Abstract
Immunogenicity and protective efficacy of three Campylobacter jejuni flagellum-secreted proteins, FlaC, FspA1, and FspA2, were compared by use of a mouse model. Mice were immunized intranasally with each protein with or without LTR192G as the adjuvant and challenged intranasally with C. jejuni 81-176 or CG8486. All three proteins were immunogenic, although FspA1 induced the highest levels of serum immunoglobulin G (IgG) and fecal IgA. Although immunogenic, FlaC provided only 18% protection against disease from C. jejuni 81-176. Immunization with FspA1 resulted in 57.8% protection without adjuvant or 63.8% protection with adjuvant against homologous challenge with 81-176. Alternatively, immunization with FspA2 provided 38.4% (without adjuvant) or 47.2% (with adjuvant) protection against disease from homologous challenge with CG8486. In contrast to FspA2, FspA1 provided some heterologous protection against C. jejuni CG8486 when delivered with (31.2%) or without (44.8%) LTR192G. These results suggest that FspA1 may be a good subunit vaccine candidate against C. jejuni disease.
Campylobacter jejuni is an important worldwide cause of bacterial diarrhea (7, 10), and in some geographical regions it is the primary cause of traveler's diarrhea (31, 33, 38). In addition to diarrheal disease, C. jejuni infection is associated with extraintestinal complications including reactive arthritis (13, 25) and Guillain-Barré syndrome (16, 18). Guillain-Barré syndrome polyneuropathy is due to molecular mimicry between the outer lipooligosaccharide cores of C. jejuni and human gangliosides (21). The significant level of morbidity associated with Campylobacter infection among children in the developing world, travelers, and deploying military personnel (1, 4, 6, 27, 32) drives efforts to develop vaccines against this infection. Direct evidence of acquired protective immunity to Campylobacter following natural infection (5, 7, 37) provides the rationale for efforts to develop vaccines. A further impetus to vaccine development is the alarmingly high levels of multiple antibiotic resistance noted among clinical isolates (9, 22, 30, 38).
Campylobacter vaccine approaches investigated are similar to those made with other enteric pathogens ranging from whole-bacterial-cell products through recombinantly produced proteins. Campylobacter vaccine studies using mice have included oral delivery of killed whole cells (2), whole-cell lysates (29), protein delivery by Salmonella vectors (34), nasal delivery of recombinant flagellin (17), or parenteral delivery of recombinant proteins (26). At present none has been developed into an effective human vaccine.
Vaccine development against C. jejuni has been hindered due to a number of factors that include a lack of understanding of the nature of acquired immunity and a lack of small-animal models suitable for vaccine evaluation. Moreover, C. jejuni has been particularly enigmatic in terms of molecular pathogenesis, and a limited number of virulence factors that might be useful for subunit vaccine candidates have been identified. C. jejuni flagella play multiple roles in pathogenesis (12) and prior studies have shown that a flagellin-based vaccine was immunogenic and protective in animal models (17). However, the heterogeneity of flagellin among C. jejuni strains and the high conservation of some domains of C. jejuni flagellin with other bacterial flagellins (20) suggest that this may not be an ideal subunit vaccine. C. jejuni strains lack specialized type three secretion systems and, instead, flagella secrete a number of nonflagellar protein substrates that modulate virulence. These secreted proteins appear to belong to one of two classes. The first class is composed of about eight Cia (Campylobacter invasion antigen) proteins, including CiaB, which have been shown to be required for the invasion of intestinal epithelial cells by some strains of C. jejuni (11, 14, 15, 28). Mutation of ciaB results in the loss of secretion of all cia proteins (14, 15, 28). Although the Cia proteins are synthesized in nonmotile mutants, secretion requires a full-length flagellar filament (15). The second class of secreted proteins includes FlaC and FspA, both of which are secreted through the flagellar filament without special signals (23, 35). FlaC shows homology to flagellins but is not required for motility. Mutation of flaC in some strains resulted in a decrease in invasion frequency (23, 35). Another of these flagellum-secreted proteins is FspA, a protein that appears to be C. jejuni specific but shows considerable diversity among strains (24). For example, FspA1 from strain 81-176 shows 33% identity to FspA2 from strain CG8486 (24). Moreover, recombinant FspA2 induces rapid apoptosis of intestinal epithelial cells in vitro, while FspA1 has no detectable effect. The role of the different FspA alleles on disease in vivo remains uncertain. Here, we compared the immunogenicities and protective efficacies of FlaC, FspA1, and FspA2 by use of a mouse intranasal model of C. jejuni infection.
MATERIALS AND METHODS
Mice.
Female BALB/c mice aged from 6 to 8 weeks were purchased (Jackson Laboratory, Bar Harbor, ME) and housed in laminar flow cages for 8 to 12 days before use. Food and water were provided ad libitum. The experiments were conducted according to the principles set forth in the Guide for the Care and Use of Laboratory Animals, Institute of Laboratory Animals Resources, National Research Council (19).
Bacterial strains and growth conditions.
The characteristics of C. jejuni strains 81-176 and CG8486 and their growth conditions for mouse challenge have been described previously (3, 24).
Protein expression and purification.
