Milligan GN. Clearance of herpes simplex virus type 2 (HSV-2) by CD8+ T cells requires IFN-γ and either perforin- or Fas- mediated mechanisms. J. Virol. 2005;79:14546–14554. doi: 10.1128/JVI.79.23.14546-14554.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eis Hubinger A, Schmidt DS, Schneweis KE. Anti-glycoprotein B monoclonal antibody protects T cell-depleted mice against herpes simplex virus infection by inhibition of virus replication at the inoculated mucous membranes. J. Gen. Virol. 1993;74:379–385. doi: 10.1099/0022-1317-74-3-379. [DOI] [PubMed] [Google Scholar]
- Erlich KS, Mills J. Passive immunotherapy for encephalitis caused by herpes simplex virus. Rev. Infect. Dis. 1986;8 Suppl 4:S439–S445. doi: 10.1093/clinids/8.supplement_4.s439. [DOI] [PubMed] [Google Scholar]
- Gangappa TJ, Kanangat S, Rouse BT. On the essential involvement of neutrophils in the immunopathologic disease: herpetic stromal keratitis. J. Immunol. 1997;158:1383–1391. [PubMed] [Google Scholar]
- Kitamura D, Roes R, Kuhn R, Rajewsky K. A B cell-deficient mouse by targeted disruption of the membrane exon of the immunoglobulin μ chain gene. Nature. 1991;350:423–426. doi: 10.1038/350423a0. [DOI] [PubMed] [Google Scholar]
- Kohl S. Role of antibody-dependent cellular cytotoxicity in defense against herpes simplex virus infections. Rev. Infect. Dis. 1991;13:108–114. doi: 10.1093/clinids/13.1.108. [DOI] [PubMed] [Google Scholar]
- Kohl S, Charlebois ED, Sigouroudinia M, Goldbeck C, Hartog K, Sekulovich RE, Langenberg AG, Burke RL. Limited antibody- dependent cellular cytotoxicity antibody response induced by a herpes simplex virus type 2 subunit vaccine. J. Infect. Dis. 2000;181:335–339. doi: 10.1086/315208. [DOI] [PubMed] [Google Scholar]
- Kuklin NA, Daheshia M, Chun S, Rouse BT. Role of mucosal immunity in herpes simplex virus infection. J. Immunol. 1998;160:5998–6003. [PubMed] [Google Scholar]
- LeBlanc RA, Pesnicak L, Godleski m, Straus SE. Treatment of HSV-1 infection with immunolglobulin or acyclovir: comparison of their effects on viral spread, latency, and reactivation. Virology. 1999;262:230–236. doi: 10.1006/viro.1999.9891. [DOI] [PubMed] [Google Scholar]
- Linehan MM, Richman S, Krummenacher C, Eisenberg RJ, Cohen GH, Iwasaki A. In vivo role of nectin-1 in entry of herpes simplex virus type 1 (HSV-1) and HSV-2 through the vaginal mucosa. J. Virol. 2004;78:2530–2536. doi: 10.1128/JVI.78.5.2530-2536.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McDermott MR, Smiley JR, Leslie P, Brais J, Rudzroga HE, Bienenstock J. Immunity in the female genital tract after intravaginal vaccination of mice with an attenuated strain of herpes simplex virus type 2. J. Virol. 1984;51:747–753. doi: 10.1128/jvi.51.3.747-753.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McKendall RR. IgG-mediated viral clearance in experimental infection with herpes simplex virus type 1: role for neutralization and Fc-dependent functions but not C’ cytolysis and C5 chemotaxis. J. Infect. Dis. 1985;151:464–470. doi: 10.1093/infdis/151.3.464. [DOI] [PubMed] [Google Scholar]
- Mester JC, Glorioso JC, Rouse BT. Protection against zosteriform spread of herpes simplex virus by monoclonal antibodies. J. Infect. Dis. 1991;163:263–269. doi: 10.1093/infdis/163.2.263. [DOI] [PubMed] [Google Scholar]
- Mikloska Z, Sanna PP, Cunningham AL. Neutralizing antibodies inhibit axonal spread of herpes simplex virus type 1 to epidermal cells in vitro. J. Virol. 1999;73:5934–5944. doi: 10.1128/jvi.73.7.5934-5944.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milligan GN. Neutrophils aid in protection of the vaginal mucosae of immune mice against challenge with herpes simplex virus type 2. J. Virol. 1999;73:6380–6386. doi: 10.1128/jvi.73.8.6380-6386.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milligan GN, Bernstein DI. Analysis of herpes simplex virus-specific T cells in the murine female genital tract following genital infection with herpes simplex virus type 2. Virology. 1995;212:481–489. doi: 10.1006/viro.1995.1506. [DOI] [PubMed] [Google Scholar]
