Abstract
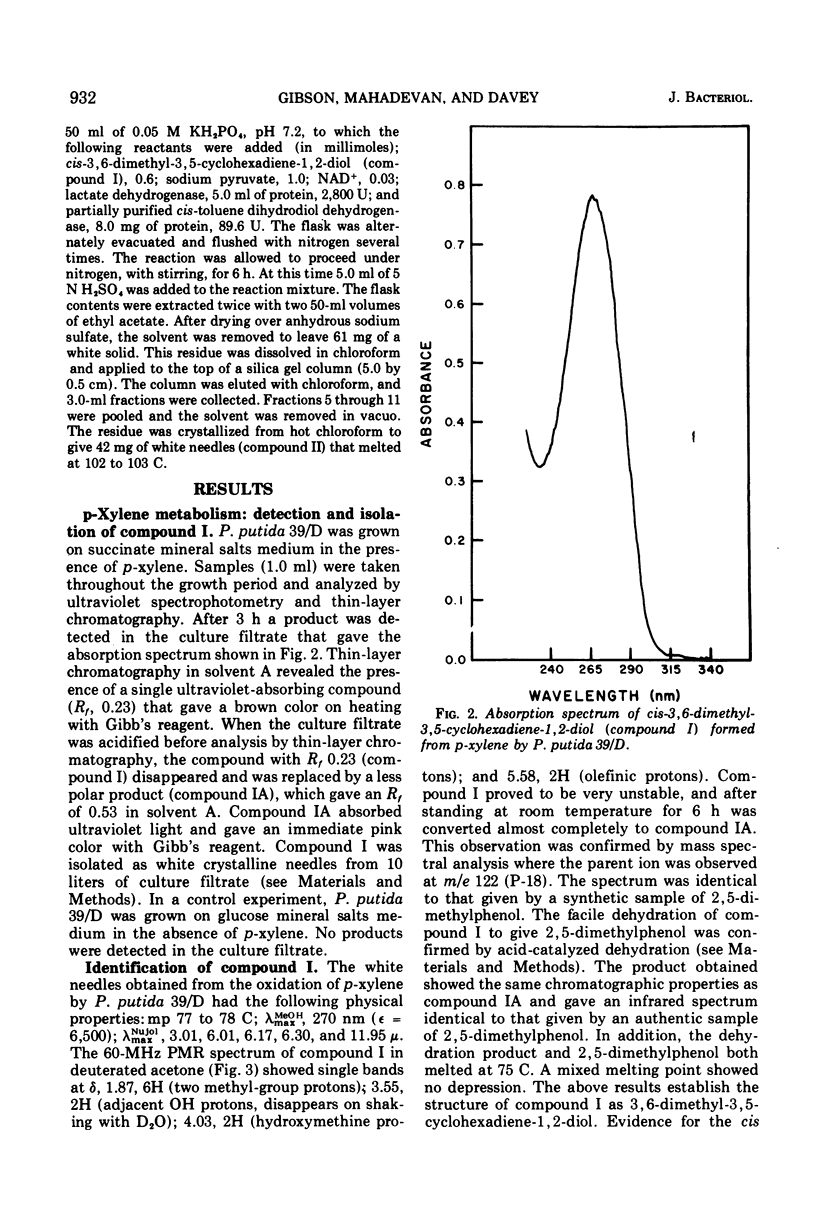
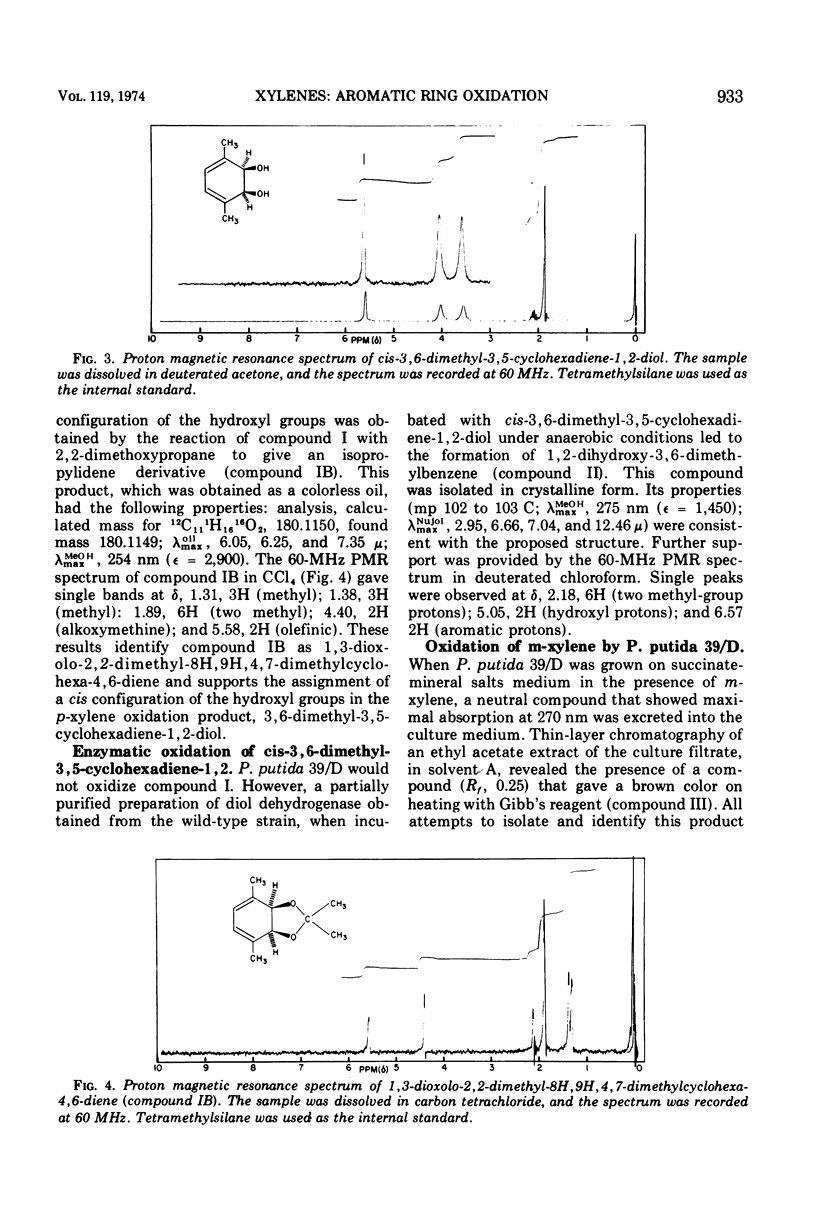
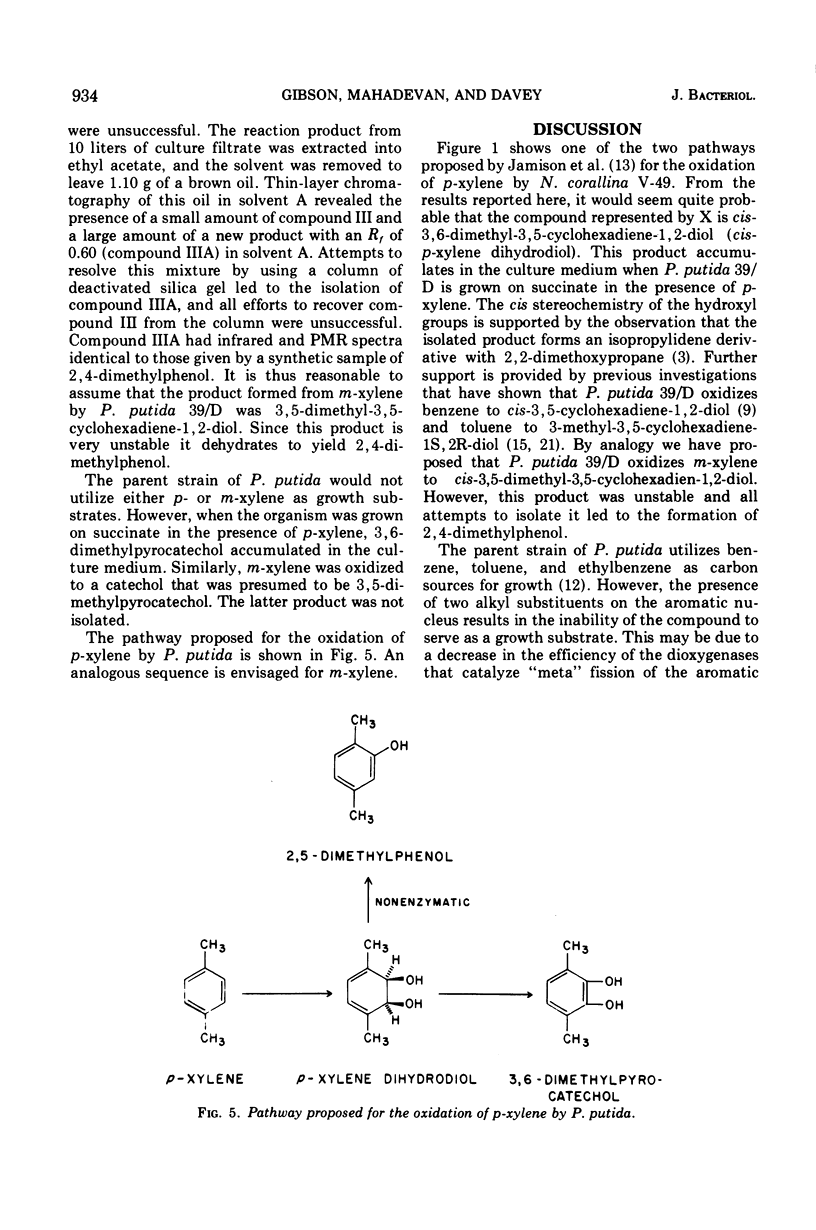
Pseudomonas putida 39/D oxidized p-xylene to cis-3,6-dimethyl-3,5-cyclohexadiene-1,2-diol (cis-p-xylene dihydrodiol). The latter compound was isolated in crystalline form and its physical properties were determined. The cis configuration of the hydroxyl groups in the oxidation product was inferred from its ability to form an isopropylidene derivative with 2,2-dimethoxypropane. Acid treatment of cis-p-xylene dihydrodiol resulted in the formation of 2,5-dimethylphenol. A partially purified preparation of cis-toluene dihydrodiol dehydrogenase oxidized cis-p-xylene dihydrodiol to 1,2-dihydroxy-3,6-dimethylbenzene (3,6-dimethylpyrocatechol). P. putida 39/D oxidized m-xylene to a compound whose spectral and chromatographic characteristics were consistent with the structure 3,5-dimethyl-3,5-cyclohexadiene-1,2-diol. This product was very unstable, and all attempts to isolate it led to the formation of 2,4-dimethylphenol.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abbott B. J., Gledhill W. E. The extracellular accumulation of metabolic products by hydrocarbon-degrading microorganisms. Adv Appl Microbiol. 1971;14:249–388. doi: 10.1016/s0065-2164(08)70546-x. [DOI] [PubMed] [Google Scholar]
