Abstract
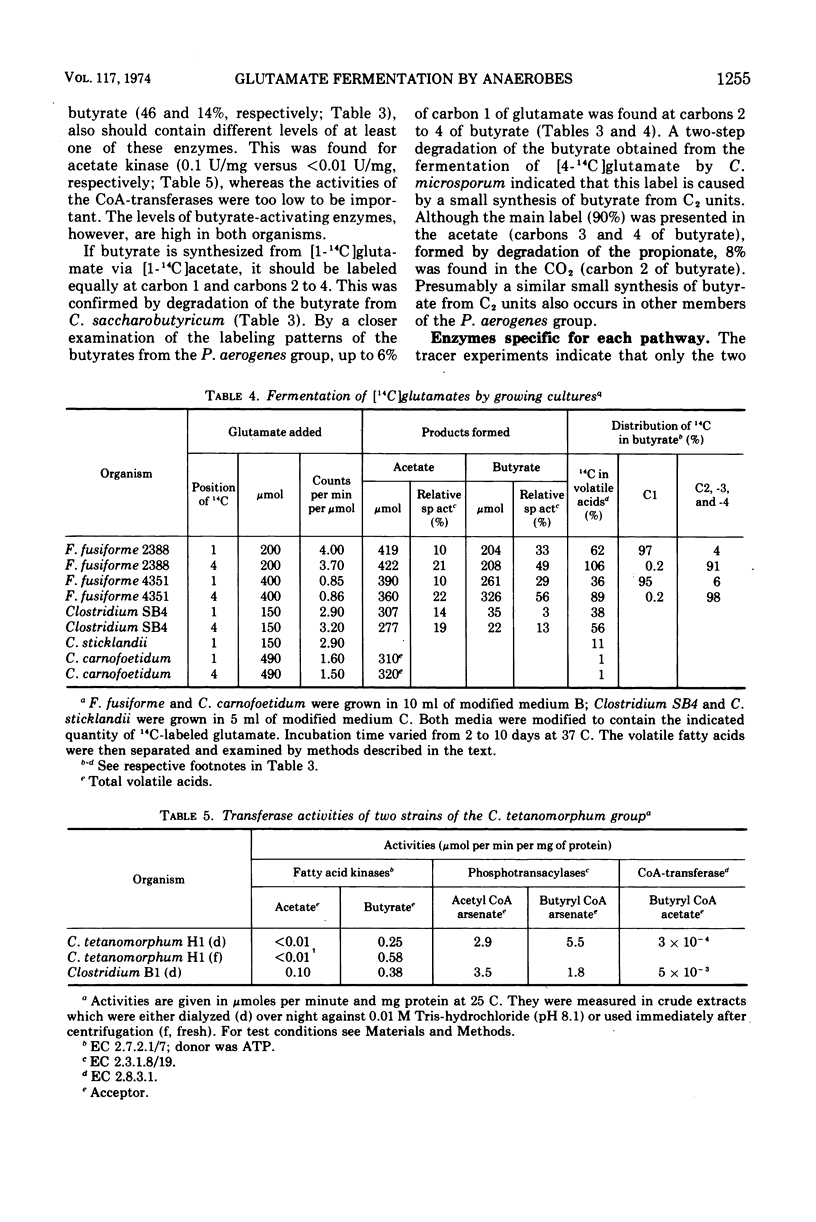
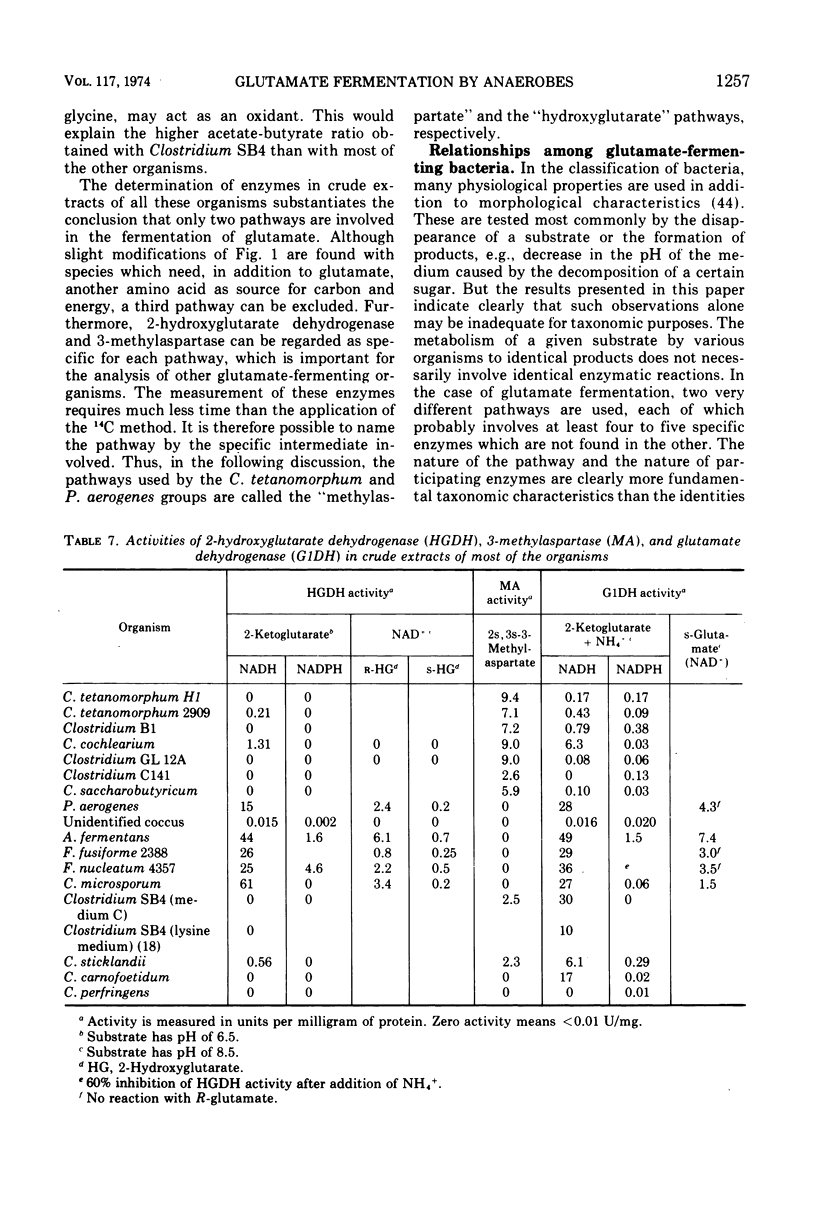
Two pathways are involved in the fermentation of glutamate to acetate, butyrate, carbon dioxide, and ammonia—the methylaspartate and the hydroxyglutarate pathways which are used by Clostridium tetanomorphum and Peptococcus aerogenes, respectively. Although these pathways give rise to the same products, they are easily distinguished by different labeling patterns of the butyrate when [4-14C]glutamate is used as substrate. Schmidt degradation of the radioactive butyrate from C. tetanomorphum yielded equally labeled propionate and carbon dioxide, whereas nearly all the radioactivity of the butyrate from P. aerogenes was recovered in the corresponding propionate. This procedure was used as a test for the pathway of glutamate fermentation by 15 strains (9 species) of anaerobic bacteria. The labeling patterns of the butyrate indicate that glutamate is fermented via the methylaspartate pathway by C. tetani, C. cochlearium, and C. saccarobutyricum, and via the hydroxyglutarate pathway by Acidaminococcus fermentans, C. microsporum, Fusobacterium nucleatum, and F. fusiformis. Enzymes specific for each pathway were assayed in crude extracts of the above organisms. 3-Methylaspartase was found only in clostridia which use the methylaspartate pathway, including Clostridium SB4 and C. sticklandii, which probably degrade glutamate to acetate and carbon dioxide by using a second amino acid as hydrogen acceptor. High levels of 2-hydroxyglutarate dehydrogenase were found exclusively in organisms that use the hydroxyglutarate pathway. The data indicate that only two pathways are involved in the fermentation of glutamate by the bacteria analyzed. The methylaspartate pathway appears to be used only by species of Clostridium, whereas the hydroxyglutarate pathway is used by representatives of several genera.
