Abstract
We previously reported in humans a novel histologic phenotype of non-gastro-esophageal reflux disease called lymphocytic esophagitis. In this work, the esophagi of 121 non-human primates (103 baboons and 18 macaques) were investigated. 45 baboons (43.7%) and 9 macaques (50%) had lymphocytic esophagitis. The lymphocytic infiltration in the squamous epithelium involved not only papillary but also inter-papillary fields. Microscopic examination around the papillae revealed a mean of 52 intraepithelial lymphocytes (IELs) per high power field. Of the remaining baboons, 29 had reflux esophagitis (GERD). Among them, 25 (24.3%) 2 (1.9%) and 2 (1.9%) had grade1, 2 and 3 esophagitis, respectively. Of the remaining macaques, 4 had GERD: 1 (5.5%) with grade 1 and 3 (16.7%) with grade 2 esophagitis. None of the macaques had grade 3 esophagitis. The IEL population in lymphocytic esophagitis was composed of T cells, a subset of natural killer cells and of helper and inflammatory T cells. This investigation in non-human primates substantiates the identity of lymphocytic esophagitis as a subset of chronic esophagitis, as well as of reflux esophagitis. The antigenic agent(s) responsible for the marked immunological reaction in lymphocytic esophagitis in non-human primates (and in humans) remains unknown.
Keywords: Esophagus, lymphocytes, chronic inflammation, esophagitis
Introduction
A multilayer of non-keratinized stratified squamous cells lines the mucosa of the esophagus. This layer protects the underlying tissues from noxious agents and irritants contained in passing solids and fluids [1]. The microenvironment on its surface is protected by the alkaline pH from both the saliva and the secretions of the esophageal glands. It is, therefore, not surprising that low pH gastric juices refluxed into its lumen provide a major insult to the esophageal epithelium. This primary gastro-esophageal reflux (GER) [2], if persistent, may severely alter the homeostasis of the esophageal microenvironment leading to mucosal inflammation followed in severe cases by ulceration and the replacement of deeper layers by fibrotic tissue [3]. The sequence of those events is known as gastro-esophageal reflux disease (GERD) [4].
Inflammatory changes in the esophagus may also be induced by other external factors such as fungi (candida albicans), bacteria, viruses, radiation, caustic substances, corticosteroid or antibiotic therapy or by diseases such as diabetes, and general debilitation [2, 5]. Acid reflux from the stomach, however, is by far the most common cause for this condition. The histological changes taking place in the squamous epithelium of the esophagus in cases with reflux esophagitis have been described by Ismail-Beiji and Pope [5]. Similar histological changes were recently found in 8 baboons having reflux esophagitis [6].
In our previous study, while reviewing the histology of the stomach in baboons [7], we noticed in one of them that the esophageal epithelium was infiltrated by a large number of lymphocytes but not by granulocytes. Looking for similar changes in the human esophagus, we recently described lymphocytic esophagitis in the esophageal biopsies of 20 patients without GERD [8].
In the present work, we reviewed the esophagi in 121 non-human primates (NHPs) to assess the frequency of lymphocytic esophagitis and of reflux esophagitis in those animals.
Material and Methods
Of the 121 esophagi (sampled at autopsy) collected in NHPs, 103 were from olive or olive/yellow hybrid baboons (Papio hamadryas anubis, P. h. cynocephalus) and the remaining 18 from macaques (Macaca fasciculata).
The NHPs were members of colonies at the Southwest National Primate Research Center, Southwest Foundation for Biomedical Research. The conditions of animal housing have been reported elsewhere [6]. Briefly the NHPs were housed in metal and concrete indoor-outdoor cages and were fed commercial monkey diets occasionally supplemented with a variety of fruit and vegetables. Water was available ad libitum.
NHPs were euthanized with a commercial barbiturate agent, because of non-gastrointestinal diseases or natural causes such as old age. All procedures were done in accordance with the Institutional Animal Care and Use Committee guidelines.
