Abstract
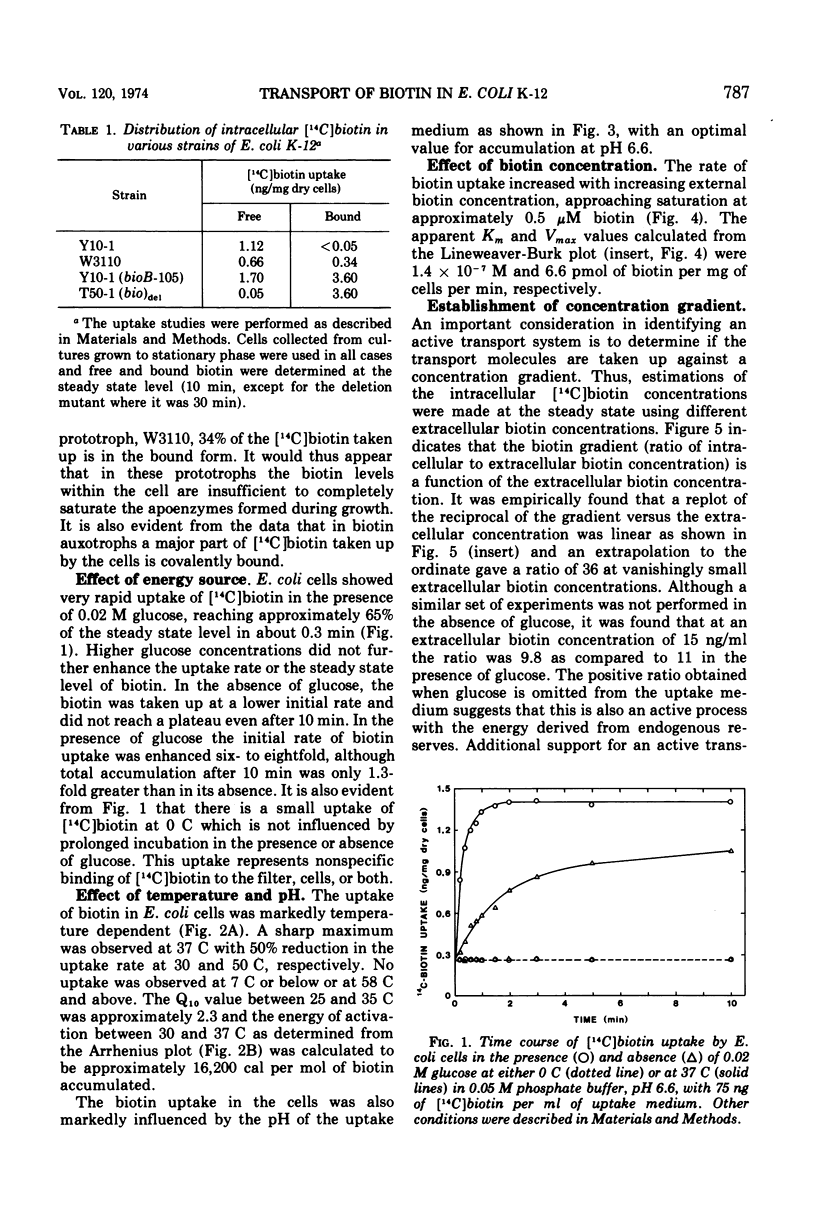
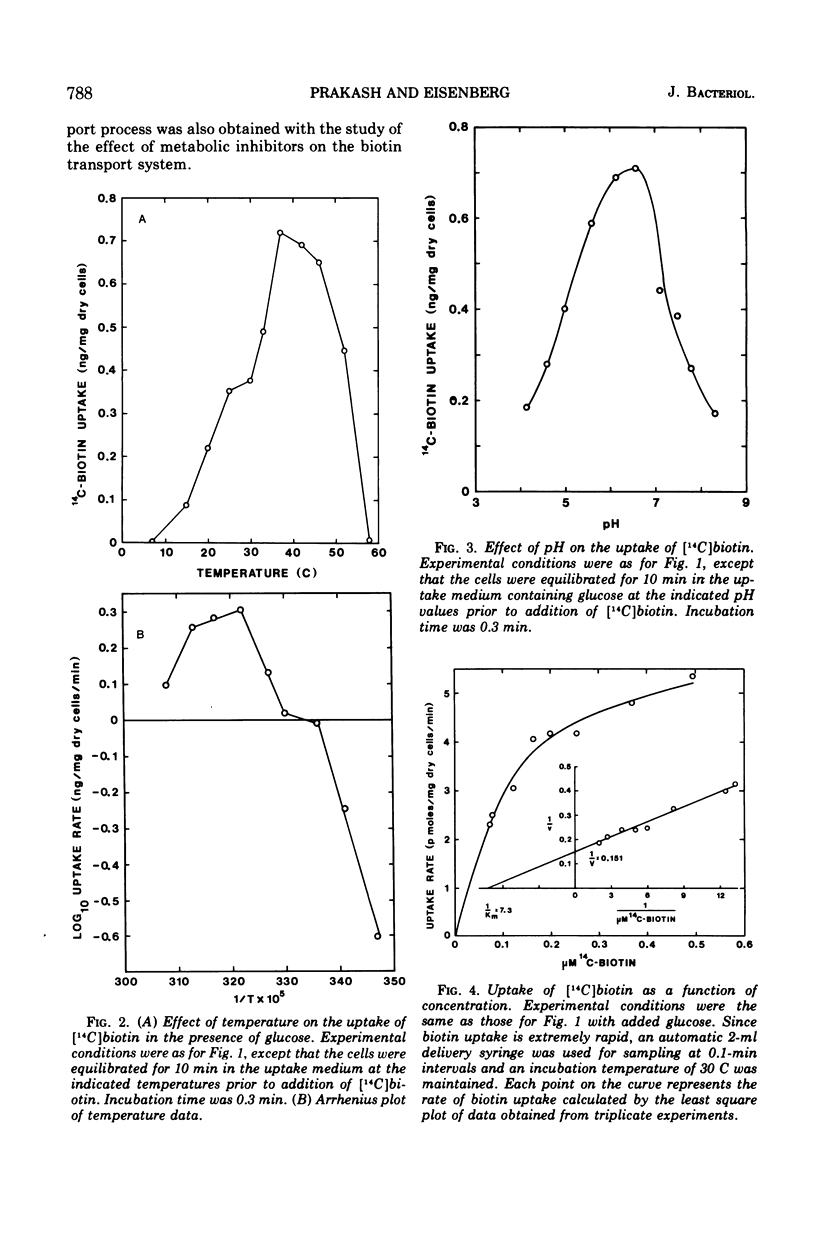
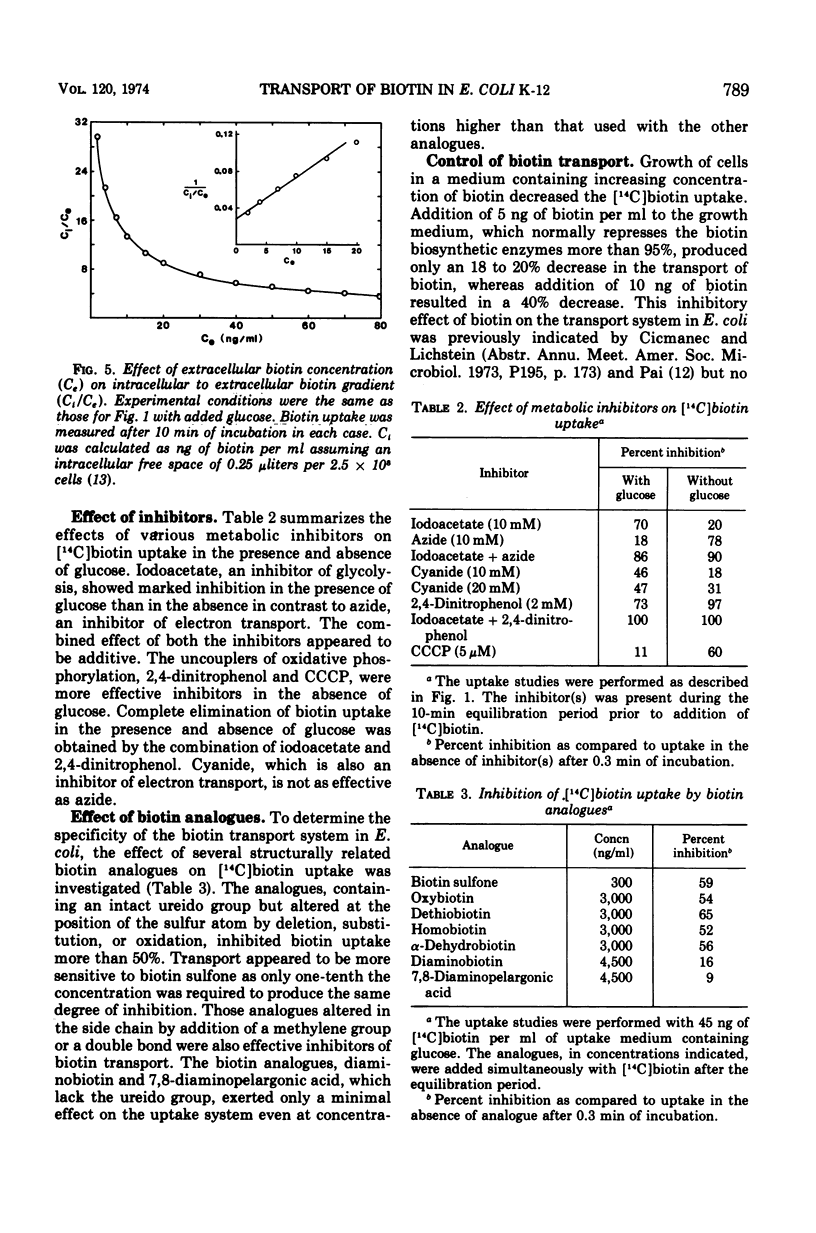
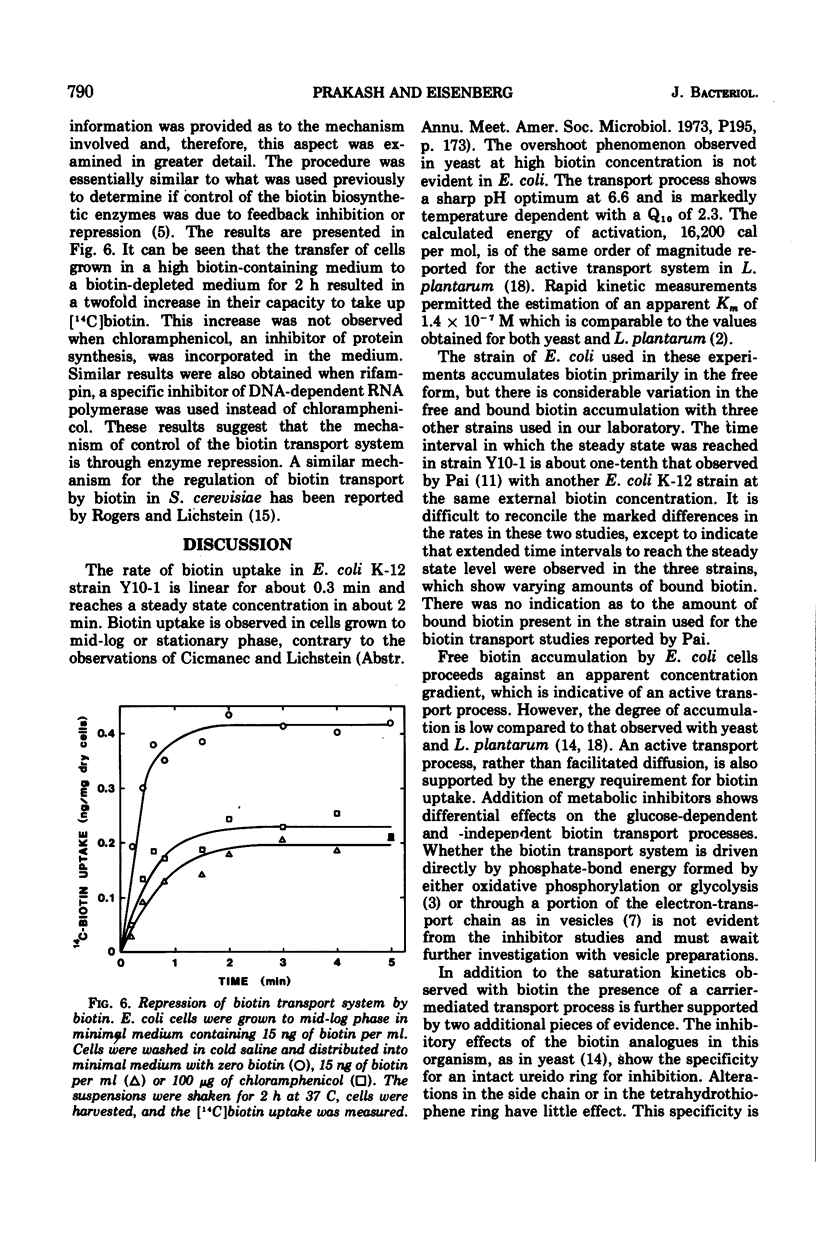
The transport of [14C]biotin into cells of a biotin prototroph, Escherichia coli K-12 strain Y10-1, was investigated. The vitamin taken up by the cells in this strain existed primarily in the free form. Addition of glucose enhanced the rate of uptake six- to eightfold and the steady level was reached in 2 to 3 min resulting in accumulation of biotin against a concentration gradient. The uptake showed marked dependence on temperature (Q10, 2.3; optimum, 37 C) and pH (optimum 6.6) and was inhibited by iodoacetate. Energy of activation for glucose-dependent uptake was calculated to be 16,200 cal per mol. The rate of biotin uptake with increasing biotin concentrations showed saturation kinetics with an apparent Km and Vmax values of 1.4 × 10−7 M and 6.6 pmol per mg of dry cells per min respectively. The cells also accumulated biotin against a concentration gradient in the absence of added glucose, although at a much lower rate. This accumulation was much more susceptible to inhibition by azide and uncouplers of oxidative phosphorylation suggesting that the energy source was supplied through the electron-transport chain. Inhibition studies with a number of biotin analogues indicated the requirement