Abstract
Evidence has shown that alcoholism leads to volume reductions in brain regions critical for associative learning using the eyeblink classical conditioning paradigm (EBCC). Evidence indicates that cerebellar shrinkage causes impairment in simple forms of EBCC, whereas changes in forebrain structures result in impairment in more complex tasks. In this study, the ability of abstinent alcoholics and matched control participants to acquire learned responses during delay discrimination and discrimination reversal was examined and related to severity of drinking history and neuropsychological performance. During discrimination learning, one tone (CS+) predicted the occurrence of an airpuff (unconditioned stimulus), and another tone (CS−) served as a neutral stimulus; then the significance of the tones was reversed. Alcoholics who learned the initial discrimination were impaired in acquiring the new CS+ after the tones reversed; this is a function that has previously been linked to forebrain structures. It is suggested that a factor important to alcoholic addiction may be the presence of alcoholic-related associative responses that interfere with the ability to learn new more adaptive associations.
Keywords: alcohol, eyeblink classical conditioning, discrimination, reversal
Chronic misuse of alcohol leads to volume reductions in brain regions critical for associative learning using the eyeblink classical conditioning (EBCC) paradigm. First, alcohol is known to cause structural alterations in the cerebellum, a structure that is both necessary and sufficient for all forms of EBCC (R. F. Thompson, 1986). Such alterations have been documented by traditional postmortem inspection (Harper & Kril, 1989) and more recently by in vivo neuroimaging studies, confirming significant volume shrinkage in the cerebellar hemispheres (Sullivan, Rosenbloom, Lim, & Pfefferbaum, 2000).
Second, in addition to alcohol-related neuropathological changes in the cerebellum, abundant evidence from different methodologies indicates that the structural alterations due to alcohol extend into prefrontal cortex and frontal circuitry. These are areas of the brain known to be essential for more complex or nonoptimal forms of EBCC. For example, using structural MRI, Sullivan and her colleagues (e.g., Sullivan et al., 2003; Sullivan & Pfefferbaum, 2005) have reported that each major node of the frontocerebellar circuit shows volume reductions and each can be independently affected. MRI studies have also revealed greater volume losses in the frontal lobes compared with other structures (Dao-Castellana et al., 1998; Jernigan et al., 1991; Kril, Halliday, Svoboda, & Cartwright, 1997). White matter changes in alcoholics have been documented using diffusion tensor imaging (Pfefferbaum & Sullivan, 2002; Pfefferbaum et al., 2000). Post-mortem evidence from Harper, Kril, and Daly (1987) showed a 22% reduction in the number of neurons in the superior frontal cortex of alcoholics.
Alcoholism also results in degradation of cognitive function. Neuropsychological deficits range from mild to severe (e.g., Korsakoff’s syndrome) and may be transient or long term. The neuropsychological processes most often affected in chronic alcoholics include executive function, working memory, visuospatial abilities, and motor function (gait and balance; Butters & Cermak, 1980; Feinberg, Haber, & Stacy, 1990; Moselhy, Georgiou, & Kahn, 2001; Nixon, 1993; Oscar-Berman, 2000; Oscar-Berman, Kirkley, Gansler, & Couture, 2004; Parsons & Leber, 1981; Sullivan, Rosenbloom, & Pfefferbaum, 2000). For example, consistent with the frontal lobe dysfunction hypothesis (Oscar-Berman & Hutner, 1993), alcoholism leads to impairments in planning and problem-solving ability (Pishkin, Lovallo, & Bourne, 1985) and response shifting and inhibition (Hildebrandt, Brokate, Eling, & Lanz, 2004) in individuals who appear to be neurologic ally intact. Sullivan, Deshmukh, De Rosa, Rosenbloom, and Pfefferbaum (2005) recently demonstrated that working memory deficits endure in both recent and long-term recovered alcoholics. Of note, there is a great deal of heterogeneity in the neuropsychological impact of chronic alcoholism. The underlying cause of this heterogeneity is most likely related to a number of factors, including but not limited to lifetime consumption, chronicity of drinking, age, gender, metabolic issues, nutrition, related medical conditions (e.g., hypertension, liver function), associated psychiatric comorbidity, and family/genetic history.
Several studies have now demonstrated deficits in classical associative learning in abstinent alcoholics (McGlinchey, Fortier, Capozzi, & Disterhoft, 2005; McGlinchey-Berroth, Carrillo, Gabrieli, Brawn, & Disterhoft, 1997; McGlinchey-Berroth et al, 1995). In the present study, we examined the performance of abstinent alcoholics in an EBCC task that requires an essential contribution from forebrain structures (Berger & Orr, 1983; Chachich & Powell, 1998; Carrillo et al., 1997): delay reversal learning following delay discrimination. Discrimination conditioning involves the presentation of two conditioned stimuli (CS), one of which (CS+) is paired with an airpuff (unconditioned stimulus [US]), and the other (CS−) is presented alone (i.e., with no consequence). During the initial phase of learning, individuals do not discriminate between the CS+ and the CS− and produce conditioned responses (CRs) to both trial types. However, over additional trials, CRs to the CS− drop off and are produced, for the most part, only during the CS+ trials. Once acquisition of the discrimination occurs, the contingencies of the two CSs can be reversed. During this more complex reversal conditioning task, the significance of the two CSs is switched by making the previously paired CS+ the CS−, and the previous CS− the CS+. Importantly, this reversal occurs unbeknownst to the participants, seamlessly, and without warning.
Several animal and human studies have demonstrated that damage to the hippocampus or the hippocampal system does not impair performance in the delay discrimination task, but either impairs (Buchanan & Powell, 1982) or completely abolishes the ability to reverse the discrimination (Berger & Orr, 1983; Carrillo et al., 1997). The failure to reverse is not due to a discrimination deficit, per se, evidenced by the fact that the initial discrimination is acquired. Rather, the inability to reverse in hippocampally damaged participants is due to continued responding to the new CS−/old CS+ during the reversal phase.
In addition to evidence supporting an essential role of the hippocampal system, frontal systems have also been demonstrated to play a role in reversal learning in animal studies. The notion of a cerebellar–thalamic–prefrontal cortex module controlling eyeblink associative learning was proposed initially by Weiss and Disterhoft (1996) and most recently by McLaughlin, Powell, and White (2002), who suggested that the medial prefrontal cortex–cerebellar–thalamic module is necessary for acquisition of the eyeblink CR during reversal conditioning. Buchanan and colleagues have demonstrated that prefrontal lesions (Buchanan & Powell, 1982) and thalamic mediodorsal nucleus lesions (which project to the medial prefrontal cortex; Buchanan, 1991; Buchanan, Penney, Tebbutt, & Powell, 1997) impaired acquisition during nonoptimal learning conditions, such as single cue trace conditioning and reversal conditioning. In support of this finding, Chachich and Powell (1998) documented that medial prefrontal lesions, but not amygdala lesions (amygdala central nucleus), impaired the ability to reverse a previously learned discrimination. Therefore, although medial prefrontal cortex and thalamic mediodorsal nucleus are active during simple EBCC learning paradigms, they do not appear to be essential (Buchanan et al., 1997). However, under nonoptimal learning conditions involving greater task complexity (e.g., trace single cue and reversal learning), these structures seem to be more critically involved. To date, the exact nature of the deficit in reversal learning secondary to prefrontal cortex damage has not been determined and either may be an impairment in inhibitory responding (increased responding to the old CS+) as seen with damage to medial temporal structures (Berger & Orr, 1983) or may be slowed (or abolished) acquisition of the new CS+ (Buchanan, 1991).
Given the neuropathological and neuropsychological evidence of cerbellar and frontal system deficits associated with alcoholism, we predicted that alcoholics would be significantly impaired in reversal learning on the basis of their combined cerebellar and frontal system pathology. To examine participants’ ability to reverse a previously learned discrimination, it was necessary for participants to demonstrate at least some acquisition of the initial discrimination (e.g., produce a greater number of CRs during reinforced as compared with nonreinforced trials). Alcoholics who could not acquire the initial discrimination were hypothesized to have extensive cerebellar damage, which could confound interpretation of their reversal performance. Therefore, alcoholics who could not learn the initial discrimination to an applied criterion were excluded from this investigation.
Method
Participants
A total of 37 individuals were recruited to participate in this study. A final sample of 14 abstinent alcoholic participants (ALC) and 14 normal control participants (NC) was included from this larger sample on the basis of discrimination learning criteria described below (Learning Criteria for Study Inclusion). The maximum number of abstinent alcoholics who met the learning criteria defined the sample size for each group. Six ALCs were excluded because they did not meet the defined learning criteria. Three NCs were excluded on the basis of the order in which they were recruited (i.e., last in, first excluded) and the completeness of their data set (i.e., excluded if missing any neuropsychological assessment data). All participants were naive to the EBCC procedures, meaning they had no prior training in eyeblink conditioning.
The participants in this study were recruited from the Geriatric Research, Education, and Clinical Center (GRECC) at the Veterans Affairs Boston Healthcare System, Boston, by way of distribution of flyers at local institutions, advertisements in local newspapers, referral from the Harvard Cooperative Program on Aging, and by referral from area hospitals. ALC participants and NC participants were screened to be free of any neurological disease or illness. Participants were also excluded for any central nervous system drugs, blackouts (NCs only), seizure disorder (NCs only), head injury, hospitalization in a psychiatric facility >1 week, or any medications for/history of severe psychiatric disorders (e.g., schizophrenia, chronic intractable obsessive–compulsive disorder, agoraphobia, current major depression). History of substance abuse or dependence other than alcohol, except nicotine (current or lifetime) and cannabis (lifetime), was cause for exclusion. Cannabis use in the year prior to testing was cause for exclusion.
