Abstract
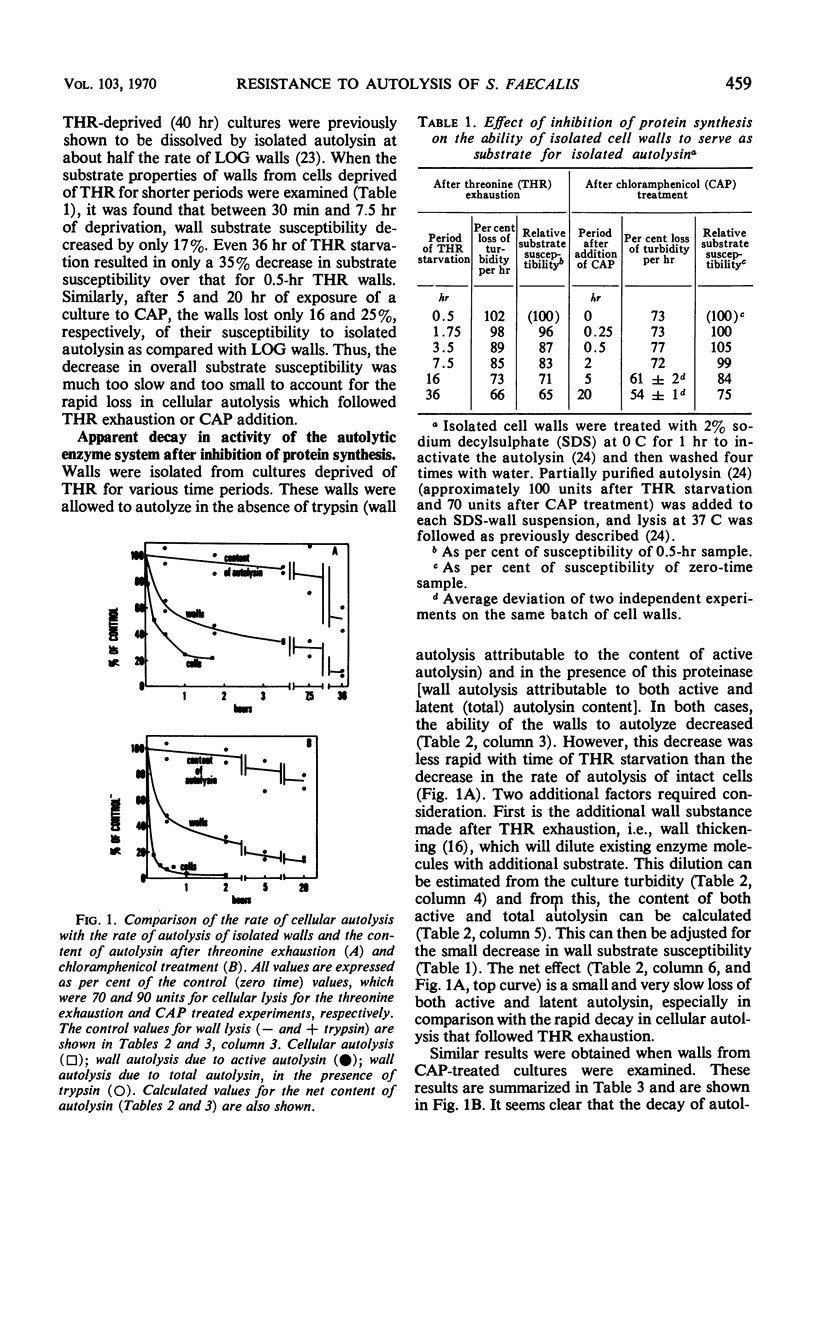
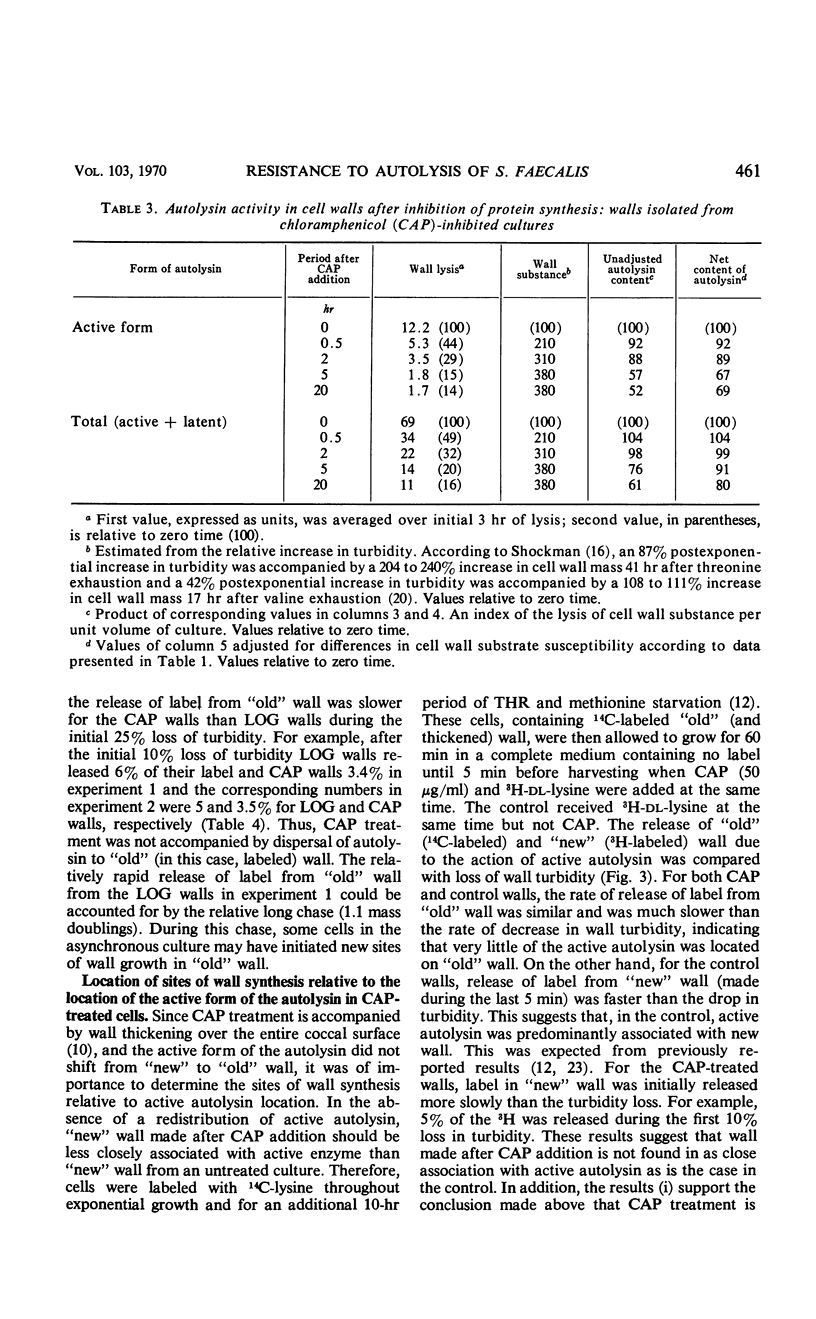
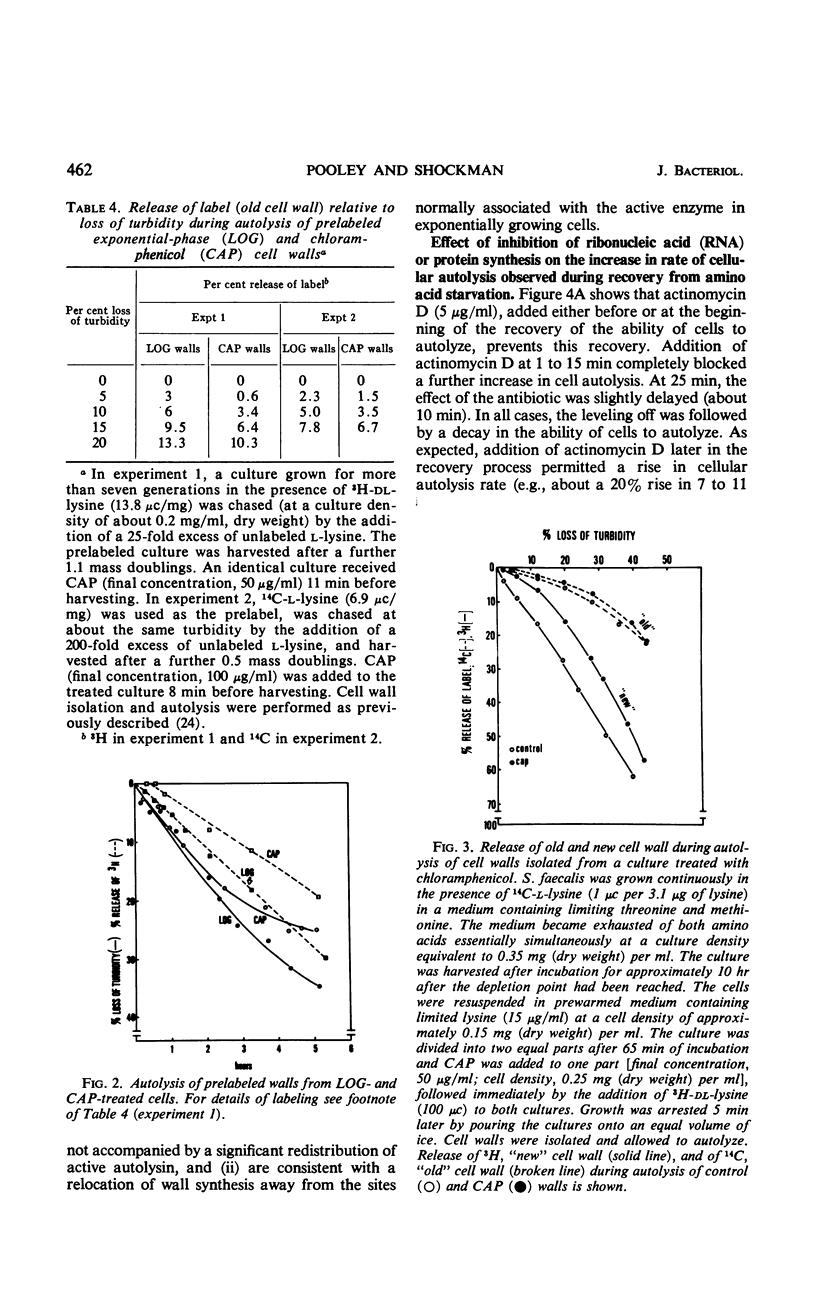
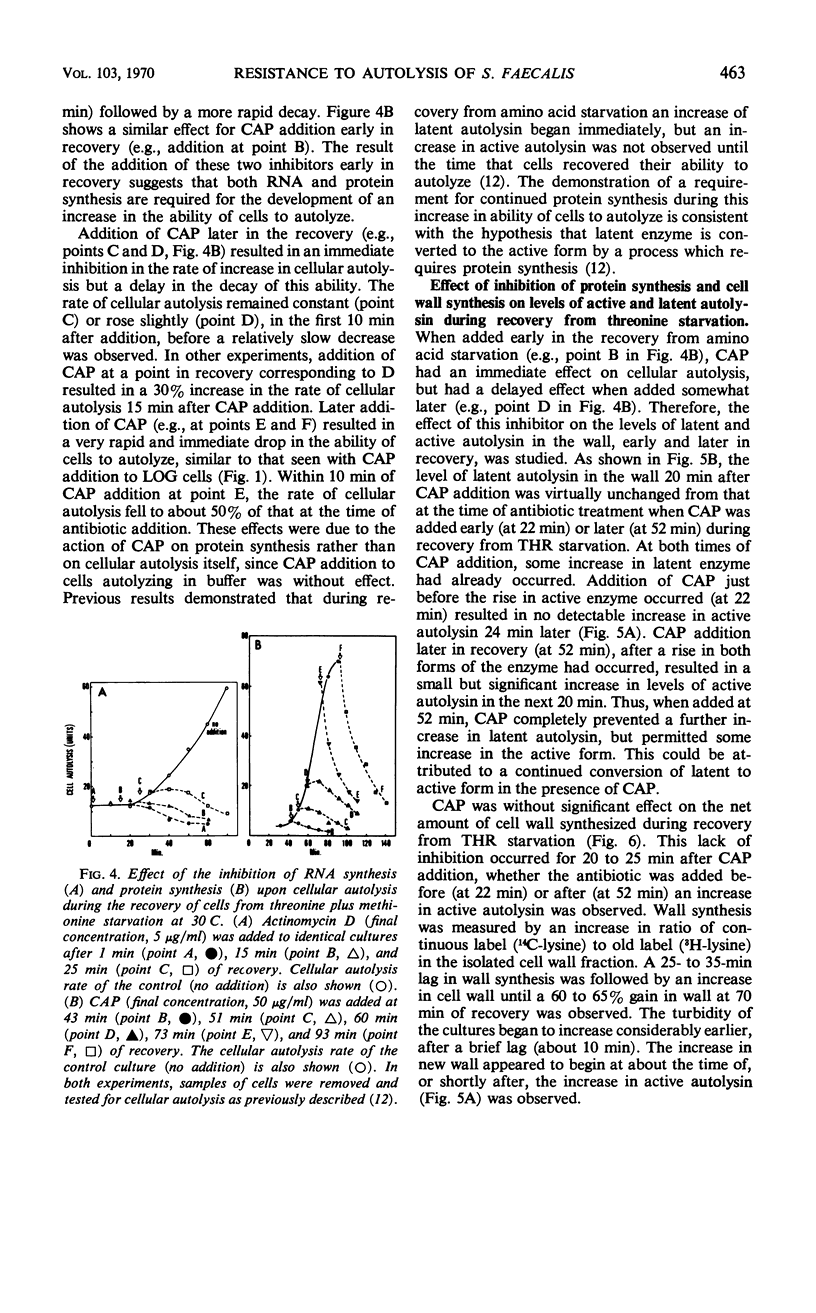
Ten minutes after inhibition of protein synthesis with chloramphenicol (CAP) the ability of cells of Streptococcus faecalis (ATCC 9790) to autolyze decreased to less than 20% of the rate for exponential-phase cells. After threonine exhaustion, the time for a 50% drop in the rate of cellular autolysis was about 20 min. These rapid increases in resistance to cellular autolysis could not be accounted for by: (i) the relatively slow and small overall decrease in susceptibility of isolated cell walls to added autolysin, or (ii) a decreased content of either the active or latent (proteinase activatable) form of the autolysin in the wall fraction. Continued wall synthesis resulted in dilution of preexisting autolysin in the isolated wall fraction. The release of labeled “old” relative to “new” wall from CAP-treated cultures showed that wall synthesis shifted away from the areas of wall previously shown to be associated with wall synthesis (extension) in exponential-phase cells. A corresponding dispersal of active autolysin activity was not observed. By using actinomycin D and CAP, a requirement for ribonucleic acid and protein synthesis early in the recovery of cells from amino acid starvation was