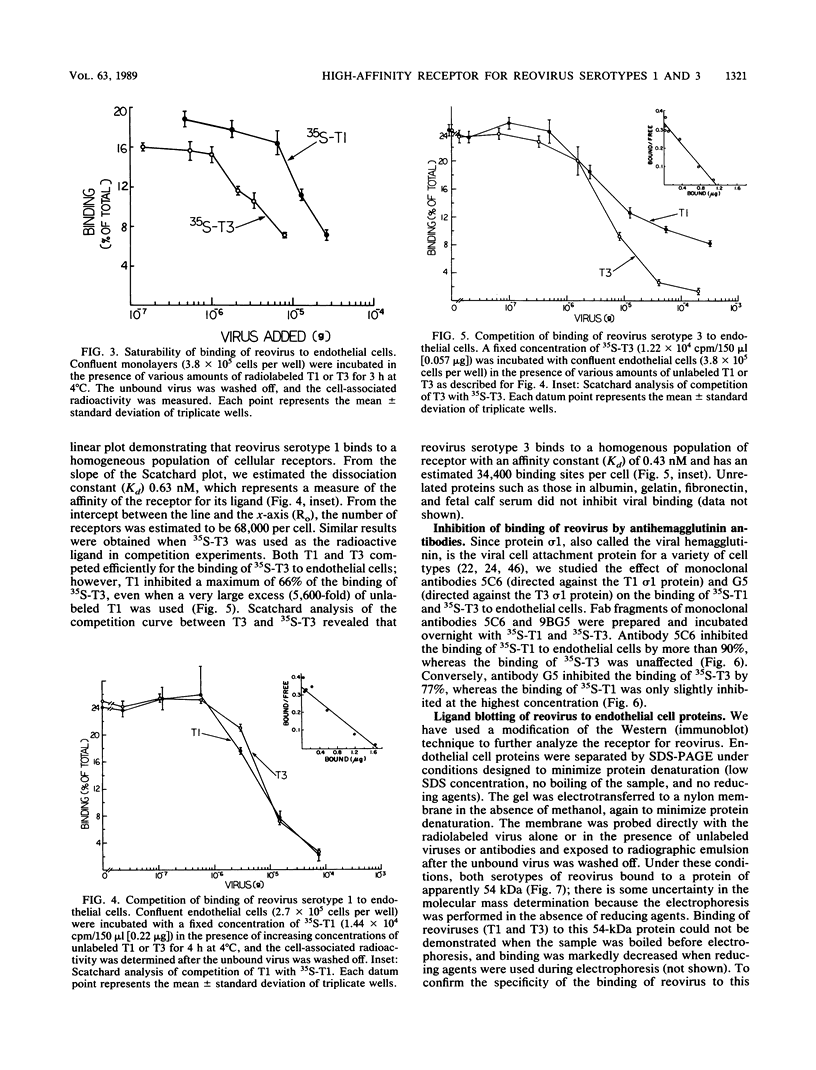
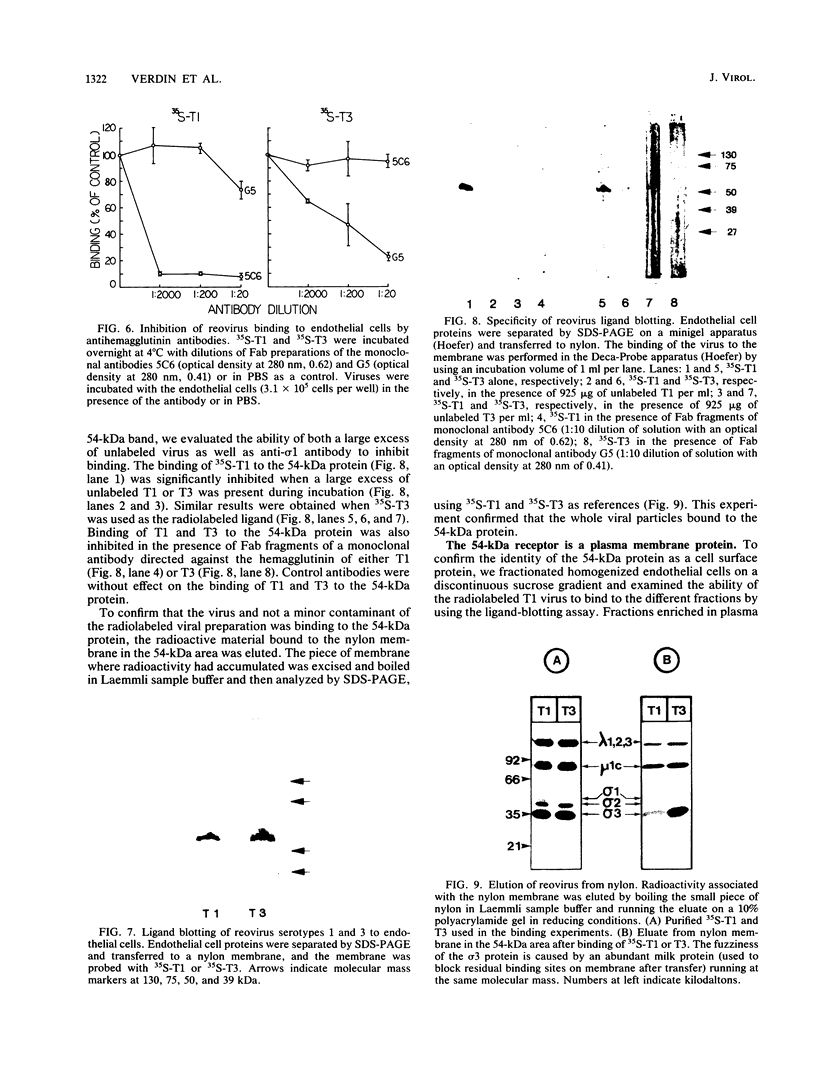
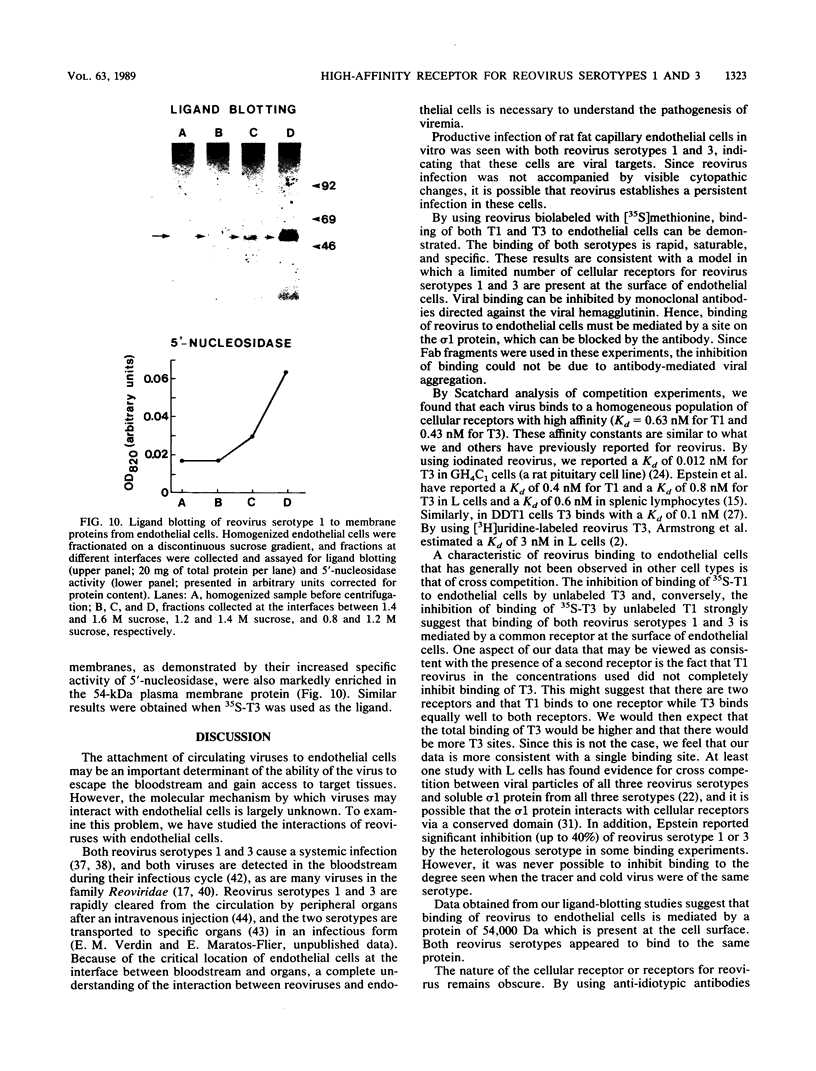
Abstract
During viremia, viruses may be cleared from the bloodstream and taken up by specific organs. The uptake of virus from the bloodstream is dependent on the association of viral particles with endothelial cells that line the luminal surfaces of large and small blood vessels. To understand the nature of this interaction, we have studied the binding of reovirus serotypes 1 and 3 to these cells in vitro. Both serotypes of reovirus productively infected endothelial cells. By using [35S]methionine-biolabeled reovirus as a tracer ligand, we found that both viruses rapidly bind to endothelial cells and that equilibrium is reached after 4 h. The binding of the radiolabeled viruses was saturable and mediated by a homogeneous population of cellular receptors with very high affinity (Kd = 0.5 nM) for the virus ligands. Both serotypes bind to the same receptor, since the attachment of each radiolabeled serotype is inhibited by both the homologous and heterologous unlabeled virus. Exposure of labeled virus to monoclonal antibodies directed against the viral hemagglutinin (sigma 1 protein) inhibited binding, demonstrating that the attachment of reovirus to endothelial cells is mediated by the hemagglutinin for both serotypes. By using a novel ligand-blotting assay, the binding of both viruses to a 54,000-dalton protein could be demonstrated. The binding of each radiolabeled serotype to this protein was inhibited by the homologous and heterologous unlabeled serotype. By using cell fractionation after homogenization, we demonstrated that this 54-kilodalton protein is a membrane protein, in agreement with its proposed role as a cell surface receptor for reovirus serotypes 1 and 3.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aggarwal B. B., Eessalu T. E., Hass P. E. Characterization of receptors for human tumour necrosis factor and their regulation by gamma-interferon. Nature. 1985 Dec 19;318(6047):665–667. doi: 10.1038/318665a0. [DOI] [PubMed] [Google Scholar]
