Abstract
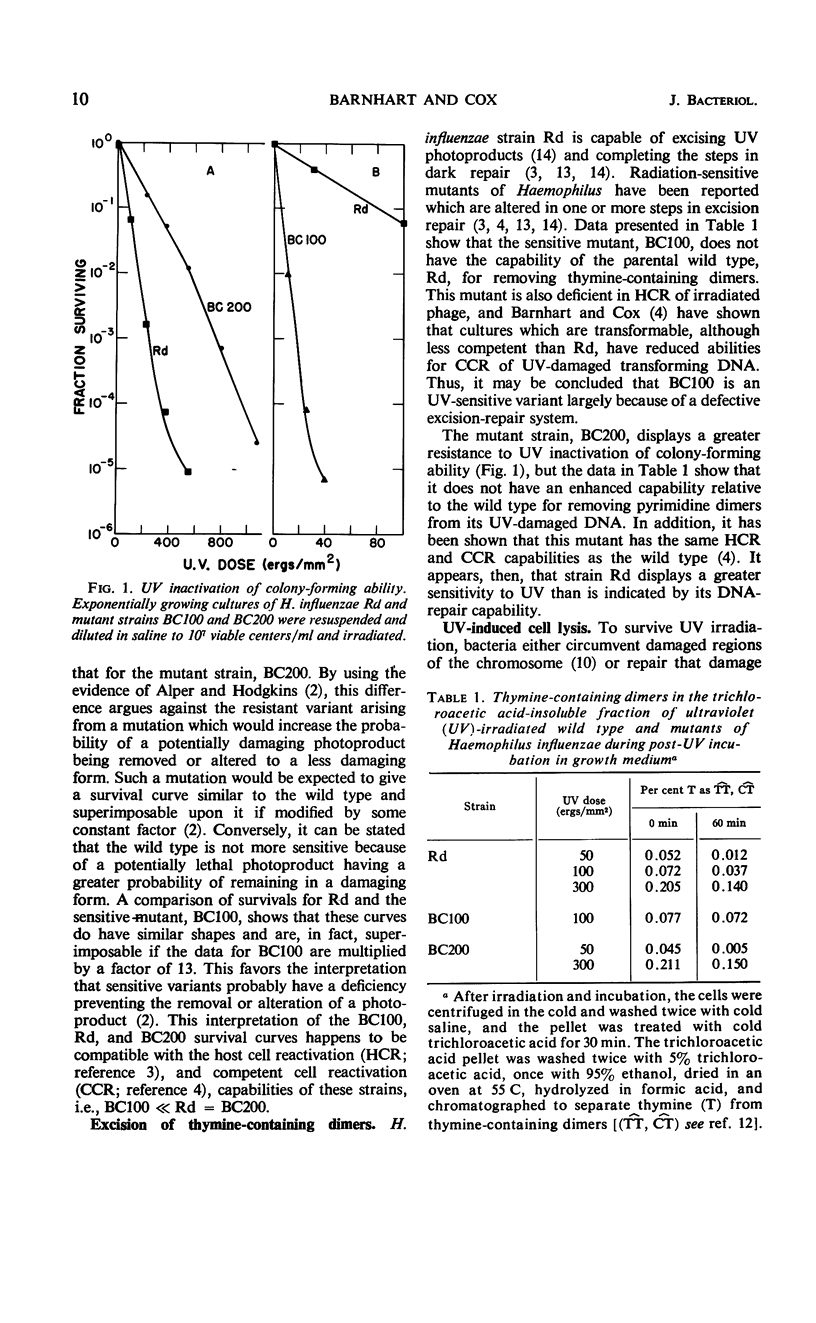
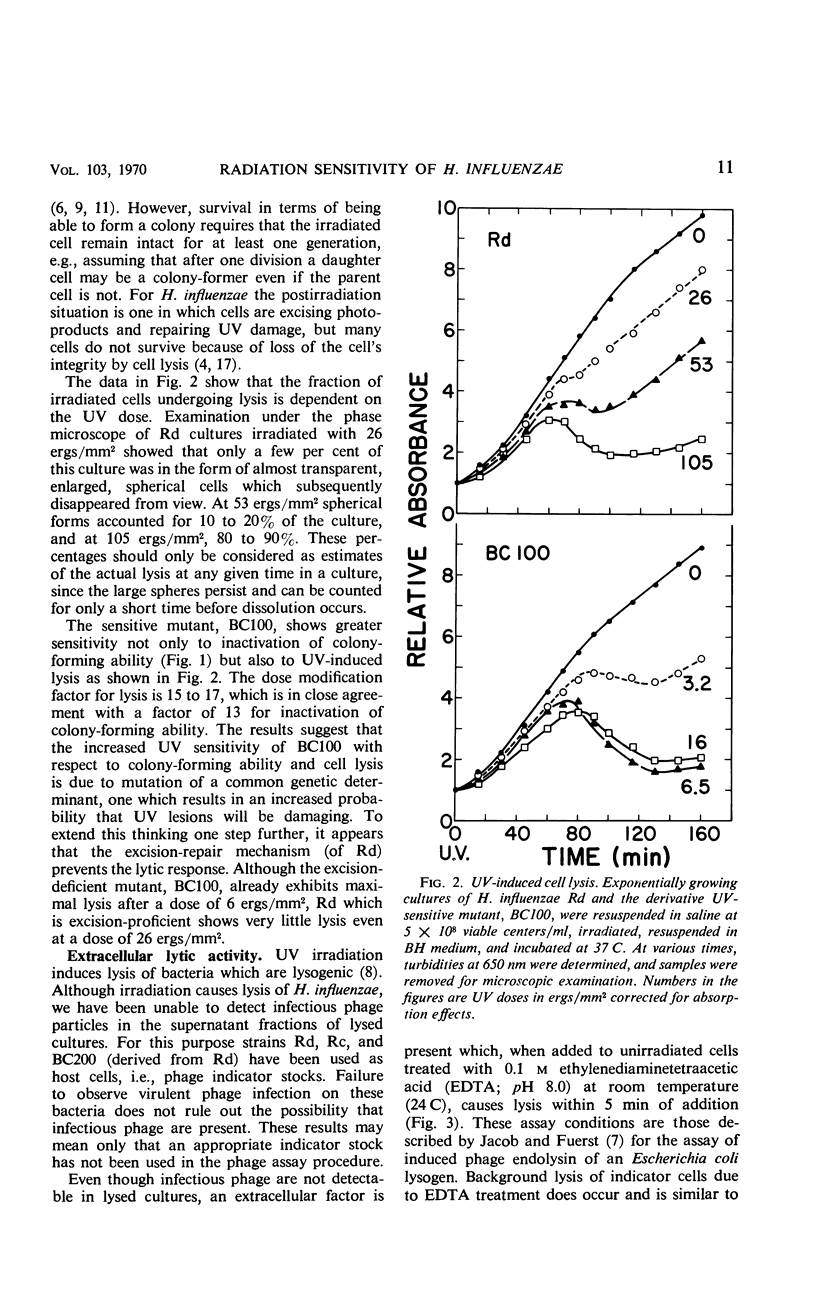
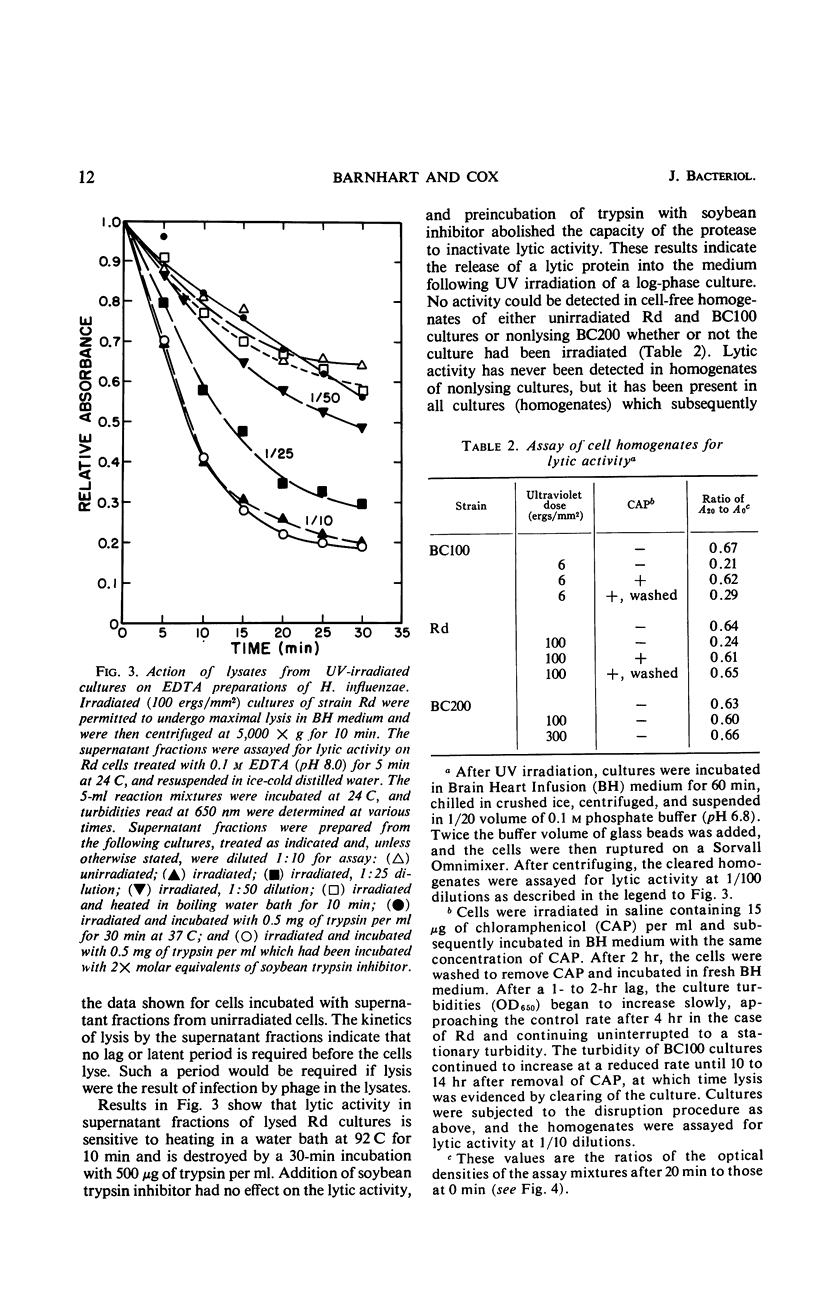
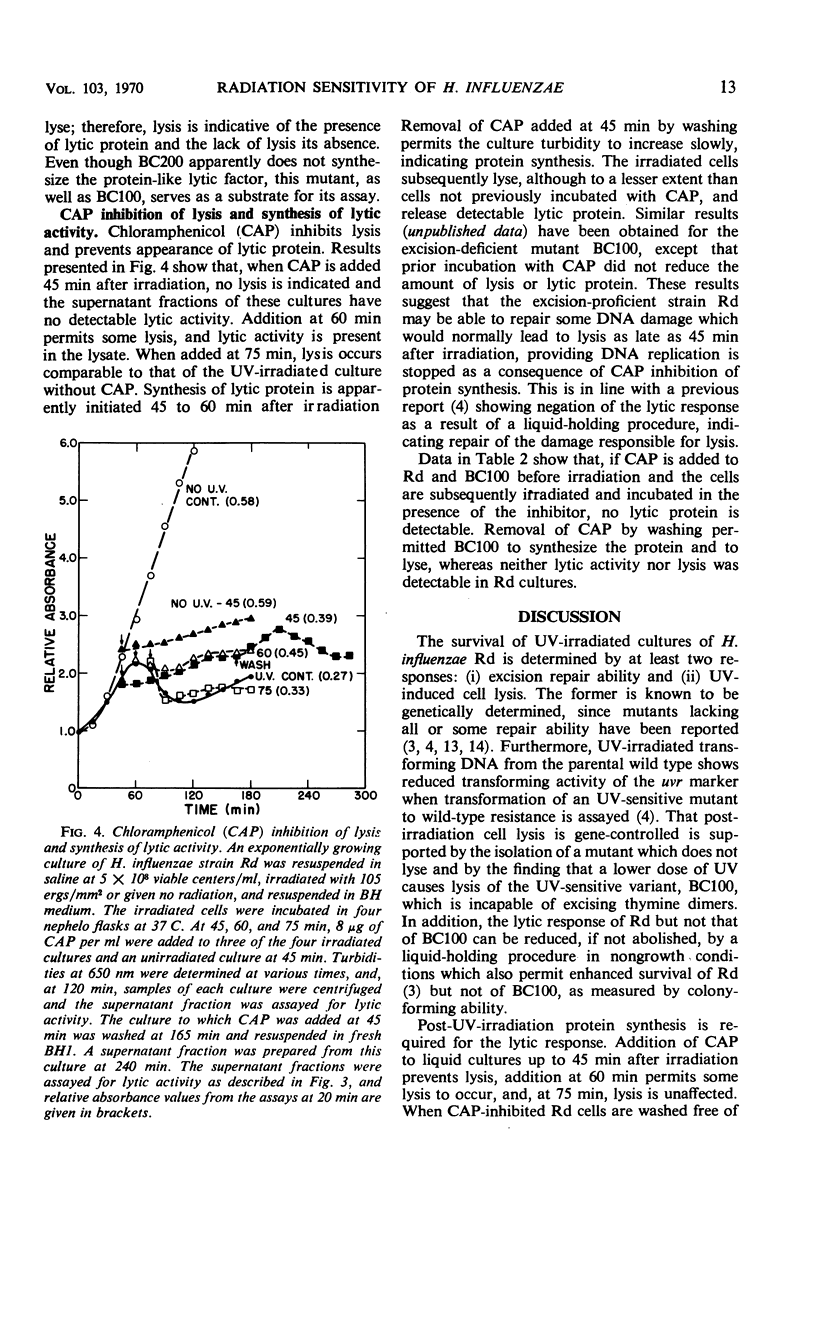
The survival of ultraviolet (UV)-irradiated cultures of Haemophilus influenzae Rd is determined by at least two responses: (i) excision-repair ability and (ii) UV-induced cell lysis. An UV-resistant mutant, BC200, has the same capabilities as the wild type, Rd, for excising dimers but does not exhibit lysis. Lytic response is dose-dependent. Relative to the wild type, a lower dose of UV causes lysis of a UV-sensitive mutant, BC100, which is incapable of excising thymine dimers. A lytic protein is present in cultures undergoing lysis. Synthesis of this protein is initiated 45 to 60 min after irradiation. Lysis appears to be due to derepression of a defective prophage which codes for an endolysin-like lytic enzyme.