Learning Criteria for Study Inclusion
To examine participants’ ability to reverse the initially learned discrimination, it was necessary for participants to demonstrate at least some acquisition of the initial discrimination. Participants were excluded from this study if they did not acquire a minimum of 20% CRs on reinforced trials during discrimination learning or if they did not achieve at least a 10% difference in percentage CRs expressed during CS+ as compared with CS− trials during discrimination learning.
Abstinent alcoholic (ALC) participants
Fourteen abstinent alcoholic individuals were included in the final sample (8 men, 6 women). The mean age of this ALC group was 47 years (SD = 7.7). The mean education in years was 13 (SD = 3.9). The mean verbal intelligence as measured by the Wechsler Adult Intelligence Scale—Third Edition (WAIS–III; Corporation, 1997) was 100 (SD = 19.5).
To meet criteria for inclusion, the ALC participants must have abstained from drinking for at least 1 month prior to participating in the study and report a history of ≥ 21 drinks per week for 5 years or longer (M. Oscar-Berman, personal communication, March 1, 2005). The mean duration of abstinence prior to testing was 6.4 years (SD = 6.7). On the Lifetime Drinking History (LDH; Skinner & Sheu, 1982), ALCs reported a significant history of alcohol abuse that ranged in duration from 12 years to 40 years. The mean length of abuse was 24 years (SD = 8.2). This measure yields an estimate of total lifetime exposure to alcohol using standard drink conversions (grams absolute alcohol) via two methods: (a) total lifetime drinks and (b) and total lifetime drinks controlling for weight (body weight in kg). ALCs reported an average lifetime total volume of alcohol exposure of 4,481 drinks or 867 g/kg when corrected for body weight. During all drinking phases, ALCs reported a mean of 9 (SD = 5.4) standard drinks per drinking day and a mean maximum of 11 (SD = 6.0) standard drinks per drinking day. For a profile of each alcoholic participant’s drinking history, see Table 1.
Table 1.
Drinking Characteristics of the Abstinent Alcoholic (ALC) Group
| LDH measures
|
||||||||
|---|---|---|---|---|---|---|---|---|
| Participant | Years of abuse | Months sober | DIS | SMAST | Total lifetime drinks | Total lifetime drinks (g/kg)a | Average drinks/day | Maximum drinks/day |
| ALC001 | 12 | 12 | 1 | 6 | 640.0 | 127.66 | 10.00 | 20.00 |
| ALC002 | 21 | 24 | 2 | 10 | 3,259.3 | 568.78 | 7.14 | 11.93 |
| ALC003 | 21 | 216 | 1 | 5 | 759.6 | 216.45 | 2.25 | 3.25 |
| ALC004 | 33 | 36 | 2 | 13 | 5,220.0 | 765.71 | 8.63 | 10.88 |
| ALC005 | 27 | 6 | 2 | 11 | 14,224.5 | 2,745.79 | 17.33 | 18.17 |
| ALC006 | 40 | 24 | 1 | 4 | 2,497.8 | 442.91 | 2.75 | 4.75 |
| ALC007 | 28 | 84 | 2 | 5 | 5,541.5 | 787.27 | 11.48 | 13.68 |
| ALC008 | 12 | 36 | 1 | 9 | 1,444.8 | 331.10 | 10.00 | 10.00 |
| ALC009 | 15 | 180 | 0 | 5 | 600.0 | 138.10 | 5.00 | 7.00 |
| ALC010 | 18 | 228 | 2 | 3 | 2,082.4 | 323.91 | 7.00 | 12.00 |
| ALC011 | 31 | 108 | 1 | 5 | 1,089.2 | 326.52 | 3.00 | 3.25 |
| ALC012 | 30 | 3 | 0 | — | 18,963.0 | 4,202.76 | 21.00 | 22.00 |
| ALC013 | 22 | 5 | 2 | 9 | 5,620.0 | 1,016.17 | 9.20 | 15.6 |
| ALC014 | 22 | 108 | 2 | — | 796.3 | 147.75 | 4.75 | 7.25 |
Note. DIS = Diagnostic Interview Schedule for DSM-IV (DIS-IV): Alcohol dependence = 2, abuse = 1, no diagnosis = 0; SMAST = Self-Administered Short Michigan Alcoholism Screening Test: 0–1 = nonalcoholic profile, 2 = possible alcoholic profile, and ≥ 3 = alcoholic profile; LDH = Lifetime Drinking History. Drinking descriptors on the LDH are presented for all drinking phases. The LDH is designed to aggregate all drinking phases across the lifespan. Therefore, this instrument assesses all time periods (not just phases of heavy drinking) in which a participant reported using alcohol regardless of quantity of use. Note that 2 ALC participants were not administered SMAST (lost to follow-up).
Adjusted for weight.
Seven ALCs met Diagnostic and Statistical Manual of Mental Disorders (4th ed.; DSM-IV; American Psychiatric Association, 1994) diagnostic criteria for alcohol dependence on the Diagnostic Interview Schedule for DSM-IV (DIS-IV; Robins, Cunningham, & Gallagher, 1996), and 5 met criteria for alcohol abuse. The entire 90–120-min DIS computerized instrument was administered to participants. Unfortunately, some participants appeared to have difficulty attending to the entire computer interview (as demonstrated by inconsistencies between self-reported history and DIS responses) and answered unreliably during the substance abuse module, which came during the latter part of the interview. If possible, these individuals returned to the laboratory for follow-up DIS Substance Abuse Module administration in which they answered only the 28 substance abuse-related items of the DIS. Two individuals were lost to follow-up and the Substance Abuse Module could not be readministered. For these participants, available Short Michigan Alcoholism Screening Test (SMAST; Selzer, Vinokur, & van Rooijen, 1975), LDH, and questionnaire data were used to confirm alcohol history. These individuals reported a history consistent with alcohol abuse as defined by ≥ 21 drinks/week for a minimum of 5 years and self-identified as alcoholics. One participant who did not meet criteria for alcohol abuse/dependence on the DIS (ALC009, see Table 1) had a positive SMAST (score of 5). The remaining participant with a negative diagnosis based on the DIS (ALC012) reported alcohol consumption of >20 drinks per day for a 30-year period.
On the SMAST, a self-reported measure of alcoholic behavior, ALCs reported scores ranging from 3 to 13, with a mean score of 7 (SD = 3.2). Selzer et al. (1975) suggest that a score of 0–1 on the SMAST represents a nonalcoholic profile, a score of 2 indicates a possible alcoholic profile, and a score of 3 or higher represents an alcoholic profile.
Normal control (NC) participants
Fourteen NC participants (8 men, 6 women) were included in the final sample. All NC participants denied a significant history of alcohol or other substance abuse. This was supported by results on the computerized DIS-IV, on which none of the NC participants met criteria for alcohol dependence or abuse. The NC group was matched to the ALC group with regard to age (M = 50, SD = 8.8), education (M = 15, SD = 2.1), and verbal intelligence as measured by the WAIS-HI (M = 109, SD = 14.3). Results of t tests indicated that the ALC and NC participants were not significantly different on each of these measures (ps > .18).
Apparatus
The apparatus used was a modified version of that used for eyeblink conditioning in the rabbit (Akase, Thompson, & Disterhoft, 1994; L. T. Thompson, Moyer, Akase, & Disterhoft, 1994), and one that we have used in previous eyeblink conditioning studies with humans (Fortier et al., 2003; McGlinchey et al., 2005). Eyeblink responses were measured via surface electromyography (EMG) electrodes (Nicolet, NY) placed over the orbicularis oculi muscle of the right eye. An adjustable headband was worn to support the airpuff delivery nozzle.
Data were acquired by a custom data acquisition system developed using National Instruments Lab VIEW (National Instruments, Austin, TX). EMG data were acquired at 5 kHz and filtered at 2 kHz using a low-pass Bessel filter. Stimulus presentation and data acquisition were controlled by custom software written in Lab-VIEW. The digitized EMG signal was rectified (absolute value of the amplitude) and integrated using a decay time constant of 10 ms. The integrated–rectified signal is well correlated with the eyelid closure measured using reflectance eyelid detectors (Knuttinen et al., 2001).
Stimuli
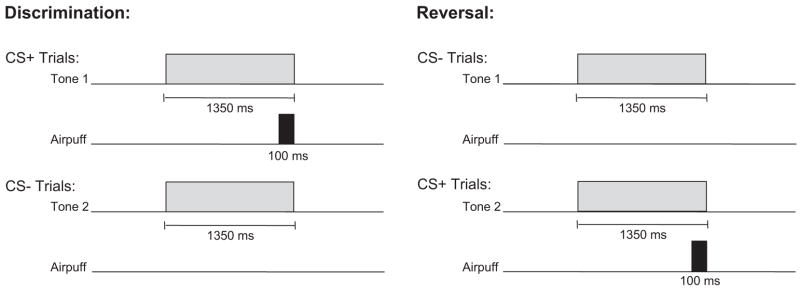
Figure 1 displays a schematic of the time course of each trial type. As shown, there were two different tones (high and low) to signal the onset of a reinforced (CS+) or nonreinforced (CS−) trial. Assignment of the tone to these two conditions was counter-balanced across subjects. For half of the participants, discrimination learning consisted of a 1,350-ms 85 dB, 1000 Hz tone CS + and a 1,350-ms 85 dB, 5000 Hz tone CS− that were delivered binaurally over headphones. The significance of the tones was reversed for the remaining participants (5000 Hz CS+ and 1000 Hz CS−). All other parameters remained constant. The US was presented only on CS+ trials and consisted of a 100-ms corneal airpuff that coterminated with the CS +. The magnitude of the airpuff was 3 psi for all participants. Participants were presented with 30 of each trial type randomly intermixed. Presentation of trial type was determined by computer-generated pseudorandomized series such that no more than 3 reinforced or nonreinforced trials could occur in succession. During reversal learning, the CS− became the CS +, and the CS+ became the CS−. The transition from discrimination training to reversal training was seamless and uninterrupted. Participants were again presented with 30 trials of each type randomly intermixed. A total of 120 EBCC learning trials were presented, including 60 discrimination trials and 60 reversal trials, half reinforced (CS+) and half nonreinforced (CS−).
