Abstract
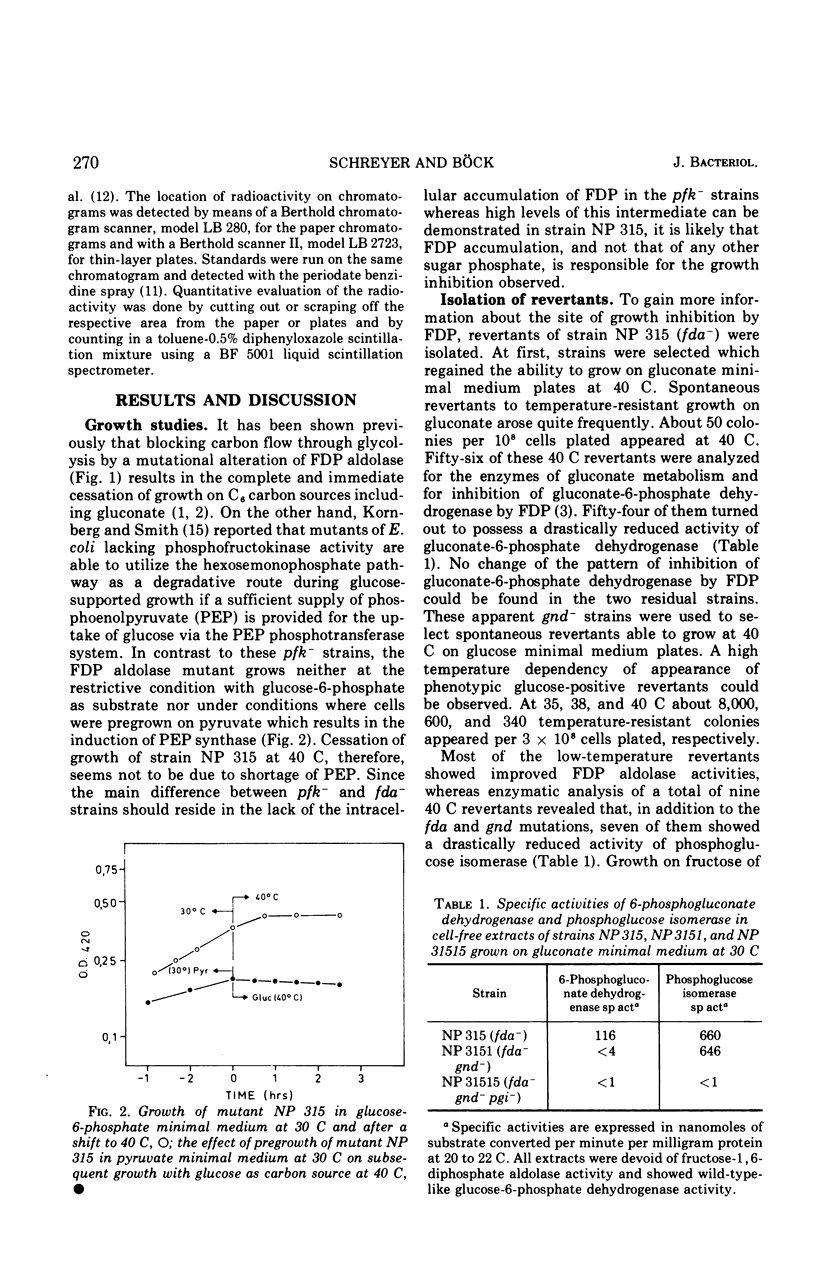
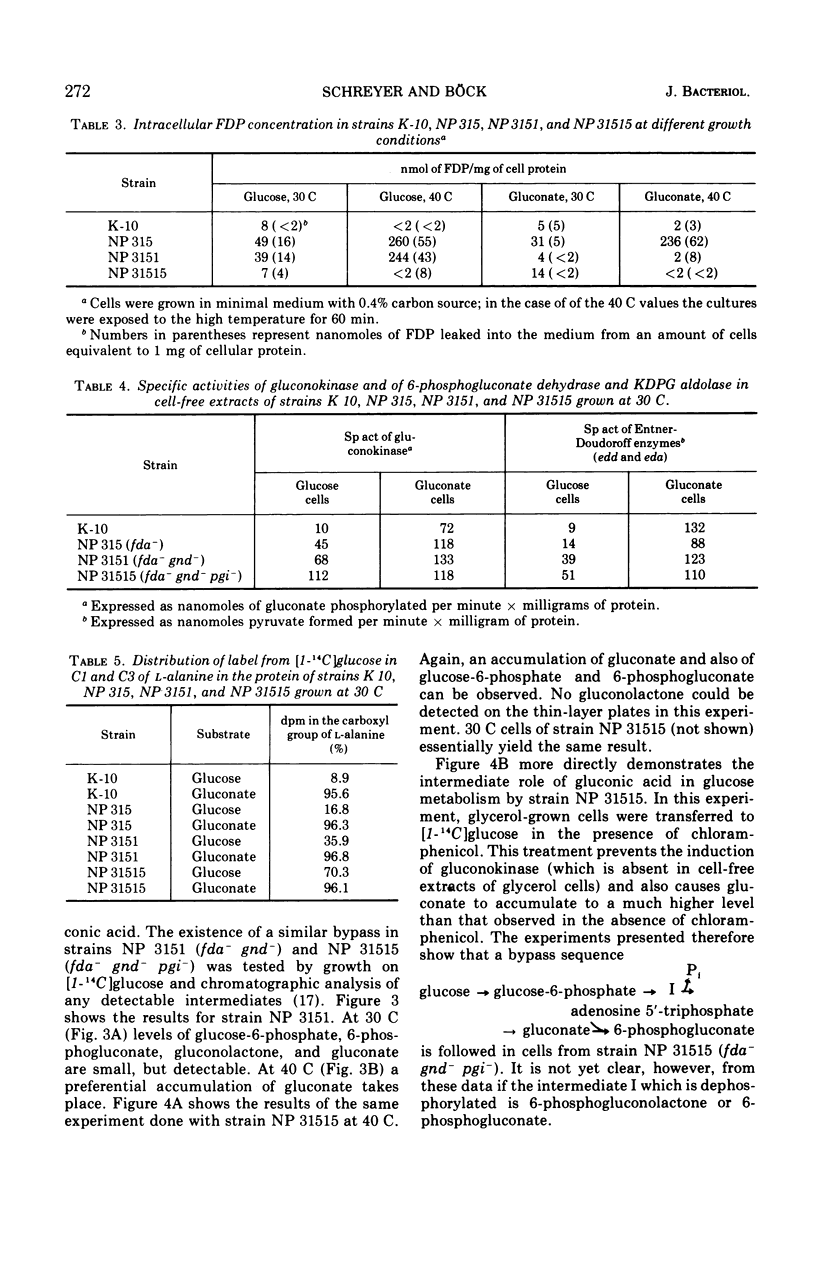
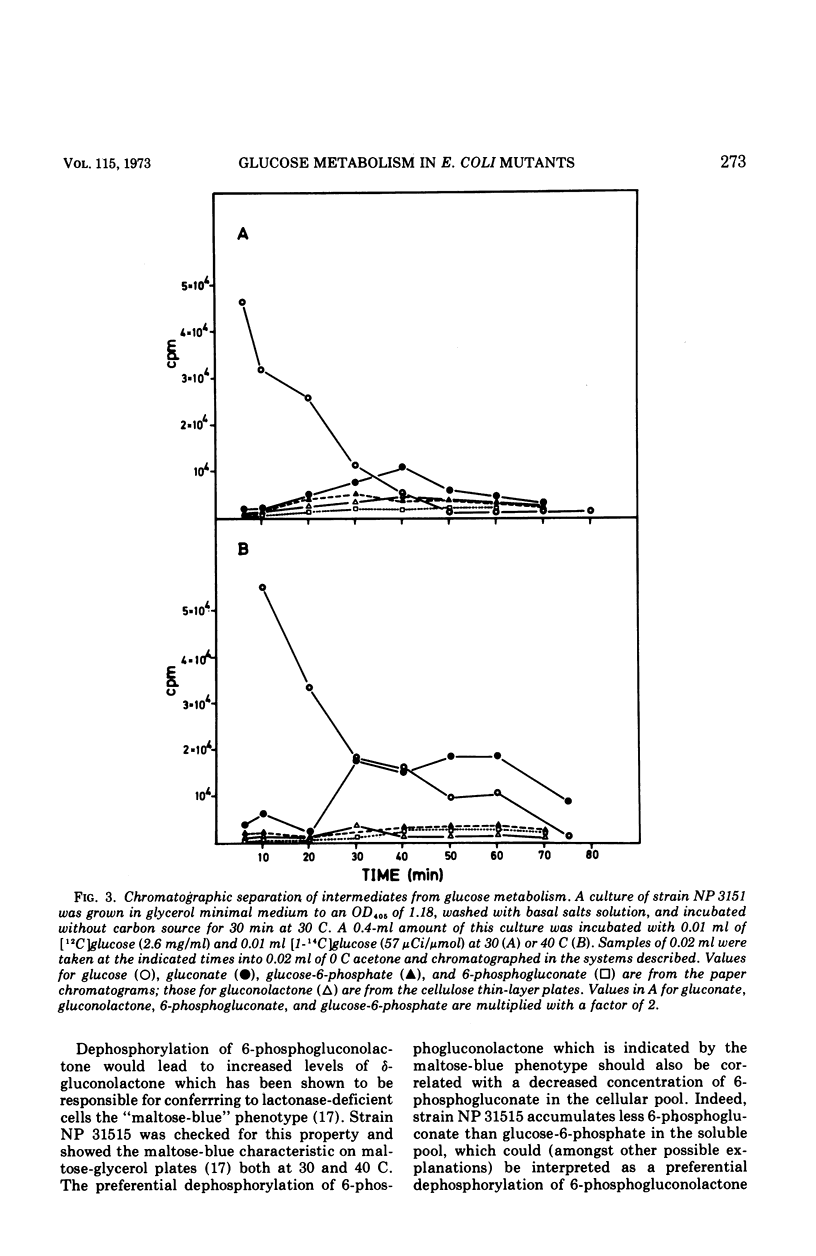
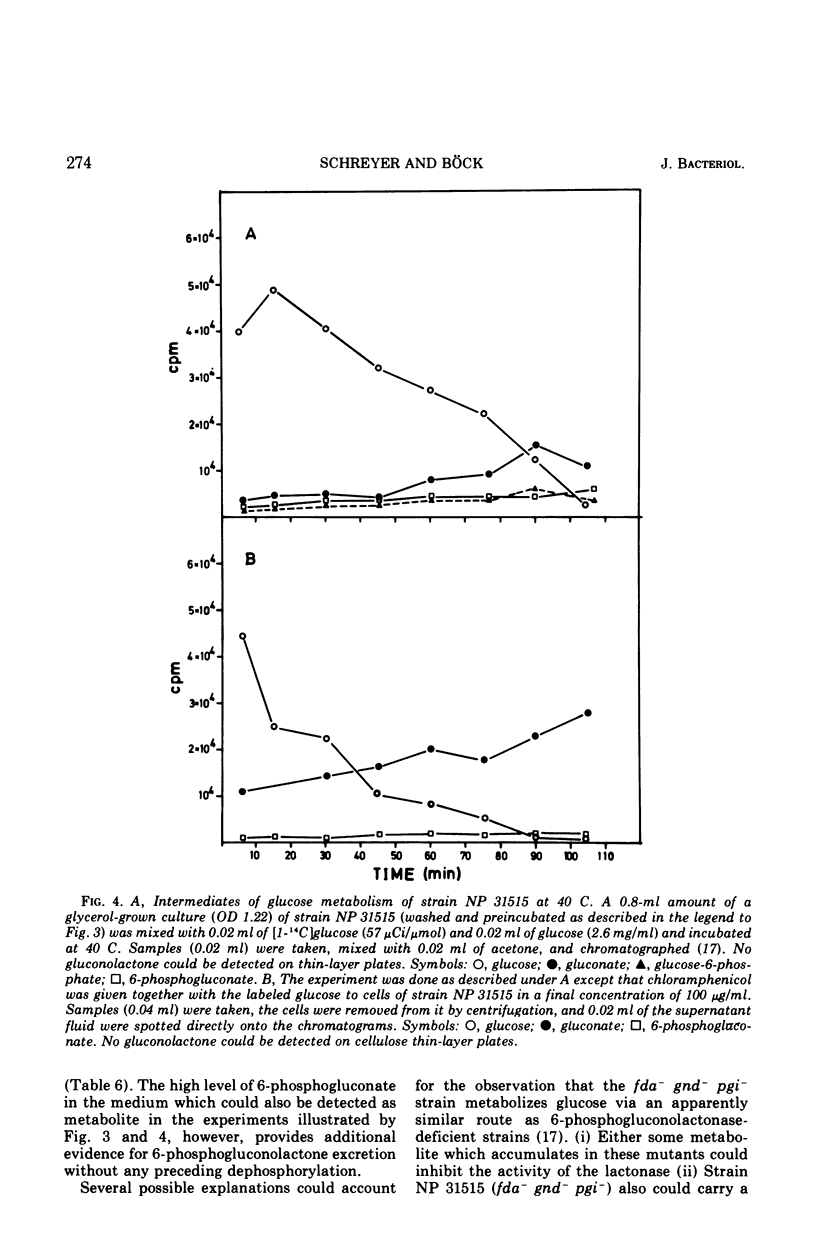
Strain NP 315 of Escherichia coli possesses a thermolabile fructose-1, 6-diphosphate (FDP) aldolase; its growth on carbohydrate substrates is inhibited probably as a consequence of the accumulation of high intracellular levels of FDP. Studies of one class of phenotypic revertants of strain NP 315 which have regained their ability to grow on C6 substrates at 40 C showed that in these strains the buildup of the inhibitory FDP pool is prevented by additional mutations in enzymes catalyzing the conversion of the substrate offered in the medium to FDP. For example, mutations affecting 6-phosphogluconate dehydrogenase activity (gnd−) may be selected in great number without any mutagenesis and enrichment simply by isolating revertants of strain NP 315 able to grow on gluconate at 40 C. Similarly, an additional mutation in phosphoglucose isomerase (pgi−) restores the ability of these fda−gnd− strains to grow on glucose at 40 C. Glucose metabolism of these fda−gnd−pgi− strains was investigated. The enzymes of the Entner-Doudoroff pathway are induced to an appreciable extent upon growth of these mutants on glucose