Abstract
The objective of this manuscript is to provide a comprehensive review of the epidemiologic evidence linking the continuum of adiposity, hyperinsulinemia, and diabetes with Alzheimer’s disease. The mechanisms for these associations remain to be elucidated, but may include direct actions from insulin, advanced products of glycosilation, cerebrovascular disease, and products of adipose tissue metabolism. Elevated adiposity in middle age is related to a higher risk of Alzheimer’s disease. The evidence relating adiposity in old age to Alzheimer’s disease is conflicting. Several studies have shown that hyperinsulinemia, a consequence of higher adiposity and insulin resistance, is also related to a higher risk of Alzheimer’s disease. Hyperinsulinemia is a risk factor for diabetes, and numerous studies have shown a relation of diabetes with higher Alzheimer’s disease risk. Most studies fail the take into account the continuum linking these risk factors which may result in underestimation of their importance in Alzheimer’s disease. The implication of these associations is that a large proportion of the world population may be at increased risk of Alzheimer’s disease given the trends for increasing prevalence of overweight, obesity, hyperinsulinemia, and diabetes. However, if proven causal, these associations also present a unique opportunity for prevention and treatment of Alzheimer’s disease.
Keywords: adiposity, overweight, obesity, hyperinsulinemia, insulin, glucose, diabetes, alzheimer’s disease, cognitive impairment
1. Burden of Alzheimer’s disease
Alzheimer’s disease is the most common form of dementia, accounting for between 70% to over 90% of all cases(Ritchie and Lovestone, 2002), and its prevalence is expected to quadruple by the year 2047 in the United States (Brookmeyer et al., 1998). As much as 50% of the population aged 85 years and older, the fastest growing segment of the population, may have Alzheimer’s disease (Evans et al., 1989). The risk factors for Alzheimer’s disease can be classified as genetic and non-genetic. Three genes have been identified in familial early onset Alzheimer’s disease, Amyloid Precursor Protein (APP), Presenilin 1, and Presenilin 2 (Selkoe, 1997). These genes affect less than 5% of cases of Alzheimer’s disease, have full penetrance and expressivity, and usually affect persons in middle age (Cummings, 2004). This review will address risk factors for late onset Alzheimer’s disease. Robust risk factors that have been identified for late onset Alzheimer’s disease include older age, lower education, and the APOE-ε4 allele(Cummings, 2004). Importantly, APOE ε4 has been found to modulate the effect of other putative risk factors (Haan et al., 1999), such as diabetes and hyperinsulinemia (Luchsinger et al., 2004; Peila et al., 2002). Current treatment options for Alzheimer’s disease only address symptoms, and no treatments are available that focus on delaying the actual disease process (Sano, 2003). One of the currently accepted hypothesis of the pathogenesis of Alzheimer’s disease is that the main culprit is the accumulation of amyloid β in the brain, resulting in synapse disruption and neuronal destruction (Selkoe, 1997; 2000). Thus, putative treatments or prevention measures for Alzheimer’s disease must target the deposition of Aβ and have the potential of preventing or delaying the onset of disease, not just symptoms (Sano, 2003). Mild Cognitive impairment (mild cognitive impairment) has been used to describe a transitional state between normal cognitive function and Alzheimer’s disease dementia (Luis et al., 2003; Petersen et al., 1999), and has thus been targeted for interventions (Petersen et al., 2005). Individuals with mild cognitive impairment do not have dementia but have memory complaints without loss of function in their daily activities(Petersen et al., 1999). While general cognitive performance is well preserved, memory performance on standardized tests falls below expectations for age and education. Persons with mild cognitive impairment progress to Alzheimer’s disease at the rate of nearly 10% to 15% per year (Luis et al., 2003) compared to 1 to 2% in elderly persons with normal cognition(Petersen et al., 1999). Mild cognitive impairment can be classified in amnestic and non-amnestic mild cognitive impairment. It is believed that amnestic mild cognitive impairment is an early stage of Alzheimer’s disease, while non-amnestic mild cognitive impairment, such as executive mild cognitive impairment, is less specific to Alzheimer’s disease(Luis et al., 2003). The prevalence of amnestic mild cognitive impairment varies between 3% and 20% depending on the criteria applied (Busse et al., 2003), and increases from about 1% in persons 60 years old to 25% at age 85 (Yesavage et al., 2002). Alzheimer’s disease can be studied in epidemiologic studies through the outcomes of memory impairment or decline, amnestic mild cognitive impairment, and Alzheimer’s dementia, and this review will cover evidence examining these outcomes.
2. Definition and burden of adiposity, hyperinsulinemia, and diabetes
There is a concerning epidemic of obesity, insulin resistance and diabetes in the world (Hill and Bessesen, 2003). With the aging of the population and greater longevity, the long term consequences of these conditions are serious and burdensome. Adiposity refers to the amount of adipose (fat) tissue in the body (Reaven and Laws, 1999). Some refer to adiposity as “fatness” or obesity. Adiposity is a continuum, and the normal or ideal threshold of adiposity is not clear. However, as adiposity increases it is associated with higher risk of insulin resistance, diabetes, hypertension, dyslipidemia, cardiovascular disease, degenerative joint disease, cancer, and respiratory diseases (Pi-Sunyer, 2002; Poirier et al., 2006). Definitions of a high level of adiposity have been devised using existing measures and according to their relationship with adverse outcomes(1998). Adiposity is usually measured indirectly with anthropological measures(Mueller et al., 1991) such as the body mass index (body mass index), defined as weight in kilograms divided by height in meters squared (k/m2). body mass index is strongly correlated with total body fat tissue and is a good indirect measure of adiposity (Pi-Sunyer, 2002), although this correlation decreases in older age (Baumgartner et al., 1995). Another commonly used measure of adiposity is waist circumference. Waist circumference is meant to measure the accumulation of adipose tissue in the abdomen, the largest depot of adipose tissue, and thus, perhaps it is a more direct measure of adiposity compared to body mass index (Mueller et al., 1991; Wahrenberg et al., 2005). Elevated waist circumference is also related to a higher risk of diabetes, hypertension, dyslipidemia, and heart disease, and some studies have shown that it is a better predictor of adverse cardiovascular outcomes compared to body mass index (Janssen et al., 2004), and some have advocated its use as the best measure of adiposity (Mueller et al., 1991). A commonly used cutoff to define elevated waist circumference is 102 cm for men and 88 cm for women (Janssen et al., 2004). Other less frequently used anthropologic measures of adiposity include skinfolds and waist to hip ratio (Mueller et al., 1991). Overweight and obesity (Flegal et al., 2002) and elevated waist circumference(Ford et al., 2003) are increasing in adults in the United States. More concerning, these trends are also observed in children and adolescents (Hedley et al., 2004). Two thirds of the United States population are overweight or obese (Hedley et al., 2004); 30% are obese, and the prevalence of obesity is higher in women than men. Insulin sensitivity is the ability of insulin to dispose of a glucose load. Insulin resistance refers to the resistance of tissues that dispose of glucose to the actions of insulin. Insulin resistance results in an increase in insulin secretion in the pancreas in order to overcome that insulin resistance. Fasting insulin levels are used in epidemiological studies as indicators of the risk of type 2 diabetes (Charles et al., 1991; Haffner et al., 1990; Lillioja et al., 1993; Lundgren et al., 1990). Fasting insulin is accepted as a measure of insulin resistance that is highly correlated with more complicated measures of insulin resistance such as the euglycemic clamp (Laakso, 1993), and the homeostasis model assessment (Haffner et al., 1997).
Glucose intolerance and diabetes are abnormal elevations of blood glucose that put people at risk for microvascular (nephropathy, neuropathy, retinopathy) and macrovascular disease (coronary artery disease, cerebrovascular disease, peripheral vascular disease)(DeFronzo, 2000). The American Diabetes Association currently defines diabetes as a fasting glucose elevation > 126 mg/dl, and glucose intolerance as an elevation of glucose between 110 and 126 mg/dl (ClarkJr. et al., 2000). It is difficult to establish an absolute threshold for the definition of glucose intolerance and diabetes. Previously, the definition of diabetes was a fasting glucose > 140 mg/dl, and people currently defined as having diabetes were then considered non-diabetic (Luchsinger, 2001). It is likely that the diabetes definition will change again and persons currently considered to have glucose intolerance will be considered to be diabetic. This underlines the caveats of using cutoffs to define conditions that have continuous (linear or non-linear) associations with disease: depending on the cutoff use, persons at risk may be classified as normal or abnormal (and vice versa). This is true for measures of adiposity, insulin resistance, and measures of glucose tolerance.
