Abstract
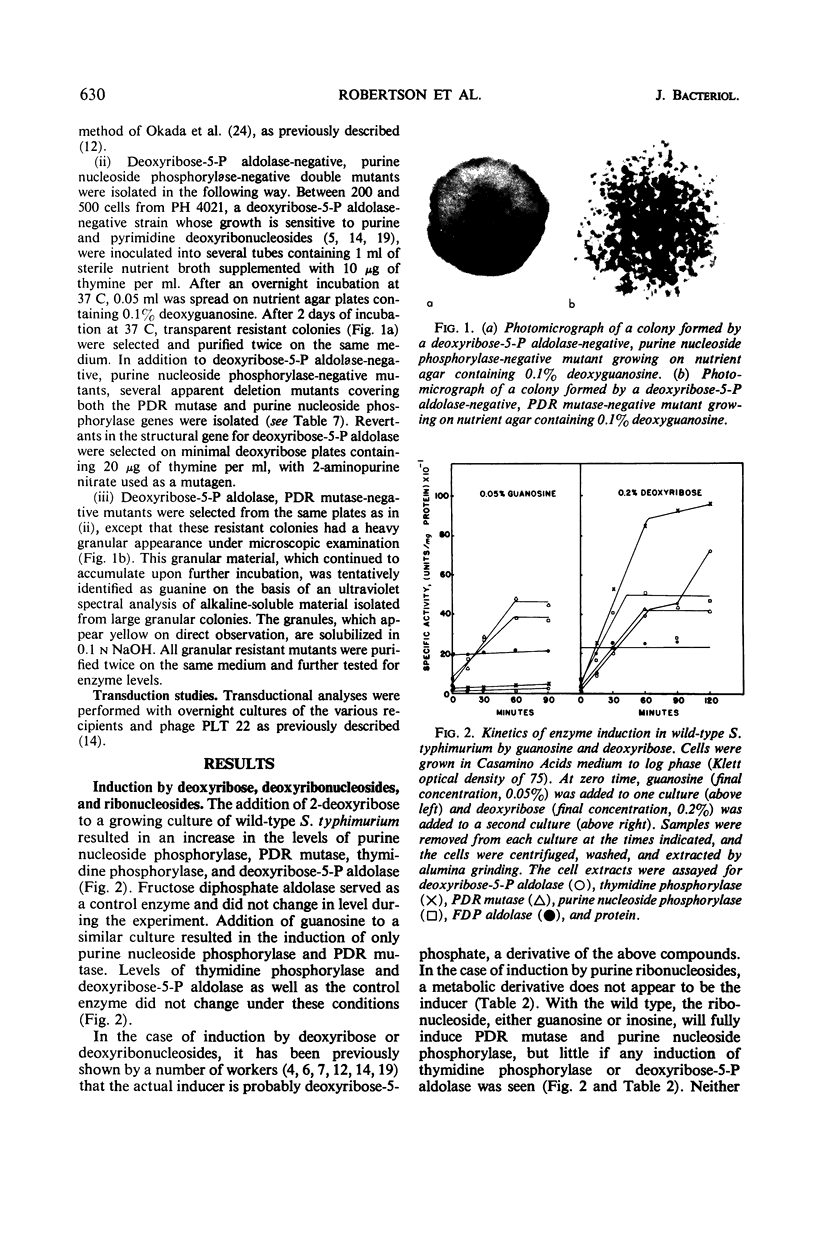
Four enzymes involved in ribonucleoside and deoxyribonucleoside catabolism (deoxyribose-5-P aldolase, thymidine phosphorylase, phosphodeoxyribomutase, and purine nucleoside phosphorylase) are coded for by four closely linked structural genes on the Salmonella chromosome. The genetic order of these genes is (deoC-deoA-deoB-deoD)-serB-thr. Studies on polarity mutants and induction patterns indicate that the deoB and deoD genes may constitute a single operon and that the deoC and deoA genes may constitute a second closely linked operon.
