Abstract
Background
Angiotensin converting enzyme (ACE)‐inhibitor overdose may result in severe hypotension. Existing data do not adequately deal with the likely onset of haemodynamic effects, which has implications for the appropriate duration of monitoring in the acute‐care setting. Therefore, the relationship between the interval after an ACE‐inhibitor overdose and onset of hypotension was examined.
Methods
A retrospective case review of patients who attended our institution after an ACE‐inhibitor overdose between 1 January 2000 and 31 December 2005 was carried out. Data collected were heart rate, blood pressure, electrolytes, electrocardiogram variables, and interval between ingestion and lowest recorded blood pressure.
Results
33 patients (24 men) who presented after an ACE‐inhibitor overdose were identified. Median (interquartile range (IQR)) age was 49 (42–56) years, and stated quantity ingested was 20 (7–42) times the defined daily dose. The median (IQR) interval between ACE‐inhibitor ingestion and lowest recorded systolic blood pressure was 4.5 (3.8–5.5) h, diastolic blood pressure was 3.8 (3.3–6.5) h and mean blood pressure was 4.2 (3.5–5.5) h. Heart rate did not increase substantially in response.
Conclusion
The lowest blood pressure was recorded within a short interval after an ACE‐inhibitor overdose, irrespective of therapeutic intervention. Patients in whom hypotension has not occurred within 6 h of ingestion can be considered for discharge.
Angiotensin converting enzyme (ACE) inhibitors have assumed an increasingly prominent role in the treatment of hypertension, chronic heart failure and cardiovascular risk prevention over the past decade.1,2 The number of different ACE inhibitors has expanded and their overall accessibility has increased. An ACE‐inhibitor overdose may cause severe hypotension and has been reported after ingestion of captopril, enalapril, lisinopril and quinapril.3,4,5,6,7,8,9,10 Hypotension may be prolonged or fatal in severe cases.4,5 Patients with moderate to severe hypotension require close observation in a critical care environment, administration of intravenous fluids and inotropic support.11,12,13 Administration of intravenous angiotensin II may restore blood pressure where hypotension is refractory to other pressor agents,8,10 and naloxone and aminophylline have been effective in certain situations.6,14
Despite the potential to cause severe hypotension, the haemodynamic effects after an ACE‐inhibitor overdose are generally mild, and most patients do not require any specific treatment.11,12,13 Relatively few data are available to guide the optimal period required to detect hypotension in patients who present to hospital after an ACE‐inhibitor overdose. In the UK, advice for managing poisoned patients is provided by the National Poisons Information Service (NPIS) via TOXBASE, a web‐based resource accessible to emergency departments and other healthcare workers (http://www.spib.axl.co.uk).15 Box 1 summarises the current NPIS guidance, which indicates that patients should be observed for at least 6 h after an ACE‐inhibitor overdose, and for at least 24 h if hypotensive or symptomatic. The present study was designed to establish the time between ingestion and onset of hypotension in patients attending hospital after an ACE‐inhibitor overdose. The aim was to determine an appropriate period of observation to allow the detection of hypotension and thereby inform NPIS guidelines for this patient group.
Methods
A local registry was used to identify patients who had attended the emergency department of Royal Infirmary of Edinburgh after an ACE‐inhibitor overdose. Our operating policy is that patients who present to the emergency department after suspected drug ingestion are admitted to our toxicology unit for ongoing medical care and psychiatric review. Patients may be admitted to the high dependency unit, if non‐invasive ventilatory support is likely to be required, or to the intensive treatment unit if invasive ventilatory support, haemodialysis or other critical care intervention is likely to be required. All patients presenting to the emergency department were included.
The study design was a retrospective case review using a standardised data collection sheet. No patient‐identifiable data were collected, to preserve confidentiality. Information recorded was date and time of ingestion, type and quantity of ACE inhibitor ingested, type and amount of co‐ingested drugs or alcohol, pulse rate, systolic blood pressure (SBP), diastolic blood pressure (DBP), electrocardiographic intervals, serum electrolytes and creatinine. The quantity of ACE inhibitor ingested was expressed as a multiple of the World Health Organization defined daily dose for lisinopril (10 mg), ramipril (2.5 mg), enalapril (10 mg), perindopril (4 mg) and captopril (50 mg).16 Mean blood pressure (MBP) was calculated as DBP + ⅓ (SBP−DBP).
Box 1 Current National Poisons Information Service recommendations for management of acute angiotensin converting enzyme‐inhibitor overdose
Consider activated charcoal (50 g for adults; 1 g/kg for children) if the patient presents within 1 h.
