Abstract
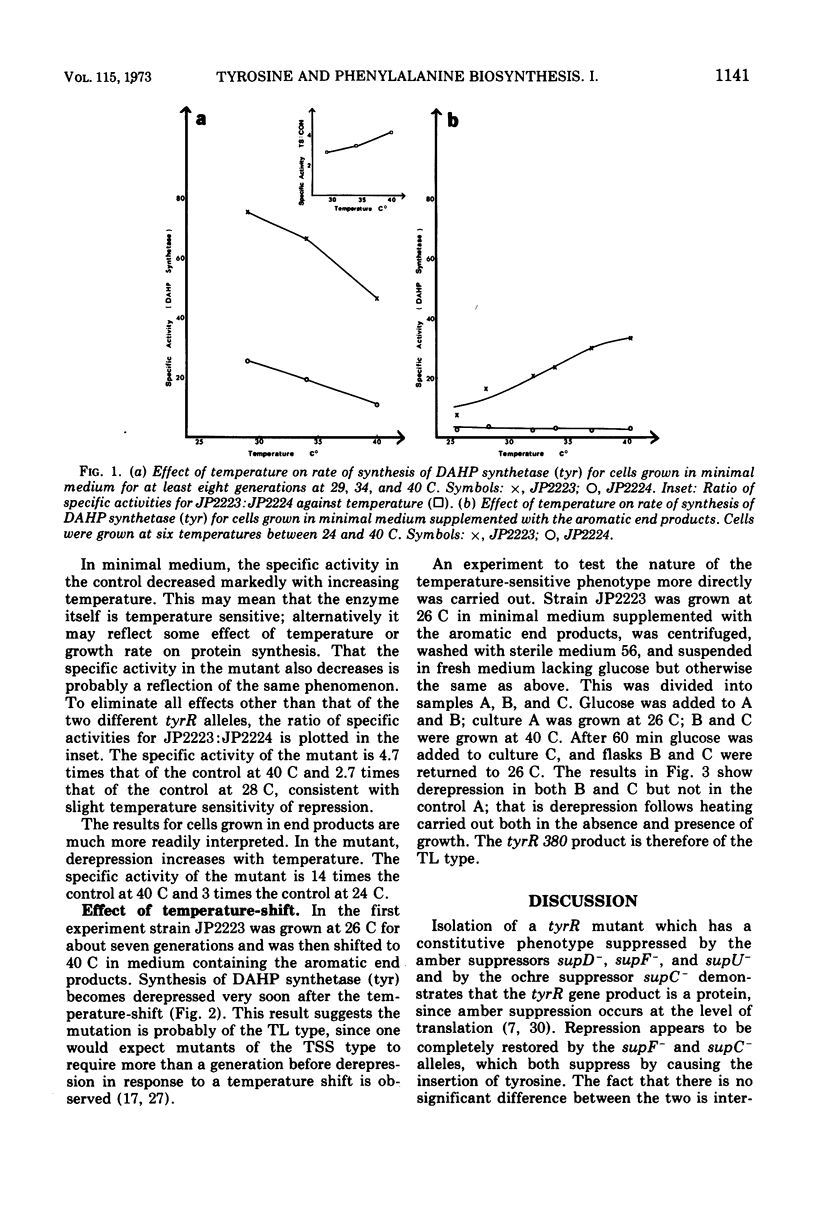
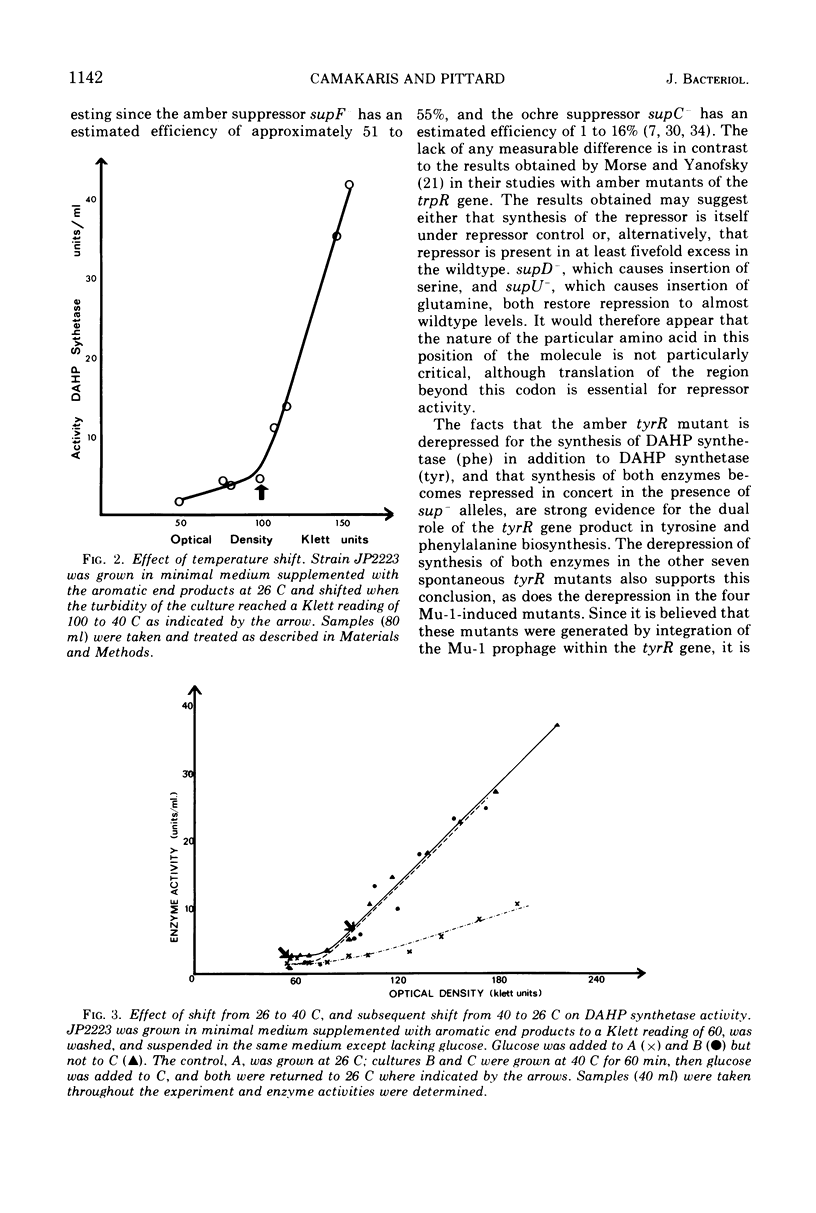
A spontaneous amber tyrR mutant has been isolated in which constitutive synthesis of 3-deoxy-d-arabinoheptulosonic acid 7-phosphate (DAHP) synthetase (tyr) and DAHP synthetase (phe) is suppressible by supC−, supD−, supF− and supU−. This finding suggests the tyrR gene product is a protein. Derepression of DAHP synthetase (phe) in this and in seven other spontaneous tyrR mutants and in four Mu-1-induced tyrR mutants provides further evidence for the involvement of the tyrR gene product in phenylalanine biosynthesis. Evidence that the tyrR product is a component of repressor, rather than an enzyme involved in its synthesis or modification, comes from a study of a temperature-sensitive tyrR mutant. This mutant is of the thermolabile type, since derepression occurs rapidly and in the presence and absence of growth.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Boram W., Abelson J. Bacteriophage Mu integration: on the mechanism of Mu-induced mutations. J Mol Biol. 1971 Nov 28;62(1):171–178. doi: 10.1016/0022-2836(71)90137-9. [DOI] [PubMed] [Google Scholar]
- Brown K. D. Regulation of aromatic amino acid biosynthesis Escherichia coli K12. Genetics. 1968 Sep;60(1):31–48. doi: 10.1093/genetics/60.1.31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown K. D., Somerville R. L. Repression of aromatic amino acid biosynthesis in Escherichia coli K-12. J Bacteriol. 1971 Oct;108(1):386–399. doi: 10.1128/jb.108.1.386-399.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doy C. H., Brown K. D. Control of aromatic biosynthesis: the multiplicity of 7-phospho-2-oxo-3-deoxy-D-arabino-heptonate D-erythrose-4-phosphate-lyase (pyruvate-phosphorylating) in Escherichia coli W. Biochim Biophys Acta. 1965 Jul 8;104(2):377–389. doi: 10.1016/0304-4165(65)90343-0. [DOI] [PubMed] [Google Scholar]
