Abstract
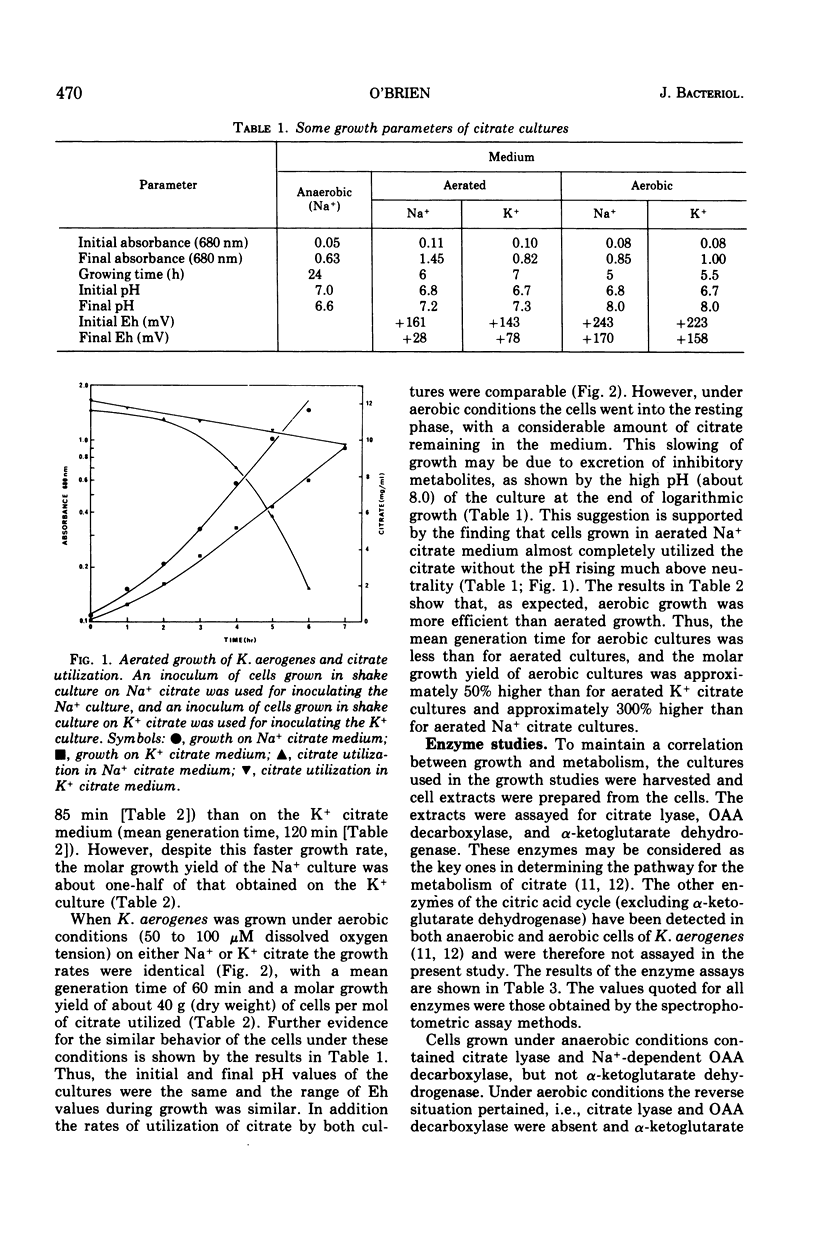
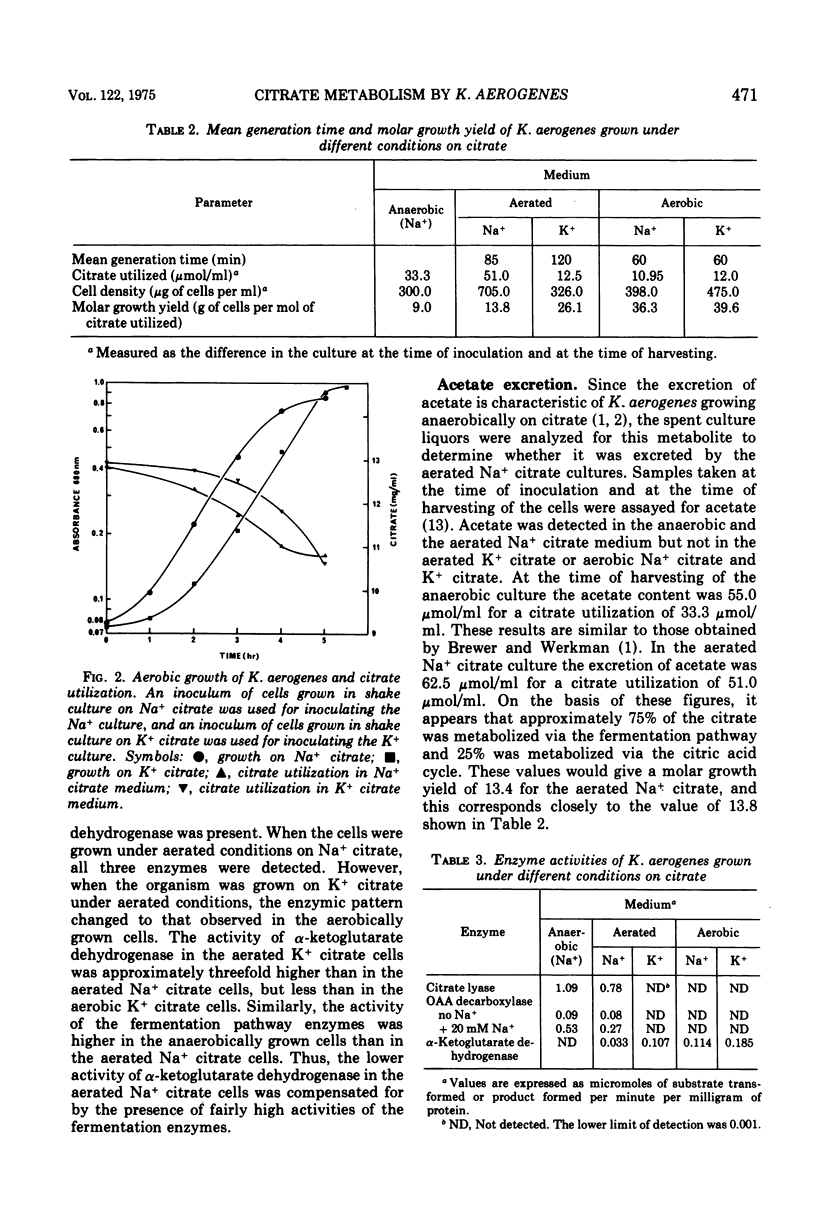
Anaerobic growth of Klebsiella aerogenes NCDO 711 (NCTC 418) on citrate was dependent on the presence of Na+ in the medium, and fermentation of citrate was mediated via the fermentation pathway enzymes, citrate lyase and a Na+-dependent oxalacetate decarboxylase. This confirms the previous findings on strain NCTC 418. Growth under aerobic conditions was independent of Na+. The mean generation time for cells grown aerobically on either Na+ or K+ citrate medium was about 60 min, with a molar growth yield of about 40 g (dry weight) of cells per mol of citrate utilized. Citrate was apparently metabolized aerobically in both the Na+ and K+ citrate cells via the citric acid cycle, since cell extracts contained alpha-ketoglutarate dehydrogenase but not the citrate fermentation enzymes. The presence of theother enzymes of the citric acid cycle in K. aerogenes was shown in earlier studies. Under aerated conditions (no detectable oxygen tension in the culture), growth was faster on the Na+ citrate medium (mean generation time, 85 min) than on the K+ citrate medium (mean generation time, 120 min). Both cultures grew slower than under aerobic conditions, presumably because of oxygen limitation. Despite the faster growth rate, the molar growth yield of the aerated Na+ citrate culture was one-half that observed for the aerated K+ citrate culture. Citrate was metabolized via the citric acid cycle in cells grown in the K+ citrate medium under aerated conditions since alpha-ketoglutarate dehydrogenase, but not the fermentation enzymes, was detected in