Abstract
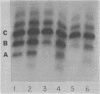
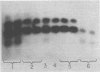
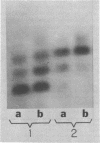
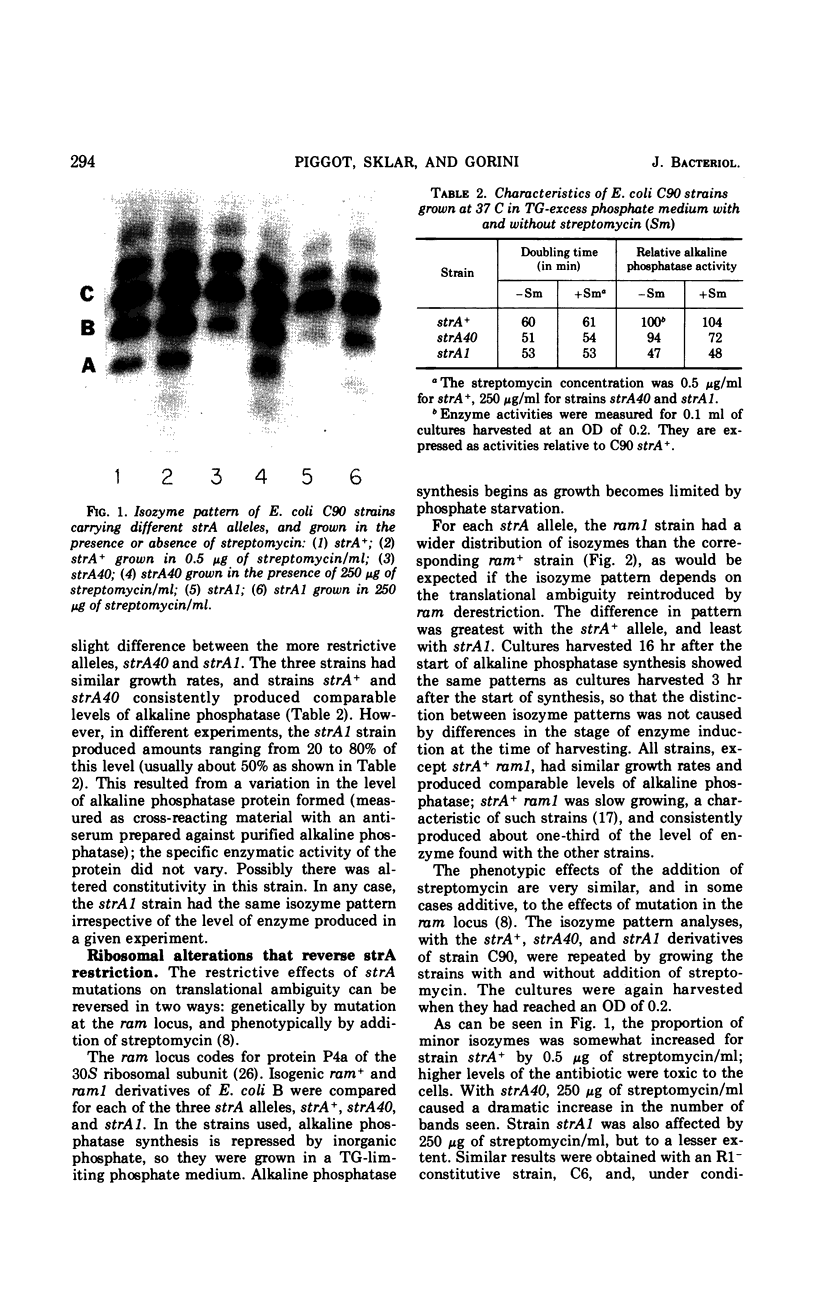
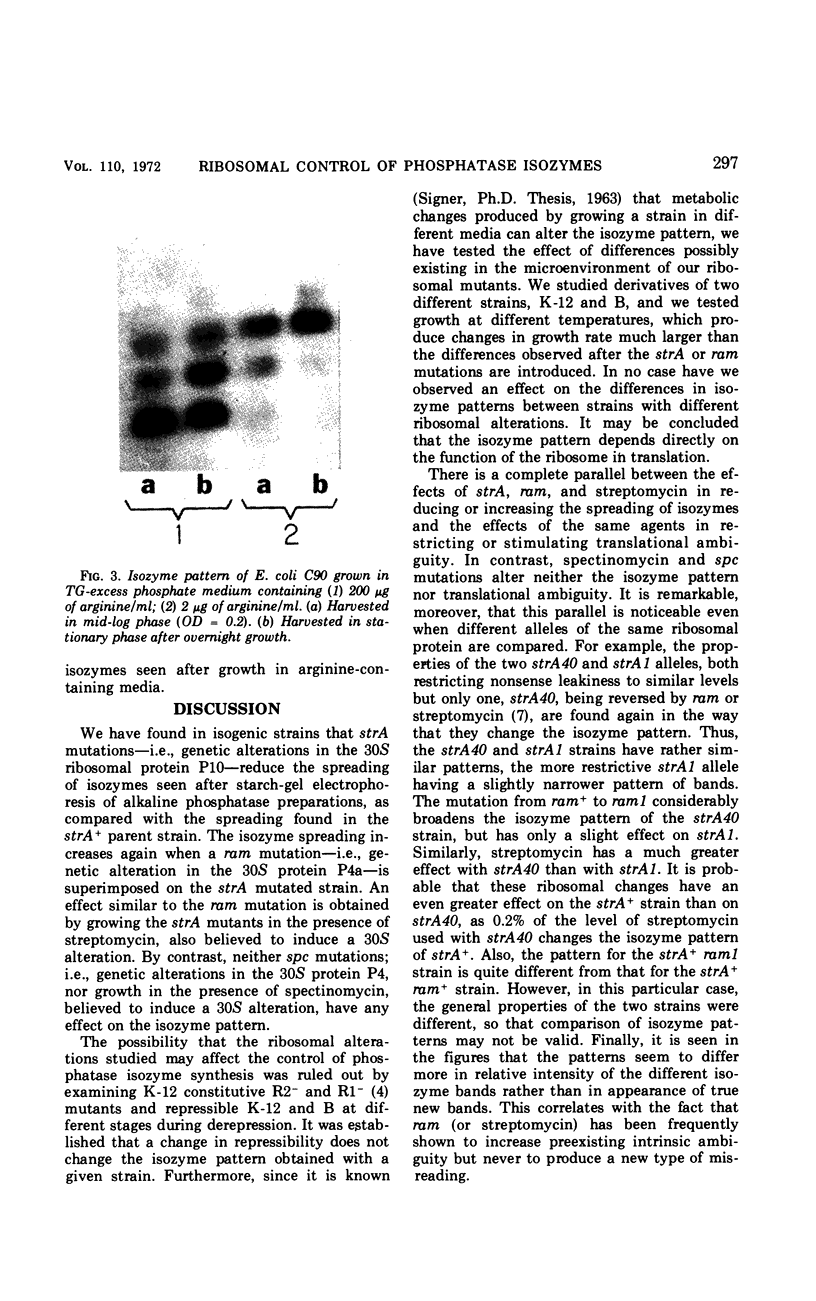
Different patterns of isozymes were obtained by starch-gel electrophoresis of alkaline phosphatase from Escherichia coli strains differing only by strA or ram mutations, or both, in the 30S ribosomal subunit. The isozyme spread was reduced in strA and increased in ram strains; this strictly parallels the restriction and enhancement of translational ambiguity produced by these mutations. Streptomycin present during growth had an effect similar to ram on both isozymes and ambiguity. The three isozymes analyzed have different N-terminal residues: aspartic acid, valine, and threonine. Different patterns of isozymes were also obtained in a wild-type strain through the specific action of exogenous arginine. A link between the mechanism of the effect of arginine and that of the ribosome is not obvious. The possibility is discussed that in both cases, although by different mechanisms, N-terminals are formed with different sensitivity to limited degradative attack.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anderson P., Davies J., Davis B. D. Effect of spectinomycin on polypeptide synthesis in extracts of Escherichia coli. J Mol Biol. 1967 Oct 14;29(1):203–215. doi: 10.1016/0022-2836(67)90191-x. [DOI] [PubMed] [Google Scholar]
- Cohen S., Porter R. R. Heterogeneity of the peptide chains of gamma-globulin. Biochem J. 1964 Feb;90(2):278–284. doi: 10.1042/bj0900278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crowshaw K., Jessup S. J., Ramwell P. W. Thin-layer chromatography of 1-dimethylaminonaphthalene-5-sulphonyl derivatives of amino acids present in superfusates of cat cerebral cortex. Biochem J. 1967 Apr;103(1):79–85. doi: 10.1042/bj1030079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- ECHOLS H., GAREN A., GAREN S., TORRIANI A. Genetic control of repression of alkaline phosphatase in E. coli. J Mol Biol. 1961 Aug;3:425–438. doi: 10.1016/s0022-2836(61)80055-7. [DOI] [PubMed] [Google Scholar]