- Bayly R. C., Dagley S., Gibson D. T. The metabolism of cresols by species of Pseudomonas. Biochem J. 1966 Nov;101(2):293–301. doi: 10.1042/bj1010293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Daly J. W., Jerina D. M., Witkop B. Arene oxides and the NIH shift: the metabolism, toxicity and carcinogenicity of aromatic compounds. Experientia. 1972 Oct 15;28(10):1129–1149. doi: 10.1007/BF01946135. [DOI] [PubMed] [Google Scholar]
- Davey J. F., Gibson D. T. Bacterial metabolism of para- and meta-xylene: oxidation of a methyl substituent. J Bacteriol. 1974 Sep;119(3):923–929. doi: 10.1128/jb.119.3.923-929.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis R. S., Hossler F. E., Stone R. W. Metabolism of p- and m-xylene by species of Pseudomonas. Can J Microbiol. 1968 Sep;14(9):1005–1009. doi: 10.1139/m68-166. [DOI] [PubMed] [Google Scholar]
- De Frenne E., Eberspächer J., Lingens F. The bacterial degradation of 5-amino-4-chloro-2-phenyl-3(2H)-pyridazinone. Eur J Biochem. 1973 Mar 1;33(2):357–363. doi: 10.1111/j.1432-1033.1973.tb02690.x. [DOI] [PubMed] [Google Scholar]
- Gibson D. T., Cardini G. E., Maseles F. C., Kallio R. E. Incorporation of oxygen-18 into benzene by Pseudomonas putida. Biochemistry. 1970 Mar 31;9(7):1631–1635. doi: 10.1021/bi00809a024. [DOI] [PubMed] [Google Scholar]