- Milligan GN, Bernstein DI, Bourne N. T lymphocytes are required for protection of the vaginal mucosae and sensory ganglia of immune mice against reinfection with herpes simplex virus type 2. J. Immunol. 1998;160:6093–6100. [PubMed] [Google Scholar]
- Milligan GN, Bourne N, Dudley KL. Role of polymorphonuclear leukocytes in resolution of HSV-2 infection of the mouse vagina. J. Reprod. Immunol. 2001;49:49–65. doi: 10.1016/s0165-0378(00)00080-2. [DOI] [PubMed] [Google Scholar]
- Milligan GN, Dudley-McClain KL, Chu D-F, Young CF. Efficacy of genital T cell responses to herpes simplex virus type 2 resulting from immunization of the nasal mucosa. Virology. 2004;318:507–515. doi: 10.1016/j.virol.2003.10.010. [DOI] [PubMed] [Google Scholar]
- Mombaerts P, Iacomini J, Johnson RS, Herrup K, Tonegawa S, Papaioannou VE. RAG-1 deficient mice have no mature B and T lymphocytes. Cell. 1992;68:869–877. doi: 10.1016/0092-8674(92)90030-g. [DOI] [PubMed] [Google Scholar]
- Morrison LA, Zhu L, Thebeau LG. Vaccine-induced serum immunoglobin contributes to protection from herpes simplex virus type 2 genital infection in the presence of immune T cells. J. Virol. 2001;75:1195–1204. doi: 10.1128/JVI.75.3.1195-1204.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagashunmugam T, Lubinski J, Wang L, Goldstein LT, Weeks BS, Sundaresan P, Kang EH, Dubin G, Friedman HM. In vivo immune evasion mediated by the herpes simplex virus type 1 immunoglobulin G Fc Receptor. J. Virol. 1998;72:5351–5359. doi: 10.1128/jvi.72.7.5351-5359.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nimmerjahn F, Bruhns P, Horiuchi K, Ravetch JV. FcγRIV: A novel FcR with Distinct IgG subclass specificity. Immunity. 2005;23:41–51. doi: 10.1016/j.immuni.2005.05.010. [DOI] [PubMed] [Google Scholar]
- Oakes JE, Lausch RN. Role of Fc fragments in Antibody-mediated recovery from ocular and subcutaneous herpes simplex virus infections. Infect. Immun. 1981;33:109–114. doi: 10.1128/iai.33.1.109-114.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ravetch JV, Bolland S. IgG Fc Receptors. Annu. Rev. Immunol. 2001;19:275–290. doi: 10.1146/annurev.immunol.19.1.275. [DOI] [PubMed] [Google Scholar]
- Sanna PP, Deerinck TJ, Ellisman MH. Localization of a passively transferred human recombinant monoclonal antibody to herpes simplex virus glycoprotein D to infected nerve fibers and sensory neurons in vivo. J. Virol. 1999;73:8817–8823. doi: 10.1128/jvi.73.10.8817-8823.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanna PP, De Logu A, Williamson RA, Hom Y-L, Straus SE, Bloom FE, Burton DR. Protection of nude mice by passive immunization with a type-common human recombinant monoclonal antibody against HSV. Virology. 1996;215:101–106. doi: 10.1006/viro.1996.0011. [DOI] [PubMed] [Google Scholar]
- Schneweis KE, Brado M, Ebers B, Friedrich A, Olbrich M, Schuler W. Immunological mechanisms giving rise to latency of herpes simplex virus in the spinal ganglia of the mouse. Med. Microbiol. Immunol. (Berl) 1988;177:1–8. doi: 10.1007/BF00190305. [DOI] [PubMed] [Google Scholar]
- Stanberry LR, Bernstein D, Kit S, Myers MG. Genital reinfection after recovery from initial genital infection with herpes simplex type 2 in guinea pigs. J. Infect. Dis. 1986;153:1055–1061. doi: 10.1093/infdis/153.6.1055. [DOI] [PubMed] [Google Scholar]
- Stanberry LR, Spruance SL, Cunningham AL, Bernstein DI, Mindel A, Sacks S, Tyring S, Aoki FY, Slaoui M, Denis M, Vandepeliere P, Dubin G. Glycoprotein D adjuvant vaccine to prevent genital herpes. N. Eng. J. Med. 2002;21:1652–1661. doi: 10.1056/NEJMoa011915. [DOI] [PubMed] [Google Scholar]
- Tumpey TM, Chen SH, Oakes JE, Lausch RN. Neutrophil-mediated suppression of virus replication after herpes simplex virus type 1 infection of the murine cornea. J. Virol. 1996;70:898–904. doi: 10.1128/jvi.70.2.898-904.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]