- Armstrong G. D., Paul R. W., Lee P. W. Studies on reovirus receptors of L cells: virus binding characteristics and comparison with reovirus receptors of erythrocytes. Virology. 1984 Oct 15;138(1):37–48. doi: 10.1016/0042-6822(84)90145-4. [DOI] [PubMed] [Google Scholar]
- Avruch J., Wallach D. F. Preparation and properties of plasma membrane and endoplasmic reticulum fragments from isolated rat fat cells. Biochim Biophys Acta. 1971 Apr 13;233(2):334–347. doi: 10.1016/0005-2736(71)90331-2. [DOI] [PubMed] [Google Scholar]
- Banskota N. K., Carpentier J. L., King G. L. Processing and release of insulin and insulin-like growth factor I by macro- and microvascular endothelial cells. Endocrinology. 1986 Nov;119(5):1904–1913. doi: 10.1210/endo-119-5-1904. [DOI] [PubMed] [Google Scholar]
- Bassel-Duby R., Spriggs D. R., Tyler K. L., Fields B. N. Identification of attenuating mutations on the reovirus type 3 S1 double-stranded RNA segment with a rapid sequencing technique. J Virol. 1986 Oct;60(1):64–67. doi: 10.1128/jvi.60.1.64-67.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bellamy A. R., Shapiro L., August J. T., Joklik W. K. Studies on reovirus RNA. I. Characterization of reovirus genome RNA. J Mol Biol. 1967 Oct 14;29(1):1–17. doi: 10.1016/0022-2836(67)90177-5. [DOI] [PubMed] [Google Scholar]
- Burstin S. J., Spriggs D. R., Fields B. N. Evidence for functional domains on the reovirus type 3 hemagglutinin. Virology. 1982 Feb;117(1):146–155. doi: 10.1016/0042-6822(82)90514-1. [DOI] [PubMed] [Google Scholar]
- Cashdollar L. W., Chmelo R. A., Wiener J. R., Joklik W. K. Sequences of the S1 genes of the three serotypes of reovirus. Proc Natl Acad Sci U S A. 1985 Jan;82(1):24–28. doi: 10.1073/pnas.82.1.24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choi A. H., Lee P. W. Does the beta-adrenergic receptor function as a reovirus receptor? Virology. 1988 Mar;163(1):191–197. doi: 10.1016/0042-6822(88)90246-2. [DOI] [PubMed] [Google Scholar]
