Abstract
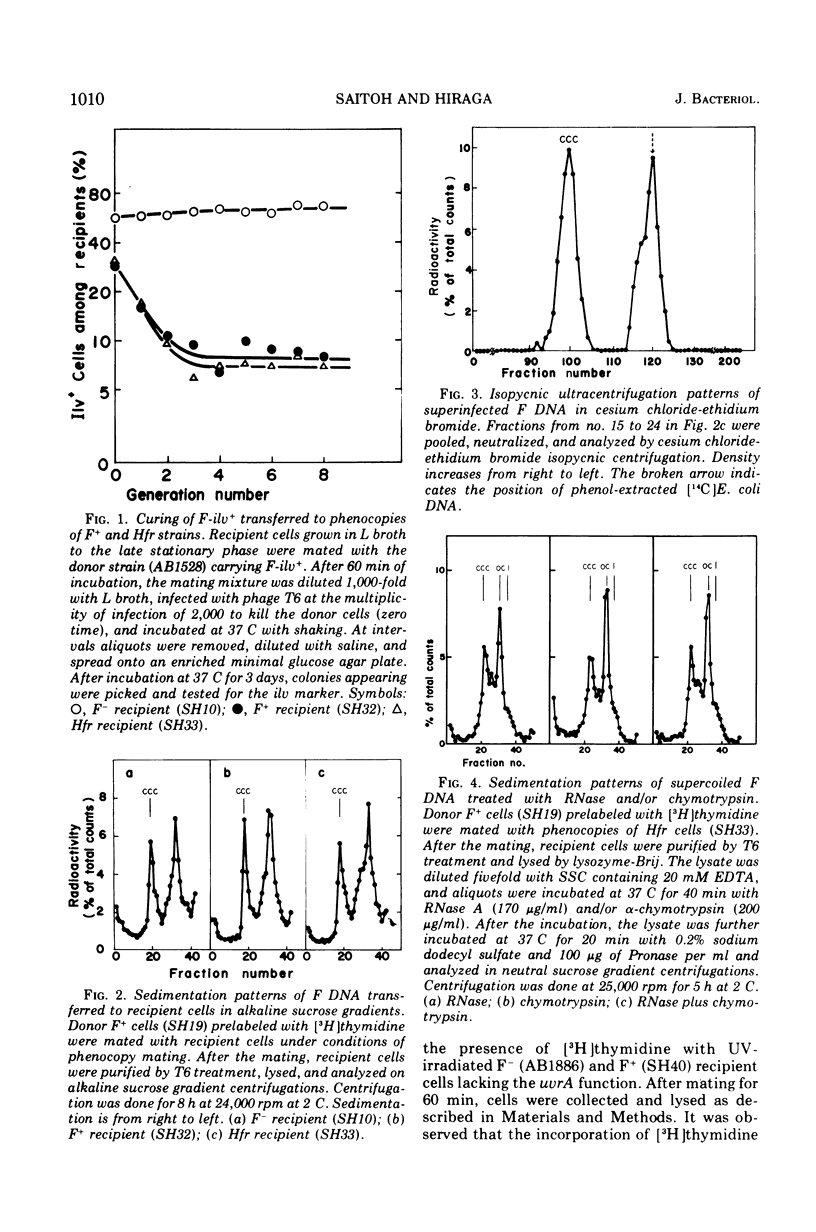
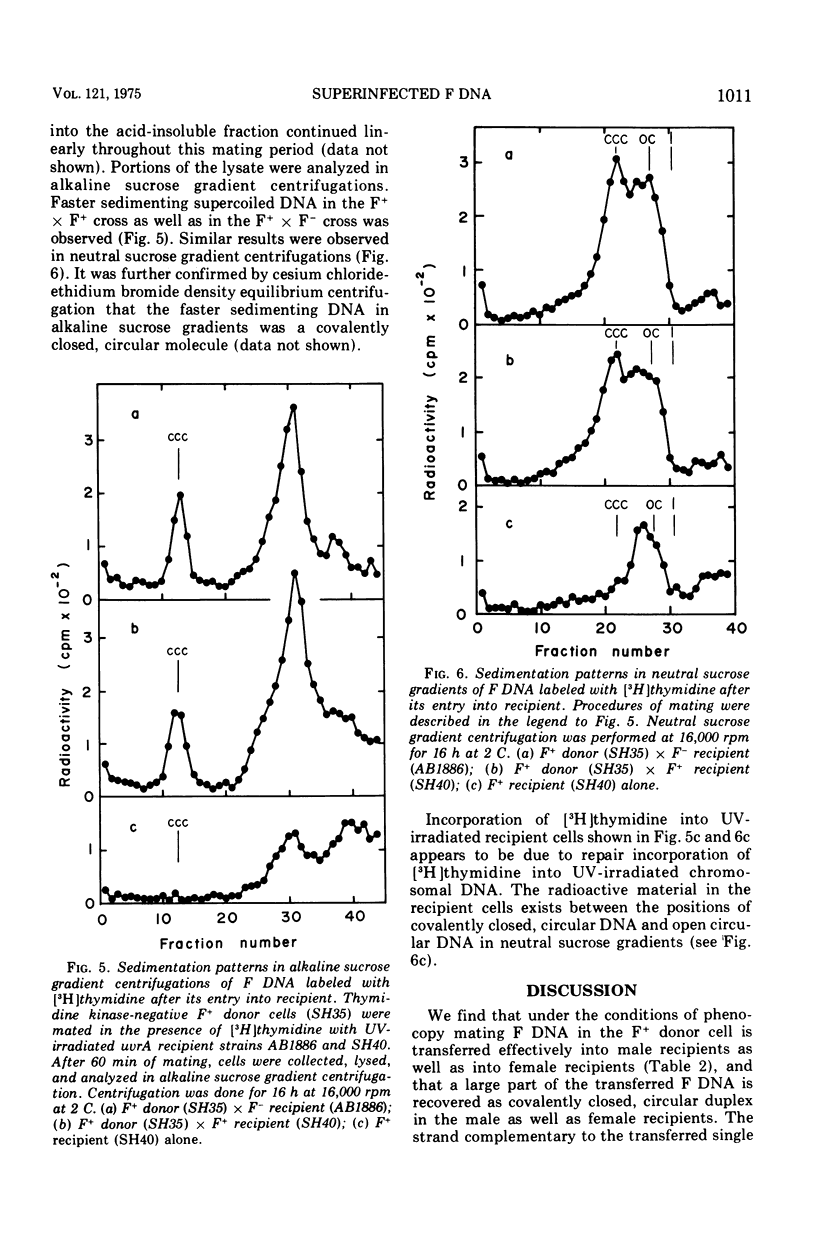
When F+ donor cells of Escherichia coli are conjugated with F-, F+, or Hfr recipients under the conditions of phenocopy mating, the male recipients are found capable of accepting the F episome as effectively as the F- recipients. The F deoxyribonucleic acid (DNA) superinfected into the male recipients is converted to the covalently closed, circular duplex form, as in the F- recipients. It is also found that the synthesis of the strand complementary to the transferred single strand and its subsequent conversion to the covalently closed, circular duplex occur effectively in male recipients as well as in female recipients. Under these mating conditions, F-ilv+ episome superinfected to F+ and Hfr cells is diluted out during growth, whereas F-ilv+ transferred into F-cells is replicated and established in almost all progeny cells. These results suggest that the incompatibility of the F episome is not due to the reduction in the rate of the conversion of transferred single-straned F DNA to covalently closed, circular duplex, but, rather, to an inhibition of further replication of the covalently closed, circular F DNA.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Dubnau E., Maas W. K. Inhibition of replication of an F'lac episome in Hfr cells of Escherichia coli. J Bacteriol. 1968 Feb;95(2):531–539. doi: 10.1128/jb.95.2.531-539.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Echols H. PROPERTIES OF F' STRAINS OF ESCHERICHIA COLI SUPERINFECTED WITH F-LACTOSE AND F-GALATOSE EPISOMES. J Bacteriol. 