3. The continuum of adiposity, hyperinsulinemia, glucose intolerance and diabetes
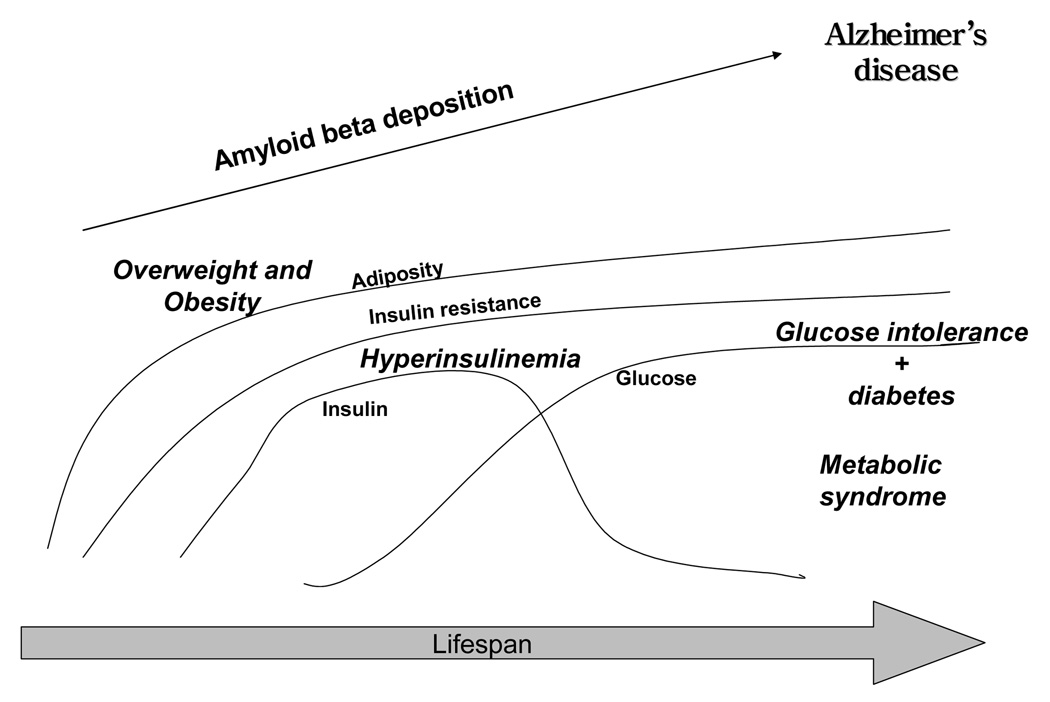
Adiposity, hyperinsulinemia, glucose intolerance, and diabetes, are often treated as separate constructs, and have been separately related to the risk of Alzheimer’s disease (Luchsinger and Mayeux, 2004a). However, they are related sequentially and often occur simultaneously, and understanding this relationship is fundamental in the study of the role of adiposity, insulin resistance, and diabetes in Alzheimer’s disease. Keeping glucose in normal levels is achieved by the balance between the ability of peripheral tissues (muscle, adipose tissue, liver) to take glucose into cells, and the pancreas’ ability to secrete insulin, the hormone in charge of glucose tissue uptake (DeFronzo, 2000). Thus, abnormal glucose levels are caused by a resistance of tissues to the action of insulin (insulin resistance), and by the pancreas’ inability to secrete enough insulin at normal levels or higher than normal insulin levels (hyperinsulinemia) to overcome insulin resistance in tissues (Festa et al., 2006). Insulin resistance increases with age, and the organism maintains normal glucose levels as long as it can produce enough insulin (hyperinsulinemia). Some individuals are less capable than others to mount sustained hyperinsulinemia and will develop glucose intolerance and diabetes (Festa et al., 2006). Other individuals with insulin resistance will maintain normal glucose levels at the expense of hyperinsulinemia but their pancreas will eventually “burn out”, will not be able to sustain hyperinsulinemia, and will develop glucose intolerance and diabetes (Festa et al., 2006). Others will continue having insulin resistance, may have or not have glucose intolerance, will not develop diabetes, but will have hyperinsulinemia and suffer its consequences. The most frequent modifiable determinant of insulin resistance and hyperinsulinemia is adiposity (Reaven, 2005; Reaven and Laws, 1999), although adipose tissue is not the only factor. Insulin resistance can reside in other tissues, including muscle, liver, and the pancreas itself (Accili, 2004). The natural history linking adiposity to insulin resistance to hyperinsulinemia to glucose intolerance and diabetes for most persons could be summarized in the following way (Figure). Elevations of adiposity result in insulin resistance, causing the pancreas to increase insulin to abnormal levels to sustain normal glucose, and if and when the pancreas can no longer sustain hyperinsulinemia, glucose intolerance and diabetes will ensue. However, the overlap between these processes is not complete (Ferrannini and Balkau, 2002). Not all persons with higher adiposity will develop insulin resistance and hyperinsulinemia, but most will. Not all persons with insulin resistance and hyperinsulinemia will develop glucose intolerance and diabetes, and this depends on genetic and other susceptibility factors that are not completely understood (Accili, 2004; Ferrannini and Balkau, 2002). Some adults develop diabetes without going through insulin resistance and hyperinsulinemia, but it is thought that most will.
Figure.
Natural history of the continuum of adiposity, insulin resistance, hyperinsulinemia, glucose intolerance and diabetes. Increased adiposity causes insulin resistance and hyperinsulinemia. Insulin levels may decrease over time due to pancreatic failure, resulting in glucose intolerance and diabetes. Adiposity, hyperinsulinemia, glucose intolerance and diabetes could increase brain amyloid beta deposition leading to Alzheimer’s disease individually or in aggregate in addition to causing cerebrovascular disease.
The susceptibility to adiposity, that is, the risk of developing the above described sequence in response to adiposity, varies by gender (Pi-Sunyer, 2002) and particularly by ethnicity. For example, Chinese and southeast Asians are more susceptible than Europeans to developing insulin resistance with comparable increases of adiposity (Reaven and Laws, 1999). The distribution of factors related to insulin resistance and the metabolic syndrome, including adiposity, is different in Whites and Blacks (Kraja et al., 2005). Thus, conventional ways to classify adiposity may not capture its relation with adverse outcomes and this should be taken into account. High adiposity and hyperinsulinemia are both accompanied by dyslipidemia, hypertension, and inflammation (Reaven, 2005), and these should also be taken into account. One implication of this continuum from a research standpoint is that if we examine diabetes as an exposure, we classify as not exposed persons without diabetes who may have increased adiposity, hyperinsulinemia, and even glucose intolerance. This kind of misclassification would result in an underestimation of the true relation between diabetes and Alzheimer’s disease if it is random (not conditional on exposure and disease).
Another implication of the continuum described above is that when an epidemiologic study finds a relation between the components of this continuum and Alzheimer’s disease we cannot be certain if we are looking at a surrogate marker of one of the other components (e.g. diabetes is a marker of past adiposity or hyperinsulinemia, obesity is a marker of hyperinsulinemia) or if the important exposure is the one we are examining. The answer could be that there is an aggregate effect of all the components of the continuum. The metabolic syndrome, an increasingly popular term in clinical practice and research, and reported to be associated with a higher risk of cognitive decline (Yaffe et al., 2004b) is a constellation of adiposity, hypertension, glucose intolerance, and dyslipidemia, that is associated mainly with insulin resistance and hyperinsulinemia (Grundy et al., 2005; Luchsinger, 2006). However, the definition of the metabolic syndrome is somewhat arbitrary, intended to capture the clustering of cardiovascular risk factors particularly in middle aged populations, and its validity in elderly populations at risk for Alzheimer’s disease is not clear (Luchsinger, 2006). The difficulty of arriving at precise metabolic syndrome criteria is reflected by the fact that over the years at least 6 different definitions have been developed that share several characteristics (Grundy et al., 2005).
4. Non-genetic risk factors for Alzheimer’s disease and their relation to adiposity, hyperinsulinemia and diabetes
Among demographic characteristics, old age (Cummings, 2004), low education (Scarmeas and Stern, 2003; Scarmeas et al., 2003), and being Caribbean -Hispanic or African American (Tang et al., 2001) have been related to a higher risk of Alzheimer’s disease in New York City. Weight decreases with aging and frailty (Morley, 2001), and body mass index in older age may not reflect that of middle age. In the United States, higher adiposity has been related to lower education and socioeconomic position (Luchsinger, 2001). Being overweight or obese, are an indication of high adiposity, and are more prevalent in Blacks and Hispanics compared to Whites (Flegal et al., 2002), who have been reported to have a higher risk of Alzheimer’s disease (Tang et al., 1996). Thus, it is possible that the association between adiposity and Alzheimer’s disease is confounded by age, socioeconomic and educational status, and ethnicity.
Among environmental risk factors, diet (Luchsinger and Mayeux, 2004b), physical activity (Scarmeas et al., 2003), and vascular risk factors (Luchsinger and Mayeux, 2004a) have attracted increasing interest. The evidence for various dietary factors is conflicting (Luchsinger and Mayeux, 2004b) and no solid conclusions can be drawn at this time. In a cohort study of aging in Northern New York City the strongest dietary risk factor for Alzheimer’s disease is higher caloric and fat intake (Luchsinger et al., 2002), and higher caloric and fat intake are related to increased adiposity (Pi-Sunyer, 2002; Poirier et al., 2006), hyperinsulinemia, and diabetes. Several studies have found that increased physical activity is inversely related to Alzheimer’s disease (Larson et al., 2006). High physical activity is typically accompanied by low adiposity (Pi-Sunyer, 2002), lower hyperinsulinemia, and lower diabetes risk which may be the explanation for the beneficial effects.