demonstrated for the recovery in the ability of cells to autolyze. Evidence was obtained which suggests that a protein is involved in the conversion of latent to active autolysin. During recovery from amino acid starvation, increase in wall synthesis and content of active autolysin was delayed (25 to 35 min), whereas an increase in turbidity and latent enzyme content began within 10 min. After treatment with CAP at 22 or 52 min of recovery, a further increase in levels of both active and latent autolysin was severely inhibited; however, the increase in rate of wall synthesis was indistinguishable from that of an untreated control. This suggests that an increase in rate of wall synthesis does not depend on an increase in level of active autolysin.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- CHUNG K. L., HAWIRKO R. Z., ISAAC P. K. CELL WALL REPLICATION. I. CELL WALL GROWTH OF BACILLUS CEREUS AND BACILLUS MEGATERIUM. Can J Microbiol. 1964 Feb;10:43–48. doi: 10.1139/m64-007. [DOI] [PubMed] [Google Scholar]
- CHUNG K. L., HAWIRKO R. Z., ISAAC P. K. CELL WALL REPLICATION. II. CELL WALL GROWTH AND CROSS WALL FORMATION OF ESCHERICHIA COLI AND STREPTOCOCCUS FAECALIS. Can J Microbiol. 1964 Jun;10:473–482. doi: 10.1139/m64-057. [DOI] [PubMed] [Google Scholar]
- COLE R. M., HAHN J. J. Cell wall replication in Streptococcus pyogenes. Science. 1962 Mar 2;135(3505):722–724. doi: 10.1126/science.135.3505.722. [DOI] [PubMed] [Google Scholar]