extracts prepared from these cells. Metabolism of citrate in the Na+ citrate medium under aerated conditions occurred via both the fermentation pathway (approximately 75 percent) and the citric acid cycle (about 25 percent), as evidenced by (i) the presence of the fermentation enzymes and alpha-ketoglutarate dehydrogenase in extracts of cells grown under these conditions, (ii) a molar growth yield which was intermediate between that obtained for anaerobic and aerated K+ citrate cultures, and (iii) the excretion of acetate, which also occurred in anaerobic cultures but not in aerated K+ citrate or aerobic cultures.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- DAGLEY S., DAWES E. A. Critic acid metabolism of Aerobacter aerogenes. J Bacteriol. 1953 Sep;66(3):259–265. doi: 10.1128/jb.66.3.259-265.1953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DAGLEY S., DAWES E. A. Dissimilation of citric acid by bacterial extracts. Nature. 1953 Aug 15;172(4372):345–346. doi: 10.1038/172345a0. [DOI] [PubMed] [Google Scholar]
- Eagon R. G., Wilkerson L. S. A potassium-dependent citric acid transport system in Aerobacter aerogenes. Biochem Biophys Res Commun. 1972 Mar 10;46(5):1944–1950. doi: 10.1016/0006-291x(72)90074-5. [DOI] [PubMed] [Google Scholar]
- O'Brien R. W., Frost G. M., Stern J. R. Enzymatic analysis of the requirement for sodium in aerobic growth of Salmonella typhimurium on citrate. J Bacteriol. 1969 Aug;99(2):395–400. doi: 10.1128/jb.99.2.395-400.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Brien R. W., Geisler J. Citrate metabolism in Aerobacter cloacae. J Bacteriol. 1974 Sep;119(3):661–665. doi: 10.1128/jb.119.3.661-665.