In terms of vascular risk factors, hypertension, dyslipidemia, diabetes, hyperinsulinemia, the metabolic syndrome, homocysteine, smoking, and heart disease are potential risk factors for Alzheimer’s disease (Luchsinger and Mayeux, 2004a). High adiposity, hyperinsulinemia and diabetes are clearly related to the metabolic syndrome (Grundy et al., 2005), dyslipidemia (Morgan and Capuzzi, 2003), and hypertension (Chobanian et al., 2003), and these can be reversed or prevented by weight loss, the reduction of insulin resistance, and the prevention of diabetes (Diabetes Prevention Program Research Group, 2002; Orchard et al., 2005; Poirier et al., 2006). Smoking, another potential risk factor for Alzheimer’s disease (Luchsinger and Mayeux, 2004a) is related to weight loss and low weight and thus may produce the appearance of a relation between low body mass index and Alzheimer’s disease through confounding. This type of confounding may partially explain U-shape associations found between body mass index and other outcomes such as mortality (Allison et al., 1997)
In summary, many of the putative risk factors for Alzheimer’s disease are related to the continuum of high adiposity, hyperinsulinemia, and diabetes. When studying the relation between adiposity and Alzheimer’s disease special care should be taken to identify which factors are confounders and which are in the causal pathway to Alzheimer’s disease.
5. Potential mechanisms linking adiposity, hyperinsulinemia and diabetes to Alzheimer’s disease
Providing a comprehensive review of mechanisms linking adiposity, hyperinsulinemia, and diabetes to Alzheimer’s disease is not the main goal of this article. I provide a brief mention of potential mechanisms to put in context the review of the epidemiological evidence.
5.1 Hyperinsulinemia
As described previously, one of the main consequences of adiposity is insulin resistance and hyperinsulinemia (Reaven and Laws, 1999). The role of insulin in Alzheimer’s disease has attracted increasing attention (Strachan, 2003). Insulin can cross the blood brain barrier from the periphery to the central nervous system and compete with Aβ for insulin degrading enzyme (IDE) in the brain, including the hippocampus (Farris et al., 2003). Insulin is also produced in the brain, and may have alternatively have a beneficial effect in amyloid clearance (Reger et al., 2006). Peripheral hyperinsulinemia may inhibit brain insulin production which, in turn results in impaired amyloid clearance and a higher risk of Alzheimer’s disease (Reger et al., 2006). Thus, it is possible that decreasing peripheral hyperinsulinemia and increasing brain insulin levels have the same beneficial effect on Alzheimer’s disease. A study found that rosiglitazone, which decreases insulin resistance and decreases peripheral insulin levels used in the treatment of diabetes may also be beneficial in Alzheimer’s disease (Risner et al., 2006). Interestingly, intranasal insulin, delivered with direct access to the brain without accessing the periphery has a similar effect (Reger et al., 2006). Manipulation of insulin levels in humans has been demonstrated to affect cognition and levels of amyloid β in the cerebrospinal fluid (Watson et al., 2006; Watson and Craft, 2004), supporting the potential direct role of insulin in Alzheimer’s disease.
5.2 Advanced products of glycosilation
Advanced glycosilation products are direct products of glucose intolerance and diabetes and are responsible for their related end organ damage (Yamagishi et al., 2005). Advanced glycosilation products can be identified immunohistochemically in senile plaques and neurofibrillary tangles, the pathologic hallmarks of Alzheimer’s disease (Cummings, 2004). Glycation of amyloid β enhances its aggregation in vitro. Furthermore, receptors for advanced glycosilation products have been found to be specific cell surface receptors for amyloid β, thus potentially causing neuronal damage (Yamagishi et al., 2005).
5.3. Adipokines and cytokines
Adipose tissue used to be conceived as a passive depot of energy in the form of fats. Recent evidence shows that adipose tissue is active and produces a series of substances that are important in metabolism (adipokines), and inflammation (cytokines). The adipokines include adiponectin (Trujillo and Scherer, 2005), leptin(Yu and Ginsberg, 2005), and resistin (Yu and Ginsberg, 2005), and the inflammatory cytokines include Tumor Necrosis Factor-α, and Interleukin-6 (Yu and Ginsberg, 2005), all correlated with insulin resistance and hyperinsulinemia. It is unclear at this point whether adipokines and cytokines produced by adipose tissue are directly related to Alzheimer’s disease or whether they are only markers of insulin resistance and hyperinsulinemia. This distinction may be important in the exploration of therapeutic targets.
5.4. Vascular risk factors and cerebrovascular disease
Cerebrovascular disease and stroke are related to a higher risk of Alzheimer’s disease (Honig et al., 2003; Vermeer et al., 2003). It is not clear whether cerebrovascular disease has a direct action on the amyloid cascade. It may cause brain damage aggregated to amyloid neurotoxicity that may decrease the threshold for the clinical manifestation of Alzheimer’s disease. An autopsy study showed that large vessel cerebrovascular disease, but not small vessel disease or infarcts, were related to a higher frequency of brain neuritic plaques (Honig et al., 2005), the pathologic hallmark of Alzheimer’s disease (Cummings, 2004). In the absence of evidence that cerebrovascular disease directly affects the amyloid cascade, I consider it to be an indirect pathway to Alzheimer’s disease. Adiposity, hyperinsulinemia, and diabetes (Poirier et al., 2006), and related vascular risk factors such as hypertension and dyslipidemia are related to a higher risk of cerebrovascular disease(Sacco et al., 1997). Thus, adiposity, hyperinsulinemia, and diabetes may affect Alzheimer’s disease risk indirectly through vascular risk factors and cerebrovascular disease.
6. Review of prospective epidemiological studies linking adiposity, hyperinsulinemia, and diabetes to Alzheimer’s disease
6.1. Adiposity
Few studies have explored the association between adiposity and Alzheimer’s disease and reveal conflicting findings. Elevated body mass index (overweight and obesity) in middle age is associated with higher dementia risk (Kivipelto et al., 2005; Whitmer et al., 2005a). Higher body mass index at ages 70, 75 and 79 years also predicts higher dementia risk (Gustafson et al., 2003). However, there have been reports of no association (Stewart et al., 2005) and of lower body mass index related to higher Alzheimer’s disease risk (Nourhashemi et al., 2003). These paradoxical findings could be explained by different age groups in different studies; those conducted in middle age show a relation of elevated body mass index to increased dementia risk, while those in older populations are conflicting. Another potential explanation is ethnicity. One study in Japanese Americans showed no association of high adiposity with Alzheimer’s disease (Stewart et al., 2005). A study in Northern New York City (Luchsinger et al., 2007a) found that in younger elderly (65 to 76 years of age), the association between body mass index quartiles and Alzheimer’s disease resembles a U shaped-curve, while in the oldest old (> 76 years) higher body mass index is related to a lower Alzheimer’s disease risk. This study also found that higher waist circumference is related to higher Alzheimer’s disease risk in the younger elderly, but not in the oldest. These findings seem to encapsulate the findings of other studies and caveats in measures of adiposity and the effect of aging. In addition, low body mass index may be a sign of frailty due to sarcopenia (Morley, 2001; Morley et al., 2006), the consequence of Alzheimer’s disease itself (White et al., 1996), or the consequence of hyperinsulinemia (Wedick et al., 2001), one of the putative mechanisms linking adiposity and Alzheimer’s disease.
6.2. Hyperinsulinemia
Several cross-sectional studies show an association between hyperinsulinemia and an increased risk of Alzheimer’s disease (Kuusisto et al., 1997; Razay and Wilcock, 1994; Stolk et al., 1997). Two longitudinal studies, one in elderly Japanese Americans in Hawaii (Peila et al., 2004), and another in elderly Black, Caribbean Hispanic, and Non-Hispanic Whites in New York City (Luchsinger et al., 2004) found that the risk of incident Alzheimer’s disease was higher in persons with hyperinsulinemia. These studies also found that the risk of Alzheimer’s disease related to hyperinsulinemia was higher among persons with the APOE-ε4. Another study found that higher C-peptide levels, a measure of insulin secretion (Harris et al., 2002), was related to cognitive decline in women (Okereke et al., 2005). There is a paucity of prospective epidemiologic studies exploring the relation between markers of hyperinsulinemia and Alzheimer’s disease and more are needed.
6.3. Diabetes
Diabetes has been related to a two-fold higher risk of developing mild cognitive impairment among postmenopausal women (Yaffe et al., 2004a). A multiethnic study in elderly from New York city found that diabetes was related to a higher risk of cognitive impairment-no dementia with stroke (relative risk (RR)=2.3) although the effect on cognitive impairment-no dementia without stroke (RR=1.5) was not evident after adjusting for demographic variables and the presence of Apo E-ε4 allele (Luchsinger et al., 2001). An Italian study showed a non-statistically significant increase of mild cognitive impairment with diabetes in an elderly population(Solfrizzi et al., 2004), while a Canadian study found that diabetes was related only to vascular cognitive impairment-no dementia (MacKnight et al., 2002). A study in New York City found that diabetes was related to a higher risk of both amnestic and non-amnestic mild cognitive impairment, underlining the importance of diabetes for both Alzheimer’s disease related and vascular cognitive impairment (Luchsinger et al., 2007b).