- Chung K. L. Autoradiographic studies of bacterial cell wall replication. I. Cell wall growth of Bacillus cereus in the presence of chloramphenicol. Can J Microbiol. 1967 Apr;13(4):341–350. doi: 10.1139/m67-046. [DOI] [PubMed] [Google Scholar]
- Cole R. M. Symposium on the fine structure and replication of bacteria and their parts. 3. Bacterial cell-wall replication followed by immunofluorescence. Bacteriol Rev. 1965 Sep;29(3):326–344. doi: 10.1128/br.29.3.326-344.1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giesbrecht P., Ruska H. Uber Veränderungen der Feinstrukturen von Bakterien unter der Einwirkung von Chloramphenicol. Klin Wochenschr. 1968 Jun 1;46(11):575–582. doi: 10.1007/BF01747836. [DOI] [PubMed] [Google Scholar]
- Hash J. H., Davies M. C. Electron Microscopy of Staphylococcus aureus Treated with Tetracycline. Science. 1962 Nov 16;138(3542):828–829. doi: 10.1126/science.138.3542.828. [DOI] [PubMed] [Google Scholar]
- Higgins M. L., Pooley H. M., Shockman G. D. Site of initiation of cellular autolysis in Streptococcus faecalis as seen by electron microscopy. J Bacteriol. 1970 Aug;103(2):504–512. doi: 10.1128/jb.103.2.504-512.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Higgins M. L., Shockman G. D. Early changes in the ultrastructure of Streptococcus faecalis after amino acid starvation. J Bacteriol. 