Figure 1.
Delay discrimination and reversal learning. CS+ = reinforced conditional stimulus; CS− = nonreinforced conditional stimulus.
Procedure
Participants were brought into the laboratory individually where the examiner reviewed the informed consent form with them. All participants underwent three types of assessment: (a) assessment of drinking, (b) neuropsychological assessment, and (c) EBCC. The assessments were completed in two to three testing sessions. The testing sessions were generally completed within 1 month for each participant. The longest interval between first and last sessions was 2 months. Some participants were contacted after study completion to provide additional information regarding their drinking history.
Neuropsychological assessment
All study participants received a neuropsychological test battery that targeted cognitive domains affected by alcoholism (tasks sensitive to frontal and cerebellar dysfunction) and those thought to underlie the learning and expression of classical conditioned responses in associative learning tasks, including executive function, motor function, and memory. A test of general verbal intelligence was also administered. Verbal abilities were assessed with WAIS–III (Corporation, 1997). Memory/medial temporal function was assessed with the Wechsler Memory Scale—Third Edition (WMS–III; Corporation, 1997) and the Warrington Recognition Memory Test (Warrington, 1984). Executive/frontal system function was assessed with the Trailmaking Test (Reitan, 1979), Controlled Oral Word Association Test (COWAT; Benton & deS Hamsher, 1978), Wisconsin Card Sorting Test (WCST; Heaton, 1981), Stroop Color–Word Test (Trenerry, Crosson, DeBoe, & Leber, 1989), and Ruff Figural Fluency Test (Ruff, 1996). Motor/cerebellar function was assessed with the Grooved Pegboard (Tiffen, 1968), Finger Tapping Test (Reitan, 1969; Spreen & Strauss, 1998), and an ataxia battery (Sullivan, Rosenbloom, & Pfefferbaum, 2000).
EBCC procedure
Each participant underwent an audiology screening using a Model 119 Beltone portable audiometer. The criteria of Solomon (Solomon, Pomerleau, Bennett, James, & Morse, 1989) was employed, which demanded that participants whose threshold in either ear was greater than 15 dB above normal (40 dB) be excluded. However, all participants’ thresholds fell within the normal range and thus none of the participants recruited for this study were excluded on the basis of the audiology screening results. Participants were seated in an upright chair and the examiner fitted them with the eyeblink apparatus. Throughout the session, the experimenter sat in the same room, out of the direct view of the participant, and answered questions as they arose. Each conditioning session consisted of a total of 120 conditioning trials. Prior to the onset of each trial, there was a 750-ms baseline recording period. The intertrial interval averaged 10 s, but varied randomly from 8 to 12 s.
Definitions
An eyeblink was only scored as a CR if its amplitude was at least 4 standard deviations greater than the mean baseline response amplitude. Eyeblinks with latencies less than 100 ms following CS onset were recorded as alpha responses and not considered CRs (Gormezano, 1966).
Results
The primary measures of interest were the percentage of CRs acquired during CS+ and CS− trials. Other dependent variables examined included characteristics of both the conditioned and unconditioned responses (URs): CR onset latency, CR peak latency, CR amplitude, and UR amplitude. CR onset latency refers to the time at which the CR amplitude first reached 4 standard deviations above baseline. Alternatively, CR peak latency represents the time at which the given CR reached its highest amplitude. CR peak latency likely captures the level of adaptiveness of a CR (optimally, a CR will peak just before the onset of the airpuff). The CR amplitude is measured as peak amplitude and refers to the amount of EMG muscle activity during a CR. UR amplitude is measured as peak amplitude and refers to the amount of EMG muscle activity during the UR period, and reflects the unconditioned reflex in response to the airpuff.
The overall level of acquisition of CRs was examined with a Group × Trial Type (reinforced, nonreinforced) analysis of variance (ANOVA), as were other measures of performance, including the CR and UR latency and amplitude. Linear regression analyses were performed to examine the rate of learning during reinforced trials. Correlational analyses were performed to investigate the relationship between measures of drinking severity, measures of neuropsychological performance, and measures of eyeblink conditioning.
Discrimination Learning
Conditioned response acquisition
ANOVA with the overall mean percentage of CRs as the dependent measure, group (ALC, NC) as a between-subjects factor, and trial type (CS + , CS−) as a within-subjects factor revealed a significant main effect of trial type, F(1, 26) = 24.8, p = .001, partial η2 = .488, indicating that participants produced a greater number of CRs during reinforced (CS+) as compared with nonreinforced (CS−) trials. Means comparisons confirmed a significantly greater percentage of CRs during reinforced trials as compared with nonreinforced trials for each group separately, indicating that both ALC (p = .004, d = 1.29) and NC participants (p = .003, d = 1.35) were able to respond differentially on CS+ versus CS− trials and acquired the initial discrimination (see Figure 2). ALCs produced a CR on 54% (SE = 5.0) of CS+ trials and 33% (SE = 3.6) of CS− trials. NCs produced a CR on 60% (SE = 6.0) of CS+ trials and 32% (SE = 4.9) of CS− trials.
Figure 2.
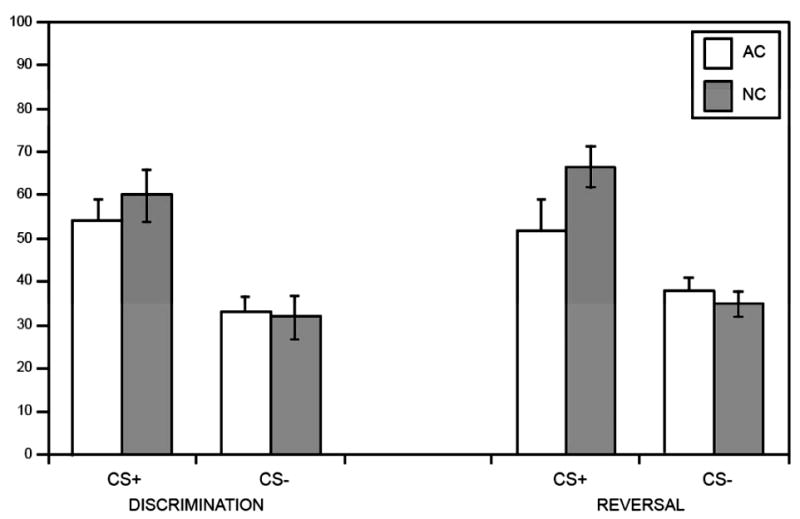
CR acquisition. Mean percentage conditioned responses (CRs) for reinforced (CS+) and nonreinforced (CS−) trials. CS+ = reinforced conditional stimulus; CS − = nonreinforced conditional stimulus; ALC = abstinent alcoholic group; NC = nonalcoholic control group.
Learning curves
The percentage of trials on which a CR occurs is expected to increase during reinforced trials during the initial phases of learning. Conversely, the percentage of CRs produced for nonreinforced trials is expected to remain stable or decrease across learning trials. As can be seen in Figure 3, when conditioning trials were collapsed into six blocks of five trials each, the NC participants demonstrated an overall increase in the percentage of CRs across the six discrimination learning blocks of reinforced trials, peaking at Blocks 5 and 6. The learning curve for the ALC participants is similar, although not as consistent as NC participants. Block-by-block comparisons between groups revealed a difference approaching significance in production of CRs during reinforced trials at Block 5 (p = .07). Percentage of CRs on nonreinforced trials remained stable across learning blocks in both groups. Block-by-block comparisons showed no difference in CR production during nonreinforced trials between groups. Including block as a within-subjects factor did not change the overall pattern of the ANOVA.
Figure 3.
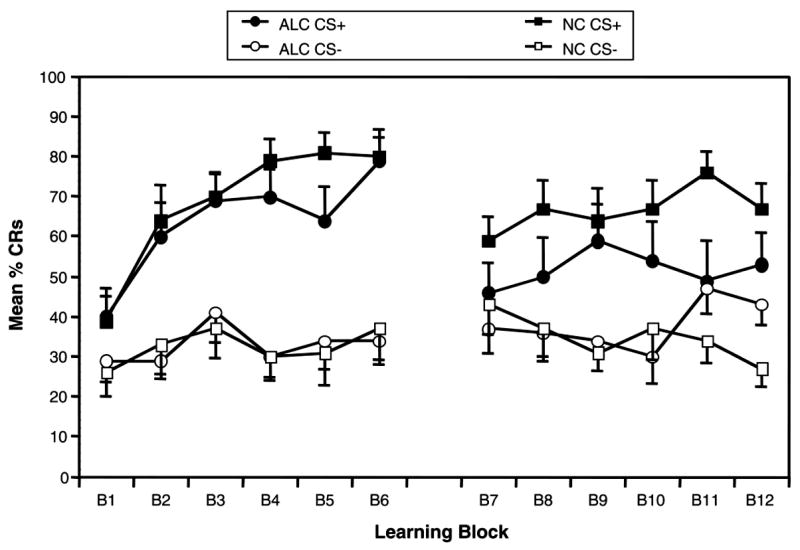
Learning curves: Conditioning trials were collapsed into six blocks of five trails each. CS+ = reinforced conditional stimulus; CS− = nonreinforced conditional stimulus; ALC = abstinent alcoholic group; NC = nonalcoholic control group; CRs = conditioned responses.