- Gibson D. T., Gschwendt B., Yeh W. K., Kobal V. M. Initial reactions in the oxidation of ethylbenzene by Pseudomonas putida. Biochemistry. 1973 Apr 10;12(8):1520–1528. doi: 10.1021/bi00732a008. [DOI] [PubMed] [Google Scholar]
- Gibson D. T., Hensley M., Yoshioka H., Mabry T. J. Formation of (+)-cis-2,3-dihydroxy-1-methylcyclohexa-4,6-diene from toluene by Pseudomonas putida. Biochemistry. 1970 Mar 31;9(7):1626–1630. doi: 10.1021/bi00809a023. [DOI] [PubMed] [Google Scholar]
- Gibson D. T., Koch J. R., Kallio R. E. Oxidative degradation of aromatic hydrocarbons by microorganisms. I. Enzymatic formation of catechol from benzene. Biochemistry. 1968 Jul;7(7):2653–2662. doi: 10.1021/bi00847a031. [DOI] [PubMed] [Google Scholar]
- Jamison V. W., Raymond R. L., Hudson J. O. Microbial Hydrocarbon Co-oxidation. III. Isolation and Characterization of an alpha, alpha'-Dimethyl-cis, cis-Muconic Acid-producing Strain of Nocardia corallina. Appl Microbiol. 1969 Jun;17(6):853–856. doi: 10.1128/am.17.6.853-856.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaubisch N., Daly J. W., Jerina D. M. Arene oxides as intermediates in the oxidative metabolism of aromatic compounds. Isomerization of methyl-substituted arene oxides. Biochemistry. 1972 Aug 1;11(16):3080–3088. doi: 10.1021/bi00766a022. [DOI] [PubMed] [Google Scholar]
