Abstract
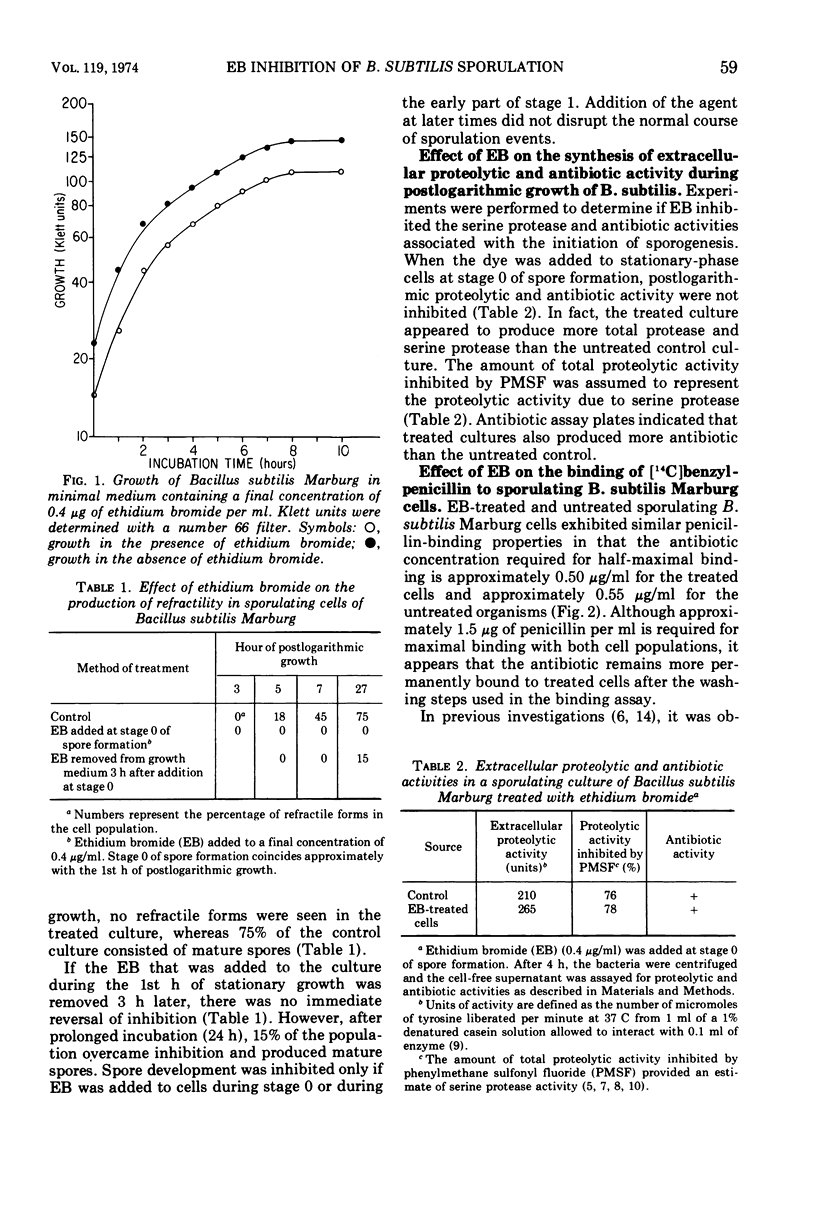
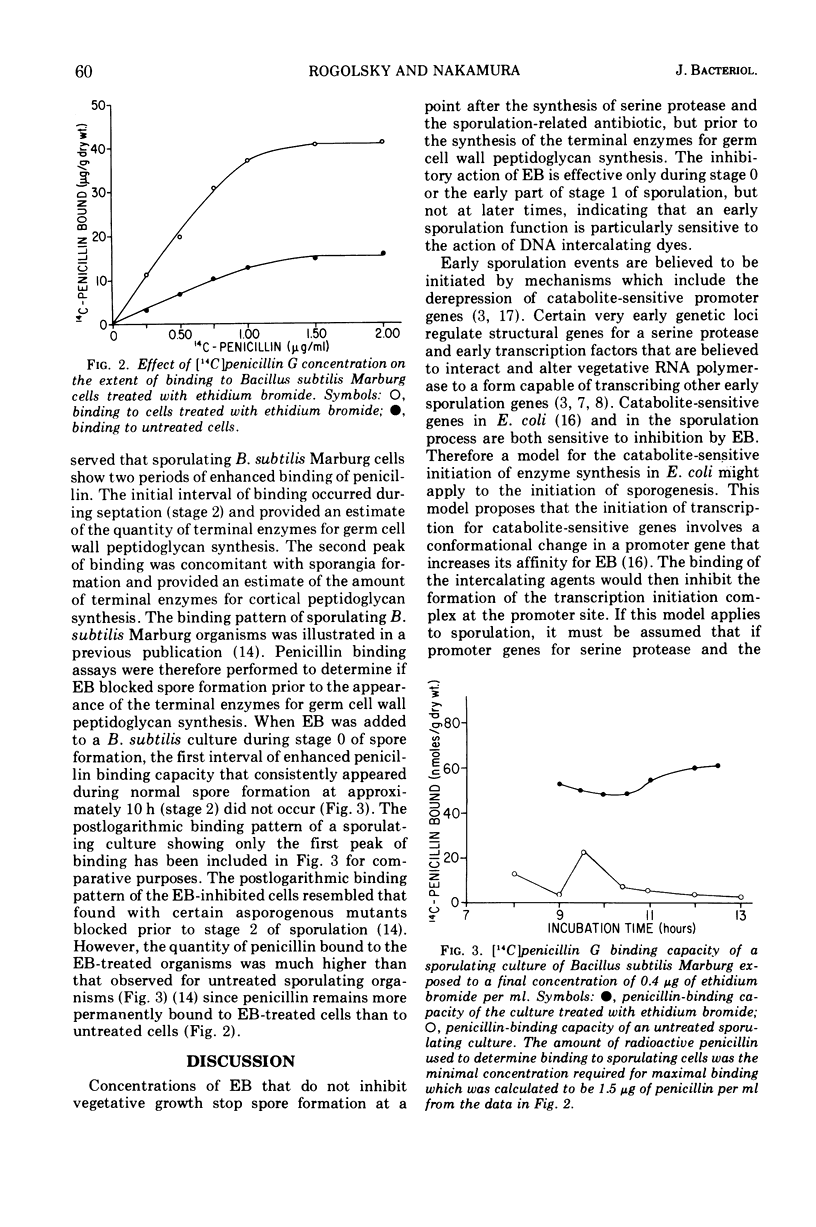
When a final concentration of 0.4 μg of ethidium bromide (EB) per ml, which is subinhibitory to vegetative growth, is added to sporulating cells of Bacillus subtilis Marburg during either stage 0 or the early part of stage 1, morphogenesis is blocked. If the given concentration of EB is added after the early part of stage 1, sporogenesis is unaffected. The synthesis of the serine protease and antibiotic, which are believed to be associated with sporulation events during the early part of stage 0, are not inhibited by EB. Enhanced binding of [14C]benzylpenicillin to sporulating cells during septation (stage 2) is a measure of the presence of terminal enzymes for germ cell wall peptidoglycan synthesis. EB does not interfere with the binding of penicillin to sporulating cells, but penicillin remains more permanently bound to EB-treated postlogarithmic cells than to untreated sporulating cells. The absence of an interval of increased penicillin binding activity during stage 2 by sporulating cells treated with EB indicates that EB blocks sporulation prior to the completion of the germ cell wall.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bouanchaud D. H., Scavizzi M. R., Chabbert Y. A. Elimination by ethidium bromide of antibiotic resistance in enterobacteria and staphylococci. J Gen Microbiol. 1968 Dec;54(3):417–425. doi: 10.1099/00221287-54-3-417. [DOI] [PubMed] [Google Scholar]