medium; further evidence for glucose degradation via this route (which normally is induced only in the presence of gluconate) was provided by following the fate of the C1 label of radioactive glucose in l-alanine. Predominant labeling of the carboxyl-carbon of l-alanine was observed, inciating a major contribution of the Entner-Doudoroff path to pyruvate formation from glucose. Chromatographic analysis of the intermediates of glucose metabolism showed further that glucose apparently is at least partly metabolized via a bypass consisting of the accumulation of extracellular gluconic acid which arises by dephosphorylation of 6-phosphogluconolactone and possibly of 6-phosphogluconate. This extracellular gluconate is then taken up and metabolized in the normal manner via the Entner-Doudoroff enzymes.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Brown A. T., Wittenberger C. L. Mechanism for regulating the distribution of glucose carbon between the Embden-Meyerhof and hexose-monophosphate pathways in Streptococcus faecalis. J Bacteriol. 1971 May;106(2):456–467. doi: 10.1128/jb.106.2.456-467.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Böck A., Neidhardt F. C. Isolation of a Mutant of Escherichia coli with a Temperature-sensitive Fructose-1,6-Diphosphate Aldolase Activity. J Bacteriol. 1966 Aug;92(2):464–469. doi: 10.1128/jb.92.2.464-469.1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Böck A., Neidhardt F. C. Properties of a Mutant of Escherichia coli with a Temperature-sensitive Fructose-1,6-Diphosphate Aldolase. J Bacteriol. 1966 Aug;92(2):470–476. doi: 10.1128/jb.92.2.470-476.1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- ENTNER N., DOUDOROFF M. Glucose and gluconic acid oxidation of Pseudomonas saccharophila. J Biol Chem. 1952 May;196(2):853–862. [PubMed] [Google Scholar]
- FRAENKEL D. G., NEIDHARDT F. C. Use of chloramphenicol to study control of RNA synthesis in bacteria. Biochim Biophys Acta. 1961 Oct 14;53:96–110. doi: 10.1016/0006-3002(61)90797-1. [DOI] [PubMed] [Google Scholar]
- Fradkin J. E., Fraenkel D. G. 2-keto-3-deoxygluconate 6-phosphate aldolase mutants of Escherichia coli. J Bacteriol. 1971 Dec;108(3):1277–1283. doi: 10.1128/jb.108.3.1277-1283.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fraenkel D. G. Selection of Escherichia coli mutants lacking glucose-6-phosphate dehydrogenase or gluconate-6-phosphate dehydrogenase. J Bacteriol. 1968 Apr;95(4):1267–1271. doi: 10.1128/jb.95.4.1267-1271.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fraenkel D. G. The accumulation of glucose 6-phosphate from glucose and its effect in an Escherichia coli mutant lacking phosphoglucose isomerase and glucose 6-phosphate dehydrogenase. J Biol Chem. 1968 Dec 25;243(24):6451–6457. [PubMed] [Google Scholar]