To examine the rate of learning or acquisition, we performed linear regression analyses for the mean percentage of CRs for each group separately for the six blocks of acquisition trials during reinforced trials. The linear regression analysis was significant for the control participants during discrimination learning, F(1, 82) = 14.4, p = .001, R2 = .15. The regression was also significant for the abstinent alcoholics during discrimination learning, F(1, 82) = 7.71, p = .007, R2 = .09.
Response latency and amplitude
Unpaired t tests of CR latency during reinforced trials revealed that none of the measures differed significantly between groups (ps > .09) during discrimination learning (see Table 2). Similarly, there were no differences between groups for CR and UR amplitude during reinforced trials (ps > .5; see Tables 2 and 3).
Table 2.
Conditioned Response Measure Means (Standard Deviations)
| CR onset latency (ms)
|
CR peak latency (ms)
|
CR amplitude (mV)
|
||||
|---|---|---|---|---|---|---|
| Group | CS + | CS − | CS + | CS − | CS+ | CS − |
| Discrimination | ||||||
| ALC | 850 (58) | 901 (52) | 985 (63) | 986 (69) | 21 (16) | 21 (18) |
| NC | 891 (69) | 884(61) | 997 (71) | 943 (48) | 18(8) | 16(9) |
| Reversal | ||||||
| ALC | 893 (68) | 888 (73) | 1,006(72) | 973 (48) | 15(9) | 13(6) |
| NC | 902 (83) | 925 (83) | 1,030(67) | 993 (82) | 17 (10) | 15(9) |
Note. CR = conditioned response; CS+ = reinforced conditioned stimulus; CS− = nonreinforced conditioned stimulus; ALC = abstinent alcoholic group; NC = nonalcoholic control group.
Table 3.
Unconditioned Response Measure Means (Standard Deviations)
| UR amplitude (mV)
|
||
|---|---|---|
| Group | CS + | CS − |
| Discrimination | ||
| ALC | 51 (14) | 16(13) |
| NC | 48(18) | 17 (30) |
| Reversal | ||
| ALC | 63 (17) | 12(6) |
| NC | 60 (23) | 13(10) |
Note. UR = unconditioned response; CS+ = reinforced conditioned stimulus; CS− = nonreinforced conditioned stimulus with no consequence; ALC = abstinent alcoholic group; NC = nonalcoholic control group.
Reversal Learning
Conditioned response acquisition
During reversal learning, the previously reinforced CS+ became the CS−, and the previously nonreinforced CS− became the CS + , requiring participants to flexibly alter their previously learned stimulus contingencies, decreasing their CR production to the old CS+ and increasing CR production to the new CS + .
ANOVA with the overall mean percentage of conditioned responses as the dependent measure, group (ALC, NC) as a between-subjects factor, and trial type (CS + , CS−) as a within-subjects factor revealed a significant main effect of trial type, F(1, 26) = 36.9, p = .001, partial η2 = .587, indicating that both ALCs and NCs produced a greater number of CRs during reinforced (CS+) as compared with nonreinforced (CS−) trials during reversal learning. However, ANOVA also revealed a significant Group (ALC, NC) × Trial Type (CS + , CS−) interaction, F(1, 26) = 5.7, p = .025, partial η2 = .180, suggesting an impairment in reversal learning in ALCs. Means comparisons confirmed a significant difference between groups for percentage CRs produced during reinforced trials, F(1, 26) = 8.03, p = .01, d = .64. As can be seen in Figure 2, NCs produced significantly more CRs during CS + trials as compared with ALCs, indicating that NCs were able to acquire the new CS+ to a greater extent than the ALCs. However, percentage CRs produced during CS− trials did not differ between groups (F < 1, d = .25), indicating that the ALCs’ impairment was specifically related to a decreased ability to acquire the new CS + , rather than a deficit in inhibiting responses to new CS− (i.e., extinguishing the old CS + ; see Figures 2 and 3).
Further comparisons demonstrated that NCs produced a significantly greater percentage of CRs during CS+ trials (M = 67, SE = 4.8) as compared with CS− trials (M = 35, SE = 2.8) during reversal learning, F(1, 26) = 35.8, p = .001, d = 2.16, indicating that NCs were able to successfully reverse the previously learned discrimination (see Figure 2). This difference was also significant for the ALCs, F(1, 26) = 6.81, p = .049, d = .64. However, the unpaired t test examining the difference score in the percentage of CRs between CS+ trials (M = 52, SE = 7.4) minus CS− trials (M = 38, SE = 3.3; i.e., the reversal discrimination difference score) across the two groups was significant, t(26) = 2.39, p = .026, d = .90. This latter finding confirms that although the ALCs were able to demonstrate some ability to reverse the previously learned discrimination, their reversal learning was impaired as compared with the NCs. This is evident in Figure 3, when conditioning trials were collapsed across learning blocks.
Learning curves
During reversal learning, the control participants demonstrated an overall increase in the percentage of CRs to the new CS+ across the six learning blocks of reinforced trials, peaking at Block 6. Production of CRs during CS− trials in the NCs was initially high, as expected, given that this was the previously learned CS + . However, across blocks, the NCs produced fewer and fewer CRs during nonreinforced trials, indicating successful extinction of the old CS+ and reversal learning. Alternatively, the ALCs produced a relatively stable percentage of CRs to both trial types during reversal learning. ALCs demonstrated some increase in CR production to the new CS+ across blocks, although not of the same magnitude as the NCs. Furthermore, although relatively stable, ALCs’ percentage CRs during CS− trials actually increased over time. Block-by-block comparisons between groups revealed significant differences in production of CRs during reinforced trials at Block 5 (p = .02) and during nonreinforced trials at Block 6 (p = .03). Including block as a within-subjects factor did not change the overall pattern of the ANOVA.
To examine the rate of learning or acquisition, we performed linear regression analyses for the mean percentage of CRs for each group separately for the six blocks of acquisition trials during reinforced trials. The linear regression analysis was not significant for either the NC participants, F(1, 82) = 1.66, p = .20, R2 = .02, or the ALCs, F(1, 82) = .134, p = .72. R2 = .002, during reversal learning.
Response latency and amplitude
Unpaired t tests of CR latency during reinforced trials revealed that none of the measures differed significantly between groups (ps > .3) during reversal learning (see Table 2). Similarly, there were no differences between groups for CR and UR amplitude during reinforced trials (ps > .5; see Tables 2 and 3).
Neuropsychological Function, Drinking Severity, and EBCC Learning
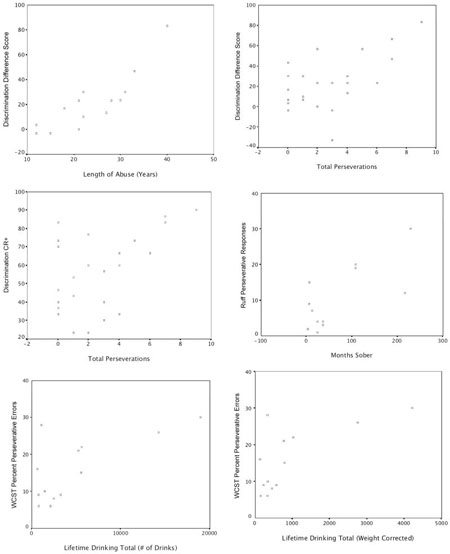
Correlational analysis of neuropsychological tests, drinking measures, and measures of EBCC are reported in Tables 4, 5, and 6. One measure of drinking severity—length of abuse—was found to significantly correlate with EBCC discrimination learning (see Table 4). To examine the amount of variance in EBCC that length of abuse explained, we calculated R2. Length of abuse accounted for nearly 79% of the variance in the EBCC discrimination score (see Table 4). Several tests of executive function were found to significantly correlate with measures assessing the severity of drinking in the alcoholic participants (see Table 5). These included total perseverations during the verbal fluency task, percent perseverative errors on the WCST, and perseverative errors on the Ruff Figural Fluency Test. There were some trends toward correlations between measures of memory function and severity of drinking, as well as measures of motor function and severity of drinking, although none were significant at the p ≤ .01 level (see Table 5).
Table 4.
Correlations Between a Measure of Alcohol Consumption and EBCC Learning Measures
| Drinking measure | EBCC learning measure | Pearson correlation | R2 | Significance |
|---|---|---|---|---|
| Length of abuse (years) | Discrimination mean % CR+ trials | .603 | .363 | .023 |
| Length of abuse (years) | Discrimination mean % CR− trials | −.632 | .399 | .015 |
| Length of abuse (years) | Discrimination score | .888 | .788 | .001* |
Note. EBCC = eyeblink classical conditioning paradigm; CR = conditioned response.
p ≤ .01.
Table 5.