Diabetes has been found consistently to be related to vascular dementia (vascular dementia), but its relation to Alzheimer’s disease is less clear. A study of Japanese subjects aged 65 years and older found that diabetes was related to a higher risk of both Alzheimer’s disease (relative risk (RR)=2.2) and vascular dementia (RR=2.8) (Yoshitake et al., 1995). A longitudinal study from the Netherlands in over 5,000 subjects aged 55 years and older without dementia at baseline found a RR of Alzheimer’s disease of 1.9 (Ott et al., 1999). This association was stronger in subjects with diabetes who reported insulin treatment. Another European study found that the risk of all cause-dementia was increased by diabetes (RR=2.6), but this relation was weaker with Alzheimer’s disease (RR=1.4) (Brayne et al., 1998). A study from Rochester, Minnesota found an RR of 2 relating diabetes and Alzheimer’s disease (Leibson et al., 1997), similar to the study from the Netherlands. A study of catholic nuns, priests, and brothers 55 years and older found that diabetes was associated with a higher risk of Alzheimer’s disease (RR=1.7) (Arvanitakis et al., 2004). The Honolulu Asia Aging study also found that diabetes in old age was related to a higher risk of Alzheimer’s disease (RR=1.8) and Alzheimer’s disease pathology on autopsy, particularly in subjects with the APOE-ε4 allele (Peila et al., 2002), the only known genetic risk factor for sporadic Alzheimer’s disease(Ritchie and Lovestone, 2002). One study from Canada found that diabetes had a weak non-statistically significant relation to Alzheimer’s disease (RR=1.3), but was related to vascular dementia (RR=2.0). A Swedish study found a similar non-significant relation to Alzheimer’s disease (RR=1.3), and a significant relation to a higher risk of vascular dementia (RR=2.6) (Xu et al., 2004). A prospective study in over 1,000 subjects from New York City who were mostly African American and Caribbean-Hispanic, with a mean age of 75 years, and without dementia at baseline found a relative risk of Alzheimer’s disease of 1.4 which was not statistically significant after adjustment for other variables, but diabetes was significantly related to higher risk of a composite outcome of Alzheimer’s disease and cognitive impairment-no dementia (Luchsinger et al., 2001). The risk of Alzheimer’s disease was also increased in those treated with insulin, indicating a higher risk of Alzheimer’s disease in subjects with long-standing diabetes. This study also found a strong association between diabetes and vascular dementia (RR=3.4). A recent reanalysis of these data with longer follow-up showed that the risk of Alzheimer’s disease associated with diabetes was stronger than previously reported (RR=4), independent of other vascular conditions (hypertension, heart disease, stroke) and not explained by misclassification of vascular dementia cases as Alzheimer’s disease (Luchsinger et al., 2005).
Few studies have examined if diabetes in middle age leads to the development of dementia in older age. One study in the United States (Whitmer et al., 2005b) and another in Israel (Schnaider Beeri et al., 2004) found that diabetes at midlife increased the risk of dementia in the elderly. However, a study in Sweden found that diabetes increased the risk of vascular dementia but not of Alzheimer’s disease (Xu et al., 2004), and that this risk was higher in the presence of hypertension and heart disease. Similarly, a study in Japanese-Americans found no association between diabetes in middle age and dementia (Curb et al., 1999).
6.4. Metabolic syndrome
There is limited evidence on the association between the metabolic syndrome and dementia in the elderly. One study in 2,632 black and white elders found that the metabolic syndrome was associated with a higher risk of cognitive decline, particularly among those with high inflammatory markers (Yaffe et al., 2004b). A cross-sectional study in Europeans found that Alzheimer’s disease prevalence was higher in persons with the metabolic syndrome (Vanhanen et al., 2006). The discrepancy between these studies could be due to the fact that our study was conducted in an older population, ethnically diverse, and with a high prevalence of vascular risk factors (Luchsinger et al., 2005). In Japanese Americans the metabolic syndrome in middle age was associated with vascular dementia, but not Alzheimer’s disease (Kalmijn et al., 2000). We found in Northern New York City that the metabolic syndrome was not related to Alzheimer’s disease risk, while diabetes and hyperinsulinemia were (Muller et al., 2007).
7. Caveats in epidemiologic studies relating adiposity, hyperinsulinemia, and diabetes with Alzheimer’s disease
As reviewed, the evidence linking diabetes and dementia is much stronger for vascular dementia than Alzheimer’s disease. This begs the question of whether the findings for Alzheimer’s disease are not due to misclassification of vascular dementia as Alzheimer’s disease. One of the great controversies in the dementia field is whether vascular dementia can present slowly mimicking the usual presentation of Alzheimer’s disease.
Another caveat is that most epidemiologic studies of cognition have occurred in the elderly. Measures of adiposity and risk factors that are part of the metabolic syndrome change with age in ways that may underestimate the effects of high adiposity, dyslipidemia, and blood pressure. In fact, high adiposity in the oldest old may be a marker of health and is related to decreased mortality (Stevens, 2000).
Since these risk factors increase morbidity and mortality, survival bias is an issue in cohorts of elderly persons. Persons with the worst adiposity, hyperinsulinemia, and diabetes may die before inclusion into a study, before having the opportunity to develop dementia, or are to sick to be included in studies. This review describes these conditions in a continuum, but in reality they may be quite heterogeneous, particularly in the elderly, in whom they do not always overlap, and more complicated sub-phenotypes with different pathological significance may exist (Ferrannini and Balkau, 2002).
As explained before, the definition of normal for adiposity, hyperinsulinemia, and diabetes is somewhat arbitrary, and may differ by gender and ethnic background. This lends itself to misclassification of persons as having or not having a condition, and this can bias the results of epidemiologic studies.
Lastly, we attempt a reductionistic approach in separating the effects of glucose, insulin, components of the metabolic syndrome (hypertension, dyslipidemia) and possibly products of adipose tissue (adipokines, cytokines) on the risk of cognitive impairment. We also try to separate the associations of these conditions with Alzheimer’s disease and vascular cognitive impairment. There is such overlap in these conditions that this reductionistic approach is a very difficult if not an impossible task. However, we should not be discouraged in isolating the mechanisms relating these conditions to Alzheimer’s disease, in particular because of the potential for interventions including specific drugs, but should always take into account that the overlaps invariably exist.
8. Putting it all together
One way to put the literature reviewed in perspective is to see how the continuum of adiposity, hyperinsulinemia and diabetes relates to Alzheimer’s disease in the same cohort. The table summarizes the findings linking the components of this continuum to Alzheimer’s disease in the Washington Heights Inwood Columbia Aging Project, a longitudinal study of aging in persons 65 years and older in New York City. We found that higher body mass index has a U-shape association with Alzheimer’s disease in persons 65 to 75 years, while there is no association in persons 76 years and older (Luchsinger et al., 2007a). We found that higher waist circumference was associated with Alzheimer’s disease also in persons 65 to 75 years, while there was no association in persons 76 years and older. We found that elevated waist circumference was a stronger risk factor for vascular dementia than for Alzheimer’s disease. For hyperinsulinemia we found that Alzheimer’s disease risk increased with higher insulin quartile, and that this association was stronger for persons with the APOE-ε4 allele (Luchsinger et al., 2004). Diabetes has been related to a higher risk of amnestic mild cognitive impairment, non-amnestic mild cognitive impairment, Alzheimer’s disease, and vascular dementia (Luchsinger et al., 2005; Luchsinger et al., 2007b; Luchsinger et al., 2001). We found that two definitions of the metabolic syndrome, one that included diabetes and one that included hyperinsulinemia, were not related to Alzheimer’s disease risk (Muller et al., 2007). However, the individual components of hyperinsulinemia and diabetes as defined within the metabolic syndrome definition were associated with a higher risk of Alzheimer’s disease. In general these findings were robust when only probable Alzheimer’s disease was examined as the outcome, suggesting that misclassification of vascular dementia as Alzheimer’s disease did not explain the positive findings. Some of these findings may seem conflicting, in particular those for measures of adiposity and the metabolic syndrome, but they make sense when we take into account some of the biases mentioned previously. It is known that measures of adiposity lose their predictive ability with aging (Stevens et al., 1998) and our results by age group reflect that. In terms of the metabolic syndrome, it is a useful concept in middle age but its use may be limited in the elderly (Luchsinger, 2006). In fact, diabetes and hyperinsulinemia, which are robust predictors for Alzheimer’s disease in our data, are the very constructs that the metabolic syndrome tries to capture.
Table.
Summary of studies relating the continuum of adiposity, hyperinsulinemia, and diabetes to Alzheimer’s disease (Alzheimer’s disease) in the Washington Heights Inwood Columbia Aging project in New York City.