for an intact ureido ring. The biotin uptake was inhibited in cells grown in biotin-containing medium and was shown to be the result of repression of the transport system, suggesting the control of the biotin transport.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Becker J. M., Lichstein H. C. Transport overshoot during biotin uptake by Saccharomyces cerevisiae. Biochim Biophys Acta. 1972 Sep 1;282(1):409–420. doi: 10.1016/0005-2736(72)90346-x. [DOI] [PubMed] [Google Scholar]
- Becker J. M., Wilchek M., Katchalski E. Irreversible inhibition of biotin transport in yeast by biotinyl-p-nitrophenyl ester. Proc Natl Acad Sci U S A. 1971 Oct;68(10):2604–2607. doi: 10.1073/pnas.68.10.2604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berger E. A. Different mechanisms of energy coupling for the active transport of proline and glutamine in Escherichia coli. Proc Natl Acad Sci U S A. 1973 May;70(5):1514–1518. doi: 10.1073/pnas.70.5.1514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eisenberg M. A. Biotin: biogenesis, transport, and their regulation. Adv Enzymol Relat Areas Mol Biol. 1973;38:317–372. doi: 10.1002/9780470122839.ch7. [DOI] [PubMed] [Google Scholar]
- Eisenberg M. A., Krell K. Dethiobiotin synthesis from 7,8-diaminolargonic acid in cell-free extracts of a biotin auxotroph of Escherichia coli K-12. J Biol Chem. 1969 Oct 25;244(20):5503–5509. [PubMed] [Google Scholar]
