Abstract
The arrangement of molecules at the interface between T cells and antigen presenting cells (APC) is known as the immunological synapse (IS). We conducted experiments with supported planar bilayers and transfected fibroblast APC to examine the IS formed by polarized Th1 and Th2 cells. Th1 cells formed typical “bull’s-eye” IS with a ring of adhesion molecules surrounding MHC/TCR interactions at all antigen concentrations tested, while Th2 cells formed multifocal IS at high concentrations of antigen. At low antigen concentrations, the majority of Th2 cells formed IS with a compact, central accumulation of MHC/TCR, but ICAM-1 was not excluded from the center of the IS. Additionally, CD45 was excluded from the center of the interface between Th1 cells and APC, while CD45 was found at the center of the multifocal IS formed by Th2 cells. Finally, phosphorylated signaling molecules colocalized with MHC/TCR to a greater extent in Th2 IS. Together, our results indicate that the IS formed by Th1 and Th2 cells are distinct in structure, with Th2 cells failing to form bull’s-eye IS.
Keywords: Th1/Th2 Cells, T Cell Receptors, Adhesion Molecules
Introduction
Immunological synapses (IS) are stable cell-cell junctions formed between antigen presenting cells (APC) and T cells during antigen recognition. Mature IS are characterized by a ring of ICAM-1/LFA-1 interactions, known as the peripheral supramolecular activation cluster (pSMAC), surrounding a central SMAC (cSMAC) of peptide MHC/TCR (1–4). Maintenance of radial symmetry is important for IS stability (5). A number of other molecules, including CD28 and PKC-θ are known to concentrate within the cSMAC (1,6), while CD45 (7, 8) and some phosphorylated signaling proteins, including Lck and Zap-70 (7,9,10), reside mainly outside of the cSMAC of the mature IS.
The IS has been implicated in a number of functions, yet these have been difficult to prove due to the complexity of manipulating this phenomenon specifically. Although IS formation correlates with activation, the cSMAC is not required for the initiation of TCR signaling, since signaling is initiated before the cSMAC is formed (9). Experiments with T cells from CD2AP−/− mice, which are incapable of forming mature IS, indicated that the function of the cSMAC may be to enhance signaling while at the same time promoting TCR downmodulation (10). Additionally, it has been shown that the lipid lysobisphosphatidic acid, which is known to exist at sites where membrane proteins are being sorted for degradation, accumulates in the cSMAC (11). The ability to signal without forming a cSMAC is consistent with recent data showing that TCR signaling is initiated in small TCR microclusters that form rapidly after contact and sustain signaling through continual formation in the periphery of the IS (11–13).
Another possible function of the IS is the delivery of effector function. IS formed by CD8+ T cells have separate domains within the cSMAC containing signaling molecules and lytic granules (14). These data suggested that the peripheral ring of adhesion molecules functions to prevent lytic granules from harming bystander cells (15, 16). However, it has been shown that CD8+ cells can kill sensitive targets without forming mature IS (17–19). Recently, it has been determined that cytokines like IL-2, IFN-γ and IL-10 are directly secreted into the IS, whereas chemokines, TNF and IL-4 are directed away from the IS (20). Thus, both Th1 and Th2 cytokines can be released into the IS or away from the IS.
Upon antigen recognition, CD4+ T cells proliferate and differentiate into effector cells. Depending upon the strength of stimulation and cytokines present at the time of stimulation, CD4+ cells can differentiate into either Th1 or Th2 cells (21–23). Th1 cells secrete IFN-γ and lymphotoxin, which serve to activate macrophages and induce inflammation by recruiting other leukocytes. Th1 cells also express FasL and can kill target cells. Th2 cells secrete IL-4 and promote allergic reactions in epithelial tissues. A recent study has shown that Th2 cells, but not Th1 cells, fail to cluster TCR at the cell-cell interface when forming conjugates with primary B cells due to increased expression of CTLA-4 (24). Although it has been shown that co-clustering of TCR with CD4 and lipid rafts is much more common in Th1 than Th2 cells (25), the organization of IS formed by polarized Th1 and Th2 cells has not been thoroughly studied.
In this study, we used a supported planar bilayer system and transfected fibroblast APC to study IS formed by polarized Th1 and Th2 cells. We report that Th1 cells form synapses with a compact accumulation of MHC/TCR surrounded by a ring of adhesion molecules. Unlike Th1 cells, Th2 cells predominantly form multifocal synapses at high antigen concentrations with multiple small accumulations of MHC/TCR with ICAM-1 interspersed throughout the interface. At low concentrations of antigen, the majority of Th2 cells form IS with a central, compact accumulation of MHC/TCR, but ICAM-1 is not excluded from the cSMAC, to form a ring structure, as in Th1 cells. We propose that the morphological differences in IS between Th1 cells and Th2 cells, specifically the formation of a “gasket” structure in Th1 cells, correlates with differences in the delivery of effector functions by these distinct types of cells.
Materials and Methods
Animals
Heterozygous AD10 TCR transgenic mice on a B10.BR background, specific for pigeon cytochrome c 88–104 and reactive against moth cytochrome c 88–103 (MCC), were provided by S. Hedrick (University of California at San Diego, La Jolla, CA) by way of P. Marrack (National Jewish Medical Center, Denver, CO). Mice were housed in specific-pathogen free conditions at Oregon Health & Science University according to institutional standards.
Antibodies
The antibodies used for immunofluorescence were as follows: anti-LFA-1 (I21/7; Southern Biotech, Birmingham, AL), anti-CD45 (30-F11; eBioscience, San Diego, CA), biotinylated anti-phosphotyrosine (4G10; Upstate, Charlottesville, VA), Texas Red-conjugated goat anti-rat IgG, Cy-5-conjugated goat anti-rat IgG and streptavidin-conjugated Cy5 (Jackson Immunoresearch, West Grove, PA).
APCs
MCC:GFP fibroblasts expressing B7-1, ICAM-1, wild type I-Ek α-chain and β-chain along with GFP-labeled I-Ek β-chain covalently attached to MCC have been described previously (26).
In vitro T cell polarization
Th1 conditions: AD10 splenocytes were used after removal of red blood cells by hypotonic lysis. Splenocytes were cultured in RPMI 1640, supplemented as previously described (22), with 2.5 μM moth cytochrome c (MCC) 88–103 (KAERADLIAYLKQATK) in the presence of 5 ng/ml IL-12 (Cell Sciences, Canton, MA) and 20 μg/ml anti-IL-4 (11B11).
Th2 conditions: After removal of red blood cells by hypotonic lysis, CD3+ cells were purified by negative selection with Mouse T Cell Enrichment Columns (R&D Systems, Minneapolis, MN). CD3+ cells were incubated at 106/ml with irradiated B10.BR splenocytes (5:1) in the presence of 2.5 μM MCC 88–103, 100 ng/ml IL-4 (as a transfected plasmacytoma culture supernatant) and 50 μg/ml anti-IFNγ(XMG 1.2). 80 U/ml IL-2 was added on day 2 of culture. After 6–7 days in culture, Th2 cells were restimulated with irradiated B10.BR splenocytes and peptide. Th2 lines were maintained for up to five weeks. Intracellular cytokine staining of acutely activated Th1 and Th2 cells for IFNγ and IL-4 confirmed polarization.