| Adiposity (Luchsinger et al., 2007a) | Compared to persons in the first quartile of body mass index, persons in the third quartile had a lower dementia and Alzheimer’s disease risk, and persons in the second quartile had a lower vascular dementia (vascular dementia) risk. The association between body mass index and dementia resembled a U-shape in those < 76 years, while dementia risk decreased with higher body mass index in those ≥ 76 years. The 4th quartile of waist circumference was related to a higher vascular dementia risk in the whole sample, and to dementia and Alzheimer’s disease in persons < 76 years. |
| Hyperinsulinemia (Luchsinger et al., 2004) | The risk of Alzheimer’s disease doubled in the 39% of the sample with hyperinsulinemia (hazard ratio = 2.1; 95% CI: 1.5, 2.9) and was highest in people without diabetes. The hazard ratio relating presence of hyperinsulinemia or diabetes in 50% of our sample to Alzheimer’s disease was 2.2 (95% CI: 1.5,3.1). The hazard ratio of Alzheimer’s disease for the highest quartile of insulin compared to the lowest was 1.7 (95% CI: 1.0,2.7; p for trend = 0.009). The risk of Alzheimer’s disease attributable to the presence of hyperinsulinemia or diabetes was 39%.Hyperinsulinemia was also related to a statistically significant decline in memory related cognitive scores, but not to decline in other cognitive domains. |
| Diabetes (Luchsinger et al., 2005; Luchsinger et al., 2007b) | Diabetes was related to a significantly higher risk of all-cause mild cognitive impairment (hazard ratio= 1.4; 95% CI: 1.0,1.8) and amnestic mild cognitive impairment (hazard ratio= 1.5; 95% CI: 1.0,2.2) after adjustment for all covariates. Diabetes was also related to a higher risk of non-amnestic mild cognitive impairment (1.4; 95% CI: 1.0,1.9), but this association was appreciably attenuated after adjustment for socio-economic variables and vascular risk factors (1.2; 95% CI: 0.9,1.8). The hazard ratio relating diabetes with Alzheimer’s disease was 2.4 (95 % CI: 1.8, 3.2). |
| Metabolic syndrome(Muller et al., 2007) | metabolic syndrome was not associated with incident dementia. Of the components of the metabolic syndrome, diabetes and hyperinsulinemia were associated with an increased incident Alzheimer’s disease (hazard ratio; 95%CI respectively 1.4; 1.0–2.1 and 1.4; 0.9–2.7) and vascular dementia risk(hazard ratio; 95%CI respectively 1.9; 1.1–3.1 and 2.3; 1.1–4.7). |
9. Implications for prevention and treatment of Alzheimer’s disease
There is very strong evidence that adiposity, hyperinsulinemia, and diabetes are related to Alzheimer’s disease. However, this evidence comes short of being considered as proof of causation until we understand the mechanisms and some of the caveats discussed in this review. If the relation between these conditions and Alzheimer’s disease were to be causal, the public health implications are enormous. As explained before, 2/3 of the adult population of the United States are overweight or obese, and the short term trend is for this to worsen. These trends are also being observed worldwide. With increasing life expectancy we are likely to increasingly see the cognitive consequences of increased adiposity, hyperinsulinemia, and diabetes in old age. We estimated that in New York City the presence of diabetes or hyperinsulinemia in elderly people could account for 39% of cases of Alzheimer’s disease (Luchsinger et al., 2004). However, the other implication is that a large proportion of cases of Alzheimer’s disease could be preventable or treatable. Large intervention studies with lifestyle interventions and drugs like metformin have shown that it is feasible to decrease hyperinsulinemia and the risk of diabetes (The Diabetes Prevention Program Research, 2005) and is possible that these interventions could extend to decreasing the risk of Alzheimer’s disease. On this basis, drugs like rosiglitazone have shown preliminary (Watson et al., 2005) promise in the treatment of Alzheimer’s disease, although concerns about its safety may make limit this drug’s usefulness (Nathan, 2007; Nathan and Berkwits, 2007). Elucidating the mechanisms linking adiposity, hyperinsulinemia and diabetes to Alzheimer’s disease will help identify specific targets for treatment and more research is needed in this regard.
Acknowledgments
Support for this work was provided by grants from the National Institutes of Health National Institute on Aging (AG07232), by the Alzheimer’s Association (IIRG-05-15053), and by the Florence and Herbert Irving Clinical Research Scholar’s Award.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Clinical Guidelines on the Identification, Evaluation, and Treatment of Overweight and Obesity in Adults--The Evidence Report. National Institutes of Health. Obesity. Res. 1998;6:51S-209. [PubMed]
- Accili D. Lilly Lecture 2003: The Struggle for Mastery in Insulin Action: From Triumvirate to Republic. Diabetes. 2004;53:1633–1642. doi: 10.2337/diabetes.53.7.1633. [DOI] [PubMed] [Google Scholar]
- Allison DB, Faith MS, Heo M, Kotler DP. Hypothesis concerning the U-shaped relation between body mass index and mortality. Am. J. Epidemiol. 1997;146:339–349. doi: 10.1093/oxfordjournals.aje.a009275. [DOI] [PubMed] [Google Scholar]
- Arvanitakis Z, Wilson RS, Bienias JL, Evans DA, Bennett DA. Diabetes mellitus and risk of Alzheimer disease and decline in cognitive function. Arch. Neurol. 2004;61:661–666. doi: 10.1001/archneur.61.5.661. [DOI] [PubMed] [Google Scholar]
- Baumgartner RN, Heymsfield SB, Roche AF. Human body composition and the epidemiology of chronic disease. Obes. Res. 1995;3:73–95. doi: 10.1002/j.1550-8528.1995.tb00124.x. [DOI] [PubMed] [Google Scholar]
- Brayne C, Gill C, Huppert FA, Barkley C, Gehlhaar E, Girling DM, O'Connor DW, Paykel ES. Vascular risks and incident dementia: results from a cohort study of the very old. Dement. Geriatr. Cogn. Disord. 1998;9:175–180. doi: 10.1159/000017043. [DOI] [PubMed] [Google Scholar]
- Brookmeyer R, Gray S, Kawas C. Projections of Alzheimer's disease in the United States and the public health impact of delaying disease onset. Am. J. Public. Health. 1998;88:1337–1342. doi: 10.2105/ajph.88.9.1337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Busse A, Bischkopf J, Riedel-Heller SG, Angermeyer MC. Mild cognitive impairment: prevalence and incidence according to different diagnostic criteria. Results of the Leipzig Longitudinal Study of the Aged (LEILA75+) Br. J. Psychiatry. 2003;182:449–454. [PubMed] [Google Scholar]
- Charles MA, Fontbonne A, Thibult N, Warnet JM, Rosselin GE, Eschwege E. Risk factors for NIDDM in white population. Paris prospective study. Diabetes. 1991;40:796–799. doi: 10.2337/diab.40.7.796. [DOI] [PubMed] [Google Scholar]
- Chobanian AV, Bakris GL, Black HR, Cushman WC, Green LA, Izzo JL, Jr, Jones DW, Materson BJ, Oparil S, Wright JT, Jr, Roccella EJ National Heart, L.; Blood Institute Joint National Committee on Prevention D.E.; Treatment of High Blood, P.; National High Blood Pressure Education Program Coordinating, C. The Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure: the JNC 7 report. JAMA. 2003;289:2560–2572. doi: 10.1001/jama.289.19.2560. [DOI] [PubMed] [Google Scholar]
- ClarkJr MJ, Sterrett JJ, Carson DS. Diabetes Guidelines: A Summary and Comparison of the Recommendations of the American Diabetes Association, Veterans Health Administration, and American Association of Clinical Endocrinologists. Clin. Ther. 2000;22:899–910. doi: 10.1016/S0149-2918(00)80063-6. [DOI] [PubMed] [Google Scholar]
- Cummings JL. Alzheimer's Disease. N. Engl. J. Med. 2004;351:56–67. doi: 10.1056/NEJMra040223. [DOI] [PubMed] [Google Scholar]
- Curb JD, Rodriguez BL, Abbott RD, Petrovitch H, Ross GW, Masaki KH, Foley D, Blanchette PL, Harris T, Chen R, White LR. Longitudinal association of vascular and Alzheimer's dementias, diabetes, and glucose tolerance. Neurology. 1999;52:971–975. doi: 10.1212/wnl.52.5.971. [DOI] [PubMed] [Google Scholar]