- Garen A. Sense and nonsense in the genetic code. Three exceptional triplets can serve as both chain-terminating signals and amino acid codons. Science. 1968 Apr 12;160(3824):149–159. doi: 10.1126/science.160.3824.149. [DOI] [PubMed] [Google Scholar]
- Gibson F., Pittard J. Pathways of biosynthesis of aromatic amino acids and vitamins and their control in microorganisms. Bacteriol Rev. 1968 Dec;32(4 Pt 2):465–492. [PMC free article] [PubMed] [Google Scholar]
- Gilbert W., Müller-Hill B. Isolation of the lac repressor. Proc Natl Acad Sci U S A. 1966 Dec;56(6):1891–1898. doi: 10.1073/pnas.56.6.1891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heinonen J., Artz S. W., Zalkin H. Regulation of the tyrosine biosynthetic enzymes in Salmonella typhimurium: analysis of the involvement of tyrosyl-transfer ribonucleic acid and tyrosyl-transfer ribonucleic acid synthetase. J Bacteriol. 1972 Dec;112(3):1254–1263. doi: 10.1128/jb.112.3.1254-1263.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Im S. W., Davidson H., Pittard J. Phenylalanine and tyrosine biosynthesis in Escherichia coli K-12: mutants derepressed for 3-deoxy-D-arabinoheptulosonic acid 7-phosphate synthetase (phe), 3-deoxy-D-arabinoheptulosonic acid 7-phosphate synthetase (tyr), chorismate mutase T-prephenate dehydrogenase, and transaminase A. J Bacteriol. 1971 Oct;108(1):400–409. doi: 10.1128/jb.108.1.400-409.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ito K., Hiraga S., Yura T. Temperature-sensitive repression of the tryptophan operon in Escherichia coli. J Bacteriol. 1969 Jul;99(1):279–286. doi: 10.1128/jb.99.1.279-286.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- JACOB F., MONOD J. Genetic regulatory mechanisms in the synthesis of proteins. J Mol Biol. 1961 Jun;3:318–356. doi: 10.1016/s0022-2836(61)80072-7. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- MONOD J., COHEN-BAZIRE G., COHN M. Sur la biosynthèse de la beta-galactosidase (lactase) chez Escherichia coli; la spécificité de l'induction. Biochim Biophys Acta. 1951 Nov;7(4):585–599. doi: 10.1016/0006-3002(51)90072-8. [DOI] [PubMed] [Google Scholar]