Correlations Between Neuropsychological Tests and Alcohol Consumption
| Neuropsychological test | Drinking measure | Pearson correlation | R2 | Significance |
|---|---|---|---|---|
| Memory/medial temporal | ||||
| WMS–III | ||||
| Auditory recognition delayed index | LDH total volume | −.611 | .373 | .035 |
| LDH total weight corrected | −.647 | .419 | .023 | |
| Auditory recognition delayed total raw score | LDH total volume | −.593 | .352 | .042 |
| LDH total weight corrected | −.620 | .384 | .031 | |
| Auditory recognition delayed total scaled score | LDH total volume | −.611 | .373 | .035 |
| LDH total weight corrected | −.647 | .419 | .023 | |
| Visual reproduction raw score | SMAST score | .679 | .461 | .031 |
| Visual reproduction recognition raw score | SMAST score | .702 | .493 | .024 |
| Visual reproduction scaled score | SMAST score | .725 | .526 | .018 |
| Spatial Span | Average drinks/day | −.577 | .333 | .050 |
| Warrington Recognition Memory Test | SMAST score | .636 | .404 | .048 |
|
| ||||
| Executive/frontal | ||||
| Trails B errors | SMAST score | −.675 | .456 | .023 |
| Months sober | .609 | .371 | .027 | |
| Verbal fluency total perseverations | Length of abuse (years) | .839 | .704 | .001* |
| WCST | ||||
| Percent perseverative errors | LDH total volume | .682 | .465 | .010* |
| LDH total weight corrected | .685 | .469 | .010* | |
| Average drinks/day | .596 | .339 | .032 | |
| Ruff Figural Fluency perseverative errors | Months sober | .726 | .527 | .008* |
| WAIS–III similarities raw score | SMAST score | .679 | .461 | .031 |
|
| ||||
| Motor/cerebellar | ||||
| Finger Tapping | ||||
| Dominant hand | SMAST score | .677 | .458 | .032 |
| Nondominant hand | Length of abuse (years) | .657 | .432 | .020 |
| Ataxia measures | ||||
| Stand on one foot (left) with eyes open | Months sober | −.701 | .491 | .024 |
| Walk on line, heel-to-toe with eyes open | Length of abuse (years) | −.706 | .498 | .015 |
Note. WMS–III = Wechsler Memory Scale—Third Edition; LDH = Lifetime Drinking History; SMAST = Self-Administered Short Michigan Alcoholism Screening Test; WCST = Wisconsin Card Sorting Task; WAIS–III = Wechsler Adult Intelligence Scale—Third Edition.
p ≤ .01.
Table 6.
Correlations Between Neuropsychological Tests and Measures of EBCC Learning
| Neuropsychological test | EBCC learning measure | Pearson correlation | R2 | Significance |
|---|---|---|---|---|
| Memory/medial temporal | ||||
| WMS–III | ||||
| Logical Memory I raw score | Discrimination % CRs on CS− trials | .443 | .196 | .027 |
|
| ||||
| Executive/frontal | ||||
| Verbal fluency total perseverations | Discrimination score | .556 | .309 | .004* |
| Discrimination % CRs on CS+ trials | .511 | .261 | .009* | |
| Reversal score | .450 | .203 | .024 | |
| Category fluency (animals) | Reversal % CRs on CS− trials | .399 | .159 | .048 |
| WCST categories completed | Reversal % CRs on CS− trials | −.409 | .167 | .042 |
| WCST percent perseverative errors | Reversal score | −.407 | .166 | .043 |
| Stroop Interference Trial | Discrimination % CRs on CS− trials | −.442 | .195 | .039 |
| Stroop Interference Trial | Discrimination score | .458 | .210 | .032 |
|
| ||||
| Motor/cerebellar | ||||
| Finger Tapping | ||||
| Nondominant hand | Discrimination score | .478 | .228 | .018 |
| Nondominant hand | Discrimination % CRs on CS+ trials | .424 | .180 | .039 |
| Ataxia measure | ||||
| Sharpened Romberg, stand heel-to-toe with eyes open | Reversal % CRs on CS+ trials | .414 | .171 | .050 |
Note. EBCC = eyeblink classical conditioning paradigm; WMS–III = Wechsler Memory Scale—Third Edition; WCST = Wisconsin Card Sorting Task.
p ≤ .01.
One test of neuropsychological function was also found to significantly correlate with EBCC performance: total perseverations on the verbal fluency task (see Table 6). This measure was significantly correlated with discrimination learning (ps ≤ .01). To examine the amount of variance in EBCC that the various neuropsychological tests explained, we calculated R2 for each measure (see Table 6). Total perseverations on the verbal fluency task, the most significant predictor of EBCC learning, accounted for approximately 30% of the variability in the EBCC discrimination score and 26% of the variability in the percentage of CRs during reinforced discrimination learning trials. Several other tests of neuropsychological function within the domains of executive and motor function showed trends toward significant correlation with EBCC performance, including reversal learning (ps ≤ .05; see Table 6). Scatterplots are provided in the Appendix for the significant (ps ≤ .01) relationships observed in the correlational analysis of drinking severity, neuropsychological test performance, and EBCC.
Only two tests in the neuropsychological test battery were significantly different among groups (ps < .05): the Warrington Recognition Memory Test and percent perseverative responses on the WCST.
Discussion
The primary finding from this study is that in the complex task of reversing a previously learned delay discrimination, abstinent alcoholics were significantly impaired compared with a nonalcoholic control group. This finding is heightened by the fact that the two groups were matched (roughly) in their acquisition of the initial delay discrimination. The ability to reverse a learned discrimination has been linked by both animal and human studies to forebrain structures, in particular the hippocampal system (Berger & Orr, 1983; Carrillo et al., 1997) and prefrontal cortex (Chachich & Powell, 1998). The reversal impairment following prefrontal lesions and thalamic mediodorsal nucleus lesions in nonhuman animals involved slowed acquisition of reversal contingencies. Rabbits were able to acquire the reversed discrimination but more slowly than normal animals (Buchanan, 1991; D. A. Powell, personal communication, February 28, 2007). The selective impairment in reversal learning in abstinent alcoholics is therefore in line with the animal literature (Buchanan et al., 1997; Chachich & Powell, 1998). Given alcohol’s documented neurotoxic predisposition for cerebellar and frontal brain regions, the current study also lends support for the notion that a cerebellar–thalamic–prefrontal cortex module controls eyeblink associative learning during nonoptimal conditions or more complex tasks such as reversal learning (McLaughlin et al., 2002; Weiss & Disterhoft, 1996).
Interestingly, the nature of the reversal learning impairment observed in abstinent alcoholics appears to be different from that associated with hippocampal system damage. Recall that the abstinent alcoholics’ reversal deficit was due to an inability to produce a normal percentage of CRs during CS+ trials rather than a deficit in inhibiting responses to new CS− (i.e., extinguishing the old CS+), as seen with hippocampal damage. Alcoholics appear to have a selective impairment in the ability to flexibly manipulate previously learned associations. In particular, they are impaired in producing a positive response to a previously acquired neutral or inhibited response. We therefore conclude that frontal system damage, as seen in alcoholics, disrupts the ability to differentially respond at a high rate to a stimulus previously responded to at a low rate.
Proactive interference occurs when previously learned information reduces the ability to acquire new, related information. Resolution of interference, or the ability to learn new information without interference from past, related information, has been linked to basal forebrain structures, including the medial septal nucleus and diagonal band (De Rosa & Hasselmo, 2000; De Rosa, Hasselmo, & Baxter, 2001). These structures are rich in cholinergic pathways connecting forebrain and brainstem areas, as well as hippocampal and septal regions (Gaykema, Luiten, Nyakas, & Traber, 1990; Rye, Wainer, Mesulam, Mufson, & Saper, 1984). Alcoholism has been demonstrated to exert neurotoxic effects on the cholinergic basal forebrain system, including muscarinic receptors in its target structures (Arendt, 1994; Arendt et al., 1988, 1989; Freund & Ballinger, 1989; Nordberg, Larsson, Perdahl, & Winblad, 1982). In healthy individuals, these cholinergic interactions have been hypothesized to optimize cortical information processing (Baxter & Chiba, 1999). Specifically, they may mediate the detection or selection of stimuli and subsequent behavioral responses to such stimuli for extended processing, allowing for appropriate allocation of processing resources and enhanced flexibility to make the appropriate choices in their presence (Everitt & Robbins, 1997; Sarter & Bruno, 1997). The cholinergic basal fore-brain system may have a critical neuromodulatory contribution to associative learning that allows for flexibility of learned responses (De Rosa & Sullivan, 2003). De Rosa et al. (2001) showed that acetylcholine acts on the memory system in rats by minimizing interference from previously learned information, allowing more efficiency in processing new and old memory traces simultaneously.
Disruption of the cholinergic basal forebrain system secondary to chronic alcoholism may explain the deficit in the ability to flexibly manipulate previously learned stimuli that we observed. In fact, a recent functional neuroimaging study conducted by De Rosa, Desmond, Anderson, Pfefferbaum, and Sullivan (2004) demonstrated that, whereas neurologic ally intact individuals recruited the basal forebrain network during a learning task designed to elicit proactive interference resolution, alcoholic participants did not activate basal forebrain but instead relied on anterior cortical sites to complete the task. This notion is also supported by a recent study by Sullivan et al. (2005), who concluded that basal forebrain structure (medial septal/diagonal band) compromise is consistent with a cholinergic mechanism for working memory deficits observed in alcoholics.