1963 Feb;85(2):262–268. doi: 10.1128/jb.85.2.262-268.1963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freifelder D. R., Freifelder D. Studies on Escherichia coli sex factors. I. Specific labeling of F'Lac DNA. J Mol Biol. 1968 Feb 28;32(1):15–23. doi: 10.1016/0022-2836(68)90141-1. [DOI] [PubMed] [Google Scholar]
- Hiraga S., Igarashi K., Yura T. A deoxythymidine kinase-deficient mutant of Escherichia coli. I. Isolation and some properties. Biochim Biophys Acta. 1967 Aug 22;145(1):41–51. doi: 10.1016/0005-2787(67)90652-1. [DOI] [PubMed] [Google Scholar]
- Hiraga S., Saitoh T. F deoxyribonucleic acid transferred to recipient cells in the presence of rifampin. J Bacteriol. 1975 Mar;121(3):1000–1006. doi: 10.1128/jb.121.3.1000-1006.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Igarashi K., Hiraga S., Yura T. A deoxythymidine kinase deficient mutant of Escherichia coli. II. Mapping and transduction studies with phage phi 80. Genetics. 1967 Nov;57(3):643–654. doi: 10.1093/genetics/57.3.643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LENNOX E. S. Transduction of linked genetic characters of the host by bacteriophage P1. Virology. 1955 Jul;1(2):190–206. doi: 10.1016/0042-6822(55)90016-7. [DOI] [PubMed] [Google Scholar]
- LeBlanc D. J., Falkow S. Studies on superinfection immunity among transmissible plasmids in Escherichia coli. J Mol Biol. 1973 Mar 15;74(4):689–701. doi: 10.1016/0022-2836(73)90057-0. [DOI] [PubMed] [Google Scholar]
- MAAS R. EXCLUSION OF A FLAC EPISOME BY AN HFR GENE. Proc Natl Acad Sci U S A. 1963 Dec;50:1051–1055. doi: 10.1073/pnas.50.6.1051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MAAS R., MAAS W. K. Introduction of a gene from Escherichia coli B into HFR and F-strains