- Kobal V. M., Gibson D. T., Davis R. E., Garza A. X-ray determination of the absolute stereochemistry of the initial oxidation product formed from toluene by Pseudomonas puida 39-D. J Am Chem Soc. 1973 Jun 27;95(13):4420–4421. doi: 10.1021/ja00794a048. [DOI] [PubMed] [Google Scholar]
- Raymond R. L., Jamison V. W. Biochemical activities of Nocardia. Adv Appl Microbiol. 1971;14:93–122. doi: 10.1016/s0065-2164(08)70541-0. [DOI] [PubMed] [Google Scholar]
- Raymond R. L., Jamison V. W., Hudson J. O. Microbial hydrocarbon co-oxidation. I. Oxidation of mono- and dicyclic hydrocarbons by soil isolates of the genus Nocardia. Appl Microbiol. 1967 Jul;15(4):857–865. doi: 10.1128/am.15.4.857-865.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reiner A. M., Hegeman G. D. Metabolism of benzoic acid by bacteria. Accumulation of (-)-3,5-cyclohexadiene-1,2-diol-1-carboxylic acid by mutant strain of Alcaligenes eutrophus. Biochemistry. 1971 Jun 22;10(13):2530–2536. doi: 10.1021/bi00789a017. [DOI] [PubMed] [Google Scholar]
- Stanier R. Y., Palleroni N. J., Doudoroff M. The aerobic pseudomonads: a taxonomic study. J Gen Microbiol. 1966 May;43(2):159–271. doi: 10.1099/00221287-43-2-159. [DOI] [PubMed] [Google Scholar]
- Ziffer H., Jerina D. M., Gibson D. T., Kobal V. M. Absolute stereochemistry of the (+)-cis-1,2-dihydroxy-3-methylcyclohexa-3,5-diene produced from toluene by Pseudomonas putida. J Am Chem Soc. 1973 Jun 13;95(12):4048–4049. doi: 10.1021/ja00793a036. [DOI] [PubMed] [Google Scholar]