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Brien R. W., Morris J. G. Oxygen and the growth and metabolism of Clostridium acetobutylicum. J Gen Microbiol. 1971 Nov;68(3):307–318. doi: 10.1099/00221287-68-3-307. [DOI] [PubMed] [Google Scholar]
- O'Brien R. W., Stern J. R. Requirement for sodium in the anaerobic growth of Aerobacter aerogenes on citrate. J Bacteriol. 1969 May;98(2):388–393. doi: 10.1128/jb.98.2.388-393.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Brien R. W., Stern J. R. Role of sodium in determining alternate pathways of aerobic citrate catabolism in Aerobacter aerogenes. J Bacteriol. 1969 Aug;99(2):389–394. doi: 10.1128/jb.99.2.389-394.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stern J. R. Oxalacetate decarboxylase of Aerobacter aerogenes. I. Inhibition by avidin and requirement for sodium ion. Biochemistry. 1967 Nov;6(11):3545–3551. doi: 10.1021/bi00863a028. [DOI] [PubMed] [Google Scholar]
- Villarreal-Moguel E. I., Ruiz-Herrera J. Induction and properties of the citrate transport system in Aerobacter aerogenes. J Bacteriol. 1969 May;98(2):552–558. doi: 10.1128/jb.98.2.552-558.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilkerson L. S., Eagon R. G. Effect of sodium on the transport and utilization of citric acid by Aerobacter (Enterobacter) aerogenes. J Bacteriol. 1974 Oct;120(1):121–124. doi: 10.1128/jb.120.1.121-124.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilkerson L. S., Eagon R. G. Transport of citric acid by Aerobacter aerogenes. Arch Biochem Biophys. 1972 Mar;149(1):209–221. doi: 10.1016/0003-9861(72)90316-5. [DOI] [PubMed] [Google Scholar]