At necropsy, longitudinal tissue samples from the esophagus were fixed in 10% neutral buffered formalin, processed conventionally, embedded in paraffin, cut at 5 μm, stained with hematoxylin and eosin (H&E), and evaluated by light microscopy.
In addition, immunohistochemistry was done in 4 animals with lymphocytic esophagitis using CD3 (dilution 1:400, Neo Markers, Inc, Freemont, CA, USA) to label T lymphocytes, CD2 (dilution 1:60, Thermo Fisher Scientific, Freemont, CA, USA) to label T cells and a subset of natural killer cells and CD4 (dilution 1:20, Novocastra, New Castle u. Tyne, UK) to label helper and inflammatory T cells.
Lymphocytic esophagitis is characterized by a high number of intraepithelial lymphocytes (IELs) in the epithelium around the papillae (which is centred by a papillary vessel) and in the epithelium between the papillary folds. There is absence of granulocytic (neutrophils and/or eosinophils) infiltration [8]. The papillary folds are usually taller than normal but lacked the basal cell hyperplasia seen in reflux esophagitis [5]. Counting of IELs was performed at 20× in the most affected epithelial areas, without knowledge of the clinical chart including symptoms, in the 121 primates.
According to Ismail-Beiji and Pope [5], reflux esophagitis or gastro-esophageal reflux disease (GERD) can be divided into 3 distinct histological subgroups in human. Grade 1 GERD is characterized by basal cell hyperplasia and abnormally tall (i.e. deep) papillae, while grade 2 reflux esophagitis is characterized by intraepithelial granulocytic infiltration, namely neutrophils, and/or eosinophils in addition to those seen grade 1. If the insulting factor persists or is strong, the epithelium becomes gradually eroded or ulcerated, hence grade 3 GERD. In our previous publication [2], similar criteria were applied and used to classify and grade cases with GERD in baboons.
Results
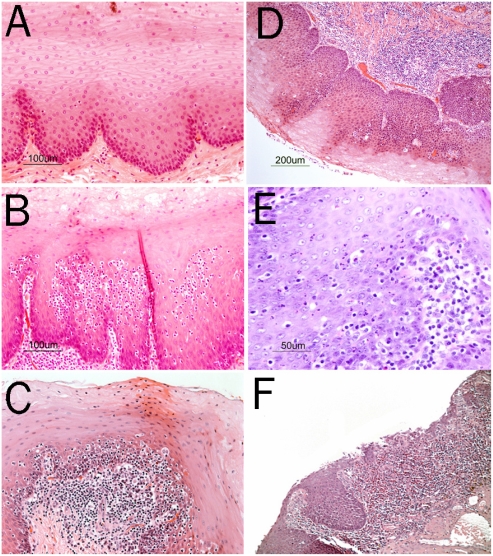
Figure 1 shows the histological differences between the normal esophagus, lymphocytic esophagitis and reflux esophagitis in baboons and in macaques.
Figure 1.
A. Normal squamous epithelium of the esophagus in a baboon (H&E, original magnification 20×). B and C. Lymphocytic esophagitis in baboons showing infiltration of the squamous epithelium of the esophagus around the papillae by a high numbers of lymphocytes as well as chronic inflammation in the lamina propria (H&E, original magnification 20×). D. Grade 1 reflux esophagitis in a baboon (D) with tall papillae and basal-cell proliferation and few intraepithelial lymphocytes. Chronic inflammation in the lamina propria is also seen (H&E, original magnification 10×). Grade 2 reflux esophagitis in a baboon (E) with intraepithelial granulocytes and chronic inflammation in the lamina propria (H&E, original magnification 20×). Grade 3 reflux esophagitis in a baboon (F) with mucosal ulceration and severe chronic inflammation in the lamina propria (H&E, original magnification 20×).
Of the 121 consecutive esophagi examined 49 (40.5%) had lymphocytic esophagitis. Among those, 45 were from baboons (43.7%) and 9 were from macaques (50%). There was no difference between these two species.
