Abstract
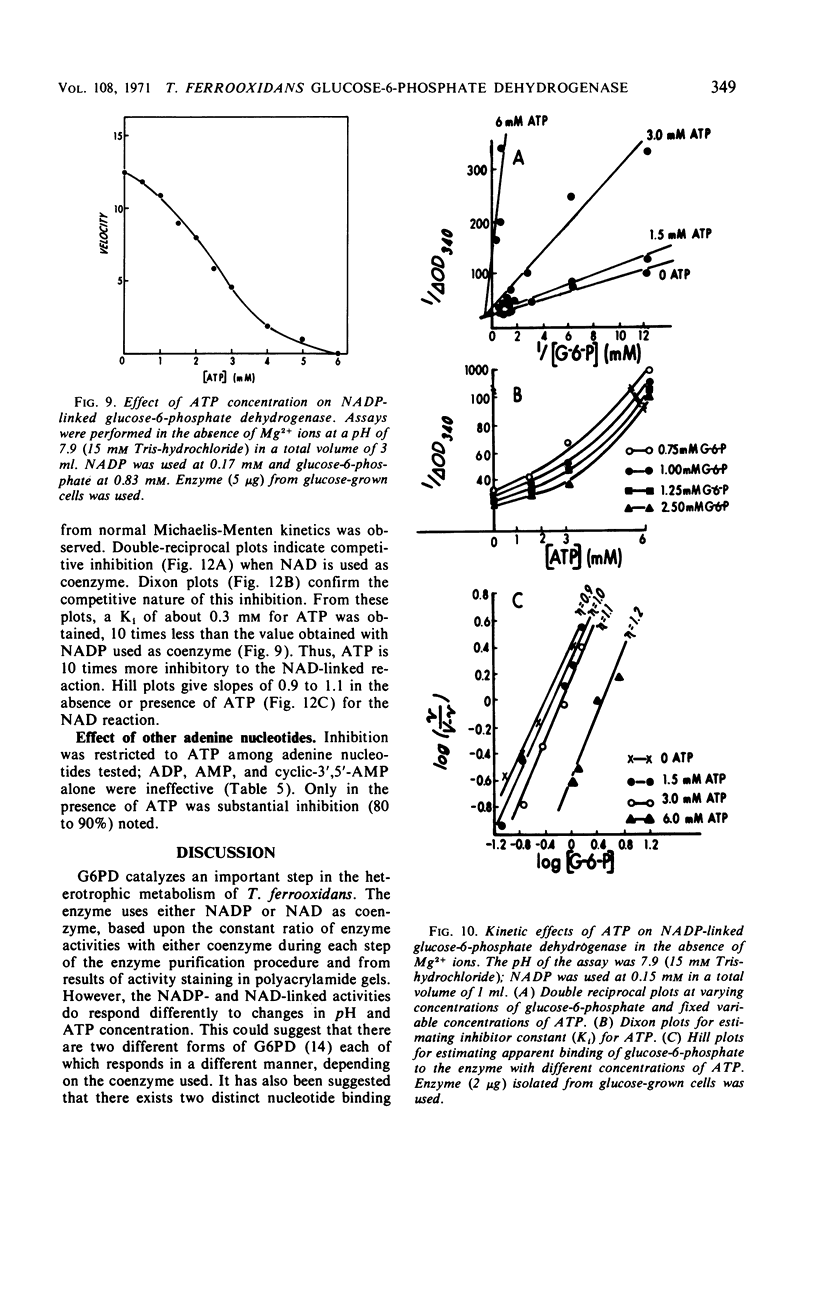
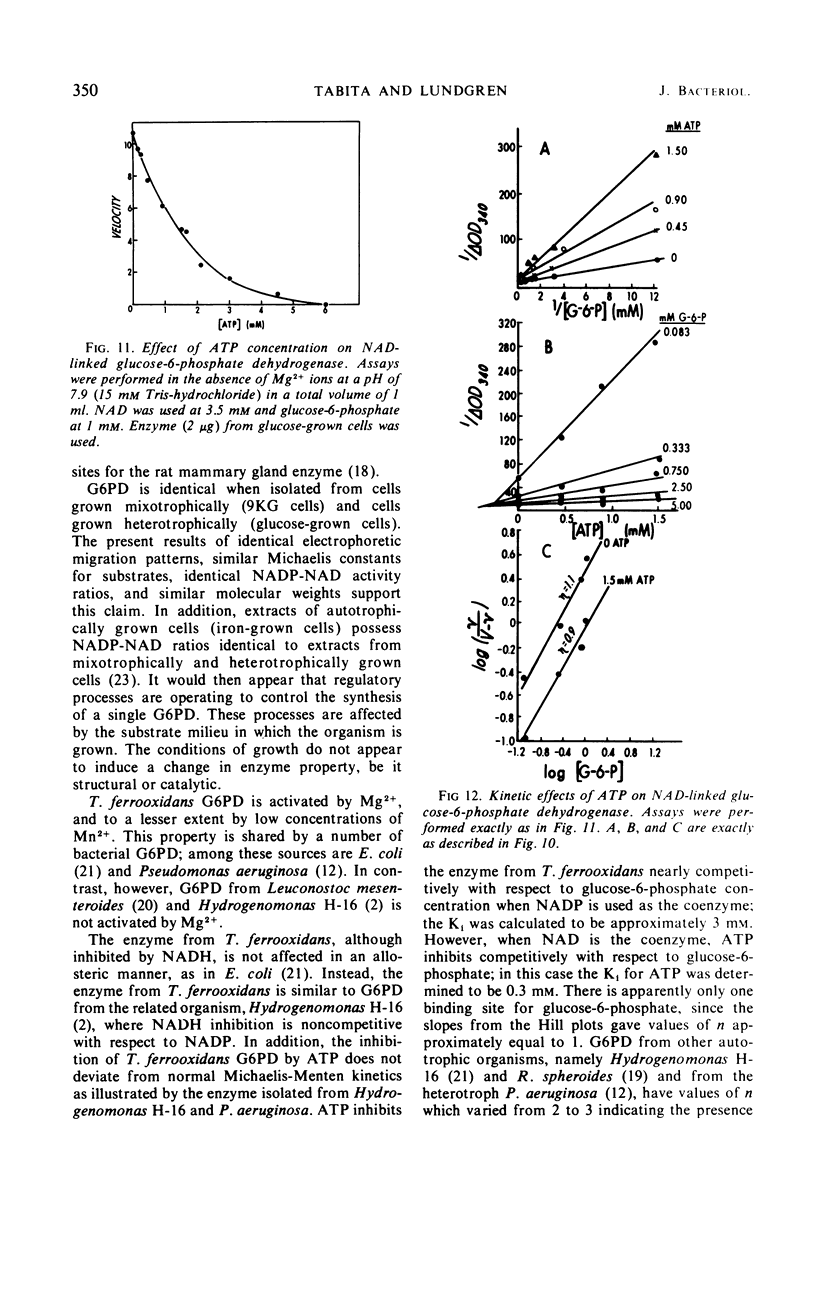
Glucose-6-phosphate dehydrogenase was partially purified from both glucose-grown and iron-glucose-grown Thiobacillus ferrooxidans. The enzyme possesses a dual nucleotide specificity for either nicotinamide adenine dinucleotide phosphate (NADP) or nicotinamide adenine dinucleotide (NAD) and has a molecular weight of 110,000 as determined by gel electrophoresis. Evidence is presented that T. ferrooxidans glucose-6-phosphate dehydrogenase is identical when isolated from cells grown mixotrophically (iron-glucose grown) or cells grown heterotrophically (glucose-grown cells). The enzyme is activated by Mg2+, and to a lesser extent by low concentrations of Mn2+. Reduced NAD inhibits the enzyme from T. ferrooxidans. No deviation from normal Michaelis-Menten kinetics was observed in velocity versus substrate concentration experiments. Adenosine triphosphate exerted a profound inhibition of the enzyme; the effect was 10 times more pronounced in the presence of NAD as compared to NADP. The physiological significance of this inhibition is discussed.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Avigad G. Inhibition of glucose 6-phosphate dehydrogenase by adenosine 5'-triphosphate. Proc Natl Acad Sci U S A. 1966 Nov;56(5):1543–1547. doi: 10.1073/pnas.56.5.1543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blackkolb F., Schlegel H. G. Regulation der Glucose-6-phosphate-Dehydrogenase aus Hydrogenomonas H 16 durch ATP und NADH. Arch Mikrobiol. 1968;63(2):177–196. [PubMed] [Google Scholar]
- COHEN L. H., NOELL W. K. Glucose catabolism of rabbit retina before and after development of visual function. J Neurochem. 1960 May;5:253–276. doi: 10.1111/j.1471-4159.1960.tb13363.x. [DOI] [PubMed] [Google Scholar]