- DeFronzo RA. Pharmacologic therapy for type 2 diabetes mellitus. Ann. Intern. Med. 2000;133:73–74. doi: 10.7326/0003-4819-133-1-200007040-00016. [DOI] [PubMed] [Google Scholar]
- Diabetes Prevention Program Research Group. Reduction in the Incidence of Type 2 Diabetes with Lifestyle Intervention or Metformin. N Engl J Med. 2002;346:393–403. doi: 10.1056/NEJMoa012512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evans DA, Funkenstein HH, Albert MS, Scherr PA, Cook NR, Chown MJ, Hebert LE, Hennekens CH, Taylor JO. Prevalence of Alzheimer's disease in a community population of older persons. Higher than previously reported. JAMA. 1989;262:2551–2556. [PubMed] [Google Scholar]
- Farris W, Mansourian S, Chang Y, Lindsley L, Eckman EA, Frosch MP, Eckman CB, Tanzi RE, Selkoe DJ, Guenette S. Insulin-degrading enzyme regulates the levels of insulin, amyloid beta-protein, and the beta-amyloid precursor protein intracellular domain in vivo. Proc. Natl. Acad. Sci. U S A. 2003;100:4162–4167. doi: 10.1073/pnas.0230450100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferrannini E, Balkau B. Insulin: in search of a syndrome. Diabet. Med. 2002;19:724–729. doi: 10.1046/j.1464-5491.2002.00794.x. [DOI] [PubMed] [Google Scholar]
- Festa A, Williams K, D'Agostino R, Jr, Wagenknecht LE, Haffner SM. The Natural Course of {beta}-Cell Function in Nondiabetic and Diabetic Individuals: The Insulin Resistance Atherosclerosis Study. Diabetes. 2006;55:1114–1120. doi: 10.2337/diabetes.55.04.06.db05-1100. [DOI] [PubMed] [Google Scholar]
- Flegal KM, Carroll MD, C.L O, C.L. J. Prevalence and trends in obesity among US adults, 1999–2000. JAMA. 2002;288:1723–1727. doi: 10.1001/jama.288.14.1723. [DOI] [PubMed] [Google Scholar]
- Ford ES, Mokdad AH, Giles WH. Trends in Waist Circumference among U.S. Adults. Obes. Res. 2003;11:1223–1231. doi: 10.1038/oby.2003.168. [DOI] [PubMed] [Google Scholar]
- Grundy SM, Cleeman JI, Daniels SR, Donato KA, Eckel RH, Franklin BA, Gordon DJ, Krauss RM, Savage PJ, Smith SC, Jr, Spertus JA, Costa F. Diagnosis and Management of the Metabolic Syndrome: An American Heart Association/National Heart, Lung, and Blood Institute Scientific Statement. Circulation. 2005;112:2735–2752. doi: 10.1161/CIRCULATIONAHA.105.169404. [DOI] [PubMed] [Google Scholar]
- Gustafson D, Rothenberg E, Blennow K, Steen B, Skoog I. An 18-Year Follow-up of Overweight and Risk of Alzheimer Disease. Arch. Intern. Med. 2003;163:1524–1528. doi: 10.1001/archinte.163.13.1524. [DOI] [PubMed] [Google Scholar]
- Haan MN, Shemanski L, Jagust WJ, Manolio TA, Kuller L. The role of APOE epsilon4 in modulating effects of other risk factors for cognitive decline in elderly persons. JAMA. 1999;282:40–46. doi: 10.1001/jama.282.1.40. [DOI] [PubMed] [Google Scholar]
- Haffner SM, Miettinen H, Stern MP. The homeostasis model in the San Antonio Heart Study. Diabetes. Care. 1997;20:1087–1092. doi: 10.2337/diacare.20.7.1087. [DOI] [PubMed] [Google Scholar]
- Haffner SM, Stern MP, Mitchell BD, Hazuda HP, Patterson JK. Incidence of type II diabetes in Mexican Americans predicted by fasting insulin and glucose levels, obesity, and body-fat distribution. Diabetes. 1990;39:283–288. doi: 10.2337/diab.39.3.283. [DOI] [PubMed] [Google Scholar]
- Harris MI, Cowie CC, Gu K, Francis ME, Flegal K, Eberhardt MS. Higher fasting insulin but lower fasting C-peptide levels in African Americans in the US population. Diabetes/Metabolism Research Reviews. 2002;18:149–155. doi: 10.1002/dmrr.273. [DOI] [PubMed] [Google Scholar]
- Hedley AA, Ogden CL, Johnson CL, Carroll MD, Curtin LR, Flegal KM. Prevalence of Overweight and Obesity Among US Children, Adolescents, and Adults, 1999–2002. JAMA. 2004;291:2847–2850. doi: 10.1001/jama.291.23.2847. [DOI] [PubMed] [Google Scholar]
- Hill JO, Bessesen D. What to do about the metabolic syndrome? Arch. Intern. Med. 2003;163:395–397. doi: 10.1001/archinte.163.4.395. [DOI] [PubMed] [Google Scholar]
- Honig LS, Kukull W, Mayeux R. Atherosclerosis and AD: Analysis of data from the US National Alzheimer's Coordinating Center. Neurology. 2005;64:494–500. doi: 10.1212/01.WNL.0000150886.50187.30. [DOI] [PubMed] [Google Scholar]
- Honig LS, Tang MX, Albert S, Costa R, Luchsinger J, Manly J, Stern Y, Mayeux R. Stroke and the risk of Alzheimer disease. Arch. Neurol. 2003;60:1707–1712. doi: 10.1001/archneur.60.12.1707. [DOI] [PubMed] [Google Scholar]
- Janssen I, Katzmarzyk PT, Ross R. Waist circumference and not body mass index explains obesity-related health risk. Am. J. Clin. Nutr. 2004;79:379–384. doi: 10.1093/ajcn/79.3.379. [DOI] [PubMed] [Google Scholar]
- Kalmijn S, Foley D, White L, Burchfiel CM, Curb JD, Petrovitch H, Ross GW, Havlik RJ, Launer LJ. Metabolic cardiovascular syndrome and risk of dementia in Japanese- American elderly men. The Honolulu-Asia aging study. Arterioscler. Thromb. Vasc. Biol. 2000;20:2255–2260. doi: 10.1161/01.atv.20.10.2255. [DOI] [PubMed] [Google Scholar]
- Kivipelto M, Ngandu T, Fratiglioni L, Viitanen M, Kareholt I, Winblad B, Helkala EL, Tuomilehto J, Soininen H, Nissinen A. Obesity and vascular risk factors at midlife and the risk of dementia and Alzheimer disease. Arch. Neurol. 2005;62:1556–1560. doi: 10.1001/archneur.62.10.1556. [DOI] [PubMed] [Google Scholar]
- Kraja AT, Hunt SC, Pankow JS, Myers RH, Heiss G, Lewis CE, Rao D, Province MA. An evaluation of the metabolic syndrome in the HyperGEN study. Nutr. Metab. (Lond) 2005;2:2. doi: 10.1186/1743-7075-2-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuusisto J, Koivisto K, Mykkanen L, Helkala EL, Vanhanen M, Hanninen T, Kervinen K, Kesaniemi YA, Riekkinen PJ, Laakso M. Association between features of the insulin resistance syndrome and Alzheimer's disease independently of apolipoprotein E4 phenotype: cross sectional population based study. BMJ. 1997;315:1045–1049. doi: 10.1136/bmj.315.7115.1045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laakso M. How good a marker is insulin level for insulin resistance? Am. J. Epidemiol. 1993;137:959–965. doi: 10.1093/oxfordjournals.aje.a116768. [DOI] [PubMed] [Google Scholar]
- Larson EB, Wang L, Bowen JD, McCormick WC, Teri L, Crane P, Kukull W. Exercise Is Associated with Reduced Risk for Incident Dementia among Persons 65 Years of Age and Older. Ann. Intern. Med. 2006;144:73–81. doi: 10.7326/0003-4819-144-2-200601170-00004. [DOI] [PubMed] [Google Scholar]
- Leibson CL, Rocca WA, Hanson VA, Cha R, Kokmen E, O'Brien PC, Palumbo PJ. Risk of dementia among persons with diabetes mellitus: a population- based cohort study. Am. J Epidemiol. 1997;145:301–308. doi: 10.1093/oxfordjournals.aje.a009106. [DOI] [PubMed] [Google Scholar]
- Lillioja S, Mott DM, Spraul M, Ferraro R, Foley JE, Ravussin E, Knowler WC, Bennett PH, Bogardus C. Insulin resistance and insulin secretory dysfunction as precursors of non-insulin-dependent diabetes mellitus. Prospective studies of Pima Indians. N. Engl. J. Med. 1993;329:1988–1992. doi: 10.1056/NEJM199312303292703. [DOI] [PubMed] [Google Scholar]
- Luchsinger J, Mayeux R. Cardiovascular risk factors and Alzheimer's disease. Curr Atheroscler Rep. 2004a;6:261–266. doi: 10.1007/s11883-004-0056-z. [DOI] [PubMed] [Google Scholar]
- Luchsinger JA. Diabetes. In: Aguirre-Molina M, C.W. M, R.E. Z, editors. Health issues in the Latino community. San Francisco: Jossey-Bass; 2001. pp. 277–300. [Google Scholar]
- Luchsinger JA. A work in progress: the metabolic syndrome. Sci. Aging. Knowledge. Environ. 2006:pe19. doi: 10.1126/sageke.2006.10.pe19. [DOI] [PubMed] [Google Scholar]
- Luchsinger JA, Mayeux R. Dietary factors and Alzheimer's disease. The Lancet Neurology. 2004b;3:579–587. doi: 10.1016/S1474-4422(04)00878-6. [DOI] [PubMed] [Google Scholar]
- Luchsinger JA, Patel B, Tang MX, Schupf N, Mayeux R. Measures of adiposity and dementia risk in elderly persons. Arch. Neurol. 2007a;64:392–398. doi: 10.1001/archneur.64.3.392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luchsinger JA, Reitz C, Honig LS, Tang MX, Shea S, Mayeux R. Aggregation of vascular risk factors and risk of incident Alzheimer disease. Neurology. 2005;65:545–551. doi: 10.1212/01.wnl.0000172914.08967.dc. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luchsinger JA, Reitz C, Patel B, Tang M-X, Manly JJ, Mayeux R. Relation of Diabetes to Mild Cognitive Impairment. Arch Neurol. 2007b;64:570–575. doi: 10.1001/archneur.64.4.570. [DOI] [PubMed] [Google Scholar]