of Escherichia coli K-12. Proc Natl Acad Sci U S A. 1962 Nov 15;48:1887–1893. doi: 10.1073/pnas.48.11.1887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohki M., Tomizawa J. Asymmetric transfer of DNA strands in bacterial conjugation. Cold Spring Harb Symp Quant Biol. 1968;33:651–658. doi: 10.1101/sqb.1968.033.01.074. [DOI] [PubMed] [Google Scholar]
- Palchoudhury S. R., Iyer V. N. Compatibility between two F' factors in an Escherichia coli strain bearing a chromosomal mutation affecting DNA synthesis. J Mol Biol. 1971 Apr 28;57(2):319–333. doi: 10.1016/0022-2836(71)90349-4. [DOI] [PubMed] [Google Scholar]
- SCAIFE J., GROSS J. D. Inhibition of multiplication of an Flac factor in Hfr cells of Escherichia coli K-12. Biochem Biophys Res Commun. 1962 May 11;7:403–407. doi: 10.1016/0006-291x(62)90324-8. [DOI] [PubMed] [Google Scholar]
- San Blas F., Thompson R., Broda P. An Escherichia coli K12 mutant apparently carrying two autonomous F-prime factors. Mol Gen Genet. 1974 May 21;130(2):153–163. doi: 10.1007/BF00269086. [DOI] [PubMed] [Google Scholar]
- Taylor A. L., Trotter C. D. Linkage map of Escherichia coli strain K-12. Bacteriol Rev. 1972 Dec;36(4):504–524. doi: 10.1128/br.36.4.504-524.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- VOGEL H. J., BONNER D. M. Acetylornithinase of Escherichia coli: partial purification and some properties. J Biol Chem. 1956 Jan;218(1):97–106. [PubMed] [Google Scholar]
- Vapnek D., Rupp W. D. Asymmetric segregation of the complementary sex-factor DNA strands during conjugation in Escherichia coli. J Mol Biol. 1970 Nov 14;53(3):287–303. doi: 10.1016/0022-2836(70)90066-5. [DOI] [PubMed] [Google Scholar]