1970 Jul;103(1):244–253. doi: 10.1128/jb.103.1.244-253.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Higgins M. L., Shockman G. D. Model for cell wall growth of Streptococcus faecalis. J Bacteriol. 1970 Feb;101(2):643–648. doi: 10.1128/jb.101.2.643-648.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MITCHELL P., MOYLE J. Autolytic release and osmotic properties of protoplasts from Staphylococcus aureus. J Gen Microbiol. 1957 Feb;16(1):184–194. doi: 10.1099/00221287-16-1-184. [DOI] [PubMed] [Google Scholar]
- Pooley H. M., Shockman G. D. Relationship between the latent form and the active form of the autolytic enzyme of Streptococcus faecalis. J Bacteriol. 1969 Nov;100(2):617–624. doi: 10.1128/jb.100.2.617-624.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SHOCKMAN G. D. Bacterial cell wall synthesis: the effect of threonine depletion. J Biol Chem. 1959 Sep;234:2340–2342. [PubMed] [Google Scholar]
- SHOCKMAN G. D., KOLB J. J., TOENNIES G. Relations between bacterial cell wall synthesis, growth phase, and autolysis. J Biol Chem. 1958 Feb;230(2):961–977. [PubMed] [Google Scholar]
- SHOCKMAN G. D. Reversal of cycloserine inhibition by D-alanine. Proc Soc Exp Biol Med. 1959 Aug-Sep;101:693–695. doi: 10.3181/00379727-101-25064. [DOI] [PubMed] [Google Scholar]