The flaC and fspA1 genes from C. jejuni 81-176 and the fspA2 gene from C. jejuni CG8486 were expressed in Escherichia coli as hexahistidine-tagged proteins in pET-19b as described previously (23). Strains of BL21(DE3) containing each clone were grown in Luria broth containing 100 μg/ml ampicillin and proteins were purified by nickel chromatography under native conditions. The recombinant protein yields for FlaC, FspA1, and FspA2 were 3.5, 10.6, and 6.2 mg/g of bacterial cells, respectively. Each recombinant protein was ≥94% pure as determined by densitometry scans and contained ≤100 ng of endotoxin/mg of protein, as determined by Limulus amoebocyte lysate gel clot assay (Cape Code Inc., Falmouth, MA). A recombinant form of a mutant heat labile toxin of enterotoxigenic E. coli, LTR192G (8), was manufactured at the bioproduction facility at Walter Reed Army Institute of Research, Silver Spring, MD, and used as the adjuvant.
Vaccination.
Mice were lightly anesthetized with isoflurane, and 30 μl of phosphate-buffered saline (PBS) alone or PBS containing 5 μg (7 to 10 mice per group) or 25 or 100 μg (10 to 18 mice per group) of protein was applied a drop (5 to 6 μl) at a time to the external nares (3). A total of three vaccinations for each dose level of each protein were delivered at 2-week intervals. To determine efficacy, animals receiving 100 μg of each protein alone or with 1 μg of LTR192G as the adjuvant were challenged with homologous or heterologous strains of C. jejuni. Following vaccination, animals were observed for two consecutive days for the development of vaccine-associated side effects.
Sample collection and processing.
Blood was collected 18 to 20 days after the third vaccination and the serum was separated and stored at −30°C until assayed for immunoglobulin A (IgA) and IgG. Antigen-specific and total IgA was measured in fecal pellets collected 7 days after the last vaccination. Five to seven fresh fecal pellets (100 to 120 mg) were collected from each mouse at each sampling interval. For each mg of sample, 100 μl of IgA extraction buffer was added (PBS containing 0.05% Tween 20, 0.5% fetal calf serum, 1 mg/ml EDTA, 1 mg/ml phenylmethylsulfonyl fluoride, and 200 μg/ml trypsin soybean inhibitor). After 10 to 15 min of incubation on ice, a homogeneous suspension was prepared and then centrifuged at 23,000 × g for 15 min at 4°C. Supernatant devoid of fecal debris was collected and stored in aliquots at −70°C until assayed.
Measurement of immune responses.
Total and Campylobacter antigen-specific IgA in fecal extracts were determined by direct enzyme-linked immunosorbent assay (ELISA) (2, 3). For total IgA, alternating wells of MaxiSorp 96-well immunoplates (Nunc) were coated with 100 μl of coating buffer containing 2 μg/ml goat anti-mouse IgA (Kirkegaard and Perry Laboratories) or 10 μg/ml bovine serum albumin (Sigma Chemical, St. Louis, MO) and incubated at 37°C for 1 h followed by an overnight incubation at 4°C. Concentrations (3 to 300 ng/ml) of purified mouse IgA (Bethyl Laboratories, Montgomery, TX) were used as a standard. Dilutions of samples and standards were added (100 μl/well) in duplicate wells and incubated at 37°C for 2 h. Plates were then washed and 100 ml of buffer containing 0.25 μg/ml of horseradish peroxidase-conjugated goat anti-mouse IgA (Kirkegaard and Perry Laboratories, Gaithersburg, MD) was added. Following 2 h of incubation, captured antibodies were detected using 2,2′-azino-di(3-ethyl-benzthiazoline-6-sulfonate) peroxidase substrate and by measuring the optical density at 405 nm. The total IgA in each sample was adjusted to 150 ± 25 μg/ml.
FspA1-, FspA2-, or FlaC-specific serum IgA, IgG, and fecal IgA were determined by ELISA as previously described (2, 3). Plates were coated with 100 μl of 0.3 μg/ml of each antigen or 10 μg/ml of bovine serum albumin in coating buffer, and peroxidase-conjugated goat anti-mouse IgA (α chain; 0.25 mg/ml) and IgG (γ chain; 0.125 mg/ml) were used as detecting antibodies. Antigen-specific endpoint titers (reciprocal of highest dilution showing a net optical density at 405 nm of 0.15 to 0.29) were ln transformed; averages were calculated and presented as geometric means and standard deviations. The antigen-specific serum IgG subclasses with horseradish peroxidase-conjugated goat anti-mouse antibodies (Bethyl Laboratories, Montgomery, TX) were used to determine IgG1, IgG2a, IgG2b, and IgG3 levels by direct ELISA, and then IgG1/(IgG2a + IgG2b) ratios were calculated (36).
Challenge with C. jejuni.
Twenty-eight days following the last vaccination, mice were intranasally challenged with 3 × 109 CFU of C. jejuni 81-176 or CG8486. The details of challenge procedures are described elsewhere (3). Following challenge, animals were observed for 5 days, and an illness score was assigned to individual mice with a minor modification (apparently healthy, 0; ruffled fur, 1; ruffled fur plus hunched back, 2) from what was previously described (3). The mean illness indices of the group were used to determine average vaccine efficacies as follows: (illness index control mice − illness index vaccinated mice)/(illness index control mice) × 100. Following challenge, the fecal excretion of C. jejuni was monitored for nine consecutive days by use of Campylobacter select medium (CVA; Remel) as described previously (2, 3).
Statistics.
Comparisons were made using an analysis of variance with a Tukey-Kramer correction to adjust for multiple comparisons.
RESULTS
Safety.