- GAREN A., GAREN S. Complementation in vivo between structural mutants of alkaline phosphatase from E. coli. J Mol Biol. 1963 Jul;7:13–22. doi: 10.1016/s0022-2836(63)80015-7. [DOI] [PubMed] [Google Scholar]
- GOMORI G. Alkaline phosphatase of cell nuclei. J Lab Clin Med. 1951 Apr;37(4):526–531. [PubMed] [Google Scholar]
- GORINI L., KATAJA E. PHENOTYPIC REPAIR BY STREPTOMYCIN OF DEFECTIVE GENOTYPES IN E. COLI. Proc Natl Acad Sci U S A. 1964 Mar;51:487–493. doi: 10.1073/pnas.51.3.487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gorini L. Informational suppression. Annu Rev Genet. 1970;4:107–134. doi: 10.1146/annurev.ge.04.120170.000543. [DOI] [PubMed] [Google Scholar]
- Gorini L. The contrasting role of strA and ram gene products in ribosomal functioning. Cold Spring Harb Symp Quant Biol. 1969;34:101–109. doi: 10.1101/sqb.1969.034.01.016. [DOI] [PubMed] [Google Scholar]
- LEVINTHAL C., SIGNER E. R., FETHEROLF K. Reactivation and hybridization of reduced alkaline phosphatase. Proc Natl Acad Sci U S A. 1962 Jul 15;48:1230–1237. doi: 10.1073/pnas.48.7.1230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lazdunski C., Lazdunski M. Les isophosphatases alcalines d' Escherichia coli. Séparation, propriétés cinétiques et structurales. Biochim Biophys Acta. 1967 Oct 23;147(2):280–288. [PubMed] [Google Scholar]
- MALAMY M. H., HORECKER B. L. PURIFICATION AND CRYSTALLIZATION OF THE ALKALINE PHOSPHATASE OF ESCHERICHIA COLI. Biochemistry. 1964 Dec;3:1893–1897. doi: 10.1021/bi00900a018. [DOI] [PubMed] [Google Scholar]
- Natori S., Garen A. Molecular heterogeneity in the amino-terminal region of alkaline phosphatase. J Mol Biol. 1970 May 14;49(3):577–588. doi: 10.1016/0022-2836(70)90282-2. [DOI] [PubMed] [Google Scholar]
- Neu H. C., Heppel L. A. On the surface localization of enzymes in E. coli. Biochem Biophys Res Commun. 1964 Oct 14;17(3):215–219. doi: 10.1016/0006-291x(64)90386-9. [DOI] [PubMed] [Google Scholar]