Ensure clear airway and adequate ventilation.
Observe for a minimum of 6 h after ingestion if asymptomatic.
If symptomatic or hypotensive, observe for at least 24 h. If significant hypotension is documented, observe for 36 h as hypotensive episodes may recur up to 36 h after ingestion in some cases.
Monitor blood pressure and pulse; measure electrolytes.
Haemodynamic observations and laboratory investigations were expressed with respect to time after ingestion of ACE inhibitor. The time intervals between ACE inhibitor ingestion and minimum recorded SBP, DBP and MBP (tmin), and maximum recorded heart rate (tmax) were recorded in all patients. Data were also considered with respect to particular ACE inhibitors, and after excluding patients who had coingested other antihypertensive drugs. Additionally, MBP was expressed relative to the lowest value, to minimise the confounding effect of different baseline blood pressures.
The realistic setting in which observations were recorded meant that time‐points varied between patients. Therefore, a form of simple imputation was used to minimise the potential effect of unbalanced data on time‐trend analyses.17,18 These were standardised to 0.5, 1, 1.5, 2, 2.5, 3, 3.5, 4, 4.5, 5, 6, 7, 8, 10, 12 and 16 h after ingestion, and data were generated using a linear regression relationship established from data immediately before and after appropriate time‐points. This model minimises the effect of outliers and therefore underestimates the true magnitude of haemodynamic responses.19 However, it was selected to allow a clearer interpretation of the time‐course and onset of haemodynamic effects, which was more relevant to the aims of this study. Statistical analyses were carried out using StatsDirect Statistical Software.
Results
We identified 33 patients who had attended hospital after ACE‐inhibitor overdose, of whom 24 (73%) were men. The median (interquartile range) age was 49 (42–56) years. Eighteen patients had been receiving treatment with ACE inhibitors for hypertension (55%), 5 for previous myocardial infarction (15%) and 10 patients had ingested another person's drugs (30%). The ACE inhibitor ingested was enalapril in 11 patients, ramipril in 10 patients, lisinopril in 9 patients, perindopril in 2 patients and captopril in 1 patient; the quantity ingested was 20 (7–42) times as high as the defined daily dose. Co‐ingested agents were alcohol in 13 (39%), non‐steroidal anti‐inflammatory drugs in 8 (24%), β blockers in 7 (21%), paracetamol in 4 (12%), calcium channel blockers in 3 (9%) and diuretics in 3 (9%) patients.
Table 1 shows that median intervals between ACE‐inhibitor ingestion and lowest recorded SBP, DBP and MBP were 4.5, 3.8 and 4.2 h, respectively.
Table 1 Interval between ingestion and minimum systolic blood pressure, minimum diastolic blood pressure, minimum mean blood pressure and maximum heart rate.
| Whole study population: | |||||
|---|---|---|---|---|---|
| ACE inhibitor | n | tmin SBP | tmin DBP | tmin MBP | tmax HR |
| Enalapril | 11 | 4.8 (3.9–5.0) | 4.5 (3.3–5.0) | 4.8 (3.7–5.0) | 4.5 (2.5–7.5) |
| Ramipril | 10 | 5.3 (4.6–5.8) | 4.9 (4.0–6.5) | 4.9 (4.0–6.5) | 7.9 (7.2–9.7) |
| Lisinopril | 9 | 4.0 (3.8–7.0) | 5.5 (3.8–7.0) | 4.0 (3.8–6.0) | 6.5 (5.2–11.5) |
| Perindopril | 2 | 4.0 (3.4–4.6) | 4.4 (3.3–5.4) | 3.7 (3.0–4.5) | 1.4 (1.3–1.5) |
| Captopril | 1 | 7.5 (–) | 3.5 (–) | 7.5 (–) | 11.5 (–) |
| All | 33 | 4.8 (3.8–6.0) | 4.5 (3.3–6.0) | 4.8 (3.3–5.5) | 6.0 (3.0–10.0) |
| Population after exclusion of patients who had ingested other antihypertensives | |||||
|---|---|---|---|---|---|
| ACE inhibitor | n | tmin SBP | tmin DBP | tmin MBP | tmax HR |
| All | 20 | 4.5 (3.8–5.5) | 3.8 (3.3–6.5) | 4.2 (3.5–5.5) | 5.5 (2.7–9.0) |
ACE, angiotensin converting enzyme; DBP, diastolic blood pressure; HR, heart rate; MBP, mean blood pressure; SBP, systolic blood pressure.