No apparent side effects were noted for mice receiving up to 100 μg of any of the three proteins intranasally. Delivery of 100 μg of FspA1 with 1 μg of LTR192G caused a mild ruffled-fur appearance in all mice that lasted <24 h.
Immune responses. (i) FlaC.
Vaccination with an amount of FlaC lower than 100 μg without the adjuvant induced levels of antigen-specific serum IgG and IgA that were indistinguishable from those of PBS recipients (Table 1). Compared to what was seen for FlaC alone, significantly higher levels of serum IgA and IgG were detected in ≥90% of the animals when 25 μg or more of FlaC was delivered with the adjuvant. Vaccination with PBS or various doses of FlaC with or without the adjuvant did not alter the total amount of secretory IgA (sIgA) in stool. FlaC tested alone at all doses failed to stimulate significant (compared to PBS, P > 0.05) levels of antigen-specific fecal IgA. However, compared to PBS or FlaC without adjuvant, ≥25 μg of FlaC delivered with the adjuvant induced significantly higher levels of fecal IgA (P < 0.001). A clear vaccine dose-dependent response was seen for the magnitude of the response as well as for the numbers of responders for each dose.
TABLE 1.
Systemic and mucosal immune responses in mice vaccinated with escalating doses of recombinant FlaC
| FlaC immunization dose (μg) | Presence of LTR192Ga | Serum response to FlaC [geometric mean ± SD (% responders)]b
|
Fecal IgA response
|
||
|---|---|---|---|---|---|
| IgG | IgA | FlaC [geometric mean ± SD (% responders)] | Totalc (geometric mean ± SD μg/ml) | ||
| 0 | −/+ | 3.4 ± 0.3 a (0) | 3.4 ± 0.3 a (0) | 1.7 ± 0.3 a (0) | 191 ± 93 |
| 5 | − | 4.6 ± 1.0 a,b (10) | 3.9 ± 0 a (0) | 1.4 ± 0 a (0) | 145 ± 79 |
| + | 5.1 ± 1.5 a,b (22) | 4.2 ± 0.6 a (10) | 1.7 ± 0.7 a (10) | 187 ± 81 | |
| 25 | − | 5.4 ± 1.4 a,b (56) | 4.5 ± 1.2 a (30) | 1.4 ± 0 a (0) | 194 ± 77 |
| + | 11.2 ± 1.8 c (100) | 6.7 ± 1.8 b (90) | 4.0 ± 1.2 b (90) | 168 ± 46 | |
| 100 | − | 8.9 ± 2.5 d (100) | 7.9 ± 1.7 b (90) | 2.1 ± 1.2 a (30) | 152 ± 90 |
| + | 13.3 ± 0.3 e (100) | 9.8 ± 1.0 c (100) | 6.0 ± 0.9 c (100) | 155 ± 84 | |
−, absent; +, present.
The responder cutoff values were determined using respective means ± 3 standard deviations of titers in mice receiving PBS. The responders were the individual animals showing an endpoint titer of FlaC-specific serum IgA or IgG of >1:100 or fecal IgA of >1:10. Group means with a different letter within a column are significantly different as defined above (P < 0.05).
% Responders for total IgA values are not applicable.
(ii) FspA.
The systemic and mucosal immune responses to homologous and heterologous antigens in mice immunized with 5 or 25 μg of FspA1 or FspA2 alone or with 1 μg of LTR192G are summarized in Table 2. No significant levels of anti-FspA1- or -FspA2-specific immune responses were detected for control animals. Immunization with 5 μg of FspA1 induced high levels of FspA1-specific serum IgG and IgA, which increased significantly with dose escalation. Coadministration of 5 or 25 μg of FspA1 with LTR192G resulted in equally enhanced levels of systemic immune responses with no clear vaccine dose response. Immunization with FspA1 at either dose resulted in 100% responder rates for all animals. In contrast, FspA2 was a relatively weaker immunogen; none (0%) or 57% of the animals receiving 25 μg of FspA2 alone resulted in the induction of anti-FspA2-specific serum IgA or IgG, respectively (Table 2). However, immune responses to 5- and 25-μg doses were significantly (P < 0.0001) enhanced in all animals when FspA2 was delivered with the adjuvant.
TABLE 2.