- SUD I. J., SCHAECHTER M. DEPENDENCE OF THE CONTENT OF CELL ENVELOPES ON THE GROWTH RATE OF BACILLUS MEGATERIUM. J Bacteriol. 1964 Dec;88:1612–1617. doi: 10.1128/jb.88.6.1612-1617.1964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwarz U., Asmus A., Frank H. Autolytic enzymes and cell division of Escherichia coli. J Mol Biol. 1969 May 14;41(3):419–429. doi: 10.1016/0022-2836(69)90285-x. [DOI] [PubMed] [Google Scholar]
- Schwarz U., Leutgeb W. In vivo studies on murein synthesis. Folia Microbiol (Praha) 1967;12(3):279–282. doi: 10.1007/BF02868744. [DOI] [PubMed] [Google Scholar]
- Shockman G. D., Cheney M. C. Autolytic enzyme system of Streptococcus faecalis. V. Nature of the autolysin-cell wall complex and its relationship to properties of the autolytic enzyme of Streptococcus faecalis. J Bacteriol. 1969 Jun;98(3):1199–1207. doi: 10.1128/jb.98.3.1199-1207.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shockman G. D., Conover M. J., Kolb J. J., Phillips P. M., Riley L. S., Toennies G. LYSIS OF STREPTOCOCCUS FAECALIS. J Bacteriol. 1961 Jan;81(1):36–43. doi: 10.1128/jb.81.1.36-43.1961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shockman G. D., Pooley H. M., Thompson J. S. Autolytic enzyme system of Streptococcus faecalis. 3. Localization of the autolysin at the sites of cell wall synthesis. J Bacteriol. 1967 Nov;94(5):1525–1530. doi: 10.1128/jb.94.5.1525-1530.