- Luchsinger JA, Tang M-X, Shea S, Mayeux R. Hyperinsulinemia and risk of Alzheimer disease. Neurology. 2004;63:1187–1192. doi: 10.1212/01.wnl.0000140292.04932.87. [DOI] [PubMed] [Google Scholar]
- Luchsinger JA, Tang MX, Shea S, Mayeux R. Caloric intake and the risk of Alzheimer disease. Arch. Neurol. 2002;59:1258–1263. doi: 10.1001/archneur.59.8.1258. [DOI] [PubMed] [Google Scholar]
- Luchsinger JA, Tang MX, Stern Y, Shea S, Mayeux R. Diabetes mellitus and risk of Alzheimer's disease and dementia with stroke in a multiethnic cohort. Am. J. Epidemiol. 2001;154:635–641. doi: 10.1093/aje/154.7.635. [DOI] [PubMed] [Google Scholar]
- Luis CA, Loewenstein DA, Acevedo A, Barker WW, Duara R. Mild cognitive impairment: directions for future research. Neurology. 2003;61:438–444. doi: 10.1212/01.wnl.0000080366.90234.7f. [DOI] [PubMed] [Google Scholar]
- Lundgren H, Bengtsson C, Blohme G, Lapidus L, Waldenstrom J. Fasting serum insulin concentration and early insulin response as risk determinants for developing diabetes. Diab. Med. 1990;7:407–413. doi: 10.1111/j.1464-5491.1990.tb01415.x. [DOI] [PubMed] [Google Scholar]
- MacKnight C, Rockwood K, Awalt E, McDowell I. Diabetes mellitus and the risk of dementia, Alzheimer's disease and vascular cognitive impairment in the Canadian Study of Health and Aging. Dement. Geriatr. Cogn. Disord. 2002;14:77–83. doi: 10.1159/000064928. [DOI] [PubMed] [Google Scholar]
- Morgan JM, Capuzzi DM. Hypercholesterolemia. The NCEP Adult Treatment Panel III Guidelines. Geriatrics. 2003;58:33–38. quiz 41. [PubMed] [Google Scholar]
- Morley JE. Anorexia, sarcopenia, and aging. Nutrition. 2001;17:660–663. doi: 10.1016/s0899-9007(01)00574-3. [DOI] [PubMed] [Google Scholar]
- Morley JE, Thomas DR, Wilson M-MG. Cachexia: pathophysiology and clinical relevance. Am. J. Clin. Nutr. 2006;83:735–743. doi: 10.1093/ajcn/83.4.735. [DOI] [PubMed] [Google Scholar]
- Mueller WH, Wear ML, Hanis CL, Emerson JB, Barton SA, Hewett-Emmett D, Schull WJ. Which measure of body fat distribution is best for epidemiologic research? Am. J. Epidemiol. 1991;133:858–869. doi: 10.1093/oxfordjournals.aje.a115966. [DOI] [PubMed] [Google Scholar]
- Muller M, Tang MX, Schupf N, Manly JJ, Mayeux R, Luchsinger JA. Metabolic syndrome and dementia risk in a multiethnic elderly cohort. Dement. Geriatr. Cogn. Disord. 2007;24:185–192. doi: 10.1159/000105927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nathan DM. Rosiglitazone and Cardiotoxicity -- Weighing the Evidence. N. Engl. J. Med. 2007 doi: 10.1056/NEJMe078117. NEJMe078117. [DOI] [PubMed] [Google Scholar]
- Nathan DM, Berkwits M. Trials that matter: rosiglitazone, ramipril, and the prevention of type 2 diabetes. Ann. Intern. Med. 2007;146:461–463. doi: 10.7326/0003-4819-146-6-200703200-00015. [DOI] [PubMed] [Google Scholar]
- Nourhashemi F, Deschamps V, Larrieu S, Letenneur L, Dartigues JF, Barberger-Gateau P, Quid PsPA. Body mass index and incidence of dementia: the PAQUID study. Neurology. 2003;60:117–119. doi: 10.1212/01.wnl.0000038910.46217.aa. [DOI] [PubMed] [Google Scholar]
- Okereke O, Hankinson SE, Hu FB, Grodstein F. Plasma C Peptide Level and Cognitive Function Among Older Women Without Diabetes Mellitus. Arch. Intern Med. 2005;165:1651–1656. doi: 10.1001/archinte.165.14.1651. [DOI] [PubMed] [Google Scholar]
- Orchard TJ, Temprosa M, Goldberg R, Haffner S, Ratner R, Marcovina S, Fowler S Diabetes Prevention Program Research Group. The Effect of Metformin and Intensive Lifestyle Intervention on the Metabolic Syndrome: The Diabetes Prevention Program Randomized Trial. Ann. Intern. Med. 2005;142:611–619. doi: 10.7326/0003-4819-142-8-200504190-00009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ott A, Stolk RP, van Harskamp F, Pols HA, Hofman A, Breteler MM. Diabetes mellitus and the risk of dementia: The Rotterdam Study. Neurology. 1999;53:1937–1942. doi: 10.1212/wnl.53.9.1937. [DOI] [PubMed] [Google Scholar]
- Peila R, Rodriguez BL, Launer LJ, Honolulu-Asia Aging S. Type 2 diabetes, APOE gene, and the risk for dementia and related pathologies: The Honolulu-Asia Aging Study. Diabetes. 2002;51:1256–1262. doi: 10.2337/diabetes.51.4.1256. [DOI] [PubMed] [Google Scholar]
- Peila R, Rodriguez BL, White LR, Launer LJ. Fasting insulin and incident dementia in an elderly population of Japanese-American men. Neurology. 2004;63:228–233. doi: 10.1212/01.wnl.0000129989.28404.9b. [DOI] [PubMed] [Google Scholar]
- Petersen RC, Smith GE, Waring SC, Ivnik RJ, Tangalos EG, Kokmen E. Mild cognitive impairment: clinical characterization and outcome. Arch. Neurol. 1999;56:303–308. doi: 10.1001/archneur.56.3.303. [DOI] [PubMed] [Google Scholar]
- Petersen RC, Thomas RG, Grundman M, Bennett D, Doody R, Ferris S, Galasko D, Jin S, Kaye J, Levey A, Pfeiffer E, Sano M, van Dyck CH, Thal LJ Alzheimer's Disease Cooperative Study Group. Vitamin E and Donepezil for the Treatment of Mild Cognitive Impairment. N. Engl. J. Med. 2005 doi: 10.1056/NEJMoa050151. NEJMoa050151. [DOI] [PubMed] [Google Scholar]
- Pi-Sunyer FX. The Obesity Epidemic: Pathophysiology and Consequences of Obesity. Obes. Res. 2002;10:97S-104. doi: 10.1038/oby.2002.202. [DOI] [PubMed] [Google Scholar]
- Poirier P, Giles TD, Bray GA, Hong Y, Stern JS, Pi-Sunyer FX, Eckel RH. Obesity and Cardiovascular Disease: Pathophysiology, Evaluation, and Effect of Weight Loss: An Update of the 1997 American Heart Association Scientific Statement on Obesity and Heart Disease From the Obesity Committee of the Council on Nutrition, Physical Activity, and Metabolism. Circulation. 2006;113:898–918. doi: 10.1161/CIRCULATIONAHA.106.171016. [DOI] [PubMed] [Google Scholar]
- Razay G, Wilcock GK. Hyperinsulinaemia and Alzheimer's disease. Age. Ageing. 1994;23:396–399. doi: 10.1093/ageing/23.5.396. [DOI] [PubMed] [Google Scholar]
- Reaven G. Insulin Resistance, Type 2 Diabetes Mellitus, and Cardiovascular Disease: The End of the Beginning. Circulation. 2005;112:3030–3032. doi: 10.1161/CIRCULATIONAHA.105.504670. [DOI] [PubMed] [Google Scholar]
- Reaven GM, Laws A. Insulin resistance: the metabolic syndrome X. Totowa, New Jersey: Humana Press; 1999. [Google Scholar]
- Reger MA, Watson GS, Frey WH, 2nd, Baker LD, Cholerton B, Keeling ML, Belongia DA, Fishel MA, Plymate SR, Schellenberg GD, Cherrier MM, Craft S. Effects of intranasal insulin on cognition in memory-impaired older adults : modulation by APOE genotype. Neurobiol. Aging. 2006;27:451–458. doi: 10.1016/j.neurobiolaging.2005.03.016. [DOI] [PubMed] [Google Scholar]
- Risner ME, Saunders AM, Altman JF, Ormandy GC, Craft S, Foley IM, Zvartau-Hind ME, Hosford DA, Roses AD. Efficacy of rosiglitazone in a genetically defined population with mild-to-moderate Alzheimer's disease. Pharmacogenomics. J. 2006;6:246–254. doi: 10.1038/sj.tpj.6500369. [DOI] [PubMed] [Google Scholar]
- Ritchie K, Lovestone S. The dementias. Lancet. 2002;360:1759–1766. doi: 10.1016/S0140-6736(02)11667-9. [DOI] [PubMed] [Google Scholar]
- Sacco RL, Benjamin EJ, Broderick JP, Dyken M, Easton JD, Feinberg WM, Goldstein LB, Gorelick PB, Howard G, Kittner SJ, Manolio TA, Whisnant JP, Wolf PA. American Heart Association Prevention Conference. IV. Prevention and Rehabilitation of Stroke. Risk factors. Stroke. 1997;28:1507–1517. doi: 10.1161/01.str.28.7.1507. [DOI] [PubMed] [Google Scholar]
- Sano M. Noncholinergic treatment options for Alzheimer's disease. J. Clin. Psychiatry. 2003;64:23–28. [PubMed] [Google Scholar]