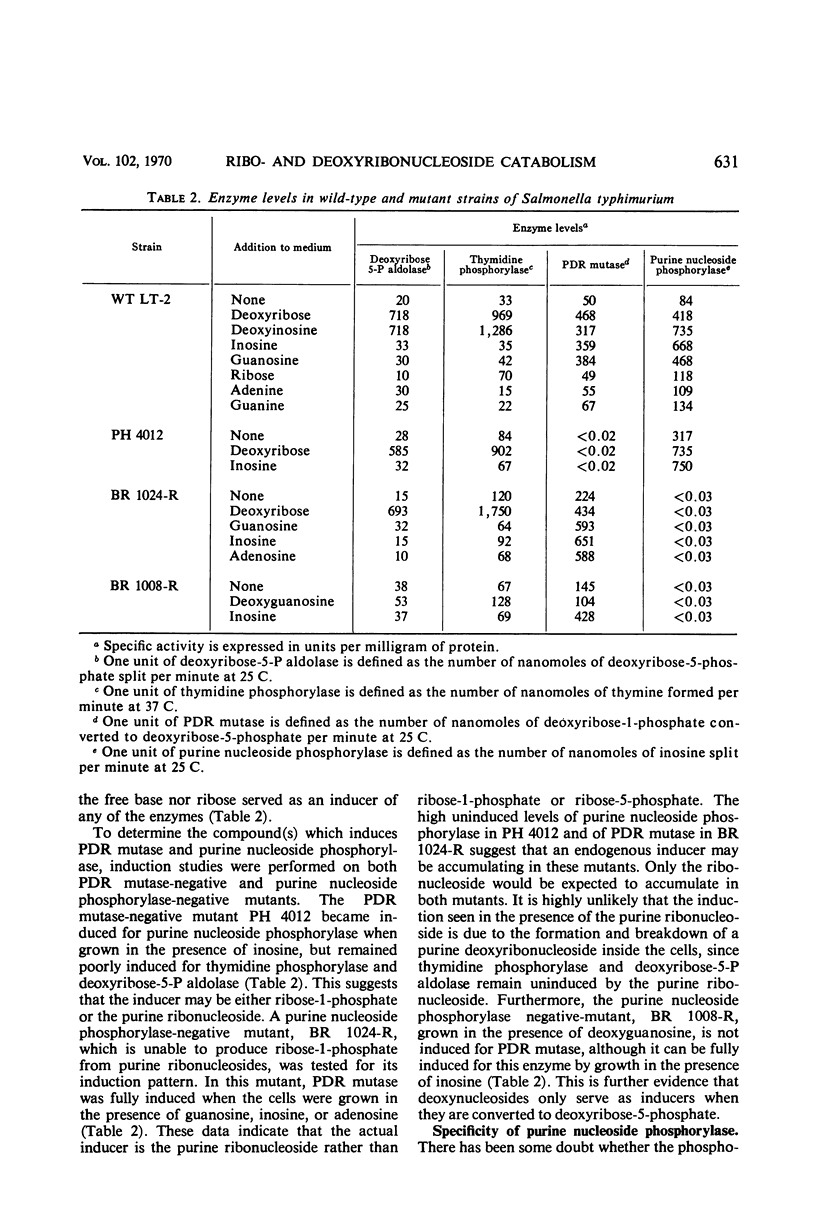
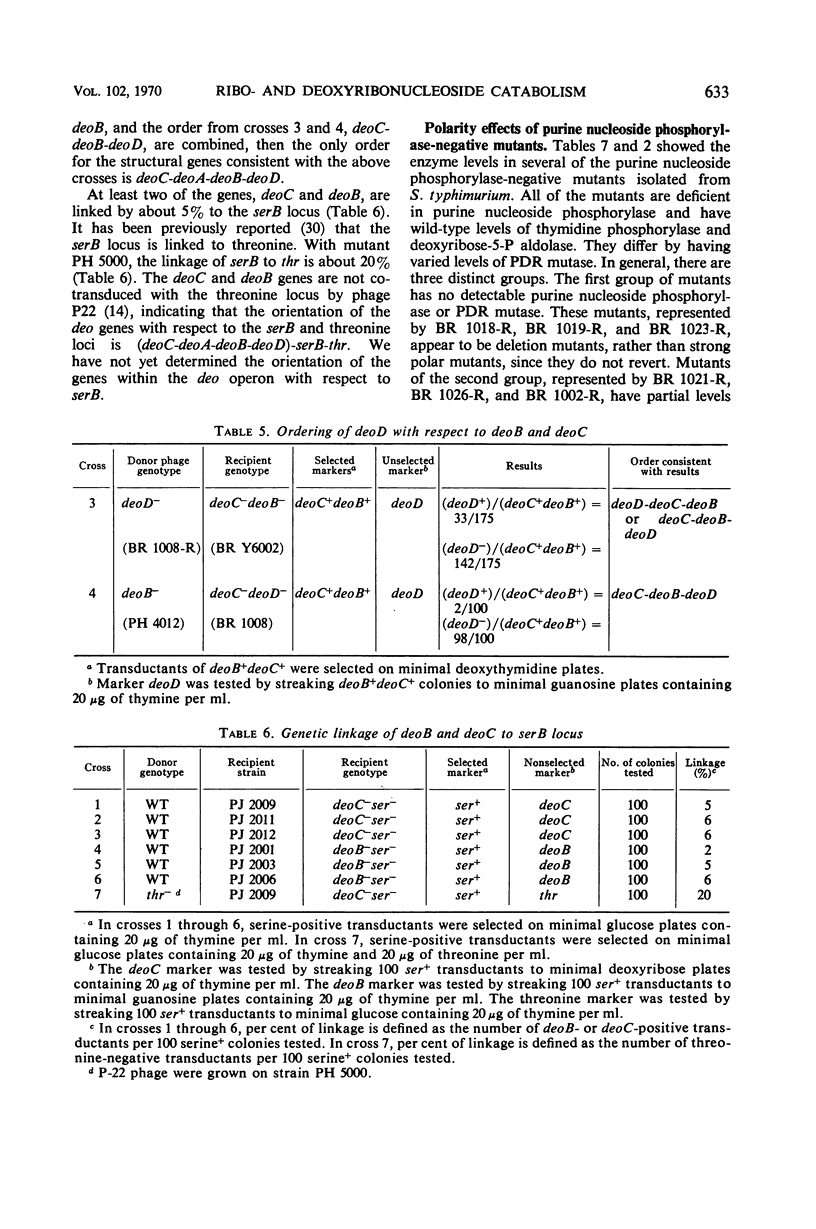
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ahmad S. I., Barth P. T., Pritchard R. H. Properties of a mutant of Escherichia coli lacking purine nucleoside phosphorylase. Biochim Biophys Acta. 1968 Jul 23;161(2):581–583. doi: 10.1016/0005-2787(68)90140-8. [DOI] [PubMed] [Google Scholar]
- Ahmad S. I., Pritchard R. H. A map of four genes specifying enzymes involved in catabolism of nucleosides and deoxynucleosides in Escherichia coli. Mol Gen Genet. 1969 Aug 15;104(4):351–359. doi: 10.1007/BF00334234. [DOI] [PubMed] [Google Scholar]
- Alikhanian S. I., Iljina T. S., Kaliaeva E. S., Kameneva S. V., Sukhodolec V. V. A genetical study of thymineless mutants of E. coli K12. Genet Res. 1966 Aug;8(1):83–100. doi: 10.1017/s0016672300009939. [DOI] [PubMed] [Google Scholar]
- Barth P. T., Beacham I. R., Ahmad S. I., Pritchard R. H. The inducer of the deoxynucleoside phosphorylases and deoxyriboaldolase in Escherichia coli. Biochim Biophys Acta. 1968 Jul 23;161(2):554–557. doi: 10.1016/0005-2787(68)90132-9. [DOI] [PubMed] [Google Scholar]
- Beacham I. R., Eisenstark A., Barth P. T., Pritchard R. H. Deoxynucleoside-sensitive mutants of Salmonella typhimurium. Mol Gen Genet. 1968;102(2):112–127. doi: 10.1007/BF01789138. [DOI] [PubMed] [Google Scholar]
- Breitman T. R., Bradford R. M. Inability of low thymine-requiring mutants of Escherichia coli lacking phosphodeoxyribomutase to be induced for deoxythymidine phosphorylase and deoxyriboaldolase. J Bacteriol. 1968 Jun;95(6):2434–2435. doi: 10.1128/jb.95.6.2434-2435.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Breitman T. R., Bradford R. M. The absence of deoxyriboaldolase activity in a thymineless mutant of Escherichia coli strain 15: a possible explanation for the low thymine requirement of some thymineless strains. Biochim Biophys Acta. 1967 Mar 29;138(1):217–220. doi: 10.1016/0005-2787(67)90610-7. [DOI] [PubMed] [Google Scholar]
