Abstract
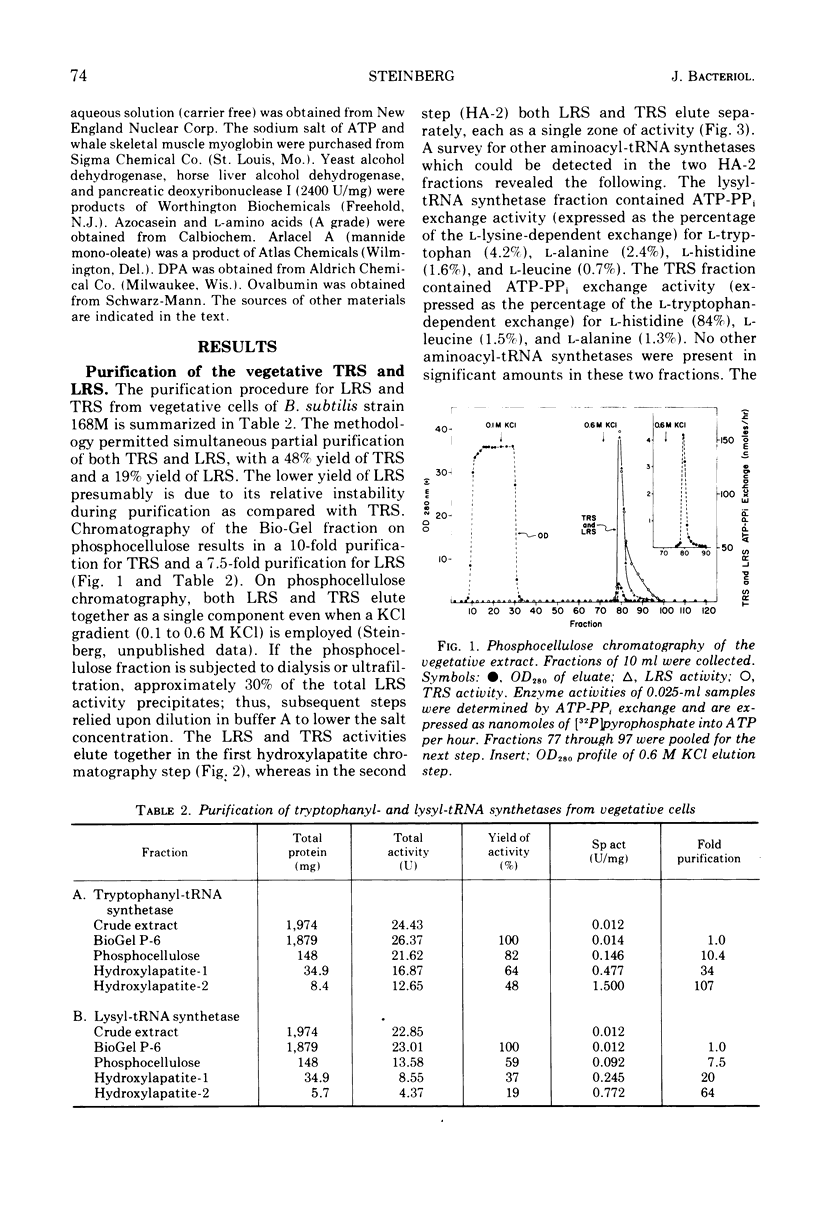
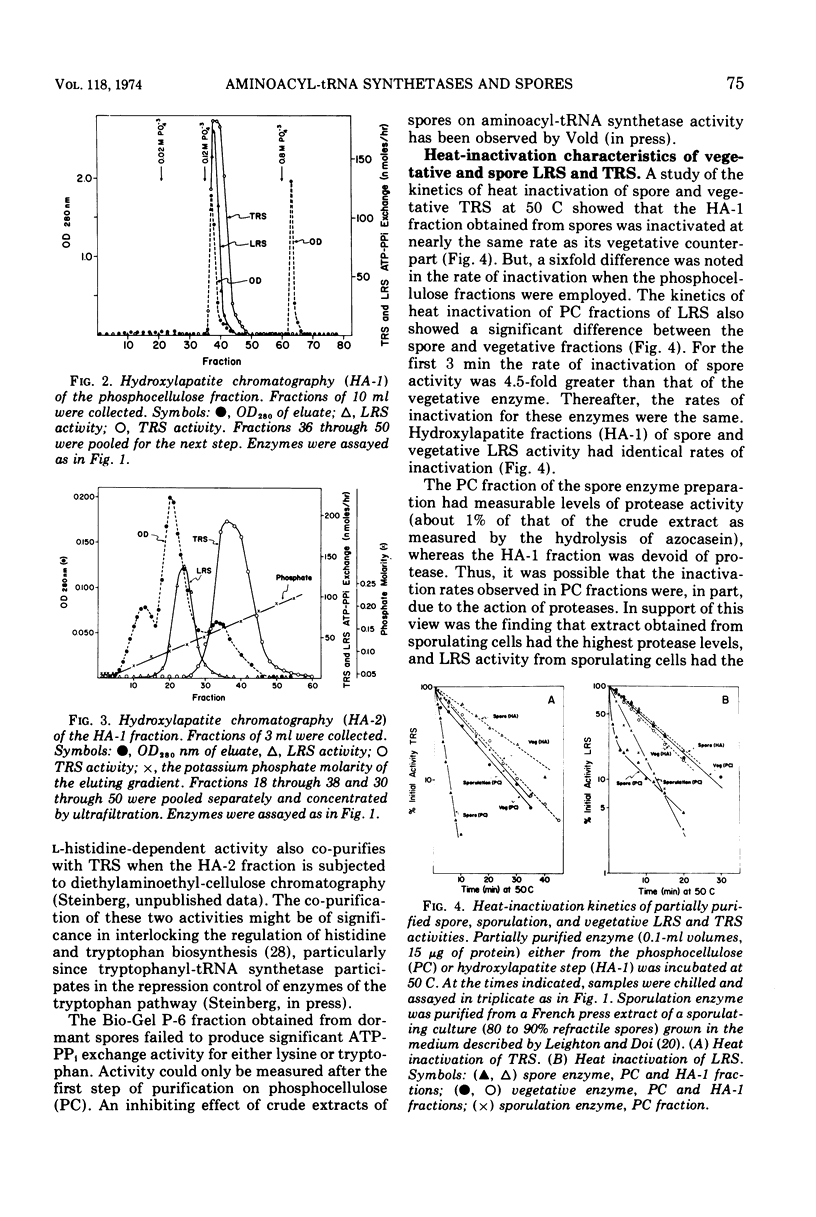
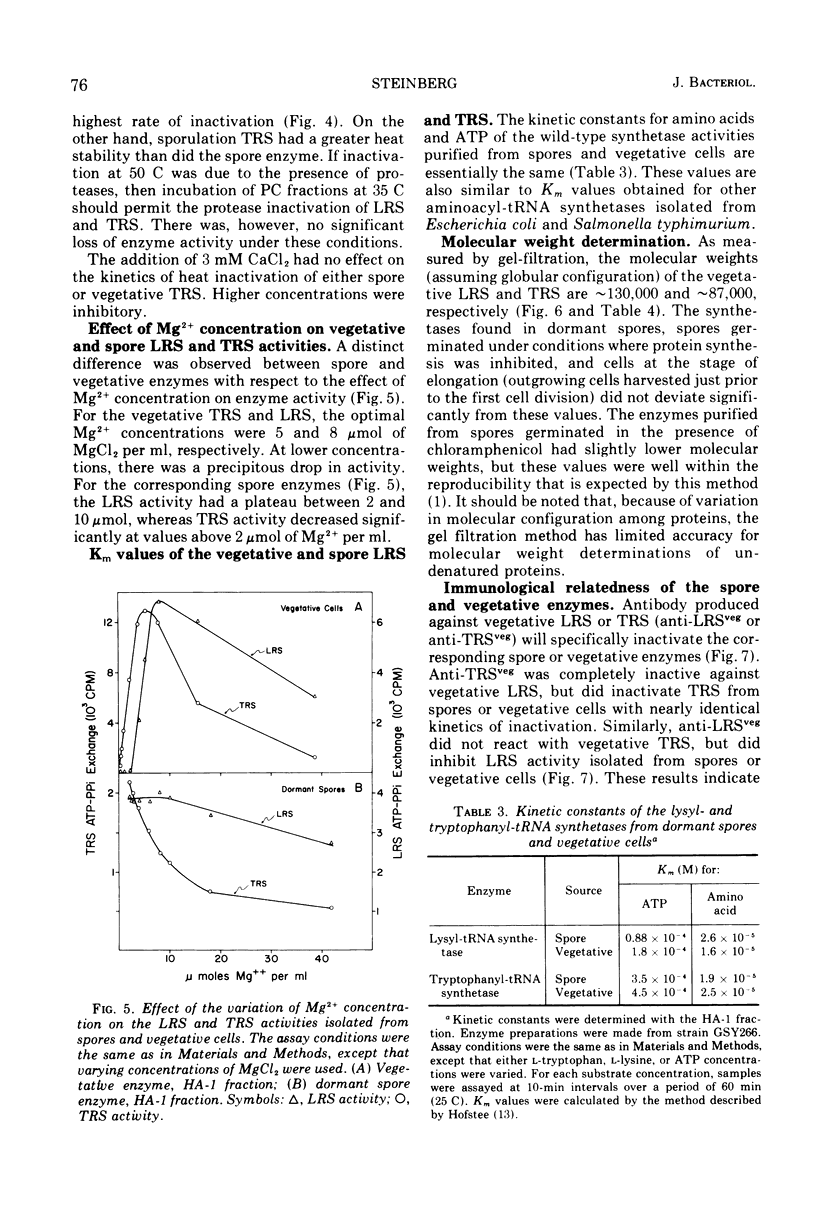
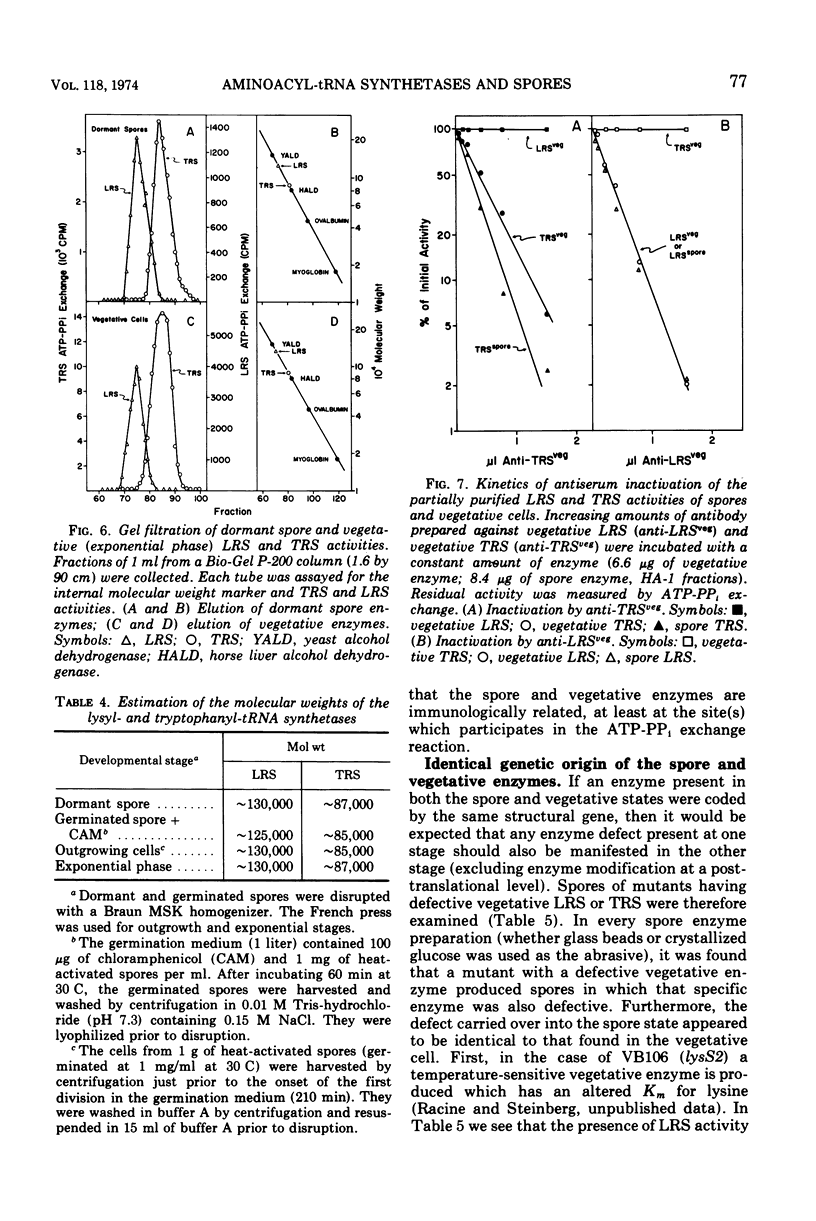
The lysyl-transfer ribonucleic acid synthetase (LRS) and tryptophanyl-transfer ribonucleic acid synthetases (TRS) (l-lysine:tRNA ligase [AMP], EC 6.1.1.6; and l-tryptophan:tRNA ligase [AMP], EC 6.1.1.2) have been purified 60- and 100-fold, respectively, from vegetative cells and spores of Bacillus subtilis. There are no significant differences between the corresponding spore and vegetative enzymes with respect to their elution characteristics from columns of phosphocellulose or hydroxylapatite, their molecular weight (~130,000 for LRS and ~87,000 for TRS as determined by gel filtration), their kinetic constants for substrates (in the amino acid-dependent adenosine triphosphate-pyrophosphate exchange reaction), and the kinetics of inactivation by heat and by antibody. The Mg2+ requirement for optimal enzyme activity of the corresponding spore and vegetative enzyme differ slightly. Mutants having defective (temperature sensitive) vegetative LRS or TRS activities produce spores in which these enzymes are also defective. The mutant spores are more heat sensitive than the parental type, but contain normal levels of dipicolinic acid. They germinate normally at the restrictive temperature (43 C), but are blocked at specific developmental stages in outgrowth. No modification in temperature sensitivity phenotype occurs during outgrowth, nor is there a change in molecular weight of the two enzymes. The implication is that the LRS and TRS activities of the vegetative and spore stages are each coded (at least in part) by the same structural gene. The temperature sensitivity of mutant spores is discussed with respect to those factors which are involved in the formation of the heat-resistant state.