- Clowes R. C. Molecular structure of bacterial plasmids. Bacteriol Rev. 1972 Sep;36(3):361–405. doi: 10.1128/br.36.3.361-405.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greenleaf A. L., Losick R. Appearance of a ribonucleic acid polymerase-binding protein in asporogenous mutants of Bacillus subtilis. J Bacteriol. 1973 Oct;116(1):290–294. doi: 10.1128/jb.116.1.290-294.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lawrence P. J., Rogolsky M., Hanh V. T. Binding of radioactive benzylpenicillin to sporulating Bacillus cultures: chemistry and fluctuations in specific binding capacity. J Bacteriol. 1971 Nov;108(2):662–667. doi: 10.1128/jb.108.2.662-667.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leighton T. J., Dor R. H., Warren R. A., Kelln R. A. The relationship of serine protease activity to RNA polymerase modification and sporulation in Bacillus subtilis. J Mol Biol. 1973 May 5;76(1):103–122. doi: 10.1016/0022-2836(73)90083-1. [DOI] [PubMed] [Google Scholar]
- Millet J. Characterization of proteinases excreted by Bacillus subtilis Marburg strain during sporulation. J Appl Bacteriol. 1970 Mar;33(1):207–219. doi: 10.1111/j.1365-2672.1970.tb05245.x. [DOI] [PubMed] [Google Scholar]
- Prestidge L., Gage V., Spizizen J. Protease activities during the course of sporulation on Bacillus subtilis. J Bacteriol. 1971 Sep;107(3):815–823. doi: 10.1128/jb.107.3.815-823.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richardson J. P. Mechanism of ethidium bromide inhibition of RNA polymerase. J Mol Biol. 1973 Aug 25;78(4):703–714. doi: 10.1016/0022-2836(73)90290-8. [DOI] [PubMed] [Google Scholar]
- Richardson J. P., Parker S. R. Letter: Effect of ethidium bromide on transcription of linear and circular DNA templates. J Mol Biol. 1973 Aug 25;78(4):715–720. doi: 10.1016/0022-2836(73)90291-x. [DOI] [PubMed] [Google Scholar]