- Dale B., Greenberg G. R. Genetic mapping of a mutation in Escherichia coli showing reduced activity of thymidine phosphorylase. J Bacteriol. 1967 Sep;94(3):778–779. doi: 10.1128/jb.94.3.778-779.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eisenstark A., Eisenstark R., Cunningham S. Genetic analysis of thymineless(thy) mutants in Salmonella typhimurium. Genetics. 1968 Apr;58(4):493–506. doi: 10.1093/genetics/58.4.493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- FRIEDKIN M., ROBERTS D. The enzymatic synthesis of nucleosides. I. Thymidine phosphorylase in mammalian tissue. J Biol Chem. 1954 Mar;207(1):245–256. [PubMed] [Google Scholar]
- GORINI L., KAUFMAN H. Selecting bacterial mutants by the penicillin method. Science. 1960 Feb 26;131(3400):604–605. doi: 10.1126/science.131.3400.604. [DOI] [PubMed] [Google Scholar]
- HOFFMANN C. E., LAMPEN J. O. Products of desoxyribose degradation by Escherichia coli. J Biol Chem. 1952 Oct;198(2):885–893. [PubMed] [Google Scholar]
- Hoffee P. A. 2-deoxyribose gene-enzyme complex in Salmonella typhimurium. I. Isolation and enzymatic characterization of 2-deoxyribose-negative mutants. J Bacteriol. 1968 Feb;95(2):449–457. doi: 10.1128/jb.95.2.449-457.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoffee P. A. 2-deoxyribose-5-phosphate aldolase of Salmonella typhimurium: purification and properties. Arch Biochem Biophys. 1968 Sep 10;126(3):795–802. doi: 10.1016/0003-9861(68)90473-6. [DOI] [PubMed] [Google Scholar]
- Hoffee P. A., Robertson B. C. 2-Deoxyribose gene-enzyme complex in Salmonella typhimurium: regulation of phosphodeoxyribomutase. J Bacteriol. 1969 Mar;97(3):1386–1396. doi: 10.1128/jb.97.3.1386-1396.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kammen H. O., Koo R. Phosphopentomutases. I. Identification of two activities in rabbit tissues. J Biol Chem. 1969 Sep 25;244(18):4888–4893. [PubMed] [Google Scholar]