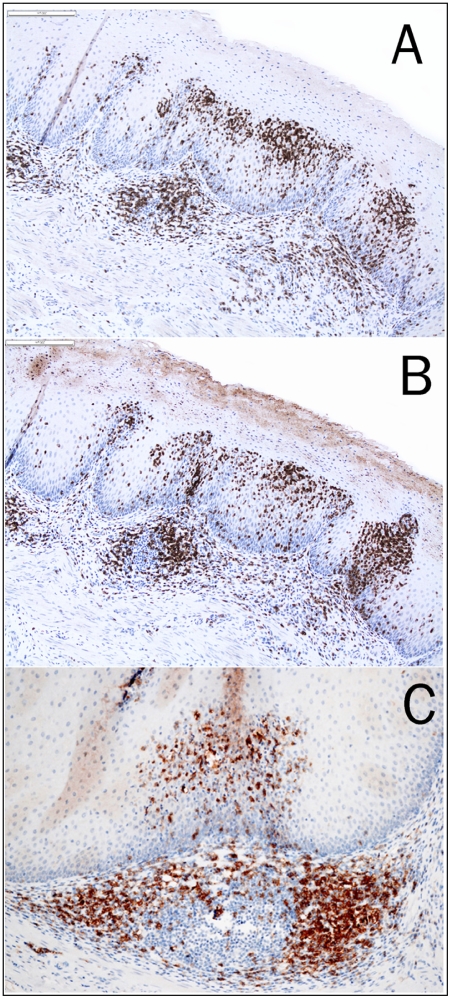
In lymphocytic esophagitis, a mean of 52 IELs per high power field (range from 38 to 58) were found in the squamous epithelium around the papillae. The lymphocytic infiltration was also seen in the epithelium between papillary folds (Figure 1C). The IELs were predominantly T cells with a T-helper phenotype (Figure 2).
Figure 2.
Immunostains for CD2 (A, 10×), CD3 (B, 10×) and CD4 (C, 20×) showing lymphocytic esophagitis with predominantly intraepithelial T cell infiltration.
33 esophagi examined had reflux esophagitis. Reflux esophagitis was present in 28.2% (n=29) of the 103 baboons, and in 22.2% (n=4) of the 18 macaques. There was no difference between these two species.
In the 103 baboons, grade 1, 2 and 3 reflux esophagitis was present in 24.3% (n=25), 1.9% (n=2) and 1.9% (n=2) of the cases. In the 18 macaques, grade 1 reflux esophagitis was seen in 5.6% (n=1) and grade 2 reflux in 16.7% (n=3) of the cases. None of the macaques had grade 3 reflux esophagitis.
Discussion
This study showed that more than two thirds (67.8%) or 82 of the 121 esophagi examined in NHPs died of reasons other than gastrointestinal symptoms had either lymphocytic or reflux esophagitis. Lymphocytic esophagitis was much more frequent in baboons (43.7%) and in macaques (50.0%) than reflux esophagitis in the same animals (28.2% and 22.2%, respectively). Interestingly, whereas reflux esophagitis grade 1 was more common in baboons (24.3%) than in macaques (5.6%), reflux esophagitis grade 2 was more common in macaques (16.7%) than in baboons (1.9%). The cause for this difference remains unclear.
In some animals with lymphocytic gastritis, the ELs were found exclusively in the epithelium around the papillae (centred by a papillary vessel) and in others both in the epithelium around the papillae and in the epithelium between the papillary folds. After crossing through the papillary vessel, IELs apparently spread laterally within the squamous epithelium.
The number and distribution of IELs in lymphocytic esophagitis in NHPs (as well as in humans [8]) contrasts with the number and distribution of IELs in reflux esophagitis [5, 6]. In fact in reflux esophagitis the number of IELs is not only much lower than in lymphocytic esophagitis [8] but the number of IELs between the papillary folds surpass that seen in the squamous epithelium surrounding the papillae. These findings suggest that in reflux esophagitis, the bulk of the IELs might have traversed the basement membrane between the papillary folds of the squamous epithelium [8].
The main criteria for diagnosing lymphocytic esophagitis were the presence of a high number of IELs and the absence of granulocytes [8]. In contrast, in grade 2 and 3 reflux esophagitis, many intraepithelial granulocytes (neutrophils and/or eosinophils) were seen within the epithelium [3].