- Scarmeas N, Stern Y. Cognitive reserve and lifestyle. J. Clin. Exp. Neuropsychol. 2003;25:625–633. doi: 10.1076/jcen.25.5.625.14576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scarmeas N, Zarahn E, Anderson KE, Habeck CG, Hilton J, Flynn J, Marder KS, Bell KL, Sackeim HA, Van Heertum RL, Moeller JR, Stern Y. Association of life activities with cerebral blood flow in Alzheimer disease: implications for the cognitive reserve hypothesis. Arch. Neurol. 2003;60:359–365. doi: 10.1001/archneur.60.3.359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schnaider Beeri M, Goldbourt U, Silverman JM, Noy S, Schmeidler J, Ravona-Springer R, Sverdlick A, Davidson M. Diabetes mellitus in midlife and the risk of dementia three decades later. Neurology. 2004;63:1902–1907. doi: 10.1212/01.wnl.0000144278.79488.dd. [DOI] [PubMed] [Google Scholar]
- Selkoe DJ. Alzheimer's disease: genotypes, phenotypes, and treatments. Science. 1997;275:630–631. doi: 10.1126/science.275.5300.630. [DOI] [PubMed] [Google Scholar]
- Selkoe DJ. The origins of Alzheimer disease: a is for amyloid. JAMA. 2000;283:1615–1617. doi: 10.1001/jama.283.12.1615. [DOI] [PubMed] [Google Scholar]
- Solfrizzi V, Panza F, Colacicco AM, D'Introno A, Capurso C, Torres F, Grigoletto F, Maggi S, Del Parigi A, Reiman EM, Caselli RJ, Scafato E, Farchi G, Capurso A Italian Longitudinal Study on Aging Working Group. Vascular risk factors, incidence of MCI, and rates of progression to dementia. Neurology. 2004;63:1882–1891. doi: 10.1212/01.wnl.0000144281.38555.e3. [DOI] [PubMed] [Google Scholar]
- Stevens J. Impact of age on associations between weight and mortality. Nutr. Rev. 2000;58:129–137. doi: 10.1111/j.1753-4887.2000.tb01847.x. [DOI] [PubMed] [Google Scholar]
- Stevens J, Cai J, Pamuk ER, Williamson DF, Thun MJ, Wood JL. The effect of age on the association between body-mass index and mortality. N. Engl. J. Med. 1998;338:1–7. doi: 10.1056/NEJM199801013380101. [DOI] [PubMed] [Google Scholar]
- Stewart R, Masaki K, Xue Q-L, Peila R, Petrovitch H, White LR, Launer LJ. A 32-Year Prospective Study of Change in Body Weight and Incident Dementia: The Honolulu-Asia Aging Study. Arch. Neurol. 2005;62:55–60. doi: 10.1001/archneur.62.1.55. [DOI] [PubMed] [Google Scholar]
- Stolk RP, Breteler MM, Ott A, Pols HA, Lamberts SW, Grobbee DE, Hofman A. Insulin and cognitive function in an elderly population. The Rotterdam Study. Diabetes. Care. 1997;20:792–795. doi: 10.2337/diacare.20.5.792. [DOI] [PubMed] [Google Scholar]
- Strachan MWJ. Insulin and cognitive function. Lancet. 2003;362:1253. doi: 10.1016/S0140-6736(03)14615-6. [DOI] [PubMed] [Google Scholar]
- Tang M-X, Maestre G, Tsai W-Y. Relative risk of Alzheimer's disease and age-at-onset base of APOE genotypes among elderly among elderly African Americans, Caucasians and Hispanics in New York City. Am. J. Hum. Genet. 1996;58:554–574. [PMC free article] [PubMed] [Google Scholar]
- Tang MX, Cross P, Andrews H, Jacobs DM, Small S, Bell K, Merchant C, Lantigua R, Costa R, Stern Y, Mayeux R. Incidence of AD in African-Americans, Caribbean Hispanics, and Caucasians in northern Manhattan. Neurology. 2001;56:49–56. doi: 10.1212/wnl.56.1.49. [DOI] [PubMed] [Google Scholar]
- The Diabetes Prevention Program Research, G. Role of Insulin Secretion and Sensitivity in the Evolution of Type 2 Diabetes in the Diabetes Prevention Program: Effects of Lifestyle Intervention and Metformin. Diabetes. 2005;54:2404–2414. doi: 10.2337/diabetes.54.8.2404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trujillo ME, Scherer PE. Adiponectin - journey from an adipocyte secretory protein to biomarker of the metabolic syndrome. J. Intern. Med. 2005;257:167–175. doi: 10.1111/j.1365-2796.2004.01426.x. [DOI] [PubMed] [Google Scholar]
- Vanhanen M, Koivisto K, Moilanen L, Helkala EL, Hanninen T, Soininen H, Kervinen K, Kesaniemi YA, Laakso M, Kuusisto J. Association of metabolic syndrome with Alzheimer disease: A population-based study. Neurology. 2006;67:843–847. doi: 10.1212/01.wnl.0000234037.91185.99. [DOI] [PubMed] [Google Scholar]
- Vermeer SE, Prins ND, den Heijer T, Hofman A, Koudstaal PJ, Breteler MM. Silent brain infarcts and the risk of dementia and cognitive decline. N. Engl. J. Med. 2003;348:1215–1222. doi: 10.1056/NEJMoa022066. [DOI] [PubMed] [Google Scholar]
- Wahrenberg H, Hertel K, Leijonhufvud B-M, Persson L-G, Toft E, Arner P. Use of waist circumference to predict insulin resistance: retrospective study. BMJ. 2005;330:1363–1364. doi: 10.1136/bmj.38429.473310.AE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watson GS, Bernhardt T, Reger MA, Cholerton BA, Baker LD, Peskind ER, Asthana S, Plymate SR, Frolich L, Craft S. Insulin effects on CSF norepinephrine and cognition in Alzheimer's disease. Neurobiol. Aging. 2006;27:38–41. doi: 10.1016/j.neurobiolaging.2004.11.011. [DOI] [PubMed] [Google Scholar]
- Watson GS, Cholerton BA, Reger MA, Baker LD, Plymate SR, Asthana S, Fishel MA, Kulstad JJ, Green PS, Cook DG, Kahn SE, Keeling ML, Craft S. Preserved Cognition in Patients With Early Alzheimer Disease and Amnestic Mild Cognitive Impairment During Treatment With Rosiglitazone: A Preliminary Study. Am. J. Geriatr. Psychiatry. 2005;13:950–958. doi: 10.1176/appi.ajgp.13.11.950. [DOI] [PubMed] [Google Scholar]
- Watson GS, Craft S. Modulation of memory by insulin and glucose: neuropsychological observations in Alzheimer's disease. Eur. J. Pharmacol. 2004;490:97–113. doi: 10.1016/j.ejphar.2004.02.048. [DOI] [PubMed] [Google Scholar]
- Wedick NM, Mayer-Davis EJ, Wingard DL, Addy CL, Barrett-Connor E. Insulin Resistance Precedes Weight Loss in Adults without Diabetes : The Rancho Bernardo Study. Am. J. Epidemiol. 2001;153:1199–1205. doi: 10.1093/aje/153.12.1199. [DOI] [PubMed] [Google Scholar]
- White H, Pieper C, Schmader K, Fillenbaum G. Weight change in Alzheimer's disease. J. Am. Geriatr. Soc. 1996;44:265–272. doi: 10.1111/j.1532-5415.1996.tb00912.x. [DOI] [PubMed] [Google Scholar]
- Whitmer RA, Gunderson EP, Barrett-Connor E, Quesenberry CP, Jr, Yaffe K. Obesity in middle age and future risk of dementia: a 27 year longitudinal population based study. BMJ. 2005a doi: 10.1136/bmj.38446.466238.E0. bmj.38446.466238.E466230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whitmer RA, Sidney S, Selby J, Johnston SC, Yaffe K. Midlife cardiovascular risk factors and risk of dementia in late life. Neurology. 2005b;64:277–281. doi: 10.1212/01.WNL.0000149519.47454.F2. [DOI] [PubMed] [Google Scholar]
- Xu WL, Qiu CX, Wahlin A, Winblad B, Fratiglioni L. Diabetes mellitus and risk of dementia in the Kungsholmen project: a 6-year follow-up study. Neurology. 2004;63:1181–1186. doi: 10.1212/01.wnl.0000140291.86406.d1. [DOI] [PubMed] [Google Scholar]
- Yaffe K, Blackwell T, Kanaya AM, Davidowitz N, Barrett-Connor E, Krueger K. Diabetes, impaired fasting glucose, and development of cognitive impairment in older women. Neurology. 2004a;63:658–663. doi: 10.1212/01.wnl.0000134666.64593.ba. [DOI] [PubMed] [Google Scholar]
- Yaffe K, Kanaya A, Lindquist K, Simonsick EM, Harris T, Shorr RI, Tylavsky FA, Newman AB. The Metabolic Syndrome, Inflammation, and Risk of Cognitive Decline. JAMA. 2004b;292:2237–2242. doi: 10.1001/jama.292.18.2237. [DOI] [PubMed] [Google Scholar]
- Yamagishi S, Nakamura K, Inoue H, Kikuchi S, Takeuchi M. Serum or cerebrospinal fluid levels of glyceraldehyde-derived advanced glycation end products (AGEs) may be a promising biomarker for early detection of Alzheimer's disease. Med. Hypotheses. 2005;64:1205–1207. doi: 10.1016/j.mehy.2005.01.016. [DOI] [PubMed] [Google Scholar]
- Yesavage JA, O'Hara R, Kraemer H, Noda A, Taylor JL, Ferris S, Gely-Nargeot MC, Rosen A, Friedman L, Sheikh J, Derouesne C. Modeling the prevalence and incidence of Alzheimer's disease and mild cognitive impairment. J. Psychiatr. Res. 2002;36:281–286. doi: 10.1016/s0022-3956(02)00020-1. [DOI] [PubMed] [Google Scholar]
- Yoshitake T, Kiyohara Y, Kato I, Ohmura T, Iwamoto H, Nakayama K, Ohmori S, Nomiyama K, Kawano H, Ueda K, et al. Incidence and risk factors of vascular dementia and Alzheimer's disease in a defined elderly Japanese population: the Hisayama Study. Neurology. 1995;45:1161–1168. doi: 10.1212/wnl.45.6.1161. [DOI] [PubMed] [Google Scholar]
- Yu YH, Ginsberg HN. Adipocyte signaling and lipid homeostasis: sequelae of insulin-resistant adipose tissue. Circ. Res. 2005;96:1042–1052. doi: 10.1161/01.RES.0000165803.47776.38. [DOI] [PubMed] [Google Scholar]