The cholinergic and frontal system dysfunction related to chronic alcoholism may have behavioral consequences related to alcoholic relapse. Cholinergic and frontal system dysfunction may further perpetuate alcoholics’ inability to maintain abstinence through an increase in proactive interference. The previously learned behavioral patterns, such as drinking triggers and maladaptive coping mechanisms, are so pervasive that they interfere with the individuals’ ability to flexibly learn new patterns of behavior. This idea is similar to Hyman’s hypothesis (Hyman, Malenka, & Nestler, 2006) that addiction represents “a pathological usurpation of neural processes that normally serve reward-related learning” (p. 565). Hyman’s work focuses on forebrain circuits involving ventral and dorsal striatum and prefrontal cortex receiving input from midbrain dopaminergic neurons. This system may be different from that hypothesized to subserve proactive interference (e.g., cholinergic). However, the idea that long-lasting addiction and relapse are a result of persistent associative memories or associative learned responses formed in circuits involving prefrontal cortex and dorsal striatum interfering with the ability to learn new, more adaptive associations is quite compatible and may help provide a framework of understanding what happens in alcoholics’ resistance to treatment.
A secondary finding from this study is the ability of the abstinent alcoholics to acquire, at least to some extent, the initial delay discrimination when specific learning criteria were applied. This finding is not surprising given subject inclusion criteria requiring a minimum difference score of 10 CRs during discrimination trials (elevating the performance of the abstinent alcoholics). If the learning criteria had not been applied, we anticipated impaired learning during the initial discrimination among alcoholics as compared with nonalcoholic controls. Our laboratory has previously reported that abstinent alcoholics with assumed cerebellar atrophy were impaired when challenged with a temporal discrimination EBCC task (McGlinchey-Berroth, Fortier, Cermak, & Disterhoft, 2002). The cerebellar atrophy associated with chronic alcoholism likely leads to a deficit in simple discrimination learning, as well as temporal discrimination learning. However, this cannot be directly addressed by the data in this study because of the subject inclusion criteria.
In selecting the neuropsychological test battery for this study, we chose the domains of memory, executive function, and motor function on the basis of empirically driven hypotheses regarding neurologic sequela of alcoholism. We realized that significant differences among groups were unlikely given the small sample size in the current study, and that our findings in this regard would likely be conservative. Nevertheless, two neuropsychological tests differed among the abstinent alcoholics and normal control participants: the Warrington Recognition Memory Test, and percentage perseverative responses on the WCST. The observed difference in Warrington Recognition Memory score may reflect a deficit in visual processing in the abstinent alcoholic participants. Sullivan and colleagues (Sullivan, Fama, Rosenbloom, & Pfefferbaum, 2002; Sullivan, Rosenbloom, & Pfefferbaum, 2000) reported deficits in the domains of executive function (male alcoholics only), verbal and nonverbal working memory (female only), visuospatial function (male and female), and motor function (male and female) in abstinent alcoholics in her examination of neuropsychological function in a large group of male and female alcoholics. Sullivan and colleagues also documented preserved declarative memory function (Sullivan, Rosenbloom, & Pfefferbaum, 2002; Sullivan, Rosenbloom, Lim, & Pfefferbaum, 2000), as well as recovery of short-term memory function with maintained sobriety (Rosenbloom, Pfefferbaum, & Sullivan, 2004) in alcoholics. Therefore, observed deficits in facial recognition may be specific to visuospatial processing deficits rather than deficits in visual memory and are consistent with other recent findings of visuospatial processing deficits in alcoholics (Fein, Torres, Price, & Di Sclafani, 2006; Pfefferbaum, Adalsteinsson, & Sullivan, 2006). The observed increase in perseverative responding on the WCST in abstinent alcoholics reflects an executive function deficit. Specifically, alcoholics demonstrated impaired ability to shift cognitive set during an abstract problem-solving task. The current study did not differentiate performance on the basis of gender because the small sample size did not allow for such a comparison.
The relationship between neuropsychological test performance and measures of EBCC learning (see Table 6) supported the hypothesized neural circuit underlying the formation of new memory traces in classical conditioning of associative relationships. Specifically, we predicted that the cerebellar–thalamic–prefrontal cortex module, as defined by Weiss and Disterhoft (1996), supported eyeblink associative learning in the discrimination reversal task. Neuropsychological test performance in the domain of executive (frontal) function was most strongly related to EBCC learning performance. As anticipated, memory function as assessed by neuropsychological test performance was not as strongly correlated with EBCC measures. Perseveration during the verbal fluency task was related to discrimination EBCC learning measures, with trends observed between other executive function measures and reversal EBCC learning. Given the known cerebellar contribution to EBCC, it is not surprising that some motor function measures were correlated with both discrimination and reversal learning. These relationships were not as a strong as those observed for executive function measures. Although largely exploratory given the small sample size, these findings indicate that frontocerebellar function as assessed by neuropsychological tests is more related to discrimination reversal learning than tests supported by medial temporal systems.
The relation between neuropsychological test performance and drinking severity was supported by correlational analyses. Perseverative responding in general (verbal fluency task, WCST, and Ruff Figural Fluency) appeared to be particularly sensitive to measures of drinking severity. Correlations between motor measures and drinking measures were expected given the documented effect of alcohol on the cerebellum. However, these relationships were not as strong as those observed between executive function and drinking severity in this small group of alcoholics.
The relation between EBCC discrimination learning and drinking severity measures was also supported by correlational analyses. However, no relationship between EBCC reversal learning and drinking severity was observed. One of the limitations of this correlational analysis involving drinking severity is that we collected drinking measures on alcoholic participants only and, therefore, the distribution of the scores was truncated. Our future studies will include drinking severity indices for normal participants as well as abstinent alcoholics.
It is important to note that our abstinent alcoholic group may have included individuals with less significant drinking histories than often studied (e.g., alcoholics with recent hospitalized detoxification). The criterion of ≥ 21 drinks per week and a minimum duration of 5 years was used to define our alcoholic group for a number of reasons. Our primary objective was to include a wide range of alcoholic profiles representative of alcoholism in a community setting. We attempted to avoid oversampling a more severe subgroup of alcoholics such as those alcoholic individuals in clinical treatment settings (Fein et al., 2002). Significant differences in EBCC learning were observed in this group, demonstrating that community-dwelling abstinent alcoholics have deficits in complex, nonoptimal classical conditioning learning paradigms.
The current study examined simple discrimination and reversal learning in abstinent alcoholics with presumed cerebellar–frontal system damage. The study revealed a significant impairment in reversal learning in abstinent alcoholics that was attributable to their inability to acquire the new CS+ to normal levels. The nature of this deficit is different from that following hippocampal system damage in which the deficit lies in the inability to inhibit responses to new CS− or extinguish the old CS +. In particular, alcoholics demonstrate proactive interference. They are impaired in producing a positive response to a previously acquired inhibitory response. This deficit can also be interpreted as an impairment in resolution of interference, a phenomenon that inhibits adaptive learning of subsequent information. Alcoholic addiction may result from the overlearning of pathological, persistent associative memories or associative learned responses that interfere with the ability to learn new, more adaptive associations. As a result, alcoholic individuals are prone to relapse on the basis of their patterns of learning.
Acknowledgments
This research was supported by National Institutes of Health (NIH)/National Institute on Alcohol Abuse and Alcoholism Grant 14205, NIH AG08796, NIH MH53673, and Veterans Affairs Merit Review Awards to Catherine Brawn Fortier and Regina McGlinchey. We thank the Harvard Cooperative Program on Aging for its help in recruiting control participants. We also appreciate the advice of Marlene Oscar-Berman in defining the inclusionary criteria for alcoholics in this study.
Appendix
Contributor Information
Catherine Brawn Fortier, Veterans Affairs Boston Healthcare System and Harvard Medical School.
Ginette LaFleche, Boston University School of Medicine and Veterans Affairs Boston Healthcare System.