- Kaback H. R., Milner L. S. Relationship of a membrane-bound D-(-)-lactic dehydrogenase to amino acid transport in isolated bacterial membrane preparations. Proc Natl Acad Sci U S A. 1970 Jul;66(3):1008–1015. doi: 10.1073/pnas.66.3.1008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krell K., Eisenberg M. A. The purification and properties of dethiobiotin synthetase. J Biol Chem. 1970 Dec 25;245(24):6558–6566. [PubMed] [Google Scholar]
- LICHSTEIN H. C., FERGUSON R. B. On the permeability of Lactobacillus arabinosus to biotin. J Biol Chem. 1958 Jul;233(1):243–244. [PubMed] [Google Scholar]
- Moss J., Lane M. D. The biotin-dependent enzymes. Adv Enzymol Relat Areas Mol Biol. 1971;35:321–442. doi: 10.1002/9780470122808.ch7. [DOI] [PubMed] [Google Scholar]
- Pai C. H. Biotin uptake in biotin regulatory mutant of Escherichia coli. J Bacteriol. 1973 Oct;116(1):494–496. doi: 10.1128/jb.116.1.494-496.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pai C. H. Mutant of Escherichia coli with derepressed levels of the biotin biosynthetic enzymes. J Bacteriol. 1972 Dec;112(3):1280–1287. doi: 10.1128/jb.112.3.1280-1287.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parnes J. R., Boos W. Unidirectional transport activity mediated by the galactose-binding protein of Escherichia coli. J Biol Chem. 1973 Jun 25;248(12):4436–4445. [PubMed] [Google Scholar]