Bilayers
GPI-anchored forms of Oregon Green 488 labeled I-Ek (200 molecules/μm2) and Cy5-labeled ICAM-1 (300 molecules/μm2) were incorporated into dioleoylphosphatidylcholine bilayers as described (4). These bilayers were supported on a coverglass in a Bioptechs flow cell, and were loaded with various concentrations of peptide for 24 hr at 37°C (4).
Live cell microscopy
107 Th1 or Th2 cells in 1 ml HEPES-buffered saline supplemented with 6 mM D-glucose, 2 mM MgCl2, 1 mM CaCl2, and 1% human serum albumin were injected onto bilayers at 37°C. Imaging was performed with a 40X or 60X objective using two Applied Precision DeltaVision systems (Issaquah, WA). These systems included an Applied Precision chassis with a motorized XYZ stage, a Nikon TE200 or Olympus IX71 inverted fluorescent microscope, halogen illumination, a CH350L or CoolSnap HQ2 camera and the DeltaVision SoftWorx software package.
Fixed cell microscopy
2.5 X 104 MCC:GFP APC were seeded onto a LabTek II (Campbell, CA) eight-chambered #1.5 (0.17 mm) coverglass in complete DMEM and incubated overnight at 37°C. After removal of DMEM, 5 X 105 Th1 or Th2 cells in RPMI 1640 were added per chamber. After a brief centrifugation, the chambers were incubated at 37°C for 10 or 30 min. The cells were fixed and stained as previously described (26). Conjugates to be imaged were chosen based on the presence of a GFP signal at the T cell:APC interface. Stacks of fluorescent images spaced 0.2 μm apart were obtained with a 1.4 NA 60X oil immersion lens on the DeltaVision system and deconvolved with an iterative, constrained algorithm. Deconvolution and 3D reconstructions were performed with the Applied Precision SoftWorx software. Colocalization studies were conducted with Imaris software from Bitplane.
Results
MHC/TCR clusters do not coalesce to form a cSMAC in Th2 IS
Previous studies using the supported planar bilayer system demonstrated the early stages of IS formation (4). Upon antigen recognition, MHC/TCR interactions are initiated in the periphery and immediately start moving towards the center of the T-cell-APC interface (4, 11–13). After 4–5 minutes, most of the MHC/TCR interactions are centrally located, and this structure with a centrally located cluster of MHC/TCR surrounded by a ring of adhesion molecules is stable for at least an hour (4).
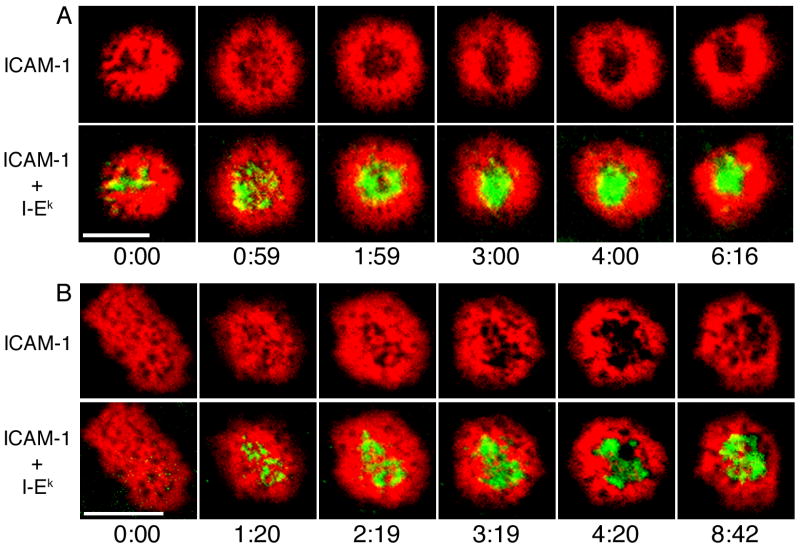
We conducted experiments with supported planar bilayers to observe IS formation by Th1 and Th2 cells. Initially upon antigen recognition, both Th1 and Th2 cells spread out and showed broad accumulation of ICAM-1 across the T cell-bilayer interface (Fig. 1). Small accumulations of MHC/TCR were randomly distributed across the interface very rapidly after the initiation of synapse formation. After 1 minute, both Th1 and Th2 cells showed clusters of MHC/TCR across the interface. Between 2 and 3 minutes, MHC/TCR clusters moved to the center of synapses formed by Th1 cells (Fig. 1A). By 5 minutes, most Th1 synapses had a compact central accumulation of MHC/TCR surrounded by a ring of ICAM-1, and this pattern was stable past 10 min. MHC/TCR clusters in most Th2 IS did not coalesce into a central accumulation (Fig. 1B). Unlike Th1 cells, the IS formed by Th2 cells had multiple distinct clusters of MHC/TCR that were maintained for at least ten minutes. These data indicate that Th2 cells do not form IS with a single, central accumulation of MHC/TCR surrounded by a ring of adhesion molecules.
Figure 1. Dynamics of IS formation in Th1 and Th2 cells.
Th1 (A) and Th2 (B) cells were injected onto supported planar bilayers containing ICAM-1-Cy5 (red) and peptide-loaded I-Ek-488 (green). Wide-field fluorescence microscopy was used to image T-cell:APC contacts. One set of ICAM-1-Cy5/I-Ek-488 images was obtained every minute. The time indicated is relative to the initial detection of T cell-bilayer contact. MHC/TCR clusters did not coalesce into a cSMAC in most Th2 IS. Data are from one representative experiment of two with n=14 for Th1 cells and n=83 for Th2 cells. Scale bars represent 5 μm.