Full text
PDF










Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anderson R. L., Wood W. A. Carbohydrate metabolism in microorganisms. Annu Rev Microbiol. 1969;23:539–578. doi: 10.1146/annurev.mi.23.100169.002543. [DOI] [PubMed] [Google Scholar]
- BARKER H. A., SMYTH R. D., WAWSZKIEWICZ E. J., LEE M. N., WILSON R. M. Enzymic preparation and characterization of an alpha-L-beta-methylaspartic acid. Arch Biochem Biophys. 1958 Dec;78(2):468–476. doi: 10.1016/0003-9861(58)90371-0. [DOI] [PubMed] [Google Scholar]
- Bergmeyer H. U., Moellering H. Enzymatische Bestimmung von Acetat. Biochem Z. 1966 Mar 28;344(2):167–189. [PubMed] [Google Scholar]
- Blair A. H., Barker H. A. Assay and purification of (+)-citramalate hydro-lyase components from Clostridium tetanomorphum. J Biol Chem. 1966 Jan 25;241(2):400–408. [PubMed] [Google Scholar]
- Buckel W., Eggerer H. Zur optischen Bestimmung von Citrat-Synthase und von Acetyl-Coenzym A. Biochem Z. 1965 Nov 5;343(1):29–43. [PubMed] [Google Scholar]
- Clifton C. E. The Utilization of Amino Acids and Related Compounds by Clostridium tetani. J Bacteriol. 1942 Aug;44(2):179–183. doi: 10.1128/jb.44.2.179-183.1942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Costilow R. N., Rochovansky O. M., Barker H. A. Isolation and identification of beta-lysine as an intermediate in lysine fermentation. J Biol Chem. 1966 Apr 10;241(7):1573–1580. [PubMed] [Google Scholar]
- Goldmark P. J., Linn S. An endonuclease activity from Escherichia coli absent from certain rec- strains. Proc Natl Acad Sci U S A. 1970 Sep;67(1):434–441. doi: 10.1073/pnas.67.1.434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hill L. R. An index to deoxyribonucleic acid base compositions of bacterial species. J Gen Microbiol. 1966 Sep;44(3):419–437. doi: 10.1099/00221287-44-3-419. [DOI] [PubMed] [Google Scholar]
- Horler D. F., McConnell W. B., Westlake D. W. Glutaconic acid, a product of the fermentation of glutamic acid by Peptococcus aerogenes. Can J Microbiol. 1966 Dec;12(6):1247–1252. doi: 10.1139/m66-166. [DOI] [PubMed] [Google Scholar]
- Horler D. F., Westlake D. W., McConnell W. B. Conversion of glutamic acid to volatile acids by Micrococcus aerogenes. Can J Microbiol. 1966 Feb;12(1):47–53. doi: 10.1139/m66-008. [DOI] [PubMed] [Google Scholar]
- JACKINS H. C., BARKER H. A. Fermentative processes of the fusiform bacteria. J Bacteriol. 1951 Feb;61(2):101–114. doi: 10.1128/jb.61.2.101-114.1951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson W. M., Westlake D. W. Purification and characterization of glutamic acid dehydrogenase and -ketoglutaric acid reductase from Peptococcus aerogenes. Can J Microbiol. 1972 Jun;18(6):881–892. doi: 10.1139/m72-136. [DOI] [PubMed] [Google Scholar]
- Johnson W. M., Westlake D. W. The production of alpha-hydroxyglutaric acid from glutamic acid by cell-free preparations of Peptococcus aerogenes. Can J Biochem. 1969 Dec;47(12):1103–1107. doi: 10.1139/o69-177. [DOI] [PubMed] [Google Scholar]
- Kew O. M., Woolfolk C. A. Preparation of glutamate dehydrogenase from Micrococcus aerogenes. Biochem Biophys Res Commun. 1970;39(6):1126–1133. doi: 10.1016/0006-291x(70)90676-5. [DOI] [PubMed] [Google Scholar]
- LEAVER F. W., WOOD H. G., STJERNHOLM R. The fermentation of three carbon substrates by Clostridium propionicum and Propionibacterium. J Bacteriol. 1955 Nov;70(5):521–530. doi: 10.1128/jb.70.5.521-530.1955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Lenz H., Buckel W., Wunderwald P., Biedermann G., Buschmeier V., Eggerer H., Cornforth J. W., Redmond J. W., Mallaby R. Stereochemistry of si-citrate synthase and ATP-citrate-lyase reactions. Eur J Biochem. 1971 Dec;24(2):207–215. doi: 10.1111/j.1432-1033.1971.tb19672.x. [DOI] [PubMed] [Google Scholar]