- DAVIS B. J. DISC ELECTROPHORESIS. II. METHOD AND APPLICATION TO HUMAN SERUM PROTEINS. Ann N Y Acad Sci. 1964 Dec 28;121:404–427. doi: 10.1111/j.1749-6632.1964.tb14213.x. [DOI] [PubMed] [Google Scholar]
- Eidels L., Preiss J. Carbohydrate metabolism in Rhodopseudomonas capsulata: enzyme titers, glucose metabolism, and polyglucose polymer synthesis. Arch Biochem Biophys. 1970 Sep;140(1):75–89. doi: 10.1016/0003-9861(70)90011-1. [DOI] [PubMed] [Google Scholar]
- GOTTSCHALK G., EBERHARDT U., SCHLEGEL H. G. VERWERTUNG VON FRUCTOSE DURCH HYDROGENOMONAS H 16. (I.) Arch Mikrobiol. 1964 Apr 2;48:95–108. [PubMed] [Google Scholar]
- Hedrick J. L., Smith A. J. Size and charge isomer separation and estimation of molecular weights of proteins by disc gel electrophoresis. Arch Biochem Biophys. 1968 Jul;126(1):155–164. doi: 10.1016/0003-9861(68)90569-9. [DOI] [PubMed] [Google Scholar]
- Kuehn G. D., McFadden B. A. Enzymes of the Entner-Doudoroff path in fructose-grown Hydrogenomonas eutropha. Can J Microbiol. 1968 Nov;14(11):1259–1260. doi: 10.1139/m68-209. [DOI] [PubMed] [Google Scholar]
- LEVY H. R. The interaction of mammary glucose 6-phosphate dehydrogenase with pyridine nucleotides and 3beta-hydroxyandrost-5-en-17-one. J Biol Chem. 1963 Feb;238:775–784. [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Lessie T., Neidhardt F. C. Adenosine triphosphate-linked control of Pseudomonas aeruginosa glucose-6-phosphate dehydrogenase. J Bacteriol. 1967 Apr;93(4):1337–1345. doi: 10.1128/jb.93.4.1337-1345.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levy H. R., Raineri R. R., Nevaldine B. H. On the structure and catalytic function of mammary glucose 6-phosphate dehydrogenase. J Biol Chem. 1966 May 25;241(10):2181–2187. [PubMed] [Google Scholar]