Th2 cells form multifocal IS
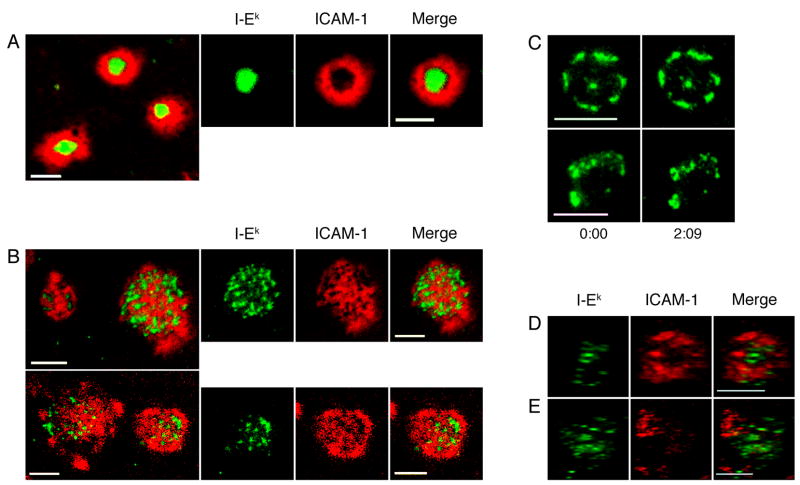
To confirm that Th2 cells do not form mature IS with a central accumulation of MHC/TCR, we observed Th1 and Th2 synapses that were 20 to 35 minutes old. As in Figure 1, we allowed Th1 or Th2 cells to settle onto supported lipid bilayers loaded with 100 μM MCC, and let the cells interact with the bilayers for 20 minutes at 37°C before commencing imaging. Th1 cells consistently formed tight, compact IS with an ICAM-1 ring surrounding a central accumulation of MHC/TCR (Fig. 2A). This structure agrees well with IS observed in a variety of systems using both CD4+ T cell blasts and CD8+ cytotoxic T lymphocytes (1, 4, 27). The pattern of MHC/TCR was only scored as compact if there was a single cluster of MHC/TCR. Multiple MHC/TCR accumulations were scored as multifocal even if all of the clusters were centrally located. Although a compact accumulation of MHC/TCR was the dominant phenotype for Th1 IS, a multifocal structure was seen in 23% of the cells (Table I).
Figure 2. Th2 cells form multifocal IS.
Th1 and Th2 cells were incubated on supported planar bilayers containing ICAM-1-Cy5 (red) and peptide-loaded I-Ek-488 (green) for 20 minutes prior to imaging. Images were then captured between 20 and 35 minutes. (A) Th1 cells consistently formed IS with a compact, monofocal accumulation of MHC/TCR. (B) The majority of Th2 cells formed multifocal IS. (C) Th2 cells were imaged over time, 20 min after IS formation. Th1 (D) and Th2 (E) cells were allowed to interact with MCC:GFP fibroblast APC for 30 minutes. Conjugates were fixed, permeabilized and stained with an antibody to LFA-1 followed by a Cy-5-conjugated secondary antibody. Following deconvolution, 3-D reconstructions of T-cell:APC interfaces were made. En face views from the 3D reconstructions of T cell:APC interfaces are shown. Data are representative of two experiments with n=20 for Th1 cells and n=19 for Th2 cells. Scale bars represent 5 μm.
Table I.
Quantitation of Th1 and Th2 IS formed on supported planar bilayersa
| Pattern of MHC/TCR in IS | Pattern of ICAM-1 in IS | |||||
|---|---|---|---|---|---|---|
| Antigen Conc. | n | Compact | Multifocal | Ring | Diffuse | |
| Th1 | 100 μM | 407 | 314 (77%) | 93 (23%) | 264 (65%) | 143 (35%) |
| 10 μM | 284 | 215 (76%) | 69 (24%) | 149 (52%) | 135 (48%) | |
| 1 μM | 518 | 388 (75%) | 130 (25%) | 289 (56%) | 229 (44%) | |
| Th2 | 100 μM | 805 | 251 (31%) | 554 (69%) | 113 (14%) | 692 (86%) |
| 10 μM | 483 | 265 (55%) | 218 (45%) | 104 (21%) | 379 (79%) | |
| 1 μM | 238 | 129 (54%) | 109 (46%) | 45 (19%) | 193 (81%) | |
Th2 cells formed synapses with the same frequency as Th1 cells, but with a markedly different structure. Instead of containing a single, compact accumulation of MHC/TCR, Th2 IS were distinctly multifocal in nature (Fig. 2B). The majority of IS formed by Th2 cells had multiple small accumulations of MHC/TCR with ICAM-1 interspersed throughout the T-cell-APC interface. A quantitative assessment showed that nearly 70% of IS formed by Th2 cells had a diffuse multifocal distribution of MHC/TCR (Table I). The number of MHC/TCR clusters varied from cell to cell with some having as few as 5 and others more than 20. We also examined the distribution of ICAM-1 in Th1 and Th2 IS. Two-thirds of the IS formed by Th1 cells have the classic “bull’s-eye” pattern with ICAM-1 excluded from the cSMAC, resulting in a ring-like structure. Th2 cells, on the other hand, only formed ring structures in 14% of the IS examined (Table I). The vast majority of Th2 IS had a diffuse distribution of ICAM-1 throughout the interface with the planar bilayer. We also imaged Th2 IS over time, 20 min after introduction to the bilayers, to determine if the location of MHC/TCR clusters was dynamic after the initial steps of IS formation. As shown in Figure 2C, the pattern of MHC/TCR clusters in Th2 IS was stable.
To confirm that Th1 and Th2 cells formed distinctly different IS, we conducted experiments with viable APC instead of supported planar bilayers. For these experiments we used B7.1 positive fibroblasts transfected with ICAM-1 and MHC/peptide. These fibroblasts have been shown to form IS with AD10 T cell blasts and to induce T cell proliferation, but it should be noted that IS formed with these fibroblasts have a patchier distribution of MHC/TCR and ICAM-1/LFA-1 than IS formed on the highly simplified planar bilayers (26). T cell:APC conjugates were stained with an antibody to LFA-1. Some of the IS formed by Th1 cells with fibroblast APC were characterized by a ring of LFA-1 surrounding the largest accumulations of MHC/TCR, as shown in Figure 2D. On the other hand, all of the Th2 cells examined formed multifocal IS with LFA-1 distributed homogenously throughout the T cell-APC interface (Fig 2E).
CD45 is not excluded from the T-cell-APC interface in Th2 cells
The large tyrosine phosphatase CD45 is involved in both the positive and negative regulation of TCR signaling (28, 29). The role of CD45 in TCR signaling makes its spatial and temporal regulation extremely important. Initially upon antigen recognition and TCR clustering, CD45 is located centrally, along with MHC/TCR (7), where it may positively regulate TCR signaling by dephosphorylating the negative regulatory tyrosine phosphorylation sites on the C-terminus of Lck (30, 31). However, by 5 to 7 min CD45 is completely excluded from the T-cell-APC cSMAC (7, 32). Recent experiments with LFA-1 deficient T cells have shown that this adhesion molecule is critical for the exclusion of CD45 from the cSMAC (33). Importantly, it has been reported that after stimulation through the TCR, CD45 is located in lipid rafts in Th1 but not Th2 cells (25). This led us to conduct experiments to examine the location of CD45 in IS formed by Th1 and Th2 cells.