FspA-specific homologous and heterologous systemic and mucosal immune responses in mice vaccinated with recombinant FspA1 or FspA2
| Vaccination protein | Dose (μg) | Presence of LTR192Ga | Response [geometric mean ± SD (% respondersb)] for:
|
Total IgAc (geometric mean ± SD μg/ml) | |||||
|---|---|---|---|---|---|---|---|---|---|
| Serum IgG to:
|
Serum IgA to:
|
Fecal IgA to:
|
|||||||
| FspA1 | FspA2 | FspA1 | FspA2 | FspA1 | FspA2 | ||||
| None | 0 | −/+ | 4.1 ± 0.5 a (10) | 3.9 ± 0 a (0) | 3.9 ± 0.2 a (0) | 3.5 ± 0.5 a (0) | 1.8 ± 0.3 a (0) | 1.8 ± 0.1 a (0) | 161 ± 49 a |
| FspA1 | 5 | − | 11.5 ± 1.4 b (100) | 6.8 ± 1.2 b (100) | 6.6 ± 1.7 b (100) | 3.3 ± 0.3 a (0) | 4.1 ± 0.6 b (100) | 1.8 ± 0.1 a,b (0) | 157 ± 66 a |
| + | 14.7 ± 0.6 c (100) | 10.7 ± 1.1 c (100) | 10.4 ± 0.5 c (100) | 4.4 ± 0.3 a (0) | 6.7 ± 0.6 c (100) | 3.1 ± 1.1 b,d (60) | 174 ± 53 a | ||
| 25 | − | 13.1 ± 1.2 d (100) | 8.6 ± 1.4 d (100) | 8.5 ± 0.9 d (100) | 3.9 ± 0 a (0) | 5.2 ± 1.0 b (100) | 2.3 ± 1.2 a,b,d,e (20) | 183 ± 44 a | |
| + | 15.6 ± 1.1 c (100) | 9.9 ± 1.1 c,e (100) | 11.2 ± 0.7 e (100) | 4.3 ± 1.1 a (20) | 7.9 ± 0.8 c (100) | 3.8 ± 1.7 a,b,d,e (70) | 168 ± 67 a | ||
| FspA2 | 5 | − | NDd | ND | ND | ND | ND | ND | ND |
| + | 10.8 ± 1.8 b (100) | 12.6 ± 0.7 e (100) | 5.6 ± 0.7 b (100) | 7.2 ± 0.9 b (100) | 4.0 ± 1.6 b (100) | 5.9 ± 0.6 g (100) | 173 ± 49 a | ||
| 25 | − | 5.9 ± 0.7 a (0) | 7.5 ± 1.7 b (57) | 3.6 ± 0.4 a (0) | 3.9 ± 0 a (0) | 1.8 ± 0 a (0) | 1.8 ± 0 a,b,d,e (0) | 131 ± 89 a | |
| + | 11.4 ± 1.2 b (100) | 13.4 ± 0.2 e,f (100) | 6.3 ± 0.6 b,c (100) | 8.5 ± 0.4 c (100) | 3.8 ± 1.1 b (100) | 5.6 ± 0.9 c,f,g (100) | 163 ± 81 a | ||
−, absent; +, present.
The responder cutoff values were determined using respective means ± 3 standard deviations of titers in mice receiving PBS. The responders were the individual animals showing an endpoint titer for serum IgG to FspA1 of >1:200 and to FspA2 of >1:50 and to both antigens for serum IgA of >1:80, and for fecal IgA of >1:8. Group means with a different letter within a column are significantly different as defined above (P < 0.05).
% Responders for total IgA values are not applicable.
ND, not determined.
The levels of fecal IgA were determined as a direct measure of mucosal immune responses. Irrespective of immunization, the total level of IgA in stool remained unchanged (Table 2). Although immunization with 5 or 25 μg of FspA1 resulted in the induction of homologous antigen-specific fecal IgA in 100% of the recipients, the magnitude of the responses was significantly (P < 0.0001) higher when similar doses were delivered with the adjuvant. In contrast to what was seen for FspA1, groups of mice receiving FspA2 alone showed levels of FspA2-specific sIgA similar to those of PBS recipients (P > 0.05). Adjuvanted FspA2 significantly (P = 0.001) enhanced FspA2-specific sIgA in stool at both dose levels.
Due to the diverse nature of FspA proteins, levels of cross-reacting immunoglobulins were determined (Table 2). Both 5 and 25 μg of FspA1 delivered without adjuvant induced cross-reacting serum IgG to FspA2 (100% responders), but 25 μg (5 μg not done) of FspA2 failed to elicit a significant IgG response to FspA1. However, both proteins induced similarly high levels of heterologous serum IgG when delivered with adjuvant. In contrast to serum IgG, 5 or 25 μg of FspA1 alone or with adjuvant did not elicit cross-reacting serum IgA, whereas adjuvanted FspA2 did. Additionally, adjuvanted FspA1 and FspA2 successfully induced fecal IgA (60 to 100% responders) to the heterologous antigen, though the levels were relatively low compared to the respective homologous responses.
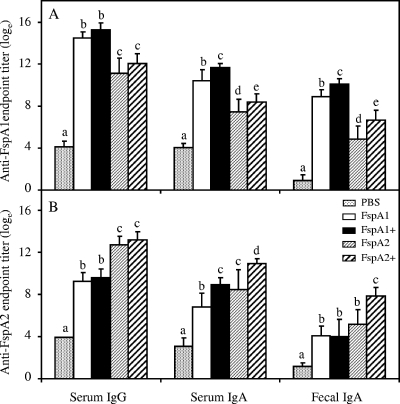
The immune responses to 100 μg of both FspA proteins are summarized in Fig. 1. Unlike what was seen for lower doses, all animals receiving 100 μg of vaccine with or without the adjuvant mounted similarly high serum IgG responses to respective and cross-reacting FspA antigens. In contrast to serum IgG, an adjuvant effect in enhancing homologous as well as heterologous FspA1- or FspA2-specific serum IgA was seen following vaccination. At this dose, serum immune responses to both antigens were high in all animals (100% response rate).
FIG. 1.
Comparative immunogenicity of 100 μg of FspA1 and FspA2 proteins with and without LTR192G. Plus signs indicate proteins that were delivered with the adjuvant LTR192G. All group mean comparisons made with PBS were significantly different (P < 0.05). No responders were observed for animals receiving PBS, while 100% of the animals were categorized as responders under all conditions except fecal IgA, for which the following responder levels were shown: for anti-FspA1 in FspA2-immunized mice, 86%; for anti-FspA2 in FspA1-immunized mice, 85%; for anti-FspA2 in FspA1-plus-LTR192G-immunized mice, 78%; and for anti-FspA2 in FspA2-immunized mice, 78%. Letters a to e indicate pairwise comparisons for each test; bars with different letters within a comparison graph are significantly different (P < 0.05).