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- ALEXANDER H. E., LEIDY G. Induction of streptomycin resistance in sensitive Hemophilus influenzae by extracts containing desoxyribonucleic acid from resistant Hemophilus influenzae. J Exp Med. 1953 Jan;97(1):17–31. doi: 10.1084/jem.97.1.17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alper T., Hodgkins B. "Excision repair" and dose-modification: questions raised by radiobiological experiments with acriflavine. Mutat Res. 1969 Jul-Aug;8(1):15–23. doi: 10.1016/0027-5107(69)90136-5. [DOI] [PubMed] [Google Scholar]
- BOYCE R. P., HOWARD-FLANDERS P. RELEASE OF ULTRAVIOLET LIGHT-INDUCED THYMINE DIMERS FROM DNA IN E. COLI K-12. Proc Natl Acad Sci U S A. 1964 Feb;51:293–300. doi: 10.1073/pnas.51.2.293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barnhart B. J., Cox S. H. Radiation-sensitive and radiation-resistant mutants of Haemophilus influenzae. J Bacteriol. 1968 Jul;96(1):280–282. doi: 10.1128/jb.96.1.280-282.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barnhart D. J., Cox S. H. Recovery of Haemophilus influenzae from ultraviolet and x-ray damage. Photochem Photobiol. 1970 Mar;11(3):147–162. doi: 10.1111/j.1751-1097.1970.tb05983.x. [DOI] [PubMed] [Google Scholar]