By ten minutes after conjugation, CD45 was excluded from the central region of the interface in the majority of the Th1-APC conjugates examined (Fig. 3A and C). With viable fibroblast APC, MHC/TCR clusters at the interface did not completely coalesce into a cSMAC in many of the conjugates. Nevertheless, MHC/TCR clusters rarely overlapped the ring of CD45 at the periphery of the Th1-APC interface. In Th2-APC conjugates, MHC/TCR clusters were diffusely spread throughout the interface. Strikingly, CD45 was not excluded from the interface between Th2 cells and APC (Fig. 3B and C). Instead CD45 was interspersed among the MHC/TCR clusters in the majority of conjugates examined. Although CD45 was not excluded from the central region of the interface in Th2 cells, minimal colocalization with MHC-TCR clusters was seen (Fig. 3D). This is consistent with recent studies that have shown that CD45 is excluded from TCR microclusters (11).
Figure 3. CD45 is not excluded from Th2 IS.
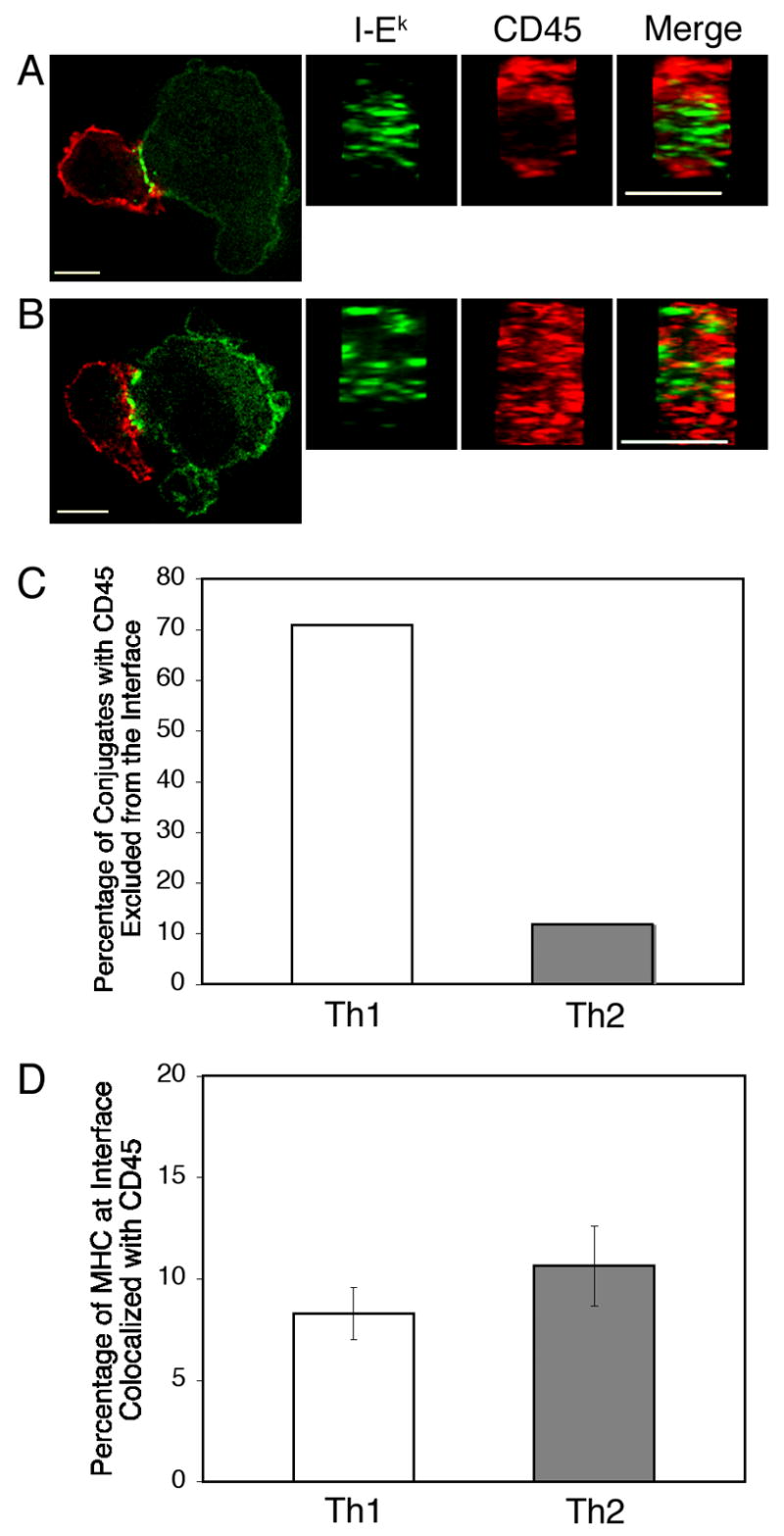
Th1 (A) and Th2 (B) cells were incubated with MCC:GFP fibroblast APC for 10 minutes, fixed, permeabilized and stained with an antibody to CD45 followed by a Texas Red-conjugated secondary antibody (red). 3-D reconstructions of T-cell:APC interfaces were made after deconvolution. (C) Conjugates were scored for the exclusion of CD45 from the center of the T cell-APC interface. (D) Although CD45 is not excluded from the center of the T cell-APC interface in Th2 cells, there is not a significant difference in the colocalization between MHC/TCR and CD45 in Th1 and Th2 cells. Colocalization analysis was conducted with Imaris software from Bitplane using the thresholding technique. The percentage of the total volume of MHC/TCR at the T cell-APC interface that was colocalized with CD45 is shown. The difference between Th1 and Th2 cells was not statistically significant (P=0.3; Student’s t Test). Data shown are representative of three independent experiments with n=32 for Th1 cells and n=33 for Th2 cells. Scale bars represent 5 μm.
Phosphotyrosine colocalizes with MHC/TCR clusters to a greater extent in Th2 IS
Recent publications have shown that continuous signaling after the formation of a mature IS, as measured by the presence of phosphorylated proteins, is largely excluded from the cSMAC (7, 9). Total internal reflection fluorescent microscopy of T cells interacting with a supported lipid bilayer demonstrated that TCR microclusters in the periphery, but not the center of an IS, colocalized with phospho-Lck, phospho-Zap-70 and phospho-Lat (12, 13).
We conducted experiments with our MCC:GFP fibroblasts to observe the pattern of pTyr staining in mature IS formed by Th1 and Th2 cells. We chose to look at 30 min conjugates to assure that the majority of IS we observed were mature. Phosphotyrosine and MHC/TCR generally occupied distinct domains in the IS formed by Th1 cells (Fig. 4A). However, there was significant colocalization at the boundary between the domains containing pTyr and MHC/TCR. We performed a quantitative colocalization analysis to determine the degree of overlap between pTyr and MHC/TCR at the T cell-APC interface. On average, approximately 30% of the MHC/TCR at the interface was colocalized with MHC/TCR in Th1 IS (Fig. 4C). In Th2 IS, the MHC/TCR and pTyr domains were not as distinct as for Th1 cells (Fig. 4B). Colocalization analysis showed that for Th2 cells, a significantly greater percentage of the MHC/TCR at the interface was colocalized with pTyr (Fig. 4C).
Figure 4. Phosphotyrosine localization is distinct in Th1 and Th2 IS.