In lymphocytic esophagitis, I EL population was composed of mostly T cells, a subset of natural killer cells and of helper and inflammatory T cells. The antigen(s) responsible for the marked immunological reaction in the squamous epithelium of the esophagus in lymphocytic esophagitis remains unknown. It remains also unclear why lymphocytic esophagitis is so frequent in our NHPs, and less frequent in humans [8].
The findings in this study raise several interesting questions. Does the orthostatic (postural?) difference between humans and primates play a role in the development of lymphocytic esophagitis? Does the diet trigger or initiate the disease? Are environmental factors decisive for the occurrence of the disease? Does stress [9] of the animals kept in captivity contribute to the development of lymphocytic esophagitis in NHPs? One possibility to explore the effect of the environment in NHPs would be to assess the frequency of lymphocytic esophagitis at other facilities engaged in primate research. The use of different food regimens at other facilities would identify whether the diet has any bearing in the triggering of lymphocytic esophagitis.
In conclusion, this investigation showed that not only reflux esophagitis but particularly lymphocytic esophagitis occurs in NHPs. The findings substantiate the identity of lymphocytic esophagitis as a subset of chronic esophagitis in human. The antigenic agent(s) responsible for this marked immunological reaction in the squamous epithelium of the esophagus in non-human primates (and in humans) remains to be investigated.
Acknowledgments
Thanks are due to the staff of the Histology Laboratory and to Priscilla Williams, Data Management, Biostatistics and Scientific Computing, at the Southwest Foundation for Biomedical Research, San Antonio, Texas, for their invaluable help. This study was supported by a grant from the Karolinska Institute, Stockholm, Sweden.
References
- 1.McColl KE. When saliva meets acid: chemical warfare at the oesophagogastric junction. Gut. 2005;54:1–3. doi: 10.1136/gut.2004.047126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Nilsson M, Johnsen R, Ye W, Hveem K, Lagergren J. Prevalence of gastro-oesophageal reflux symptoms and the influence of age and sex. Scand J Gastroenterol. 2004;39:1040–1045. doi: 10.1080/00365520410003498. [DOI] [PubMed] [Google Scholar]
- 3.Rosaida MS, Goh KL. Gastro-oesophageal reflux disease, reflux oesophagitis and non-erosive reflux disease in a multiracial Asian population: a prospective, endoscopy based study. Eur J Gastroenterol Hepatol. 2004;16:495–501. doi: 10.1097/00042737-200405000-00010. [DOI] [PubMed] [Google Scholar]
- 4.Black DD, Haggitt RC, Orenstein SR, Whitington PF. Esophagitis in infants. Morphometric histological diagnosis and correlation with measures of gastroesophageal reflux. Gastroenterology. 1990;98:1408–1414. [PubMed] [Google Scholar]
- 5.Ismail-Beigi F, Pope CE. Distribution of the histological changes of gastroesophageal reflux in the distal esophagus of man. Gastroenterology. 1974;66:1109–1113. [PubMed] [Google Scholar]
- 6.Glover EJ, Leland MM, Dick EJ, Jr, Hubbard GB. Gastroesophageal reflux disease in baboons (Papio sp.): a new animal model. J Med Primatol. 2007 doi: 10.1111/j.1600-0684.2007.00217.x. (in press) [DOI] [PubMed] [Google Scholar]
- 7.Rubio CA, Hubbard GB. Hyperplastic foveolar gastropathy and hyperplastic foveolar gastritis in baboons. In Vivo. 1996;10:507–510. [PubMed] [Google Scholar]
- 8.Rubio CA, Sjodahl K, Lagergren J. Lymphocytic esophagitis: a histologic subset of chronic esophagitis. Am J Clin Pathol. 2006;125:432–437. [PubMed] [Google Scholar]
- 9.Rubio CA, Sveander M, Lagergren J. Re-adaptation of the esophageal mucosa of rats to protracted stress. In Vivo. 2001;15:413–416. [PubMed] [Google Scholar]