Full text
PDF






Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ackers G. K. Analytical gel chromatography of proteins. Adv Protein Chem. 1970;24:343–446. doi: 10.1016/s0065-3233(08)60245-4. [DOI] [PubMed] [Google Scholar]
- Andrews P. The gel-filtration behaviour of proteins related to their molecular weights over a wide range. Biochem J. 1965 Sep;96(3):595–606. doi: 10.1042/bj0960595. [DOI] [PMC free article] [PubMed] [Google Scholar]
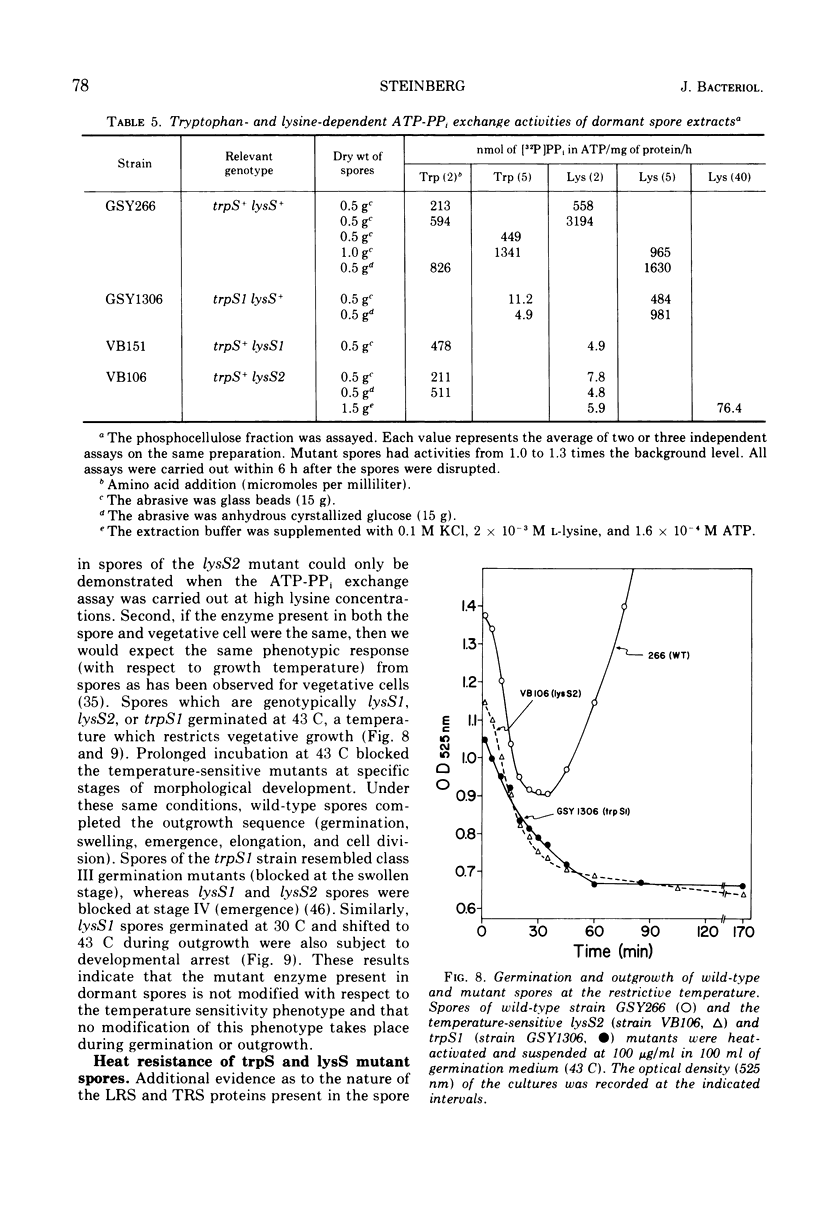
- Demain A. L., Newkirk J. F. DISSOCIATION OF SPORE GERMINATION FROM OUTGROWTH BY USE OF AUXOTROPHIC MUTANTS OF BACILLUS SUBTILIS. J Bacteriol. 1960 Jun;79(6):783–788. doi: 10.1128/jb.79.6.783-788.1960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doering J. L., Bott K. F. Differential amino acid requirements for sporulation in Bacillus subtilis. J Bacteriol. 1972 Oct;112(1):345–355. doi: 10.1128/jb.112.1.345-355.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Engelbrecht H. L., Sadoff H. L. Properties of purine nucleoside phosphorylases from spores and vegetative cells of Bacillus cereus and their modification by orthophosphate. J Biol Chem. 1969 Nov 25;244(22):6228–6232. [PubMed] [Google Scholar]
- FREESE E., PARK S. W., CASHEL M. THE DEVELOPMENTAL SIGNIFICANCE OF ALANINE DEHYDROGENASE IN BACILLUS SUBTILIS. Proc Natl Acad Sci U S A. 1964 Jun;51:1164–1172. doi: 10.1073/pnas.51.6.1164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Falaschi A., Kornberg A. Biochemical studies of bacterial sporulation. II. Deoxy- ribonucleic acid polymerase in spores of Bacillus subtilis. J Biol Chem. 1966 Apr 10;241(7):1478–1482. [PubMed] [Google Scholar]
- Fangman W. L., Nass G., Neidhardt F. C. Immunological and chemical studies of phenylalanyl sRNA synthetase from Escherichia coli. J Mol Biol. 1965 Aug;13(1):202–219. doi: 10.1016/s0022-2836(65)80090-0. [DOI] [PubMed] [Google Scholar]
- Freese E., Fortnagel P. Analysis of sporulation mutants. I. Response of uracil incorporation to carbon sources, and other mutant properties. J Bacteriol. 1967 Dec;94(6):1957–1969. doi: 10.1128/jb.94.6.1957-1969.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fukuda A., Gilvarg C. The relationship of dipicolinate and lysine biosynthesis in Bacillus megaterium. J Biol Chem. 1968 Jul 25;243(14):3871–3876. [PubMed] [Google Scholar]
- Hanson R. S., Curry M. V., Garner J. V., Halvorson H. O. Mutants of Bacillus cereus strain T that produce thermoresistant spores lacking dipicolinate and have low levels of calcium. Can J Microbiol. 1972 Jul;18(7):1139–1143. doi: 10.1139/m72-175. [DOI] [PubMed] [Google Scholar]
- Ilan J., Patel N. Mechanism of gene expression in Tenebrio molitor. Juvenile hormone determination of translational control through transfer ribonucleic acid and enzyme. J Biol Chem. 1970 Mar 25;245(6):1275–1281. [PubMed] [Google Scholar]