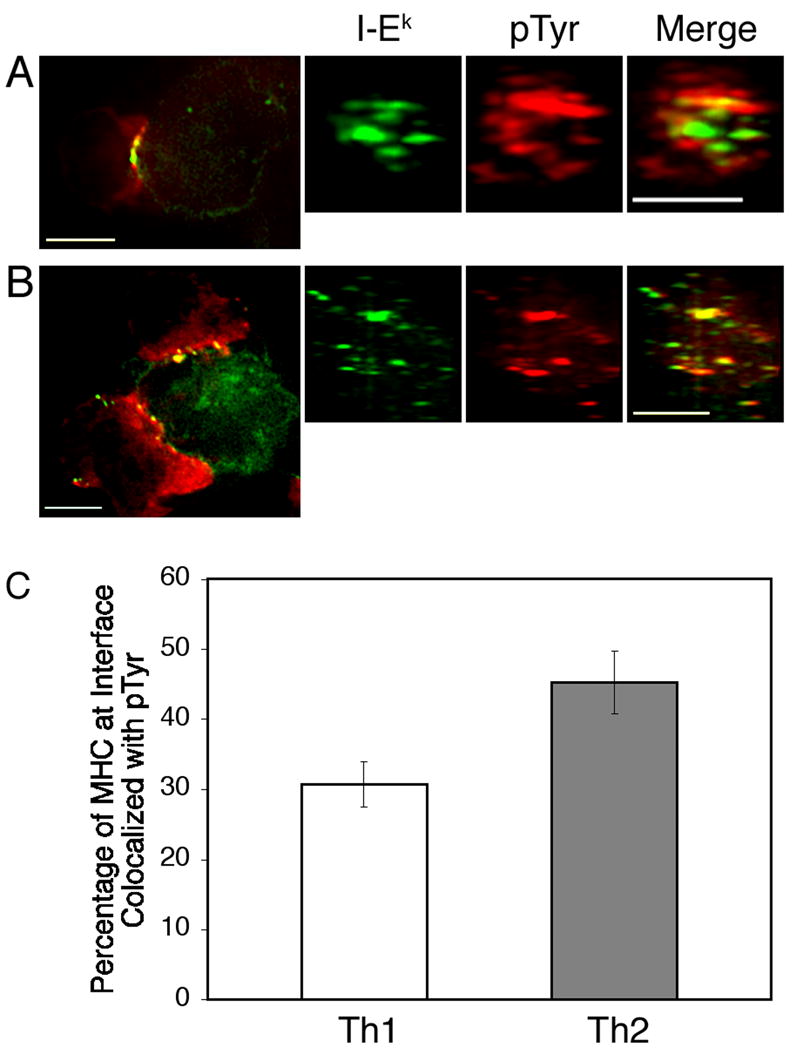
Th1 (A) and Th2 (B) cells were incubated with MCC:GFP fibroblasts for 30 minutes. The conjugates were then fixed, permeabilized and stained with a biotinylated antibody to phosphotyrosine followed by Cy-5-conjugated streptavidin (red). Following deconvolution, 3-D reconstructions of the T cell:APC interfaces were made. (C) A greater percentage of the MHC/TCR at the T cell-APC interface is colocalized with pTyr in Th2 cells. Colocalization analysis was performed as in Figure 3. The difference between Th1 and Th2 cells is significant (P = 0.01; Student’s unpaired t test) Data are representative of three independent experiments with n=32 for Th1 cells and n=28 for Th2 cells. Scale bars represent 5 μm.
A greater percentage of Th2 cells form compact IS at low antigen concentrations, but ICAM-1 does not form a ring structure
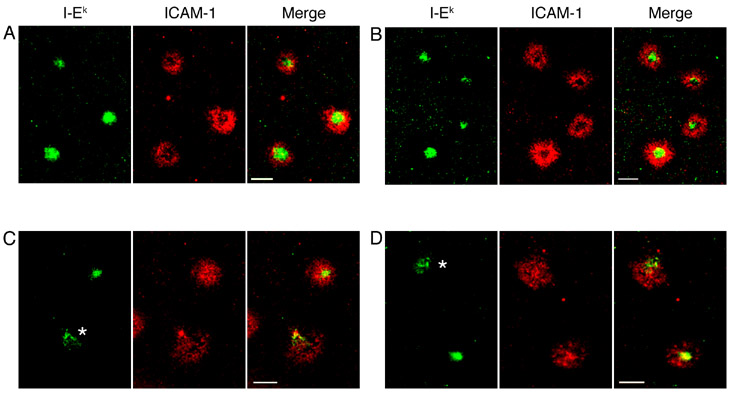
T cell blasts from AND TCR-transgenic mice on a B10.BR background, which skew towards a Th1 phenotype (34), continue to form mature IS with a central accumulation of MHC/TCR surrounded by a ring of ICAM-1 even when the number of available MHC-peptide molecules is reduced several fold (4, 11). We conducted experiments with bilayers loaded with 10- and 100-fold less peptide to determine if the IS formed by Th1 and Th2 cells were still morphologically distinct when less antigen was available. These bilayers contained 200 MHC molecules/μm2 as in the preceding experiments. Th1 cells continued to form compact IS at a similar frequency to experiments with planar bilayers loaded with 100 μM peptide. Interestingly, when the bilayers were loaded with 10- or 100-fold less antigen, a greater percentage of the IS formed by Th2 cells had a compact accumulation of MHC/TCR, although still less than for Th1 cells (Figure 5 and Table I). However, unlike Th1 IS, Th2 IS formed on bilayers loaded with 10 or 1 μM MCC did not segregate ICAM-1 into a pSMAC, forming ring-like structures. Instead, ICAM-1 was distributed diffusely across the IS in approximately 80% of the IS examined regardless of the antigen concentration (Figure 5 and Table I).
Figure 5. Th2 cells form compact IS at low antigen concentration; but ICAM-1 is not excluded from the cSMAC.
For these experiments, 10 or 1 μM MCC was used during the peptide-loading step instead of 100 μM MCC, as in the preceding figures. Th1 (A and B) or Th2 (C and D) cells were incubated on supported planar bilayers containing ICAM-1-Cy5 (red) and I-Ek-488 (green) loaded with 10 μM MCC (A and C) or 1 μM MCC (B and D) for 15 minutes prior to imaging. Images were then captured between 15 and 35 minutes. Cells marked with an asterisk were scored as multifocal for MHC/TCR. Data are from one representative experiment of three. Scale bars represent 5 μm.