- Lerud R. F., Whiteley H. R. Purification and properties of alpha-ketoglutarate reductase from Micrococcus aerogenes. J Bacteriol. 1971 May;106(2):571–577. doi: 10.1128/jb.106.2.571-577.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MOORE S., STEIN W. H. A modified ninhydrin reagent for the photometric determination of amino acids and related compounds. J Biol Chem. 1954 Dec;211(2):907–913. [PubMed] [Google Scholar]
- MOSBACH E. H., PHARES E. F., CARSON S. F. Degradation of isotopically labeled citric, alpha-ketoglutaric and glutamic acids. Arch Biochem Biophys. 1951 Sep;33(2):179–185. doi: 10.1016/0003-9861(51)90095-1. [DOI] [PubMed] [Google Scholar]
- McConnell W. B., Horler D. F., Westlake D. W. Metabolism of histidine by Peptococcus aerogenes. Can J Microbiol. 1967 Feb;13(2):143–146. doi: 10.1139/m67-020. [DOI] [PubMed] [Google Scholar]
- PHARES E. F. Degradation of labeled propionic and acetic acids. Arch Biochem Biophys. 1951 Sep;33(2):173–178. doi: 10.1016/0003-9861(51)90094-x. [DOI] [PubMed] [Google Scholar]
- Rogosa M. Acidaminococcus gen. n., Acidaminococcus fermentans sp. n., anaerobic gram-negative diplococci using amino acids as the sole energy source for growth. J Bacteriol. 1969 May;98(2):756–766. doi: 10.1128/jb.98.2.756-766.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- STADTMAN E. R. The coenzyme A transphorase system in Clostridium kluyveri. J Biol Chem. 1953 Jul;203(1):501–512. [PubMed] [Google Scholar]
- STADTMAN T. C., MCCLUNG L. S. Clostridium sticklandii nov. spec. J Bacteriol. 1957 Feb;73(2):218–219. doi: 10.1128/jb.73.2.218-219.1957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- STADTMAN T. C. On the metabolism of an amino acid fermenting Clostridium. J Bacteriol. 1954 Mar;67(3):314–320. doi: 10.1128/jb.67.3.314-320.1954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spray R. S. Semisolid Media for Cultivation and Identification of Sporulating Anaerobes. J Bacteriol. 1936 Aug;32(2):135–155. doi: 10.1128/jb.32.2.135-155.1936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stjernholm R., Wood H. G. METHYLMALONYL ISOMERASE, II. PURIFICATION AND PROPERTIES OF THE ENZYME FROM PROPIONIBACTERIA. Proc Natl Acad Sci U S A. 1961 Mar;47(3):303–313. doi: 10.1073/pnas.47.3.303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- TONOMURA B., MALKIN R., RABINOWITZ J. C. DEOXYRIBONUCLEIC ACID BASE COMPOSITION OF CLOSTRIDIAL SPECIES. J Bacteriol. 1965 May;89:1438–1439. doi: 10.1128/jb.89.5.1438-1439.1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- TWAROG R., WOLFE R. S. Enzymatic phosphorylation of butyrate. J Biol Chem. 1962 Aug;237:2474–2477. [PubMed] [Google Scholar]
- WACHSMAN J. T., BARKER H. A. Tracer experiments on glutamate fermentation by Clostridium tetanomorphum. J Biol Chem. 1955 Dec;217(2):695–702. [PubMed] [Google Scholar]
- WHITELEY H. R. Fermentation of amino acids by Micrococcus aerogenes. J Bacteriol. 1957 Sep;74(3):324–330. doi: 10.1128/jb.74.3.324-330.1957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woods D. D., Clifton C. E. Studies in the metabolism of the strict anaerobes (genus Clostridium): Hydrogen production and amino-acid utilization by Clostridium tetanomorphum. Biochem J. 1937 Oct;31(10):1774–1788. doi: 10.1042/bj0311774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woods D. D., Trim A. R. Studies in the metabolism of the strict anaerobes: The metabolism of amino-acids by Cl. welchii. Biochem J. 1942 Jun;36(5-6):501–512. doi: 10.1042/bj0360501. [DOI] [PMC free article] [PubMed] [Google Scholar]