- Mattern I. E., Pittard J. Regulation of tyrosine biosynthesis in Escherichia coli K-12: isolation and characterization of operator mutants. J Bacteriol. 1971 Jul;107(1):8–15. doi: 10.1128/jb.107.1.8-15.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McFall E., Heincz M. C. Thermosensitive regulation of D-serine deaminase synthesis in a mutant of Escherichia coli K 12. Mol Gen Genet. 1970;106(4):371–377. doi: 10.1007/BF00324054. [DOI] [PubMed] [Google Scholar]
- Minson A. C., Smith D. A. Methionine regulatory defects in Salmonella typhimurium arising from amber-suppressible mutations. J Gen Microbiol. 1972 May;70(3):471–476. doi: 10.1099/00221287-70-3-471. [DOI] [PubMed] [Google Scholar]
- Morse D. E., Yanofsky C. Amber mutants of the trpR regulatory gene. J Mol Biol. 1969 Aug 28;44(1):185–193. doi: 10.1016/0022-2836(69)90413-6. [DOI] [PubMed] [Google Scholar]
- PITTARD J. EFFECT OF INTEGRATED SEX FACTOR ON TRANSDUCTION OF CHROMOSOMAL GENES IN ESCHERICHIA COLI. J Bacteriol. 1965 Mar;89:680–686. doi: 10.1128/jb.89.3.680-686.1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pittard J., Camakaris J., Wallace B. J. Inhibition of 3-deoxy-d-arabinoheptulosonic acid-7-phosphate synthetase (trp) in Escherichia coli. J Bacteriol. 1969 Mar;97(3):1242–1247. doi: 10.1128/jb.97.3.1242-1247.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ravel J. M., White M. N., Shive W. Activation of tyrosine analogs in relation to enzyme repression. Biochem Biophys Res Commun. 1965 Jul 26;20(3):352–359. doi: 10.1016/0006-291x(65)90372-4. [DOI] [PubMed] [Google Scholar]
- Riggs A. D., Bourgeois S., Newby R. F., Cohn M. DNA binding of the lac repressor. J Mol Biol. 1968 Jul 14;34(2):365–368. doi: 10.1016/0022-2836(68)90261-1. [DOI] [PubMed] [Google Scholar]
- Riggs A. D., Bourgeois S. On the assay, isolation and characterization of the lac repressor. J Mol Biol. 1968 Jul 14;34(2):361–364. doi: 10.1016/0022-2836(68)90260-x. [DOI] [PubMed] [Google Scholar]