- Grassetti D. R., Murray J. F., Jr, Wellings J. L. Thin-layer chromatography of phosphorylated glycolysis intermediates. J Chromatogr. 1965 Jun;18(3):612–614. doi: 10.1016/s0021-9673(01)80429-5. [DOI] [PubMed] [Google Scholar]
- KOVACHEVICH R., WOOD W. A. Carbohydrate metabolism by Pseudomonas fluorescens. III. Purification and properties of a 6-phosphogluconate dehydrase. J Biol Chem. 1955 Apr;213(2):745–756. [PubMed] [Google Scholar]
- Kornberg H. L., Smith J. Role of phosphofructokinase in the utilization of glucose by Escherichia coli. Nature. 1970 Jul 4;227(5253):44–46. doi: 10.1038/227044a0. [DOI] [PubMed] [Google Scholar]
- Kupor S. R., Fraenkel D. G. Glucose metabolism in 6 phosphogluconolactonase mutants of Escherichia coli. J Biol Chem. 1972 Mar 25;247(6):1904–1910. [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Zablotny R., Fraenkel D. G. Glucose and gluconate metabolism in a mutant of Escherichia coli lacking gluconate-6-phosphate dehydrase. J Bacteriol. 1967 May;93(5):1579–1581. doi: 10.1128/jb.93.5.1579-1581.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]