- Karlström O. Mutants of Escherichia coli defective in ribonucleoside and deoxyribonucleoside catabolism. J Bacteriol. 1968 Mar;95(3):1069–1077. doi: 10.1128/jb.95.3.1069-1077.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Lomax M. S., Greenberg G. R. Characteristics of the deo operon: role in thymine utilization and sensitivity to deoxyribonucleosides. J Bacteriol. 1968 Aug;96(2):501–514. doi: 10.1128/jb.96.2.501-514.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MANSON L. A., LAMPEN J. O. The metabolism of desoxyribose nucleosides in Escherichia coli. J Biol Chem. 1951 Dec;193(2):539–547. [PubMed] [Google Scholar]
- Munch-Petersen A. On the catabolism of deoxyribonucleosides in cells and cell extracts of Escherichia coli. Eur J Biochem. 1968 Nov;6(3):432–442. doi: 10.1111/j.1432-1033.1968.tb00465.x. [DOI] [PubMed] [Google Scholar]
- Munch-Petersen A. Thymineless mutants of Escherichia coli with deficiencies in deoxyribomutase and deoxyriboaldolase. Biochim Biophys Acta. 1968 Jun 18;161(1):279–282. doi: 10.1016/0005-2787(68)90325-0. [DOI] [PubMed] [Google Scholar]
- OKADA T., HOMMA J., SONOHARA H. Improved method for obtaining thymineless mutants of Escherichia coli and Salmonella typhimurium. J Bacteriol. 1962 Sep;84:602–603. doi: 10.1128/jb.84.3.602-603.1962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okada T. Mutational Site of the Gene Controlling Quantitative Thymine Requirement in ESCHERICHIA COLI K-12. Genetics. 1966 Dec;54(6):1329–1336. doi: 10.1093/genetics/54.6.1329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- PARK J. T., JOHNSON M. J. A submicrodetermination of glucose. J Biol Chem. 1949 Nov;181(1):149–151. [PubMed] [Google Scholar]
- RACKER E. Enzymatic synthesis and breakdown of desoxyribose phosphate. J Biol Chem. 1952 May;196(1):347–365. [PubMed] [Google Scholar]
- RAZZELL W. E., CASSHYAP P. SUBSTRATE SPECIFICITY AND INDUCTION OF THYMIDINE PHOSPHORYLASE IN ESCHERICHIA COLI. J Biol Chem. 1964 Jun;239:1789–1793. [PubMed] [Google Scholar]
- SMITH C. G., BERNSTEIN I. A. Studies on phosphodeoxyribomutase. Biochim Biophys Acta. 1961 Sep 2;52:184–193. doi: 10.1016/0006-3002(61)90916-7. [DOI] [PubMed] [Google Scholar]
- Sanderson K. E. Revised linkage map of Salmonella typhimurium. Bacteriol Rev. 1967 Dec;31(4):354–372. doi: 10.1128/br.31.4.354-372.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]