John F. Disterhoft, Northwestern University, Feinberg School of Medicine
Elizabeth M. Steffen, Veterans Affairs Boston Healthcare System
Jonathan R. Venne, Veterans Affairs Boston Healthcare System
Regina E. McGlinchey, Veterans Affairs Boston Healthcare System and Harvard Medical School
References
- Akase E, Thompson LT, Disterhoft JF. A system for quantitative analysis of associative learning. Journal of Neuroscience Methods. 1994;54:119–130. doi: 10.1016/0165-0270(94)90166-x. [DOI] [PubMed] [Google Scholar]
- American Psychiatric Association. Diagnostic and statistical manual of mental disorders. 4. Washington, DC: Author; 1994. [Google Scholar]
- Arendt T. Impairment in memory function and neurodegenerative changes in the cholinergic basal forebrain system induced by chronic intake of ethanol. Journal of Neural Transmission. 1994;44(Suppl):173–187. doi: 10.1007/978-3-7091-9350-1_13. [DOI] [PubMed] [Google Scholar]
- Arendt T, Allen Y, Marchbanks RM, Schugens MM, Sinden J, Lantos PL, et al. Cholinergic system and memory in the rat: Effects of chronic ethanol, embryonic basal forebrain brain transplants and excitotoxic lesions of cholinergic basal forebrain projection system. Neuroscience. 1989;33:435–462. doi: 10.1016/0306-4522(89)90397-7. [DOI] [PubMed] [Google Scholar]
- Arendt T, Allen Y, Sinden J, Schugens MM, Marchbanks RM, Lantos PL, et al. Cholinergic-rich brain transplants reverse alcohol-induced memory deficits. Nature. 1988 March;332:448–450. doi: 10.1038/332448a0. [DOI] [PubMed] [Google Scholar]
- Baxter MG, Chiba AA. Cognitive functions of the basal forebrain. Current Opinion in Neurobiology. 1999;9:178–183. doi: 10.1016/s0959-4388(99)80024-5. [DOI] [PubMed] [Google Scholar]
- Benton AL, deS Hamsher K. Multilingual Aphasia Examination. Iowa City: University of Iowa; 1978. [Google Scholar]
- Berger T, Orr WB. Hippocampectomy selectively disrupts discrimination reversal conditioning of the rabbit nictitating membrane response. Behavioural Brain Research. 1983;8:49–68. doi: 10.1016/0166-4328(83)90171-7. [DOI] [PubMed] [Google Scholar]
- Buchanan SL. Differential and reversal Pavlovian conditioning in rabbits with mediodorsal thalamic lesions: Assessment of heart rate and eyeblink responses. Experimental Brain Research. 1991;86:174–181. doi: 10.1007/BF00231051. [DOI] [PubMed] [Google Scholar]
- Buchanan SL, Penney J, Tebbutt D, Powell BJ. Lesions of the mediodorsal nucleus of the thalamus and classical eyeblink conditioning under less-than-optimal stimulus conditions: Role of partial reinforcement and interstimulus interval. Behavioral Neuroscience. 1997;111:1075–1085. doi: 10.1037//0735-7044.111.5.1075. [DOI] [PubMed] [Google Scholar]
- Buchanan SL, Powell DA. Cingulate cortex: Its role in Pavlovian conditioning. Journal of Comparative and Physiological Psychology. 1982;96:755–777. doi: 10.1037/h0077925. [DOI] [PubMed] [Google Scholar]
- Butters N, Cermak LS. Alcoholic Korsakoff’s Syndrome: An information processing approach to amnesia. New York: Academic Press; 1980. [Google Scholar]
- Carrillo MC, Hopkins RO, Gabrieli JDE, Kesner RP, McGlinchey-Berroth R, Disterhoft JF. Discrimination and discrimination reversal in medial temporal lobe amnesics. Society for Neuroscience Abstracts. 1997;23:208. [Google Scholar]
- Chachich M, Powell DA. Both medial prefrontal and amygdala central nucleus lesions abolish heart rate classical conditioning, but only prefrontal lesions impair reversal of eyeblink differential conditioning. Neuroscience Letters. 1998;257:151–154. doi: 10.1016/s0304-3940(98)00832-5. [DOI] [PubMed] [Google Scholar]
- Corporation TP. WAIS–III/WMS–III technical manual. San Antonio, TX: Harcourt Assessment Company; 1997. [Google Scholar]
- Dao-Castellana MH, Samson Y, Legault F, Martinot JL, Aubin HJ, Crouzel C, et al. Frontal dysfunction in neurologically normal chronic alcoholic subjects: Metabolic and neuropsychological findings. Psychological Medicine. 1998;28:1039–1048. doi: 10.1017/s0033291798006849. [DOI] [PubMed] [Google Scholar]
- De Rosa E, Desmond JE, Anderson AK, Pfefferbaum A, Sullivan EV. The human basal forebrain integrates the old and the new. Neuron. 2004;41:825–837. doi: 10.1016/s0896-6273(04)00080-7. [DOI] [PubMed] [Google Scholar]
- De Rosa E, Hasselmo ME. Muscarinic cholinergic neuro-modulation reduces proactive interference between stored odor memories during associative learning in rats. Behavioral Neuroscience. 2000;114:32–41. [PubMed] [Google Scholar]
- De Rosa E, Hasselmo ME, Baxter MG. Contribution of the cholinergic basal forebrain to proactive interference from stored odor memories during associative learning in rats. Behavioral Neuroscience. 2001;115:314–327. [PubMed] [Google Scholar]
- De Rosa E, Sullivan EV. Enhanced release from proactive interference in nonamnesic alcoholic individuals: Implications for impaired associative binding. Neuropsychology. 2003;17:469–481. doi: 10.1037/0894-4105.17.3.469. [DOI] [PubMed] [Google Scholar]
- Everitt BJ, Robbins TW. Central cholinergic systems and cognition. Annual Review of Psychology. 1997;48:649–684. doi: 10.1146/annurev.psych.48.1.649. [DOI] [PubMed] [Google Scholar]
- Fein G, Di Sclafani V, Cardenas VA, Goldmann H, Tolou-Shams M, Meyerhoff DJ. Cortical gray matter loss in treatment-naive alcohol dependent individuals. Alcoholism: Clinical and Experimental Research. 2002;26:558–564. [PMC free article] [PubMed] [Google Scholar]
- Fein G, Torres J, Rice LJ, Di Sclafani V. Cognitive performance in long-term abstinent alcoholic individuals. Alcoholism, Clinical and Experimental Research. 2006;30:1538–1544. doi: 10.1111/j.1530-0277.2006.00185.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feinberg TE, Haber LD, Stacy CB. Ipsilateral extinction in the hemineglect syndrome. Archives of Neurology. 1990;47:802–804. doi: 10.1001/archneur.1990.00530070100017. [DOI] [PubMed] [Google Scholar]
- Fortier C, Disterhoft JF, Capozzi S, Kilduff P, Cronin-Golomb A, McGlinchey R. Conditional discrimination learning in patients with bilateral medial temporal lobe amnesia. Behavioral Neuroscience. 2003;117:1181–1195. doi: 10.1037/0735-7044.117.6.1181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freund G, Ballinger WE., Jr Loss of muscarinic and benzodiazepine neuroreceptors from hippocampus of alcohol abusers. Alcohol. 1989;6:23–31. doi: 10.1016/0741-8329(89)90069-4. [DOI] [PubMed] [Google Scholar]
- Gaykema RP, Luiten PG, Nyakas C, Traber J. Cortical projection patterns of the medial septum-diagonal band complex. Journal of Comparative Neurology. 1990;293:103–124. doi: 10.1002/cne.902930109. [DOI] [PubMed] [Google Scholar]
- Gormezano I. Classical conditioning. In: Sidowski JB, editor. Experimental methods and instrumentation in psychology. New York: McGraw-Hill; 1966. pp. 385–420. [Google Scholar]
- Harper C, Kril JJ. Patterns of neuronal loss in the cerebral cortex in chronic alcoholic patients. Journal of Neurological Science. 1989;92:81–89. doi: 10.1016/0022-510x(89)90177-9. [DOI] [PubMed] [Google Scholar]
- Harper C, Kril J, Daly J. Are we drinking our neurons away? British Medical Journal. 1987;294:534–536. doi: 10.1136/bmj.294.6571.534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heaton RK. Wisconsin Card Sorting Test Manual. Odessa, FL: Psychological Assessment Resources; 1981. [Google Scholar]
- Hildebrandt H, Brokate B, Eling P, Lanz M. Response shifting and inhibition, but not working memory, are impaired after long-term heavy alcohol consumption. Neuropsychology. 2004;18:203–211. doi: 10.1037/0894-4105.18.2.203. [DOI] [PubMed] [Google Scholar]
- Hyman SE, Malenka RC, Nestler EJ. Neural mechanisms of addiction: The role of reward-related learning and memory. Annual Review of Neuroscience. 2006;29:565–598. doi: 10.1146/annurev.neuro.29.051605.113009. [DOI] [PubMed] [Google Scholar]
- Jernigan TL, Butters N, DiTraglia G, Schafer K, Smith T, Irwin M, et al. Reduced cerebral grey matter observed in alcoholics using magnetic resonance imaging. Alcoholism: Clinical and Experimental Research. 1991;15:418–427. doi: 10.1111/j.1530-0277.1991.tb00540.x. [DOI] [PubMed] [Google Scholar]
- Knultinen MG, Power JM, Preston AR, Disterhoft JF. Awareness in classical differential eyeblink conditioning in young and aging humans. Behavioral Neuroscience. 2001;115:747–757. doi: 10.1037//0735-7044.115.4.747. [DOI] [PubMed] [Google Scholar]
- Kril JJ, Halliday GM, Svoboda MD, Cartwright H. The cerebral cortex is damaged in chronic alcoholics. Neuroscience. 1997;79:983–998. doi: 10.1016/s0306-4522(97)00083-3. [DOI] [PubMed] [Google Scholar]
- McGlinchey RE, Fortier CB, Capozzi SM, Disterhoft JF. Trace eyeblink conditioning in abstinent alcoholic individuals: Effects of complex task demands and prior conditioning. Neuropsychology. 2005;19:159–170. doi: 10.1037/0894-4105.19.2.159. [DOI] [PubMed] [Google Scholar]