- JANSSEN F. W., LUND A. J., ANDERSON L. E. Colorimetric assay for dipicolinic acid in bacterial spores. Science. 1958 Jan 3;127(3288):26–27. doi: 10.1126/science.127.3288.26. [DOI] [PubMed] [Google Scholar]
- Kaplan S., Anderson D. Selection of temperature-sensitive activating enzyme mutants in Escherichia coli. J Bacteriol. 1968 Mar;95(3):991–997. doi: 10.1128/jb.95.3.991-997.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kornberg A., Spudich J. A., Nelson D. L., Deutscher M. P. Origin of proteins in sporulation. Annu Rev Biochem. 1968;37:51–78. doi: 10.1146/annurev.bi.37.070168.000411. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Lazzarini R. A. Differences in lysine-sRNA from spore and vegetative cells of Bacillus subtillis. Proc Natl Acad Sci U S A. 1966 Jul;56(1):185–190. doi: 10.1073/pnas.56.1.185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lazzarini R. A., Santangelo E. Medium-dependent alteration of lysine transfer ribonucleic acid in sporulating Bacillus subtilis cells. J Bacteriol. 1967 Jul;94(1):125–130. doi: 10.1128/jb.94.1.125-130.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leighton T. J., Doi R. H. The stability of messenger ribonucleic acid during sporulation in Bacillus subtilis. J Biol Chem. 1971 May 25;246(10):3189–3195. [PubMed] [Google Scholar]
- Linn T. G., Greenleaf A. L., Shorenstein R. G., Losick R. Loss of the sigma activity of RNA polymerase of Bacillus subtilis during sporulation. Proc Natl Acad Sci U S A. 1973 Jun;70(6):1865–1869. doi: 10.1073/pnas.70.6.1865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neidhardt F. C. Roles of amino acid activating enzymes in cellular physiology. Bacteriol Rev. 1966 Dec;30(4):701–719. doi: 10.1128/br.30.4.701-719.1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prestidge L., Gage V., Spizizen J. Protease activities during the course of sporulation on Bacillus subtilis. J Bacteriol. 1971 Sep;107(3):815–823. doi: 10.1128/jb.107.3.815-823.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Printz D. B., Gross S. R. An apparent relationship between mistranslation and an altered leucyl-tRNA synthetase in a conditional lethal mutant of Neurospora crassa. Genetics. 1967 Mar;55(3):451–467. doi: 10.1093/genetics/55.3.451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roth C. W., Nester E. W. Co-ordinate control of tryptophan, histidine and tyrosine enzyme synthesis in Bacillus subtilis. J Mol Biol. 1971 Dec 28;62(3):577–589. doi: 10.1016/0022-2836(71)90157-4. [DOI] [PubMed] [Google Scholar]
- Sadoff H. L., Celikkol E., Engelbrecht H. L. Conversion of bacterial aldolase from vegetative to spore form by a sporulation-specific protease. Proc Natl Acad Sci U S A. 1970 Jul;66(3):844–849. doi: 10.1073/pnas.66.3.844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sadoff H. L., Hitchins A. D., Celikkol E. Properties of fructose 1,6-diphosphate aldolases from spores and vegetative cells of Bacillus cereus. J Bacteriol. 1969 Jun;98(3):1208–1218. doi: 10.1128/jb.98.3.1208-1218.