- Rogolsky M., Lawrence P. J., Hanh V. T. Binding of radioactive benzylpenicillin to asporogenous mutants of Bacillus subtilis during postexponential growth. J Bacteriol. 1973 Apr;114(1):220–227. doi: 10.1128/jb.114.1.220-227.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rogolsky M., Warren R., Wiley B. B., Nakamura H. T., Glasgow L. A. Nature of the genetic determinant controlling exfoliative toxin production in Staphylococcus aureus. J Bacteriol. 1974 Jan;117(1):157–165. doi: 10.1128/jb.117.1.157-165.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sankaran L., Pogell B. M. Differential inhibition of catabolite-sensitive enzyme induction by intercalating dyes. Nat New Biol. 1973 Oct 31;245(148):257–260. doi: 10.1038/newbio245257a0. [DOI] [PubMed] [Google Scholar]
- Schaeffer P., Millet J., Aubert J. P. Catabolic repression of bacterial sporulation. Proc Natl Acad Sci U S A. 1965 Sep;54(3):704–711. doi: 10.1073/pnas.54.3.704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spizizen J. TRANSFORMATION OF BIOCHEMICALLY DEFICIENT STRAINS OF BACILLUS SUBTILIS BY DEOXYRIBONUCLEATE. Proc Natl Acad Sci U S A. 1958 Oct 15;44(10):1072–1078. doi: 10.1073/pnas.44.10.1072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WARING M. J. COMPLEX FORMATION WITH DNA AND INHIBITION OF ESCHERICHIA COLI RNA POLYMERASE BY ETHIDIUM BROMIDE. Biochim Biophys Acta. 1964 Jun 22;87:358–361. doi: 10.1016/0926-6550(64)90238-5. [DOI] [PubMed] [Google Scholar]
- Waring M. J. Structural requirements for the binding of ethidium to nucleic acids. Biochim Biophys Acta. 1966 Feb 21;114(2):234–244. doi: 10.1016/0005-2787(66)90305-4. [DOI] [PubMed] [Google Scholar]