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shockman G. D. Symposium on the fine structure and replication of bacteria and their parts. IV. Unbalanced cell-wall synthesis: autolysis and cell-wall thickening. Bacteriol Rev. 1965 Sep;29(3):345–358. doi: 10.1128/br.29.3.345-358.1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shockman G. D., Thompson J. S., Conover M. J. The autolytic enzyme system of Streptococcus faecalis. II. Partial characterization of the autolysin and its substrate. Biochemistry. 1967 Apr;6(4):1054–1065. doi: 10.1021/bi00856a014. [DOI] [PubMed] [Google Scholar]
- Strominger J. L., Izaki K., Matsuhashi M., Tipper D. J. Peptidoglycan transpeptidase and D-alanine carboxypeptidase: penicillin-sensitive enzymatic reactions. Fed Proc. 1967 Jan-Feb;26(1):9–22. [PubMed] [Google Scholar]
- TOENNIES G., SHOCKMAN G. D. Quantitative amino acid assimilation in homofermentative metabolism. Arch Biochem Biophys. 1953 Aug;45(2):447–458. doi: 10.1016/s0003-9861(53)80021-4. [DOI] [PubMed] [Google Scholar]
- WEIDEL W., FRANK H., MARTIN H. H. The rigid layer of the cell wall of Escherichia coli strain B. J Gen Microbiol. 1960 Feb;22:158–166. doi: 10.1099/00221287-22-1-158. [DOI] [PubMed] [Google Scholar]
- WEIDEL W., PELZER H. BAGSHAPED MACROMOLECULES--A NEW OUTLOOK ON BACTERIAL CELL WALLS. Adv Enzymol Relat Areas Mol Biol. 1964;26:193–232. doi: 10.1002/9780470122716.ch5. [DOI] [PubMed] [Google Scholar]