- Matin A., Rittenberg S. C. Regulation of glucose metabolism in Thiobacillus intermedius. J Bacteriol. 1970 Oct;104(1):239–246. doi: 10.1128/jb.104.1.239-246.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McLean P., Brown J. Activities of some enzymes concerned with citrate and glucose metabolism in transplanted rat hepatomas. Biochem J. 1966 Mar;98(3):874–882. doi: 10.1042/bj0980874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nevaldine B. H., Levy H. R. Interaction of mammary glucose 6-phosphate dehydrogenase with o-phenanthroline and its analogues. Arch Biochem Biophys. 1967 Mar;119(1):293–302. doi: 10.1016/0003-9861(67)90459-6. [DOI] [PubMed] [Google Scholar]
- Ohmann E., Borriss R., Rindt K. P. Glucose-6-phosphat-Dehydrogenase in autotrophen Mikroorganismen. II. Die Regulation der Aktivität der Glucose-6-phosphat-Dehydrogenase in Euglena gracilis und Rhodopseudomonas spheroides. Z Allg Mikrobiol. 1970;10(1):37–53. doi: 10.1002/jobm.3630100104. [DOI] [PubMed] [Google Scholar]
- Olive C., Levy H. R. The preparation and some properties of crystalline glucose 6-phosphate dehydrogenase from Leuconostoc mesenteroides. Biochemistry. 1967 Mar;6(3):730–736. doi: 10.1021/bi00855a012. [DOI] [PubMed] [Google Scholar]
- Sanwal B. D. Regulatory mechanisms involving nicotinamide adenine nucleotides as allosteric effectors. 3. Control of glucose 6-phosphate dehydrogenase. J Biol Chem. 1970 Apr 10;245(7):1626–1631. [PubMed] [Google Scholar]
- Tabita R., Lundgren D. G. Heterotrophic metabolism of the chemolithotroph Thiobacillus ferrooxidans. J Bacteriol. 1971 Oct;108(1):334–342. doi: 10.1128/jb.108.1.334-342.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tabita R., Lundgren D. G. Utilization of glucose and the effect of organic compounds on the chemolithotroph Thiobacillus ferrooxidans. J Bacteriol. 1971 Oct;108(1):328–333. doi: 10.1128/jb.108.1.328-333.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]