- Nomura M. Bacterial ribosome. Bacteriol Rev. 1970 Sep;34(3):228–277. doi: 10.1128/br.34.3.228-277.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- ROTHMAN F., BYRNE R. Fingerprint analysis of alkaline phosphatase of Escherichia coli K12. J Mol Biol. 1963 Apr;6:330–340. doi: 10.1016/s0022-2836(63)80092-3. [DOI] [PubMed] [Google Scholar]
- Rosset R., Gorini L. A ribosomal ambiguity mutation. J Mol Biol. 1969 Jan 14;39(1):95–112. doi: 10.1016/0022-2836(69)90336-2. [DOI] [PubMed] [Google Scholar]
- SMITHIES O. Zone electrophoresis in starch gels: group variations in the serum proteins of normal human adults. Biochem J. 1955 Dec;61(4):629–641. doi: 10.1042/bj0610629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlesinger M. J., Andersen L. Multiple molecular forms of the alkaline phosphatase of Escherichia coli. Ann N Y Acad Sci. 1968 Jun 14;151(1):159–170. doi: 10.1111/j.1749-6632.1968.tb11886.x. [DOI] [PubMed] [Google Scholar]
- Schlesinger M. J. The reversible dissociation of the alkaline phosphatase of Escherichia coli. II. Properties of the subunit. J Biol Chem. 1965 Nov;240(11):4293–4298. [PubMed] [Google Scholar]
- Suzuki T., Garen A. Fragments of alkaline phosphatase from nonsense mutants. I. Isolation and characterization of fragments from amber and ochre mutants. J Mol Biol. 1969 Nov 14;45(3):549–566. doi: 10.1016/0022-2836(69)90312-x. [DOI] [PubMed] [Google Scholar]
- TORRIANI A. Influence of inorganic phosphate in the formation of phosphatases by Escherichia coli. Biochim Biophys Acta. 1960 Mar 11;38:460–469. doi: 10.1016/0006-3002(60)91281-6. [DOI] [PubMed] [Google Scholar]
- Woods K. R., Wang K. T. Separation of dansyl-amino acids by polyamide layer chromatography. Biochim Biophys Acta. 1967 Feb 21;133(2):369–370. doi: 10.1016/0005-2795(67)90078-5. [DOI] [PubMed] [Google Scholar]
- Zimmermann R. A., Garvin R. T., Gorini L. Alteration of a 30S ribosomal protein accompanying the ram mutation in Escherichia coli. Proc Natl Acad Sci U S A. 1971 Sep;68(9):2263–2267. doi: 10.1073/pnas.68.9.2263. [DOI] [PMC free article] [PubMed] [Google Scholar]