Data are presented as median (interquartile range) for the whole study population (n = 33) and after exclusion of patients who had ingested other antihypertensive drugs (n = 20).
Intervals were similar after exclusion of patients who had coingested other antihypertensive drugs, and similar between different types of ACE inhibitor. Figures 1 and 2 show the standardised time‐trend analyses for absolute and relative MBP, respectively, and fig 3 shows the standardised time‐trend analysis for absolute heart rate. Minimum blood pressure was observed within 6 h of ingestion in 28 (85%) patients. A poor correlation was seen between the stated dose of ACE inhibitor ingested and minimum MBP: Spearman's coefficient of rank correlation (r) = −0.088 (95% confidence interval (CI) for r, −0.453 to 0.302, p = 0.654).
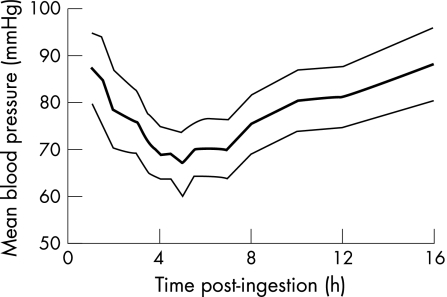
Figure 1 Time‐standardised absolute mean blood pressure, calculated as DBP + ⅓(SBP−DBP), after angiotensin converting enzyme‐inhibitor ingestion, presented as mean and 95% confidence interval.
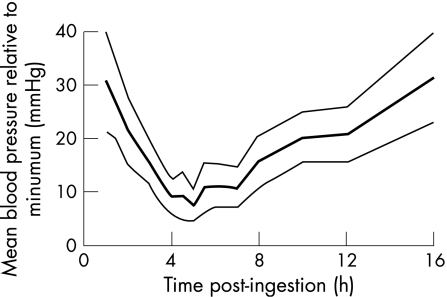
Figure 2 Time‐standardised relative mean blood pressure after angiotensin converting enzyme‐inhibitor overdose, calculated as DBP + ⅓(SBP− DBP) and subtracting the lowest recorded blood pressure. Data are presented as mean and 95% confidence interval.
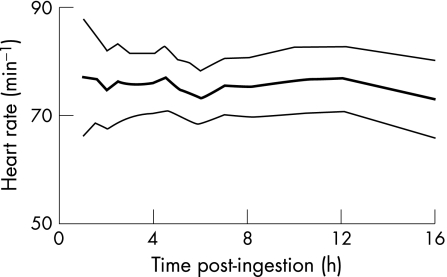
Figure 3 Time‐standardised absolute heart rate after angiotensin converting enzyme‐inhibitor ingestion, presented as mean and 95% confidence interval.
Intravenous fluids were required in most of the patients, including colloid fluids in 18 patients and crystalloids in 11 other patients. A 54‐year‐old man with angina, who was thought to have ingested ramipril 300 mg, ranitidine 4500 mg and simvastatin 600 mg, was transferred to the high dependency unit. Blood pressure had fallen to 74/36 mm Hg 4 h after ingestion and, in view of his established coronary artery disease, norepinephrine was given to maintain mean arterial pressure ⩾60 mm Hg. The norepinephrine infusion was discontinued after 12 h, and the patient transferred back to the toxicology unit shortly after. Blood pressure recovered without specific treatment in four patients. Table 2 summarises electrocardiogram and laboratory variables.
Table 2 Electrocardiogram variables and maximum recorded concentrations of serum potassium, urea and creatinine.
| Whole group (n = 33) | After exclusion of patients who coingested other antihypertensives (n = 20) | |
|---|---|---|
| ECG variables | ||
| PR (ms) | 166 (154–172) | 160 (149–170) |
| QRS (ms) | 85 (79–90) | 82 (77–86) |
| QT (ms) | 357 (335–402) | 346 (326–399) |
| QTC (ms) | 419 (399–433) | 417 (402–425) |
| Laboratory data | ||
| Potassiummax (mmol/l) | 3.9 (3.7–4.1) | 4.0 (3.9–4.1) |
| Ureamax (mmol/l) | 4.2 (2.5–4.2) | 4.1 (2.3–4.6) |
| Creatininemax (μmol/l) | 89 (75–115) | 79 (66–89) |
ECG, electrocardiogram; QTc, QT interval after correction by Bazett's formula.
Data are presented as median (interquartile range) for the whole study population (n = 33) and after exclusion of patients who had ingested other antihypertensive drugs (n = 20).