Like lower doses (data presented in Table 2), the total IgA concentrations in stool after vaccination with 100-μg doses of either vaccine formulation remained the same (168 ± 67, 194 ± 87, 198 ± 96, and 190 ± 53 μg/ml for FspA1, FspA1 plus LTR192G, FspA2, and FspA2 plus LTR192G, respectively; P > 0.05). Following immunization with FspA1 or FspA2, fecal IgA responses to homologous antigens were significantly (P < 0.001) enhanced by the adjuvant (Fig. 1). In contrast to what was seen at lower doses, heterologous antigen recognition was detected in animals immunized with 100 μg of FspA2. Furthermore, these cross-reacting anti-FspA1 sIgA antibodies were augmented when FspA2 was administered with adjuvant (Fig. 1A; fecal IgA marked as d and e). The reciprocal adjuvant-dependent enhancement of cross-reacting sIgA in FspA1 immunization was not detected. A 100% response rate was observed for both FspA1 and FspA2 proteins when homologous sIgA was detected, whereas response rates for the detection of heterologous sIgA varied from 78% to 86% (legend to Fig. 1).
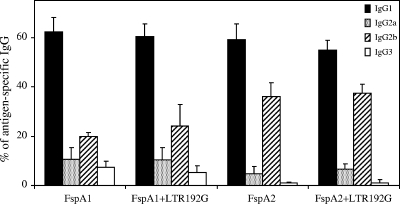
Serum IgG subclasses.
In an effort to further characterize the types of immune responses generated as the result of FspA immunizations, serum IgG subclasses to homologous antigens were determined (Fig. 2). Both FspA1 and FspA2 induced similar levels of antigen-specific IgG1. FspA2 alone or with the adjuvant failed to induce IgG3, while low levels (5 to 7%) of IgG3 were detected after FspA1 immunization. IgG2a and IgG2b levels after immunization were distinct between the groups. For FspA1, over 10% of all antigen-specific IgG corresponded to IgG2a and 20% to IgG2b (IgG2a/IgG2b ratio, 0.5), whereas for FspA2, the corresponding values were 5% and 36% (IgG2a/IgG2b ratio, 0.14). The calculated IgG subclass ratios (IgG1)/(IgG2a + IgG2b) for FspA1, adjuvanted FspA1, FspA2, and adjuvanted FspA2 were 2.13 ± 0.58, 2.08 ± 0.42, 1.51 ± 0.40, and 1.37 ± 0.52, respectively, suggesting a mixed Th1- and Th2-type response to homologous antigens following vaccination (Fig. 2).
FIG. 2.
Serum IgG subclass distribution following vaccination with FspA1 or FspA2 proteins. Immunization with either protein alone or with the adjuvant was more frequently associated with induction of IgG1 followed by IgG2b, IgG2a, and IgG3.
Protective efficacy.
Twenty-eight days following vaccination, animals immunized with 100 μg of each protein with or without LTR192G were challenged with homologous or heterologous strains of C. jejuni. The results are summarized in Table 3. FlaC failed to provide protection against C. jejuni 81-176 infection, as no significant differences in illness patterns for PBS and FlaC recipients were noted (P > 0.05). Animals immunized with FspA1 or FspA2 and challenged with the homologous strain of C. jejuni showed a significantly lower level of illness. FspA1 alone or with the adjuvant provided similar levels of protection (63.8% or 57.8%) from C. jejuni 81-176 challenge. FspA2 provided somewhat lower, albeit significant, protection against C. jejuni CG8486 challenge (FspA2 alone 47.2% versus with adjuvant 38.4%; P < 0.001). FspA1 immunization induced cross-reactive immunity to challenge with heterologous strain CG8486 (P ≤ 0.0001), but immunization with FspA2 did not provide significant protection against challenge with heterologous strain 81-176 (P = 0.13). Since mice become intestinally colonized following intranasal challenge with C. jejuni (3), fecal excretion of C. jejuni was also followed. However, immunization had no effect on either the magnitude or the duration of colonization (data not shown).
TABLE 3.
Protective efficacy of recombinant flagellum-secreted proteins against C. jejuni 81-176 or C. jejuni CG8486 challenge
| Vaccination protein | Presence of LTR192Gb | Challenge withc:
|
|||
|---|---|---|---|---|---|
| 81-176
|
CG8486
|
||||
| Illness index (mean ± SD) | Efficacy (%) | Illness index (mean ± SD) | Efficacy (%) | ||
| PBSa | −/+ | 1.16 ± 0.21 | NA | 1.25 ± 0.24 | NA |
| FspA1 | − | 0.49 ± 0.17† | 57.8 | 0.69 ± 0.21† | 44.8 |
| FspA1 | + | 0.42 ± 0.14† | 63.8 | 0.86 ± 0.28† | 31.2 |
| FspA2 | − | 0.95 ± 0.12 | 18.1 | 0.77 ± 0.27† | 38.4 |
| FspA2 | + | 1.20 ± 0.29 | −3.4 | 0.66 ± 0.18† | 47.2 |
| FlaC | − | 1.03 ± 0.27 | 11.2 | ND | NA |
| FlaC | + | 0.95 ± 0.29 | 18.1 | ND | NA |
There was no difference in challenge outcome among animals receiving PBS alone or PBS with 1 μg of LTR192G; therefore data were pooled for efficacy calculations.