- Rogers T. O., Lichstein H. C. Characterization of the biotin transport system in Saccharomyces cerevisiae. J Bacteriol. 1969 Nov;100(2):557–564. doi: 10.1128/jb.100.2.557-564.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rogers T. O., Lichstein H. C. Regulation of biotin transport in Saccharomyces cerevisiae. J Bacteriol. 1969 Nov;100(2):565–572. doi: 10.1128/jb.100.2.565-572.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rolfe B., Eisenberg M. A. Genetic and biochemical analysis of the biotin loci of Escherichia coli K-12. J Bacteriol. 1968 Aug;96(2):515–524. doi: 10.1128/jb.96.2.515-524.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- VOGEL H. J., BONNER D. M. Acetylornithinase of Escherichia coli: partial purification and some properties. J Biol Chem. 1956 Jan;218(1):97–106. [PubMed] [Google Scholar]
- Waller J. R., Lichstein H. C. Biotin transport and accumulation by cells of Lactobacillus plantarum. I. General properties of the system. J Bacteriol. 1965 Oct;90(4):843–852. doi: 10.1128/jb.90.4.843-852.1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waller J. R., Lichstein H. C. Biotin transport and accumulation by cells of Lactobacillus plantarum. II. Kinetics of the system. J Bacteriol. 1965 Oct;90(4):853–856. doi: 10.1128/jb.90.4.853-856.1965. [DOI] [PMC free article] [PubMed] [Google Scholar]