No marked electrocardiographic abnormalities occurred. A transient rise of serum creatinine concentration to >130 μmol/l occurred in six patients, and maximum concentration was 298 μmol/l. In four patients, renal impairment was evident within 6 h postingestion, whereas in two patients this emerged later. The severity of hypotension in both patients was sufficient to necessitate in‐patient care. In one patient, creatinine was 87 μmol/l at 1 h and rose to 140 μmol/l at 18 h, and in another patient creatinine was 75 μmol/l at 5 h and rose to 141 μmol/l at 16 h. Renal replacement treatment was not required in any patient. After recovery from the acute effects, the psychiatry team reviewed all patients, who were subsequently discharged home.
Discussion
The primary objective of the study was to examine the time delay between drug ingestion and onset of haemodynamic disturbance. Maximal blood pressure lowering was observed at around 5 h after ingestion, irrespective of the type of ACE inhibitor ingested and whether additional antihypertensive agents had been coingested. Monitoring of SBP, DBP or MBP seemed to be equally sensitive for detecting hypotension, which occurred within as few as 2–5 h after the overdose. Monitoring for at least 6 h after ingestion would have allowed detection of hypotension in all patients in this series and added a margin of safety to allow for uncertainty about the exact time of ACE‐inhibitor ingestion. Therefore, current NPIS recommendations to observe systemic blood pressure for at least 6 h after an ACE‐inhibitor overdose seem both safe and reasonable.
The number of patients seeking medical attention after an ACE‐inhibitor overdose is small, and constituted <0.1% of patients presenting to hospital after a drug overdose. The predominance of lisinopril, enalapril and ramipril in our patient population is reflective of prescribing practice in our hospital catchment area. Reassuringly, no life‐threatening adverse effects were encountered in any of our patients, which is consistent with expert opinion that clinical effects after an ACE‐inhibitor overdose are generally mild.11,12,13 Renal impairment is an important feature of ACE‐inhibitor toxicity and may be observed in patients who experience profound blood pressure lowering. Peak creatinine concentrations may not become apparent for several hours after ingestion and therefore consideration should be given to monitoring renal function in patients who develop severe hypotension. In this case series, efforts to ensure adequate circulating volume and systemic blood pressure allowed a spontaneous recovery of renal function.
Despite a substantial time‐dependent fall in systemic blood pressure, there was no correspondingly large increase in heart rate. Despite the ability of ACE inhibitors to lower blood pressure through vasodilatation, they do not provoke a reflex tachycardia, in contrast with other vasodilators.20 Several mechanisms have been proposed to explain this, including effects on baroreceptor sensitivity, vagal stimulation and reduced stimulation of sympathetic nerve activity.21 Therefore, tachycardia should not be expected as a feature of ACE‐inhibitor toxicity and is of limited value in determining poisoning severity in this patient group.
Some degree of caution should be exercised when interpreting the extent to which blood pressure fell after an ACE‐inhibitor overdose in this series. Maximal hypotensive effects would have been curtailed by therapeutic interventions, such as orthostatic manoeuvres, intravenous fluid administration and, in one patient, administration of norepinephrine. Therefore, the observed effects cannot be regarded as a natural pharmacodynamic profile after ingestion of a high‐dose ACE inhibitor.
A limitation of the data is that the effects of only a small number of ACE inhibitors were examined, predominantly enalapril, lisinopril and ramipril, which are in keeping with our regional formulary recommendations. Prescribing patterns vary considerably in other regions, and our findings may not apply directly to patients who have ingested other ACE inhibitors, such as benazapril, moexipril, quinapril and trandolapril.22 Nonetheless, these other ACE inhibitors achieve peak plasma concentrations at similar times (tmax) to those studied,23 and ACE inhibitors are all considered equally effective at lowering blood pressure.24
We conclude that patients who present to hospital after an ACE inhibitor overdose should be considered for discharge if hypotension has not occurred within 6 h of ingestion.
Acknowledgements
We thank Ross Davies, Scottish Poisons Information Bureau, and Lisa Galloway, Royal Infirmary of Edinburgh, for their help with identification and retrieval of patient casenotes.
Abbreviations
ACE - angiotensin converting enzyme
DBP - diastolic blood pressure
MBP - mean blood pressure
NPIS - National Poisons Information Service
SBP - systolic blood pressure
Footnotes
Competing interests: None declared.