Discussion
In this report, we have shown that polarized Th1 and Th2 cells form morphologically distinct IS. Th1 cells form IS with a compact central cluster of MHC/TCR surrounded by a ring of adhesion molecules, that closely resemble the IS described in the literature for T cell blasts. This resemblance is not surprising, considering that most of the published experiments designed to study IS formation used T cell blasts from B6 or B10 mice. Blasts from these mice tend to skew towards an IFN-γ secreting Th1 phenotype (34). Th1 IS structure was not markedly different, even when planar bilayers were loaded with 100-fold less peptide antigen. In contrast, we found that at high antigen concentrations, Th2 cells formed multifocal IS, characterized by multiple MHC/TCR accumulations. When the amount of antigen used to load I-Ek in the bilayers was reduced by 10 to 100-fold, slightly more than half of Th2 cells formed compact IS. However, even when Th2 cells formed compact IS, ICAM-1 did not form a ring. This apparent co-localization of TCR/MHC and LFA-1/ICAM-1 interactions in Th2 cells forming compact IS may reflect the close packing of many submicron TCR and LFA-1 microclusters in this region - a continuation of the multifocal paradigm to a shorter length scale. Further study will be needed to determine if this change in organization alters some of the proposed functions of the cSMAC, such as signal termination. Inactivation of signaling in the cSMAC may allow Th1 cells to maintain relatively constant signaling intensity over a wide range of antigen densities. Perhaps the biology of Th2 cells requires that signal strength is more proportional to antigen density and this might be achieved by delaying or eliminating functional cSMAC formation.
Multifocal IS have been described in several reports. Immunofluorescence and transmission electron microscopy were used to show that IS formed between naive T cells and dendritic cells are multifocal (35). IS with a multifocal structure were also seen in conjugates between naïve CD4+ T cells and APC in the absence of CD80 (36). Finally, double positive thymocytes have been shown to form multifocal IS, with pTyr stably associated with MHC/TCR foci (37). IS formed by these immature thymocytes are remarkably similar in structure to the multifocal IS formed by Th2 cells at high antigen doses. Strikingly, pTyr continues to be associated with MHC/TCR clusters for at least 30 min in both double positive thymocytes and Th2 cells. Hence, it is possible that continuous tyrosine kinase signaling at the site of MHC/TCR accumulation is a general property of multifocal synapses. Th2 cells are thought to make lengthy contacts with B cells during the delivery of T cell help (38, 39), and continuous signaling at the IS may be important for this function.
It is known that the phosphorylation of signaling proteins, including Zap-70, is less complete in Th2 compared to Th1 cells upon activation through the TCR (40, 41). A recent report has shown that Th2 cells have twofold less surface CD4 than Th1 cell (42), and we have confirmed this result with polarized AD10 cells (data not shown). When CD4 levels of Th2 cells were elevated via retroviral transduction, stimulated Th2 cells had increased levels of ζ-chain and Zap-70 phosphorylation, comparable to Th1 cells (42). Additionally, CD4 is excluded from lipid rafts in Th2-APC conjugates (25), and CD4 is necessary for T cells to arrest and form mature IS (4, 43). Therefore, the inability of Th2 cells to form mature IS when there are large quantities of MHC-peptide available, as demonstrated in this report, could be due to the lower levels of CD4 compared to Th1 cells.
Th1 cells form mature IS with a bull’s-eye stucture and have cytotoxic functions. Th2 cells are generally incapable of killing (44–46), and we have shown that a significant percentage of the IS formed by Th2 cells are multifocal, especially at high concentrations of antigen. Importantly, even at much lower antigen concentrations, where the majority of IS displayed compact accumulations of MHC/TCR, Th2 cells failed to form IS with ring-like distribution of ICAM-1. It has been hypothesized that the delivery of cytotoxic effector molecules inside the ring of adhesion molecules in IS formed between CTL and target cells could prevent damage to bystander cells (47, 48). In this model, the ring structure formed by ICAM-1-LFA-1 interactions acts as a gasket. This gasket may not be a crucial component of the IS formed by naïve CD4+ T cells, Th2 cells and thymocytes, as these cell types do not have cytotoxic function. We have recently shown that Th1 cells contain a pool of preformed CD40L that is rapidly mobilized to the cell surface upon stimulation (49). It is also possible that the ICAM-1 gasket could serve to direct secrtion of molecules involved in T cell help directly towards the source of antigen, thereby preventing activation of bystander cells. Further experimentation will be required to determine if the primary function of the bull’s-eye-like IS formed by Th1 cells is to facilitate the delivery of cytotoxic agents and/or molecules involved in T cell help to APC.
Acknowledgments
We thank Aurelie Snyder, Toby Starr and Rajat Varma for expert technical assistance and Susan Murray, Alexander Rowe, Katelynn Gardner Toren and Cortny Williams for critically reviewing the manuscript.
Abbreviations
- IS
immunological synapse
- pSMAC
peripheral supramolecular activation cluster
- cSMAC
central supramolecular activation cluster
- MCC
moth cytochrome c 88-103
- pTyr
phosphotyrosine
Footnotes
This work was supported by National Institutes of Health Grant AI050823 (DCP) and AI43542 (MLD). T.J.T. was supported by training grants EYO7123 from the National Eye Institute and T32 AI07472 from the National Institute for Allergy and Infectious Diseases.
This is an author-produced version of a manuscript accepted for publication in The Journal of Immunology (The JI). The American Association of Immunologists, Inc. (AAI), publisher of The JI, holds the copyright to this manuscript. This version of the manuscript has not yet been copyedited or subjected to editorial proofreading by The JI; hence, it may differ from the final version published in The JI (online and in print). AAI (The JI) is not liable for errors or omissions in this author-produced version of the manuscript or in any version derived from it by the U.S. National Institutes of Health or any other third party. The final, citable version of record can be found at www.jimmunol.org.
Disclosures The authors declare no conflicts of interest or financial interests.