- Co M. S., Gaulton G. N., Fields B. N., Greene M. I. Isolation and biochemical characterization of the mammalian reovirus type 3 cell-surface receptor. Proc Natl Acad Sci U S A. 1985 Mar;82(5):1494–1498. doi: 10.1073/pnas.82.5.1494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Co M. S., Gaulton G. N., Tominaga A., Homcy C. J., Fields B. N., Greene M. I. Structural similarities between the mammalian beta-adrenergic and reovirus type 3 receptors. Proc Natl Acad Sci U S A. 1985 Aug;82(16):5315–5318. doi: 10.1073/pnas.82.16.5315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dalgleish A. G., Beverley P. C., Clapham P. R., Crawford D. H., Greaves M. F., Weiss R. A. The CD4 (T4) antigen is an essential component of the receptor for the AIDS retrovirus. Nature. 1984 Dec 20;312(5996):763–767. doi: 10.1038/312763a0. [DOI] [PubMed] [Google Scholar]
- Dower S. K., Kronheim S. R., Hopp T. P., Cantrell M., Deeley M., Gillis S., Henney C. S., Urdal D. L. The cell surface receptors for interleukin-1 alpha and interleukin-1 beta are identical. Nature. 1986 Nov 20;324(6094):266–268. doi: 10.1038/324266a0. [DOI] [PubMed] [Google Scholar]
- Epstein R. L., Powers M. L., Rogart R. B., Weiner H. L. Binding of 125I-labeled reovirus to cell surface receptors. Virology. 1984 Feb;133(1):46–55. doi: 10.1016/0042-6822(84)90424-0. [DOI] [PubMed] [Google Scholar]
- Funke I., Hahn A., Rieber E. P., Weiss E., Riethmüller G. The cellular receptor (CD4) of the human immunodeficiency virus is expressed on neurons and glial cells in human brain. J Exp Med. 1987 Apr 1;165(4):1230–1235. doi: 10.1084/jem.165.4.1230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klatzmann D., Champagne E., Chamaret S., Gruest J., Guetard D., Hercend T., Gluckman J. C., Montagnier L. T-lymphocyte T4 molecule behaves as the receptor for human retrovirus LAV. Nature. 1984 Dec 20;312(5996):767–768. doi: 10.1038/312767a0. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lasky L. A., Nakamura G., Smith D. H., Fennie C., Shimasaki C., Patzer E., Berman P., Gregory T., Capon D. J. Delineation of a region of the human immunodeficiency virus type 1 gp120 glycoprotein critical for interaction with the CD4 receptor. Cell. 1987 Sep 11;50(6):975–985. doi: 10.1016/0092-8674(87)90524-1. [DOI] [PubMed] [Google Scholar]
- Lee P. W., Hayes E. C., Joklik W. K. Protein sigma 1 is the reovirus cell attachment protein. Virology. 1981 Jan 15;108(1):156–163. doi: 10.1016/0042-6822(81)90535-3. [DOI] [PubMed] [Google Scholar]
- Maratos-Flier E., Kahn C. R., Spriggs D. R., Fields B. N. Specific plasma membrane receptors for reovirus on rat pituitary cells in culture. J Clin Invest. 1983 Aug;72(2):617–621. doi: 10.1172/JCI111010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paul R. W., Lee P. W. Glycophorin is the reovirus receptor on human erythrocytes. Virology. 1987 Jul;159(1):94–101. doi: 10.1016/0042-6822(87)90351-5. [DOI] [PubMed] [Google Scholar]
- STANLEY N. F., LEAK P. J., WALTERS M. N., JOSKE R. A. MURINE INFECTIONS WITH REOVIRUS. II. THE CHRONIC DISEASE FOLLOWING REOVIRUS TYPE 3 INFECTION. Br J Exp Pathol. 1964 Apr;45:142–149. [PMC free article] [PubMed] [Google Scholar]
- STNALEY N. F., LEAK P. J. MURINE INFECTION WITH REOVIRUS TYPE 3 AND THE RUNTING SYNDROME. Nature. 1963 Sep 28;199:1309–1310. doi: 10.1038/1991309a0. [DOI] [PubMed] [Google Scholar]
- Sawutz D. G., Bassel-Duby R., Homcy C. J. High affinity binding of reovirus type 3 to cells that lack beta adrenergic receptor activity. Life Sci. 1987 Jan 26;40(4):399–406. doi: 10.1016/0024-3205(87)90142-1. [DOI] [PubMed] [Google Scholar]
- Sharpe A. H., Fields B. N. Pathogenesis of viral infections. Basic concepts derived from the reovirus model. N Engl J Med. 1985 Feb 21;312(8):486–497. doi: 10.1056/NEJM198502213120806. [DOI] [PubMed] [Google Scholar]