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spizizen J. TRANSFORMATION OF BIOCHEMICALLY DEFICIENT STRAINS OF BACILLUS SUBTILIS BY DEOXYRIBONUCLEATE. Proc Natl Acad Sci U S A. 1958 Oct 15;44(10):1072–1078. doi: 10.1073/pnas.44.10.1072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spudich J. A., Kornberg A. Biochemical studies of bacterial sporulation and germination. VI. Origin of spore core and coat proteins. J Biol Chem. 1968 Sep 10;243(17):4588–4599. [PubMed] [Google Scholar]
- Steinberg W., Anagnostopoulos C. Biochemical and genetic characterization of a temperature-sensitive, tryptophanyl-transfer ribonucleic acid synthetase mutant of Bacillus subtilis. J Bacteriol. 1971 Jan;105(1):6–19. doi: 10.1128/jb.105.1.6-19.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strehler B. L., Hendley D. D., Hirsch G. P. Evidence of a codon restriction hypothesis of cellular differentiation: multiplicity of mammalian leucyl-sRNA-specific synthetases and tissue-specific deficiency in an alanyl-sRNA synthetase. Proc Natl Acad Sci U S A. 1967 Jun;57(6):1751–1758. doi: 10.1073/pnas.57.6.1751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tono H., Kornberg A. Biochemical studies of bacterial sporulation. IV. Inorganic pyrophosphatase of vegetative cells and spores of Bacillus megaterium. J Bacteriol. 1967 Jun;93(6):1819–1824. doi: 10.1128/jb.93.6.1819-1824.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- VARY J. C., HALVORSON H. O. KINETICS OF GERMINATION OF BACILLUS SPORES. J Bacteriol. 1965 May;89:1340–1347. doi: 10.1128/jb.89.5.1340-1347.1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vallee B. L., Hoch F. L. ZINC, A COMPONENT OF YEAST ALCOHOL DEHYDROGENASE. Proc Natl Acad Sci U S A. 1955 Jun 15;41(6):327–338. doi: 10.1073/pnas.41.6.327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vold B. S. Analysis of isoaccepting transfer ribonucleic acid species of Bacillus subtilis: changes in chromatography of transfer ribonucleic acids associated with stage of development. J Bacteriol. 1973 Apr;114(1):178–182. doi: 10.1128/jb.114.1.178-182.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vold B. S. Analysis of isoaccepting transfer ribonucleic acid species of Bacillus subtilis: chromatographic differences between transfer ribonucleic acids from spores and cells in exponential growth. J Bacteriol. 1973 Feb;113(2):825–833. doi: 10.1128/jb.113.2.825-833.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vold B. S. Variations in activity of aminoacyl-tRNA synthetases as a function of development in Bacillus subtilis. Arch Biochem Biophys. 1973 Feb;154(2):691–695. doi: 10.1016/0003-9861(73)90024-6. [DOI] [PubMed] [Google Scholar]
- Williams A. L., Williams L. S. Control of arginine biosynthesis in Escherichia coli: characterization of arginyl-transfer ribonucleic acid synthetase mutants. J Bacteriol. 1973 Mar;113(3):1433–1441. doi: 10.1128/jb.113.3.1433-1441.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]