−, absent; +, present.
†, Significant difference (P < 0.05) with PBS group; NA, not applicable; ND, not determined.
DISCUSSION
Nonflagellar proteins secreted through the flagellar filament have been reported to modulate the virulence of C. jejuni (14, 15, 23, 28, 35). The potential of these flagellum-secreted proteins to induce a protective immune response was compared in a mouse intranasal model of infection. Serum IgG and IgA and fecal IgA responses to homologous antigens following immunization with FlaC, FspA1, and FspA2 with and without adjuvant were compared.
FspA1 and FspA2, two variant forms of FspA which share only 33% identity at the protein level, produced strong homologous responses in a dose-dependent manner. Compared to FspA2, FspA1 was a strong immunogen in inducing both systemic and mucosal immune responses. For both antigens, doses of ≤25 μg required adjuvant to achieve near-maximum levels of serum IgG, although at 100-μg dose levels both proteins induced similar levels of homologous systemic immune responses. In addition to homologous responses, both proteins generated cross-reacting serum antibodies, although FspA2 immunization resulted in relatively higher levels of anti-FspA1 antibodies. However, and more importantly, at 100-μg doses homologous immune responses induced by the two antigens were indistinguishable. Serum IgG subclass ratios were used as an indication of T-cell-type responses resulting from vaccination with FspA1 and FspA2 (similar evaluations for FlaC were not done). The calculated [IgG1/(IgG2a + IgG2b)] ratios were moderately low, suggesting mixed Th1- and Th2-type responses. The responses measured by IgG subclass analysis were apparently not of a predominately Th1 type, yet upon challenge a good protection against diseases was seen. The induction of gamma interferon as an indicator of T-cell activation was not assessed. We have previously shown that after infection in humans, a Th1-type response dominates (4). The role of cellular immune response in acquired immunity to Campylobacter is not fully understood.
The response pattern observed for systemic responses was not seen when the mucosal immunogenicity of FspA proteins was assessed. In this context, FspA induced antigen-specific fecal IgA responses that clearly increased as a function of both the quantity of vaccine and the vaccine formulation administered (Table 1 and Fig. 1); the highest vaccine dose delivered with the mucosal adjuvant produced the highest level of antigen-specific fecal IgA. There are only a few reports in the literature of other potential Campylobacter subunit proteins for comparison to these studies (17, 26, 34). The magnitude of the serum immune responses detected after vaccination with these flagellum-secreted proteins was comparable to those reported previously for a truncated flagellin fused to maltose binding protein (MBP-FlaA) in mice (17). However, it is difficult to compare the immune responses directly, since that vaccine has been shown to be heavily contaminated with endotoxin from the E. coli host (L. H. Lee, unpublished data). The sIgA levels reported here are higher than what was reported previously for that flagellin subunit vaccine, although there were also some differences in the technical procedures employed (fecal pellets versus intestinal lavage).
Despite a strong immune response, immunization with FlaC did not provide protection against challenge with 81-176. In contrast, immunization with FspA2 elicited an immune response that was protective only against C. jejuni strain CG8486, which secretes FspA2, and not against strain 81-176, which secretes FspA1. Immunization with FspA1 resulted in significant protection against challenge with C. jejuni 81-176 (58 to 64% efficacy) and a lower but still significant level of protection against CG8486 (31 to 45% efficacy). This difference in terms of cross-protective responses of the FspA proteins is not fully understood but may relate to differences in the folding of the two proteins; alternatively, the antigen-specific responses may be independently regulated. The protection reported here is somewhat lower than that attributed to a subunit flagellin vaccine reported previously using this same animal model (17). As mentioned above, one of the differences may be in part due to the large amount of contaminating endotoxin in the flagellin vaccine, or it may be in the type of acquired immunity generated by different proteins.
Since some of the proteins that are secreted through C. jejuni flagella are associated with virulence (14, 15, 23, 28, 35), the present study was designed to evaluate some of these proteins as potential subunit vaccines against disease. FspA2 has been shown to induce apoptosis in intestinal epithelial cells, but no phenotype has been associated with FspA1 (23). The fact that FspA1 protected against disease but not intestinal colonization by 81-176 suggests that this protein plays a role in the pathogenesis of this strain in this model. Collectively, the data suggest that FspA1 is a promising candidate for a mucosal subunit vaccine either by itself or in combination with other C. jejuni proteins.
Acknowledgments
These studies were supported by U.S. Navy Research and Development Command Work Units 62787A.001.01.EVX.1522, 63002A.001.01.HOX.1294, and 00101.HOX.3416.
The views expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Department of the Navy, the Department of Defense, or the U.S. government.
Editor: J. B. Bliska
Footnotes
Published ahead of print on 21 April 2008.