- SADLER J. R., NOVICK A. THE PROPERTIES OF REPRESSOR AND THE KINETICS OF ITS ACTION. J Mol Biol. 1965 Jun;12:305–327. doi: 10.1016/s0022-2836(65)80255-8. [DOI] [PubMed] [Google Scholar]
- Signer E. R., Beckwith J. R., Brenner S. Mapping of suppressor loci in Escherichia coli. J Mol Biol. 1965 Nov;14(1):153–166. doi: 10.1016/s0022-2836(65)80237-6. [DOI] [PubMed] [Google Scholar]
- Soll L., Berg P. Recessive lethals: a new class of nonsense suppressors in Escherichia coli. Proc Natl Acad Sci U S A. 1969 Jun;63(2):392–399. doi: 10.1073/pnas.63.2.392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stretton A. O., Kaplan S., Brenner S. Nonsense codons. Cold Spring Harb Symp Quant Biol. 1966;31:173–179. doi: 10.1101/sqb.1966.031.01.025. [DOI] [PubMed] [Google Scholar]
- UDAKA S., HORIUCHI T. MUTANTS OF ESCHERICHIA COLI HAVING TEMPERATURE SENSITIVE REGULATORY MECHANISM IN THE FORMATION OF ARGININE BIOSYNTHETIC ENZYMES. Biochem Biophys Res Commun. 1965 Apr 9;19:156–160. doi: 10.1016/0006-291x(65)90496-1. [DOI] [PubMed] [Google Scholar]
- Udaka S. Isolation of the arginine repressor in Escherichia coli. Nature. 1970 Oct 24;228(5269):336–338. doi: 10.1038/228336a0. [DOI] [PubMed] [Google Scholar]
- Wallace B. J., Pittard J. Regulator gene controlling enzymes concerned in tyrosine biosynthesis in Escherichia coli. J Bacteriol. 1969 Mar;97(3):1234–1241. doi: 10.1128/jb.97.3.1234-1241.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yanofsky C., Ito J. Nonsense codons and polarity in the tryptophan operon. J Mol Biol. 1966 Nov 14;21(2):313–334. doi: 10.1016/0022-2836(66)90102-1. [DOI] [PubMed] [Google Scholar]
- Zubay G., Morse D. E., Schrenk W. J., Miller J. H. Detection and isolation of the repressor protein for the tryptophan operon of Escherichia coli. Proc Natl Acad Sci U S A. 1972 May;69(5):1100–1103. doi: 10.1073/pnas.69.5.1100. [DOI] [PMC free article] [PubMed] [Google Scholar]