References
- 1.Monks CR, Freiberg BA, Kupfer H, Sciaky N, Kupfer A. Three-dimensional segregation of supramolecular activation clusters in T cells. Nature. 1998;395:82–86. doi: 10.1038/25764. [DOI] [PubMed] [Google Scholar]
- 2.Dustin ML, Olszowy MW, Holdorf AD, Li J, Bromley S, Desai N, Widder P, Rosenberger F, van der Merwe PA, Allen PM, Shaw AS. A novel adaptor protein orchestrates receptor patterning and cytoskeletal polarity in T-cell contacts. Cell. 1998;94:667–677. doi: 10.1016/s0092-8674(00)81608-6. [DOI] [PubMed] [Google Scholar]
- 3.Huppa JB, Davis MM. T-cell-antigen recognition and the immunological synapse. Nat Rev Immunol. 2003;3:973–983. doi: 10.1038/nri1245. [DOI] [PubMed] [Google Scholar]
- 4.Grakoui A, Bromley SK, Sumen C, Davis MM, Shaw AS, Allen PM, Dustin ML. The immunological synapse: a molecular machine controlling T cell activation. Science. 1999;285:221–227. doi: 10.1126/science.285.5425.221. [DOI] [PubMed] [Google Scholar]
- 5.Sims TN, Soos TJ, Xenias HS, Dubin-Thaler B, Hofman JM, Waite JC, Cameron TO, Thomas VK, Varma R, Wiggins CH, Sheetz MP, Littman DR, Dustin ML. Opposing effects of PKCtheta and WASp on symmetry breaking and relocation of the immunological synapse. Cell. 2007;129:773–785. doi: 10.1016/j.cell.2007.03.037. [DOI] [PubMed] [Google Scholar]
- 6.Bromley SK, Iaboni A, Davis SJ, Whitty A, Green JM, Shaw AS, Weiss A, Dustin ML. The immunological synapse and CD28-CD80 interactions. Nat Immunol. 2001;2:1159–1166. doi: 10.1038/ni737. [DOI] [PubMed] [Google Scholar]
- 7.Freiberg BA, Kupfer H, Maslanik W, Delli J, Kappler J, Zaller DM, Kupfer A. Staging and resetting T cell activation in SMACs. Nat Immunol. 2002;3:911–917. doi: 10.1038/ni836. [DOI] [PubMed] [Google Scholar]
- 8.Johnson KG, Bromley SK, Dustin ML, Thomas ML. A supramolecular basis for CD45 tyrosine phosphatase regulation in sustained T cell activation. Proc Natl Acad Sci U S A. 2000;97:10138–10143. doi: 10.1073/pnas.97.18.10138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lee KH, Holdorf AD, Dustin ML, Chan AC, Allen PM, Shaw AS. T cell receptor signaling precedes immunological synapse formation. Science. 2002;295:1539–1542. doi: 10.1126/science.1067710. [DOI] [PubMed] [Google Scholar]
- 10.Lee KH, Dinner AR, Tu C, Campi G, Raychaudhuri S, Varma R, Sims TN, Burack WR, Wu H, Wang J, Kanagawa O, Markiewicz M, Allen PM, Dustin ML, Chakraborty AK, Shaw AS. The immunological synapse balances T cell receptor signaling and degradation. Science. 2003;302:1218–1222. doi: 10.1126/science.1086507. [DOI] [PubMed] [Google Scholar]
- 11.Varma R, Campi G, Yokosuka T, Saito T, Dustin ML. T cell receptor-proximal signals are sustained in peripheral microclusters and terminated in the central supramolecular activation cluster. Immunity. 2006;25:117–127. doi: 10.1016/j.immuni.2006.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Campi G, Varma R, Dustin ML. Actin and agonist MHC-peptide complex-dependent T cell receptor microclusters as scaffolds for signaling. J Exp Med. 2005;202:1031–1036. doi: 10.1084/jem.20051182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Yokosuka T, Sakata-Sogawa K, Kobayashi W, Hiroshima M, Hashimoto-Tane A, Tokunaga M, Dustin ML, Saito T. Newly generated T cell receptor microclusters initiate and sustain T cell activation by recruitment of Zap70 and SLP-76. Nat Immunol. 2005;6:1253–1262. doi: 10.1038/ni1272. [DOI] [PubMed] [Google Scholar]
- 14.Stinchcombe JC, Bossi G, Booth S, Griffiths GM. The immunological synapse of CTL contains a secretory domain and membrane bridges. Immunity. 2001;15:751–761. doi: 10.1016/s1074-7613(01)00234-5. [DOI] [PubMed] [Google Scholar]
- 15.Stinchcombe JC, Griffiths GM. The role of the secretory immunological synapse in killing by CD8+ CTL. Semin Immunol. 2003;15:301–305. doi: 10.1016/j.smim.2003.09.003. [DOI] [PubMed] [Google Scholar]
- 16.Anikeeva N, Somersalo K, Sims TN, Thomas VK, Dustin ML, Sykulev Y. Distinct role of lymphocyte function-associated antigen-1 in mediating effective cytolytic activity by cytotoxic T lymphocytes. Proc Natl Acad Sci U S A. 2005;102:6437–6442. doi: 10.1073/pnas.0502467102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Purbhoo MA, Irvine DJ, Huppa JB, Davis MM. T cell killing does not require the formation of a stable mature immunological synapse. Nat Immunol. 2004;5:524–530. doi: 10.1038/ni1058. [DOI] [PubMed] [Google Scholar]
- 18.Faroudi M, Utzny C, Salio M, Cerundolo V, Guiraud M, Muller S, Valitutti S. Lytic versus stimulatory synapse in cytotoxic T lymphocyte/target cell interaction: manifestation of a dual activation threshold. Proc Natl Acad Sci U S A. 2003;100:14145–14150. doi: 10.1073/pnas.2334336100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.O’Keefe JP, Gajewski TF. Cutting edge: cytotoxic granule polarization and cytolysis can occur without central supramolecular activation cluster formation in CD8+ effector T cells. J Immunol. 2005;175:5581–5585. doi: 10.4049/jimmunol.175.9.5581. [DOI] [PubMed] [Google Scholar]
- 20.Huse M, Lillemeier BF, Kuhns MS, Chen DS, Davis MM. T cells use two directionally distinct pathways for cytokine secretion. Nat Immunol. 2006;7:247–255. doi: 10.1038/ni1304. [DOI] [PubMed] [Google Scholar]
- 21.Constant S, Pfeiffer C, Woodard A, Pasqualini T, Bottomly K. Extent of T cell receptor ligation can determine the functional differentiation of naive CD4+ T cells. J Exp Med. 1995;182:1591–1596. doi: 10.1084/jem.182.5.1591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.O’Garra A. Cytokines induce the development of functionally heterogeneous T helper cell subsets. Immunity. 1998;8:275–283. doi: 10.1016/s1074-7613(00)80533-6. [DOI] [PubMed] [Google Scholar]
- 23.Murphy KM, Reiner SL. The lineage decisions of helper T cells. Nat Rev Immunol. 2002;2:933–944. doi: 10.1038/nri954. [DOI] [PubMed] [Google Scholar]
- 24.Jackman RP, Balamuth F, Bottomly K. CTLA-4 differentially regulates the immunological synapse in CD4 T cell subsets. J Immunol. 2007;178:5543–5551. doi: 10.4049/jimmunol.178.9.5543. [DOI] [PubMed] [Google Scholar]
- 25.Balamuth F, Leitenberg D, Unternaehrer J, Mellman I, Bottomly K. Distinct patterns of membrane microdomain partitioning in Th1 and Th2 cells. Immunity. 2001;15:729–738. doi: 10.1016/s1074-7613(01)00223-0. [DOI] [PubMed] [Google Scholar]