REFERENCES
- 1.Allos, B. M. 2001. Campylobacter jejuni infections: update on emerging issues and trends. Clin. Infect. Dis. 321201-1206. [DOI] [PubMed] [Google Scholar]
- 2.Baqar, S., L. A. Applebee, and A. L. Bourgeois. 1995. Immunogenicity and protective efficacy of a prototype Campylobacter killed whole-cell vaccine in mice. Infect. Immun. 633731-3735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Baqar, S., A. L. Bourgeois, L. A. Applebee, A. S. Mourad, M. T. Kleinosky, Z. Mohran, and J. R. Murphy. 1996. Murine intranasal challenge model for the study of Campylobacter pathogenesis and immunity. Infect. Immun. 644933-4939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Baqar, S., B. Rice, L. Lee, A. L. Bourgeois, A. N. El Din, D. R. Tribble, G. P. Heresi, A. S. Mourad, and J. R. Murphy. 2001. Campylobacter jejuni enteritis. Clin. Infect. Dis. 33901-905. [DOI] [PubMed] [Google Scholar]
- 5.Blaser, M. J., D. J. Duncan, M. T. Osterholm, G. R. Istre, and W. L. Wang. 1983. Serologic study of two clusters of infection due to Campylobacter jejuni. J. Infect. Dis. 147820-823. [DOI] [PubMed] [Google Scholar]
- 6.Bourgeois, A. L., C. H. Gardiner, S. A. Thornton, R. A. Batchelor, D. H. Burr, J. Escamilla, P. Echeverria, N. R. Blacklow, J. E. Herrmann, and K. C. Hyams. 1993. Etiology of acute diarrhea among United States military personnel deployed to South America and west Africa. Am. J. Trop. Med. Hyg. 48243-248. [DOI] [PubMed] [Google Scholar]
- 7.Coker, A. O., R. D. Isokpehi, B. N. Thomas, K. O. Amisu, and C. L. Obi. 2002. Human campylobacteriosis in developing countries. Emerg. Infect. Dis. 8237-244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Dickinson, B. L., and J. D. Clements. 1995. Dissociation of Escherichia coli heat-labile enterotoxin adjuvanticity from ADP-ribosyltransferase activity. Infect. Immun. 631617-1623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Engberg, J., J. Neimann, E. M. Nielsen, F. M. Aerestrup, and V. Fussing. 2004. Quinolone-resistant Campylobacter infections: risk factors and clinical consequences. Emerg. Infect. Dis. 101056-1063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Frost, J. A. 2001. Current epidemiological issues in human campylobacteriosis. Symp. Ser. Soc. Appl. Microbiol. 200185S-95S. [DOI] [PubMed] [Google Scholar]
- 11.Goon, S., C. P. Ewing, M. Lorenzo, D. Pattarini, G. Majam, and P. Guerry. 2006. A σ28-regulated nonflagella gene contributes to virulence of Campylobacter jejuni 81-176. Infect. Immun. 74769-772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Guerry, P. 2007. Campylobacter flagella: not just for motility. Trends Microbiol. 15456-461. [DOI] [PubMed] [Google Scholar]
- 13.Hannu, T., L. Mattila, H. Rautelin, P. Pelkonen, P. Lahdenne, A. Siitonen, and M. Leirisalo-Repo. 2002. Campylobacter-triggered reactive arthritis: a population-based study. Rheumatology (Oxford) 41312-318. [DOI] [PubMed] [Google Scholar]
- 14.Konkel, M. E., B. J. Kim, V. Rivera-Amill, and S. G. Garvis. 1999. Bacterial secreted proteins are required for the internalization of Campylobacter jejuni into cultured mammalian cells. Mol. Microbiol. 32691-701. [DOI] [PubMed] [Google Scholar]
- 15.Konkel, M. E., J. D. Klena, V. Rivera-Amill, M. R. Monteville, D. Biswas, B. Raphael, and J. Mickelson. 2004. Secretion of virulence proteins from Campylobacter jejuni is dependent on a functional flagellar export apparatus. J. Bacteriol. 1863296-3303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kuwabara, S. 2007. Guillain-Barre syndrome. Curr. Neurol. Neurosci. Rep. 757-62. [DOI] [PubMed] [Google Scholar]
- 17.Lee, L. H., E. Burg III, S. Baqar, A. L. Bourgeois, D. H. Burr, C. P. Ewing, T. J. Trust, and P. Guerry. 1999. Evaluation of a truncated recombinant flagellin subunit vaccine against Campylobacter jejuni. Infect. Immun. 675799-5805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mishu, B., A. A. Ilyas, C. L. Koski, F. Vriesendorp, S. D. Cook, F. A. Mithen, and M. J. Blaser. 1993. Serologic evidence of previous Campylobacter jejuni infection in patients with the Guillain-Barre syndrome. Ann. Intern. Med. 118947-953. [DOI] [PubMed] [Google Scholar]
- 19.National Research Council. 1996. Guide for the care and use of laboratory animals. National Academy Press, Washington, DC.