- HARM W., RUPERT C. S. INFECTION OF TRANSFORMABLE CELLS OF HAEMOPHILUS INFLUENZAE BY BACTERIOPHAGE AND BACTERIOPHAGE DNA. Z Vererbungsl. 1963 Dec 30;94:336–348. doi: 10.1007/BF00897593. [DOI] [PubMed] [Google Scholar]
- JACOB F., FUERST C. R. The mechanism of lysis by phage studied with defective lysogenic bacteria. J Gen Microbiol. 1958 Apr;18(2):518–526. doi: 10.1099/00221287-18-2-518. [DOI] [PubMed] [Google Scholar]
- LWOFF A., SIMINOVITCH L., KJELDGAARD N. Induction de la production de bacteriophages chez une bactérie lysogène. Ann Inst Pasteur (Paris) 1950 Dec;79(6):815–859. [PubMed] [Google Scholar]
- PETTIJOHN D., HANAWALT P. EVIDENCE FOR REPAIR-REPLICATION OF ULTRAVIOLET DAMAGED DNA IN BACTERIA. J Mol Biol. 1964 Aug;9:395–410. doi: 10.1016/s0022-2836(64)80216-3. [DOI] [PubMed] [Google Scholar]
- Rupp W. D., Howard-Flanders P. Discontinuities in the DNA synthesized in an excision-defective strain of Escherichia coli following ultraviolet irradiation. J Mol Biol. 1968 Jan 28;31(2):291–304. doi: 10.1016/0022-2836(68)90445-2. [DOI] [PubMed] [Google Scholar]
- SETLOW R. B., CARRIER W. L. THE DISAPPEARANCE OF THYMINE DIMERS FROM DNA: AN ERROR-CORRECTING MECHANISM. Proc Natl Acad Sci U S A. 1964 Feb;51:226–231. doi: 10.1073/pnas.51.2.226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SETLOW R. B., SWENSON P. A., CARRIER W. L. THYMINE DIMERS AND INHIBITION OF DNA SYNTHESIS BY ULTRAVIOLET IRRADIATION OF CELLS. Science. 1963 Dec 13;142(3598):1464–1466. doi: 10.1126/science.142.3598.1464. [DOI] [PubMed] [Google Scholar]
- STUY J. H. Studies on the radiation inactivation of microorganisms. V. Deoxyribonucleic acid metabolism in ultraviolet-irradiated Haemophilus influenzae. J Bacteriol. 1959 Jul;78(1):49–58. doi: 10.1128/jb.78.1.49-58.1959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Setlow J. K., Brown D. C., Boling M. E., Mattingly A., Gordon M. P. Repair of deoxyribonucleic acid in Haemophilus influenzae. I. X-ray sensitivity of ultraviolet-sensitive mutants and their behavior as hosts to ultraviolet-irradiated bacteriophage and transforming deoxyribonucleic acid. J Bacteriol. 1968 Feb;95(2):546–558. doi: 10.1128/jb.95.2.546-558.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Setlow J. K., Randolph M. L., Boling M. E., Mattingly A., Price G., Gordon M. P. Repair of DNA in Haemophilus influenzae. II. Excision, repair of single-strand breaks, defects in transformation, and host cell modification in UV-sensitive mutants. Cold Spring Harb Symp Quant Biol. 1968;33:209–218. doi: 10.1101/sqb.1968.033.01.024. [DOI] [PubMed] [Google Scholar]
- Stachura I., Mckinley F. W., Leidy G., Alexander H. E. Incomplete bacteriophage-like particles in ultraviolet-irradiated haemophilus. J Bacteriol. 1969 May;98(2):818–820. doi: 10.1128/jb.98.2.818-820.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]


