Abstract
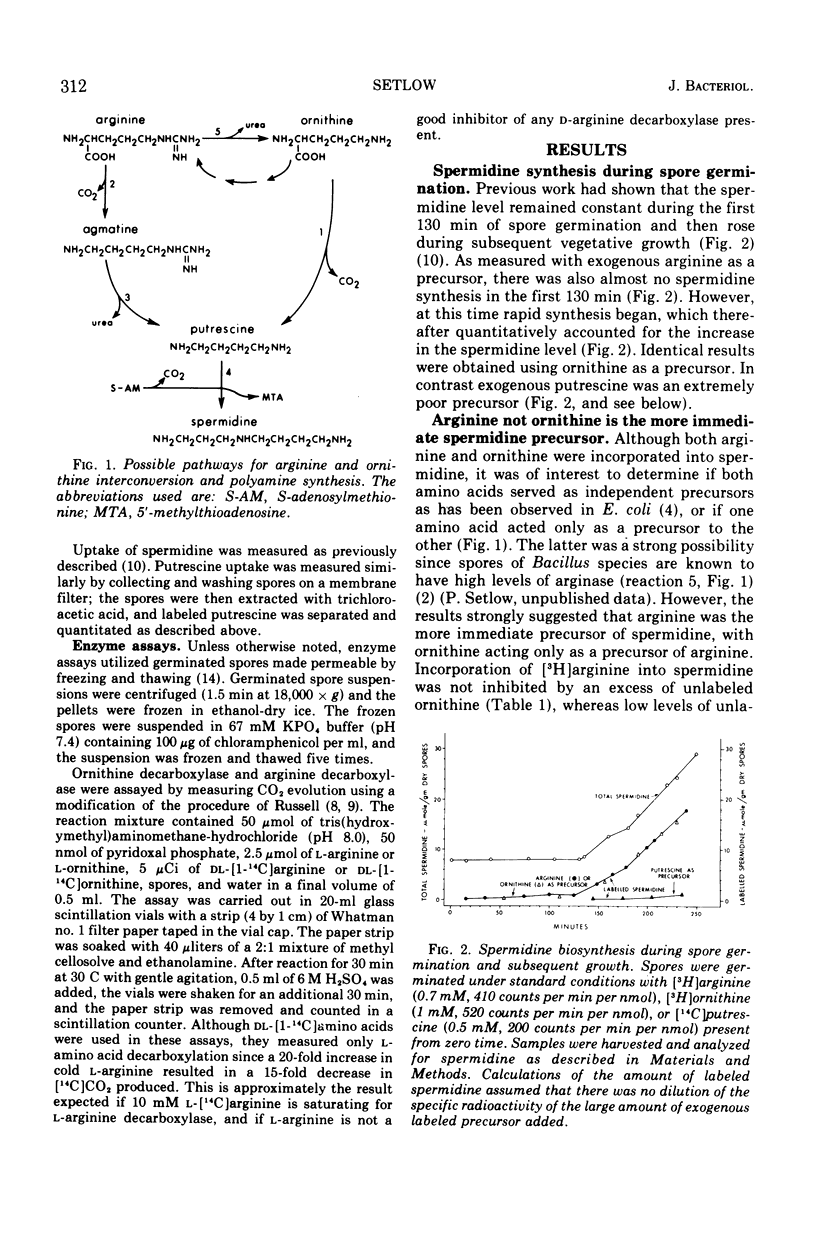
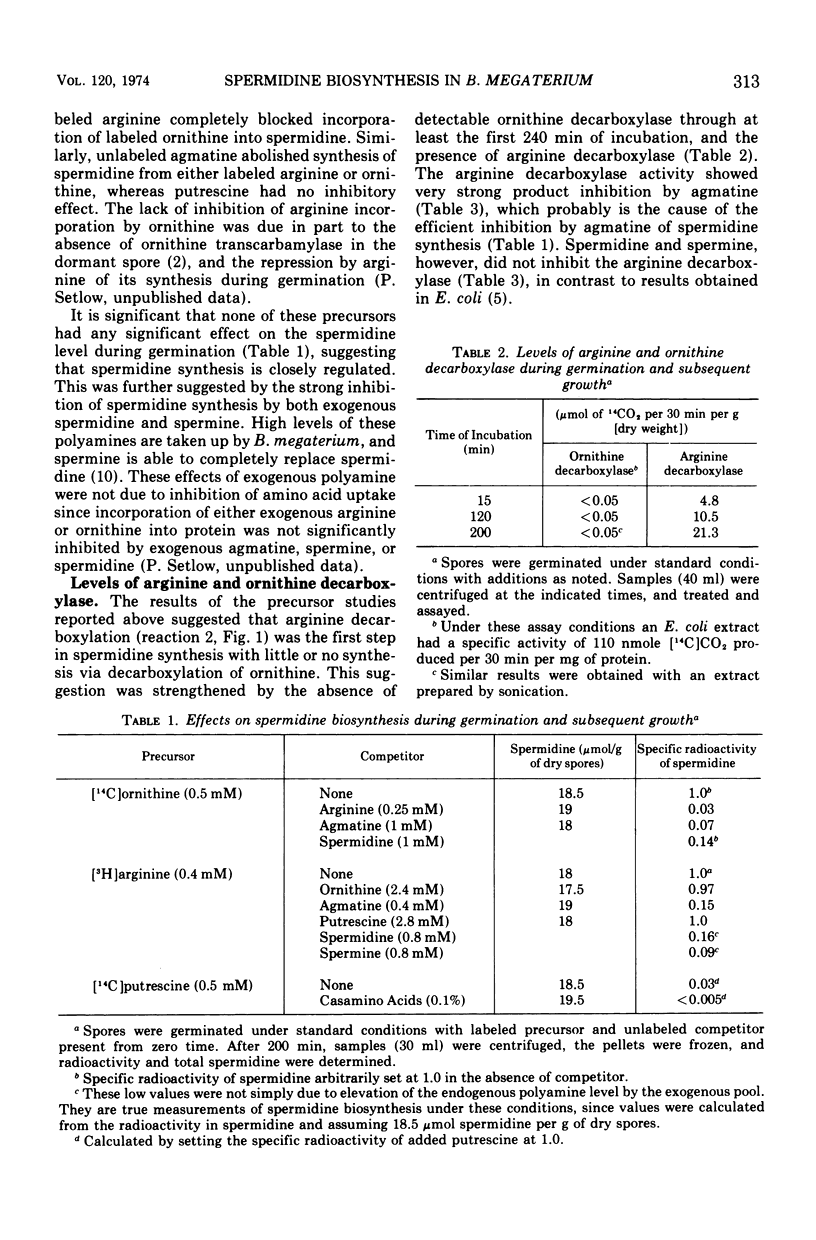
Spermidine biosynthesis was extremely low early in germination of Bacillus megaterium spores and the spermidine level remained constant. Rapid synthesis began after 130 min and thereafter accounted for the increase in spermidine level which began at this time. Biosynthesis was greatly (>84%) diminished by exogenous spermine or spermidine. Arginine and ornithine were both converted efficiently into spermidine, but arginine was the more immediate precursor as shown by isotope competition studies and by the absence of ornithine decarboxylase and the presence of arginine decarboxylase. Exogenous putrescine was not incorporated into spermidine, although it was taken up rapidly and degraded.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BACHRACH U., COHEN I. Spermidine in the bacterial cell. J Gen Microbiol. 1961 Sep;26:1–9. doi: 10.1099/00221287-26-1-1. [DOI] [PubMed] [Google Scholar]
- Deutscher M. P., Kornberg A. Biochemical studies of bacterial sporulation and germination. 8. Patterns of enzyme development during growth and sporulation of Baccillus subtilis. J Biol Chem. 1968 Sep 25;243(18):4653–4660. [PubMed] [Google Scholar]
- Michaels R., Kim K. H. Comparative studies of putrescine degradation by microorganisms. Biochim Biophys Acta. 1966 Jan 25;115(1):59–64. doi: 10.1016/0304-4165(66)90048-1. [DOI] [PubMed] [Google Scholar]
- Morris D. R., Pardee A. B. Multiple pathways of putrescine biosynthesis in Escherichia coli. J Biol Chem. 1966 Jul 10;241(13):3129–3135. [PubMed] [Google Scholar]
- Raina A., Cohen S. S. Polyamines and RNA synthesis in a polyauxotrophic strain of E. coli. Proc Natl Acad Sci U S A. 1966 Jun;55(6):1587–1593. doi: 10.1073/pnas.55.6.1587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Recsei P. A., Snell E. E. Histidine decarboxylase of Lactobacillus 30a. VI. Mechanism of action and kinetic properties. Biochemistry. 1970 Mar 31;9(7):1492–1497. doi: 10.1021/bi00809a003. [DOI] [PubMed] [Google Scholar]