- 20.Pawelec, D. P., D. Korsak, A. K. Wyszynska, E. Rozynek, J. Popowski, and E. K. Jagusztyn-Krynicka. 2000. Genetic diversity of the Campylobacter genes coding immunodominant proteins. FEMS Microbiol. Lett. 18543-49. [DOI] [PubMed] [Google Scholar]
- 21.Perera, V. N., I. Nachamkin, H. Ung, J. H. Patterson, M. J. McConville, P. J. Coloe, and B. N. Fry. 2007. Molecular mimicry in Campylobacter jejuni: role of the lipo-oligosaccharide core oligosaccharide in inducing anti-ganglioside antibodies. FEMS Immunol. Med. Microbiol. 5027-36. [DOI] [PubMed] [Google Scholar]
- 22.Pickering, L. K. 2004. Antimicrobial resistance among enteric pathogens. Semin. Pediatr. Infect. Dis. 1571-77. [DOI] [PubMed] [Google Scholar]
- 23.Poly, F., C. Ewing, S. Goon, T. E. Hickey, D. Rockabrand, G. Majam, L. Lee, J. Phan, N. J. Savarino, and P. Guerry. 2007. Heterogeneity of a Campylobacter jejuni protein that is secreted through the flagellar filament. Infect. Immun. 753859-3867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Poly, F., T. Read, D. R. Tribble, S. Baqar, M. Lorenzo, and P. Guerry. 2007. Genome sequence of a clinical isolate of Campylobacter jejuni from Thailand. Infect. Immun. 753425-3433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Pope, J. E., A. Krizova, A. X. Garg, H. Thiessen-Philbrook, and J. M. Ouimet. 2007. Campylobacter reactive arthritis: a systematic review. Semin. Arthritis Rheum. 3748-55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Prokhorova, T. A., P. N. Nielsen, J. Petersen, T. Kofoed, J. S. Crawford, C. Morsczeck, A. Boysen, and P. Schrotz-King. 2006. Novel surface polypeptides of Campylobacter jejuni as traveller's diarrhoea vaccine candidates discovered by proteomics. Vaccine 246446-6455. [DOI] [PubMed] [Google Scholar]
- 27.Riddle, M. S., J. W. Sanders, S. D. Putnam, and D. R. Tribble. 2006. Incidence, etiology, and impact of diarrhea among long-term travelers (US military and similar populations): a systematic review. Am. J. Trop. Med. Hyg. 74891-900. [PubMed] [Google Scholar]
- 28.Rivera-Amill, V., B. J. Kim, J. Seshu, and M. E. Konkel. 2001. Secretion of the virulence-associated Campylobacter invasion antigens from Campylobacter jejuni requires a stimulatory signal. J. Infect. Dis. 1831607-1616. [DOI] [PubMed] [Google Scholar]
- 29.Rollwagen, F. M., N. D. Pacheco, J. D. Clements, O. Pavlovskis, D. M. Rollins, and R. I. Walker. 1993. Killed Campylobacter elicits immune response and protection when administered with an oral adjuvant. Vaccine 111316-1320. [DOI] [PubMed] [Google Scholar]
- 30.Ruiz, J., F. Marco, I. Oliveira, J. Vila, and J. Gascon. 2007. Trends in antimicrobial resistance in Campylobacter spp. causing traveler's diarrhea. APMIS 115218-224. [DOI] [PubMed] [Google Scholar]
- 31.Sanders, J. W., D. W. Isenbarger, S. E. Walz, L. W. Pang, D. A. Scott, C. Tamminga, B. A. Oyofo, W. C. Hewitson, J. L. Sanchez, C. Pitarangsi, P. Echeverria, and D. R. Tribble. 2002. An observational clinic-based study of diarrheal illness in deployed United States military personnel in Thailand: presentation and outcome of Campylobacter infection. Am. J. Trop. Med. Hyg. 67533-538. [DOI] [PubMed] [Google Scholar]
- 32.Sanders, J. W., S. D. Putnam, P. Gould, J. Kolisnyk, N. Merced, V. Barthel, P. J. Rozmajzl, H. Shaheen, S. Fouad, and R. W. Frenck. 2005. Diarrheal illness among deployed U.S. military personnel during Operation Bright Star 2001—Egypt. Diagn. Microbiol. Infect. Dis. 5285-90. [DOI] [PubMed] [Google Scholar]
- 33.Scott, D. A. 1997. Vaccines against Campylobacter jejuni. J. Infect. Dis. 176(Suppl 2)S183-S188. [DOI] [PubMed] [Google Scholar]
- 34.Sizemore, D. R., B. Warner, J. Lawrence, A. Jones, and K. P. Killeen. 2006. Live, attenuated Salmonella typhimurium vectoring Campylobacter antigens. Vaccine 243793-3803. [DOI] [PubMed] [Google Scholar]
- 35.Song, Y. C., S. Jin, H. Louie, D. Ng, R. Lau, Y. Zhang, R. Weerasekera, S. Al Rashid, L. A. Ward, S. D. Der, and V. L. Chan. 2004. FlaC, a protein of Campylobacter jejuni TGH9011 (ATCC43431) secreted through the flagellar apparatus, binds epithelial cells and influences cell invasion. Mol. Microbiol. 53541-553. [DOI] [PubMed] [Google Scholar]
- 36.Strindelius, L., M. Filler, and I. Sjoholm. 2004. Mucosal immunization with purified flagellin from Salmonella induces systemic and mucosal immune responses in C3H/HeJ mice. Vaccine 223797-3808. [DOI] [PubMed] [Google Scholar]
- 37.Taylor, D. N., D. M. Perlman, P. D. Echeverria, U. Lexomboon, and M. J. Blaser. 1993. Campylobacter immunity and quantitative excretion rates in Thai children. J. Infect. Dis. 168754-758. [DOI] [PubMed] [Google Scholar]
- 38.Tribble, D. R., J. W. Sanders, L. W. Pang, C. Mason, C. Pitarangsi, S. Baqar, A. Armstrong, P. Hshieh, A. Fox, E. A. Maley, C. Lebron, D. J. Faix, J. V. Lawler, G. Nayak, M. Lewis, L. Bodhidatta, and D. A. Scott. 2007. Traveler's diarrhea in Thailand: randomized, double-blind trial comparing single-dose and 3-day azithromycin-based regimens with a 3-day levofloxacin regimen. Clin. Infect. Dis. 44338-346. [DOI] [PubMed] [Google Scholar]