Abstract
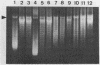
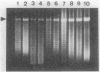
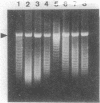
Changes in plasmid DNA supercoiling were measured following treatment of Escherichia coli cells, carrying topoisomerase mutations, with the quinolone ciprofloxacin. In quinolone-susceptible cells (top+ gyr+) as well as in topA mutants and in gyrB mutants, plasmid DNA was relaxed after the addition of ciprofloxacin. In cells partially resistant to quinolones, low ciprofloxacin levels led to an increase in negative superhelicity of plasmid DNA, whereas at higher ciprofloxacin concentrations, DNA became relaxed. Cells exhibiting partial resistance to quinolones carried either a gyrA mutation alone or a combination of gyrA and gyrB mutations. Moreover, they showed a reduction in gyrase activity, indicated by the supercoiling of a reporter plasmid. Therefore, we conclude that a low level of quinolone action and a DNA with a lower-than-normal level of superhelicity are the two essential conditions for obtaining a ciprofloxacin-promoted increase in plasmid DNA supercoiling. In contrast, deficiency in topoisomerase I is not required for this effect.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aleixandre V., Blanco M. Heterogeneity in the level of ampicillin resistance conferred by pBR322 derivatives with different DNA supercoiling. Mol Gen Genet. 1987 Aug;209(1):56–60. doi: 10.1007/BF00329836. [DOI] [PubMed] [Google Scholar]
- Aleixandre V., Urios A., Herrera G., Blanco M. New Escherichia coli gyrA and gyrB mutations which have a graded effect on DNA supercoiling. Mol Gen Genet. 1989 Oct;219(1-2):306–312. doi: 10.1007/BF00261192. [DOI] [PubMed] [Google Scholar]
- Bachmann B. J. Pedigrees of some mutant strains of Escherichia coli K-12. Bacteriol Rev. 1972 Dec;36(4):525–557. doi: 10.1128/br.36.4.525-557.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bliska J. B., Cozzarelli N. R. Use of site-specific recombination as a probe of DNA structure and metabolism in vivo. J Mol Biol. 1987 Mar 20;194(2):205–218. doi: 10.1016/0022-2836(87)90369-x. [DOI] [PubMed] [Google Scholar]
- Chang A. C., Cohen S. N. Construction and characterization of amplifiable multicopy DNA cloning vehicles derived from the P15A cryptic miniplasmid. J Bacteriol. 1978 Jun;134(3):1141–1156. doi: 10.1128/jb.134.3.1141-1156.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DiGate R. J., Marians K. J. Identification of a potent decatenating enzyme from Escherichia coli. J Biol Chem. 1988 Sep 15;263(26):13366–13373. [PubMed] [Google Scholar]
- DiNardo S., Voelkel K. A., Sternglanz R., Reynolds A. E., Wright A. Escherichia coli DNA topoisomerase I mutants have compensatory mutations in DNA gyrase genes. Cell. 1982 Nov;31(1):43–51. doi: 10.1016/0092-8674(82)90403-2. [DOI] [PubMed] [Google Scholar]
- Dorman C. J., Lynch A. S., Ni Bhriain N., Higgins C. F. DNA supercoiling in Escherichia coli: topA mutations can be suppressed by DNA amplifications involving the tolC locus. Mol Microbiol. 1989 Apr;3(4):531–540. doi: 10.1111/j.1365-2958.1989.tb00199.x. [DOI] [PubMed] [Google Scholar]
- Drlica K., Coughlin S. Inhibitors of DNA gyrase. Pharmacol Ther. 1989;44(1):107–121. doi: 10.1016/0163-7258(89)90093-4. [DOI] [PubMed] [Google Scholar]
- Drlica K., Franco R. J. Inhibitors of DNA topoisomerases. Biochemistry. 1988 Apr 5;27(7):2253–2259. doi: 10.1021/bi00407a001. [DOI] [PubMed] [Google Scholar]
- Franco R. J., Drlica K. DNA gyrase on the bacterial chromosome. Oxolinic acid-induced DNA cleavage in the dnaA-gyrB region. J Mol Biol. 1988 May 5;201(1):229–233. doi: 10.1016/0022-2836(88)90449-4. [DOI] [PubMed] [Google Scholar]
- Franco R. J., Drlica K. Gyrase inhibitors can increase gyrA expression and DNA supercoiling. J Bacteriol. 1989 Dec;171(12):6573–6579. doi: 10.1128/jb.171.12.6573-6579.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu L. F. DNA topoisomerase poisons as antitumor drugs. Annu Rev Biochem. 1989;58:351–375. doi: 10.1146/annurev.bi.58.070189.002031. [DOI] [PubMed] [Google Scholar]
- Menzel R., Gellert M. Regulation of the genes for E. coli DNA gyrase: homeostatic control of DNA supercoiling. Cell. 1983 Aug;34(1):105–113. doi: 10.1016/0092-8674(83)90140-x. [DOI] [PubMed] [Google Scholar]
- Pruss G. J. DNA topoisomerase I mutants. Increased heterogeneity in linking number and other replicon-dependent changes in DNA supercoiling. J Mol Biol. 1985 Sep 5;185(1):51–63. doi: 10.1016/0022-2836(85)90182-2. [DOI] [PubMed] [Google Scholar]
- Pruss G. J., Franco R. J., Chevalier S. G., Manes S. H., Drlica K. Effects of DNA gyrase inhibitors in Escherichia coli topoisomerase I mutants. J Bacteriol. 1986 Oct;168(1):276–282. doi: 10.1128/jb.168.1.276-282.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rose K. M. DNA topoisomerases as targets for chemotherapy. FASEB J. 1988 Jun;2(9):2474–2478. doi: 10.1096/fasebj.2.9.2836254. [DOI] [PubMed] [Google Scholar]
- Shen L. L., Mitscher L. A., Sharma P. N., O'Donnell T. J., Chu D. W., Cooper C. S., Rosen T., Pernet A. G. Mechanism of inhibition of DNA gyrase by quinolone antibacterials: a cooperative drug--DNA binding model. Biochemistry. 1989 May 2;28(9):3886–3894. doi: 10.1021/bi00435a039. [DOI] [PubMed] [Google Scholar]
- Wolfson J. S., Hooper D. C. The fluoroquinolones: structures, mechanisms of action and resistance, and spectra of activity in vitro. Antimicrob Agents Chemother. 1985 Oct;28(4):581–586. doi: 10.1128/aac.28.4.581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu H. Y., Shyy S. H., Wang J. C., Liu L. F. Transcription generates positively and negatively supercoiled domains in the template. Cell. 1988 May 6;53(3):433–440. doi: 10.1016/0092-8674(88)90163-8. [DOI] [PubMed] [Google Scholar]