- McGlinchey-Berroth R, Carrillo MC, Gabrieli JDE, Brawn CM, Disterhoft JF. Impaired trace eyeblink conditioning in bilateral medial temporal lobe amnesia. Behavioral Neuroscience. 1997;111:873–882. doi: 10.1037//0735-7044.111.5.873. [DOI] [PubMed] [Google Scholar]
- McGlinchey-Berroth R, Cermak LS, Carrillo M, Armfield S, Gabrieli JDE, Disterhoft JF. Impaired delay eyeblink conditioning in amnesic Korsakoff’s patients and recovered alcoholics. Alcoholism: Clinical and Experimental Research. 1995;19:1127–1132. doi: 10.1111/j.1530-0277.1995.tb01590.x. [DOI] [PubMed] [Google Scholar]
- McGlinchey-Berroth R, Fortier C, Cermak LS, Disterhoft JF. Temporal discrimination learning in chronic abstinent alcoholics. Alcoholism: Clinical and Experimental Research. 2002;26:804–811. [PubMed] [Google Scholar]
- McLaughlin J, Powell DA, White JD. Characterization of the neuronal changes in the medial prefrontal cortex during jaw movement and eyeblink Pavlovian conditioning in the rabbit. Behavioural Brain Research. 2002;132:117–133. doi: 10.1016/s0166-4328(01)00410-7. [DOI] [PubMed] [Google Scholar]
- Moselhy HF, Georgiou G, Kahn A. Frontal lobe changes in alcoholism: A review of the literature. Alcohol and Alcoholism. 2001;36:357–368. doi: 10.1093/alcalc/36.5.357. [DOI] [PubMed] [Google Scholar]
- Nixon SJ. Application of theoretical models to the study of alcohol-induced brain damage. In: Hunt WA, Nixon SJ, editors. Alcohol-induced brain damage. Washington, DC: National Institute on Alcohol Abuse and Alcoholism; 1993. pp. 213–228. [Google Scholar]
- Nordberg A, Larsson C, Perdahl E, Winblad B. Cholinergic activity in hippocampus in chronic alcoholism. Drug and Alcohol Dependence. 1982;10:333–344. doi: 10.1016/0376-8716(82)90035-7. [DOI] [PubMed] [Google Scholar]
- Oscar-Berman M. Neuropsychological vulnerabilities in chronic alcoholism. In: Nororha A, Eckardt M, Warren K, editors. Review of NIAAA's neuroscience and behavioral research portfolio (Research Monograph No. 34, pp. 437–471) Bethesda, MD: National Institute on Alcohol Abuse and Alcoholism; 2000. [Google Scholar]
- Oscar-Berman M, Hutner N. Frontal lobe changes after chronic alcohol ingestion. In: Hunt WA, Nixon SJ, editors. Alcohol-induced brain damage. Washington, DC: National Institute on Alcohol Abuse and Alcoholism; 1993. pp. 121–156. [Google Scholar]
- Oscar-Berman M, Kirkley SM, Gansler DA, Couture A. Comparisons of Korsakoff and non-Korsakoff alcoholics on neuropsychological tests of prefrontal brain functioning. Alcoholism: Clinical and Experimental Research. 2004;28:667–675. doi: 10.1097/01.alc.0000122761.09179.b9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parsons OA, Leber WR. The relationship between cognitive dysfunction and brain damage in alcoholics: Causal, interactive or epiphenomenal? Alcoholism: Clinical and Experimental Research. 1981;5:326–343. doi: 10.1111/j.1530-0277.1981.tb04906.x. [DOI] [PubMed] [Google Scholar]
- Pfefferbaum A, Adalsteinsson E, Sullivan EV. Supratentorial profile of white matter microstructural integrity in recovering alcoholic men and women. Biological Psychiatry. 2006;59:364–372. doi: 10.1016/j.biopsych.2005.06.025. [DOI] [PubMed] [Google Scholar]
- Pfefferbaum A, Sullivan EV. Microstructural but not macrostructural disruption of white matter in women with chronic alcoholism. Neuroimage. 2002;15:708–718. doi: 10.1006/nimg.2001.1018. [DOI] [PubMed] [Google Scholar]
- Pfefferbaum A, Sullivan EV, Hedehus M, Adalsteinsson E, Lim KO, Moselesy M. In vivo detection and functional correlates of white matter microstructural disruption in chronic alcoholism. Alcoholism: Clinical and Experimental Research. 2000;24:1214–1221. [PubMed] [Google Scholar]
- Pishkin V, Lovallo WR, Bourne LE. Chronic alcoholism in males: Cognitive deficit as a function of age of onset, age and duration. Alcoholism: Clinical and Experimental Research. 1985;9:400–406. doi: 10.1111/j.1530-0277.1985.tb05571.x. [DOI] [PubMed] [Google Scholar]
- Reitan RM. Manual for administration of neuropsycological test batteries for adults and children. 1969 Private publication. [Google Scholar]
- Reitan RM. Trail Making Test. Göttingen, Germany: Hogrefe; 1979. [Google Scholar]
- Robins LN, Marcus L, Reich W, Cunningham R, Gallagher T. Diagnostic Interview Schedule, Version TV. St. Louis, MO: Department of Psychiatry, Washington University School of Medicine; 1996. [Google Scholar]
- Rosenbloom MJ, Pfefferbaum A, Sullivan EV. Recovery of short-term memory and psychomotor speed but not postural stability with long-term sobriety in alcoholic women. Neuropsychology. 2004;18:589–597. doi: 10.1037/0894-4105.18.3.589. [DOI] [PubMed] [Google Scholar]
- Ruff R. Ruff Figural Fluency Test: Professional manual. Odessa, FL: Psychological Assessment Resources; 1996. [Google Scholar]
- Rye DB, Wainer BH, Mesulam MM, Mufson EJ, Saper CB. Cortical projections arising from the basal forebrain: A study of cholinergic and noncholinergic components employing combined retrograde tracing and immunohistochemical localization of choline acetyl-transferase. Neuroscience. 1984;13:627–643. doi: 10.1016/0306-4522(84)90083-6. [DOI] [PubMed] [Google Scholar]
- Sarter M, Bruno JP. Cognitive functions of cortical acetyl-choline: Toward a unifying hypothesis. Brain Research Reviews. 1997;23:28–46. doi: 10.1016/s0165-0173(96)00009-4. [DOI] [PubMed] [Google Scholar]
- Selzer ML, Vinokur A, van Rooijen L. A self-administered Short Michigan Alcoholism Screening Test (SMAST) Journal of Studies on Alcohol. 1975;36:117–126. doi: 10.15288/jsa.1975.36.117. [DOI] [PubMed] [Google Scholar]
- Skinner HA, Sheu WJ. Reliability of alcohol use indices: The Lifetime Drinking History and the MAST. Journal of Studies on Alcohol. 1982;43:1157–1170. doi: 10.15288/jsa.1982.43.1157. [DOI] [PubMed] [Google Scholar]
- Solomon PR, Pomerleau D, Bennett L, James J, Morse DL. Acquisition of the classically conditioned eyeblink response in humans over the life span. Psychology and Aging. 1989;4:34–41. doi: 10.1037//0882-7974.4.1.34. [DOI] [PubMed] [Google Scholar]
- Spreen O, Strauss E. A compendium of neuropsychological tests. New York: Oxford University Press; 1998. [Google Scholar]
- Sullivan EV, Deshmukh A, De Rosa E, Rosenbloom MJ, Pfefferbaum A. Striatal and forebrain nuclei volumes: Contribution to motor function and working memory deficits in alcoholism. Biological Psychiatry. 2005;57:768–776. doi: 10.1016/j.biopsych.2004.12.012. [DOI] [PubMed] [Google Scholar]
- Sullivan EV, Fama R, Rosenbloom MJ, Pfefferbaum A. A profile of neuropsychological deficits in alcoholic women. Neuropsychology. 2002;16:74–83. doi: 10.1037//0894-4105.16.1.74. [DOI] [PubMed] [Google Scholar]
- Sullivan EV, Harding AJ, Pentney R, Dlugos C, Martin PR, Parks MH, et al. Disruption of frontocerebellar circuitry and function in alcoholism. Alcoholism: Clinical and Experimental Research. 2003;27:301–309. doi: 10.1097/01.ALC.0000052584.05305.98. [DOI] [PubMed] [Google Scholar]
- Sullivan EV, Pfefferbaum A. Neurocircuitry in alcoholism: A substrate of disruption and repair. Psychopharmacology (Berl) 2005;180:583–594. doi: 10.1007/s00213-005-2267-6. [DOI] [PubMed] [Google Scholar]
- Sullivan EV, Rosenbloom MJ, Lim KO, Pfefferbaum A. Longitudinal changes in cognition, gait, and balance in abstinent and relapsed alcoholic men: Relationships to changes in brain structure. Neuropsychology. 2000;14:178–188. [PubMed] [Google Scholar]
- Sullivan EV, Rosenbloom MJ, Pfefferbaum A. Pattern of motor and cognitive deficits in detoxified alcoholic men. Alcoholism: Clinical and Experimental Research. 2000;24:611–621. [PubMed] [Google Scholar]
- Thompson LT, Moyer JR, Akase E, Disterhoft JF. A system for quantitative analysis of associative learning: Hardware interfaces with cross-species applications. Journal of Neuroscience Methods. 1994;54:109–117. doi: 10.1016/0165-0270(94)90165-1. [DOI] [PubMed] [Google Scholar]
- Thompson RF. The neurobiology of learning and memory. Science. 1986 August 29;233:941–947. doi: 10.1126/science.3738519. [DOI] [PubMed] [Google Scholar]
- Tiffen J. Purdue Pegboard examiner’s manual. Rosemont, IL: London House; 1968. [Google Scholar]
- Trenerry MR, Crosson B, DeBoe J, Leber WR. The Stroop Neuropsychological Screening Test. Odessa, FL: Psychological Assessment Resources; 1989. [Google Scholar]
- Warrington EK. Recognition Memory Test: Manual. Berkshire, England: NFER-Nelson; 1984. [Google Scholar]
- Weiss C, Disterhoft JF. Eyeblink conditioning, motor control, and the analysis of limbic–cerebellar connections. Behavioral and Brain Sciences. 1996;19:479–481. [Google Scholar]