- 26.Wetzel SA, McKeithan TW, Parker DC. Live-cell dynamics and the role of costimulation in immunological synapse formation. J Immunol. 2002;169:6092–6101. doi: 10.4049/jimmunol.169.11.6092. [DOI] [PubMed] [Google Scholar]
- 27.Potter TA, Grebe K, Freiberg B, Kupfer A. Formation of supramolecular activation clusters on fresh ex vivo CD8+ T cells after engagement of the T cell antigen receptor and CD8 by antigen-presenting cells. Proc Natl Acad Sci U S A. 2001;98:12624–12629. doi: 10.1073/pnas.221458898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Thomas ML, Brown EJ. Positive and negative regulation of Src-family membrane kinases by CD45. Immunol Today. 1999;20:406–411. doi: 10.1016/s0167-5699(99)01506-6. [DOI] [PubMed] [Google Scholar]
- 29.Hermiston ML, Xu Z, Weiss A. CD45: a critical regulator of signaling thresholds in immune cells. Annu Rev Immunol. 2003;21:107–137. doi: 10.1146/annurev.immunol.21.120601.140946. [DOI] [PubMed] [Google Scholar]
- 30.Sieh M, Bolen JB, Weiss A. CD45 specifically modulates binding of Lck to a phosphopeptide encompassing the negative regulatory tyrosine of Lck. Embo J. 1993;12:315–321. doi: 10.1002/j.1460-2075.1993.tb05659.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Cahir McFarland ED, Hurley TR, Pingel JT, Sefton BM, Shaw A, Thomas ML. Correlation between Src family member regulation by the protein-tyrosine-phosphatase CD45 and transmembrane signaling through the T-cell receptor. Proc Natl Acad Sci U S A. 1993;90:1402–1406. doi: 10.1073/pnas.90.4.1402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Leupin O, Zaru R, Laroche T, Muller S, Valitutti S. Exclusion of CD45 from the T-cell receptor signaling area in antigen-stimulated T lymphocytes. Curr Biol. 2000;10:277–280. doi: 10.1016/s0960-9822(00)00362-6. [DOI] [PubMed] [Google Scholar]
- 33.Graf B, Bushnell T, Miller J. LFA-1-mediated T cell costimulation through increased localization of TCR/class II complexes to the central supramolecular activation cluster and exclusion of CD45 from the immunological synapse. J Immunol. 2007;179:1616–1624. doi: 10.4049/jimmunol.179.3.1616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hsieh CS, Macatonia SE, O’Garra A, Murphy KM. T cell genetic background determines default T helper phenotype development in vitro. J Exp Med. 1995;181:713–721. doi: 10.1084/jem.181.2.713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Brossard C, Feuillet V, Schmitt A, Randriamampita C, Romao M, Raposo G, Trautmann A. Multifocal structure of the T cell - dendritic cell synapse. Eur J Immunol. 2005;35:1741–1753. doi: 10.1002/eji.200425857. [DOI] [PubMed] [Google Scholar]
- 36.Tseng SY, Liu M, Dustin ML. CD80 cytoplasmic domain controls localization of CD28, CTLA-4, and protein kinase C θ in the immunological synapse. J Immunol. 2005;175:7829–7836. doi: 10.4049/jimmunol.175.12.7829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hailman E, Burack WR, Shaw AS, Dustin ML, Allen PM. Immature CD4(+)CD8(+) thymocytes form a multifocal immunological synapse with sustained tyrosine phosphorylation. Immunity. 2002;16:839–848. doi: 10.1016/s1074-7613(02)00326-6. [DOI] [PubMed] [Google Scholar]
- 38.Kupfer H, Monks CR, Kupfer A. Small splenic B cells that bind to antigen-specific T helper (Th) cells and face the site of cytokine production in the Th cells selectively proliferate: immunofluorescence microscopic studies of Th-B antigen-presenting cell interactions. J Exp Med. 1994;179:1507–1515. doi: 10.1084/jem.179.5.1507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Okada T, Miller MJ, Parker I, Krummel MF, Neighbors M, Hartley SB, O’Garra A, Cahalan MD, Cyster JG. Antigen-engaged B cells undergo chemotaxis toward the T zone and form motile conjugates with helper T cells. PLoS Biol. 2005;3:e150. doi: 10.1371/journal.pbio.0030150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Tamura T, Nakano H, Nagase H, Morokata T, Igarashi O, Oshimi Y, Miyazaki S, Nariuchi H. Early activation signal transduction pathways of Th1 and Th2 cell clones stimulated with anti-CD3. Roles of protein tyrosine kinases in the signal for IL-2 and IL-4 production. J Immunol. 1995;155:4692–4701. [PubMed] [Google Scholar]
- 41.Hannier S, Bitegye C, Demotz S. Early events of TCR signaling are distinct in human Th1 and Th2 cells. J Immunol. 2002;169:1904–1911. doi: 10.4049/jimmunol.169.4.1904. [DOI] [PubMed] [Google Scholar]
- 42.Itoh Y, Wang Z, Ishida H, Eichelberg K, Fujimoto N, Makino J, Ogasawara K, Germain RN. Decreased CD4 expression by polarized T helper 2 cells contributes to suboptimal TCR-induced phosphorylation and reduced Ca2+ signaling. Eur J Immunol. 2005;35:3187–3195. doi: 10.1002/eji.200526064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Balamuth F, Brogdon JL, Bottomly K. CD4 raft association and signaling regulate molecular clustering at the immunological synapse site. J Immunol. 2004;172:5887–5892. doi: 10.4049/jimmunol.172.10.5887. [DOI] [PubMed] [Google Scholar]
- 44.Appay V, Zaunders JJ, Papagno L, Sutton J, Jaramillo A, Waters A, Easterbrook P, Grey P, Smith D, McMichael AJ, Cooper DA, Rowland-Jones SL, Kelleher AD. Characterization of CD4(+) CTLs ex vivo. J Immunol. 2002;168:5954–5958. doi: 10.4049/jimmunol.168.11.5954. [DOI] [PubMed] [Google Scholar]
- 45.Ju ST, Cui H, Panka DJ, Ettinger R, Marshak-Rothstein A. Participation of target Fas protein in apoptosis pathway induced by CD4+ Th1 and CD8+ cytotoxic T cells. Proc Natl Acad Sci U S A. 1994;91:4185–4189. doi: 10.1073/pnas.91.10.4185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Stalder T, Hahn S, Erb P. Fas antigen is the major target molecule for CD4+ T cell-mediated cytotoxicity. J Immunol. 1994;152:1127–1133. [PubMed] [Google Scholar]
- 47.Stinchcombe JC, Griffiths GM. Secretory mechanisms in cell-mediated cytotoxicity. Annu Rev Cell Dev Biol. 2007;23:495–517. doi: 10.1146/annurev.cellbio.23.090506.123521. [DOI] [PubMed] [Google Scholar]
- 48.Dustin ML. T-cell activation through immunological synapses and kinapses. Immunol Rev. 2008;221:77–89. doi: 10.1111/j.1600-065X.2008.00589.x. [DOI] [PubMed] [Google Scholar]
- 49.Koguchi Y, Thauland TJ, Slifka MK, Parker DC. Preformed CD40 ligand exists in secretory lysosomes in effector and memory CD4+ T cells and is quickly expressed on the cell surface in an antigen-specific manner. Blood. 2007;110:2520–2527. doi: 10.1182/blood-2007-03-081299. [DOI] [PMC free article] [PubMed] [Google Scholar]