References
- 1.Bennett K, Johnson H, Dack P.et al Changes in prevalence of and prescribing for ischaemic heart disease in Ireland 1990–2002. Ir J Med Sci 20051744–8. [DOI] [PubMed] [Google Scholar]
- 2.Gnani S, Gray J, Khunti K.et al Managing heart failure in primary care: first steps in implementing the National Service Framework. J Public Health 20042642–47. [DOI] [PubMed] [Google Scholar]
- 3.Augenstein W L, Kulig K W, Rumack B H. Captopril overdose resulting in hypotension. JAMA 19882593302–3305. [PubMed] [Google Scholar]
- 4.Park H, Purnell G V, Mirchandani H G. Suicide by captopril overdose. J Toxicol Clin Toxicol 199028379–382. [DOI] [PubMed] [Google Scholar]
- 5.Barr C S, Payne R, Newton R W. Profound prolonged hypotension following captopril overdose. Postgrad Med J 199167953–954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Varon J, Duncan S R. Naloxone reversal of hypotension due to captopril overdose. Ann Emerg Med 1991201125–1127. [DOI] [PubMed] [Google Scholar]
- 7.Jackson T, Corke C, Agar J. Enalapril overdose treated with angiotensin infusion. Lancet 1993341703. [DOI] [PubMed] [Google Scholar]
- 8.Newby D E, Lee M R, Gray A J.et al Enalapril overdose and the corrective effect of intravenous angiotensin II. Br J Clin Pharmacol 199540103–104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dawson A H, Harvey D, Smith A J.et al Lisinopril overdose. Lancet 1990335487–488. [DOI] [PubMed] [Google Scholar]
- 10.Trilli L E, Johnson K A. Lisinopril overdose and management with intravenous angiotensin II. Ann Pharmacother 1994281165–1168. [DOI] [PubMed] [Google Scholar]
- 11.Varughese A, Taylor A A, Nelson E B. Consequences of angiotensin‐converting enzyme inhibitor overdose. Am J Hypertens 19892355–357. [DOI] [PubMed] [Google Scholar]
- 12.Johnston G D, Smith A M. Management of overdose due to antihypertensive agents. Adverse Drug React Acute Poisoning Rev 1990975–89. [PubMed] [Google Scholar]
- 13.Lip G Y, Ferner R E. Poisoning with anti‐hypertensive drugs: angiotensin converting enzyme inhibitors. Hypertension 19959711–715. [PubMed] [Google Scholar]
- 14.Roberge R J, Rossetti M L, Rosetti J M. Aminophylline reversal of antihypertensive agent toxicity. Vet Hum Toxicol 200143285–287. [PubMed] [Google Scholar]
- 15.Bateman D N, Good A M, Laing W J.et al TOXBASE: poisons information on the internet. Emerg Med J 20021931–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.World Health Organization Collaborative Centre for Drug Statistics Methodology ATC/DDD Index 2006. http://www.whocc.no/atcddd (accessed 31 Aug 2006)
- 17.Gornbein J A, Lazaro C G, Little R J. Incomplete data in repeated measures analysis. Stat Methods Med Res 19921275–295. [DOI] [PubMed] [Google Scholar]
- 18.Schafer J L. Multiple imputation: a primer. Stat Methods Med Res 199983–15. [DOI] [PubMed] [Google Scholar]
- 19.Kenward M G, Molenberghs G. Parametric models for incomplete continuous and categorical longitudinal data. Stat Methods Med Res 1999851–83. [DOI] [PubMed] [Google Scholar]
- 20.Brown N J, Vaughan D E. Angiotensin‐converting enzyme inhibitors. Circulation 1998971411–1420. [DOI] [PubMed] [Google Scholar]
- 21.Giannettasio C, Grassi G, Seravalle G.et al Investigation of reflexes from volume and baroreceptors during converting‐enzyme inhibition in humans. Am Heart J 1989117740–745. [DOI] [PubMed] [Google Scholar]
- 22.Shrank W H, Ettner S L, Glassman P.et al A bitter pill: formulary variability and the challenge to prescribing physicians. J Am Board Fam Pract 200417401–407. [DOI] [PubMed] [Google Scholar]
- 23.Song J C, White C M. Clinical pharmacokinetics and selective pharmacodynamics of new angiotensin converting enzyme inhibitors: an update. Clin Pharmacokinet 200241207–224. [DOI] [PubMed] [Google Scholar]
- 24.Lopez‐Sendon J, Swedberg K, McMurray J.et al Expert consensus document on angiotensin converting enzyme inhibitors in cardiovascular disease. The task force on ACE‐inhibitors of the European Society of Cardiology. Eur Heart J 2004251454–1470. [DOI] [PubMed] [Google Scholar]