- Russell D. H., Medina V. J., Snyder S. H. The dynamics of synthesis and degradation of polyamines in normal and regenerating rat liver and brain. J Biol Chem. 1970 Dec 25;245(24):6732–6738. [PubMed] [Google Scholar]
- Russell D., Snyder S. H. Amine synthesis in rapidly growing tissues: ornithine decarboxylase activity in regenerating rat liver, chick embryo, and various tumors. Proc Natl Acad Sci U S A. 1968 Aug;60(4):1420–1427. doi: 10.1073/pnas.60.4.1420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Setlow P., Kornberg A. Biochemical studies of bacterial sporulation and germination. XVII. Sulfhydryl and disulfide levels in dormancy and germination. J Bacteriol. 1969 Dec;100(3):1155–1160. doi: 10.1128/jb.100.3.1155-1160.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Setlow P. Polyamine levels during growth, sporulation, and spore germination of Bacillus megaterium. J Bacteriol. 1974 Mar;117(3):1171–1177. doi: 10.1128/jb.117.3.1171-1177.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spizizen J. TRANSFORMATION OF BIOCHEMICALLY DEFICIENT STRAINS OF BACILLUS SUBTILIS BY DEOXYRIBONUCLEATE. Proc Natl Acad Sci U S A. 1958 Oct 15;44(10):1072–1078. doi: 10.1073/pnas.44.10.1072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinberg W., Halvorson H. O. Timing of enzyme synthesis during outgrowth of spores of Bacillus cereus. I. Ordered enzyme synthesis. J Bacteriol. 1968 Feb;95(2):469–478. doi: 10.1128/jb.95.2.469-478.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stevens L. The binding of spermine to the ribosomes and ribosomal ribonucleic acid from Bacillus stearothermophilus. Biochem J. 1969 Jun;113(1):117–121. doi: 10.1042/bj1130117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tabor H., Tabor C. W. Formation of 1,4-diaminobutane and of spermidine by an ornithine auxotroph of Escherichia coli grown on limiting ornithine or arginine. J Biol Chem. 1969 May 10;244(9):2286–2292. [PubMed] [Google Scholar]