- Sharpe A. H., Fields B. N. Reovirus inhibition of cellular RNA and protein synthesis: role of the S4 gene. Virology. 1982 Oct 30;122(2):381–391. doi: 10.1016/0042-6822(82)90237-9. [DOI] [PubMed] [Google Scholar]
- Shatkin A. J., Sipe J. D., Loh P. Separation of ten reovirus genome segments by polyacrylamide gel electrophoresis. J Virol. 1968 Oct;2(10):986–991. doi: 10.1128/jvi.2.10.986-991.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simionescu N. Cellular aspects of transcapillary exchange. Physiol Rev. 1983 Oct;63(4):1536–1579. doi: 10.1152/physrev.1983.63.4.1536. [DOI] [PubMed] [Google Scholar]
- Smith R. E., Zweerink H. J., Joklik W. K. Polypeptide components of virions, top component and cores of reovirus type 3. Virology. 1969 Dec;39(4):791–810. doi: 10.1016/0042-6822(69)90017-8. [DOI] [PubMed] [Google Scholar]
- Stott J. L., Oberst R. D., Channell M. B., Osburn B. I. Genome segment reassortment between two serotypes of bluetongue virus in a natural host. J Virol. 1987 Sep;61(9):2670–2674. doi: 10.1128/jvi.61.9.2670-2674.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verdin E. M., Lynn S. P., Fields B. N., Maratos-Flier E. Uptake of reovirus serotype 1 by the lungs from the bloodstream is mediated by the viral hemagglutinin. J Virol. 1988 Feb;62(2):545–551. doi: 10.1128/jvi.62.2.545-551.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Voyta J. C., Via D. P., Butterfield C. E., Zetter B. R. Identification and isolation of endothelial cells based on their increased uptake of acetylated-low density lipoprotein. J Cell Biol. 1984 Dec;99(6):2034–2040. doi: 10.1083/jcb.99.6.2034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiner H. L., Ault K. A., Fields B. N. Interaction of reovirus with cell surface receptors. I. Murine and human lymphocytes have a receptor for the hemagglutinin of reovirus type 3. J Immunol. 1980 May;124(5):2143–2148. [PubMed] [Google Scholar]
- Zetter B. R. The endothelial cells of large and small blood vessels. Diabetes. 1981;30(Suppl 2):24–28. doi: 10.2337/diab.30.2.s24. [DOI] [PubMed] [Google Scholar]