Abstract
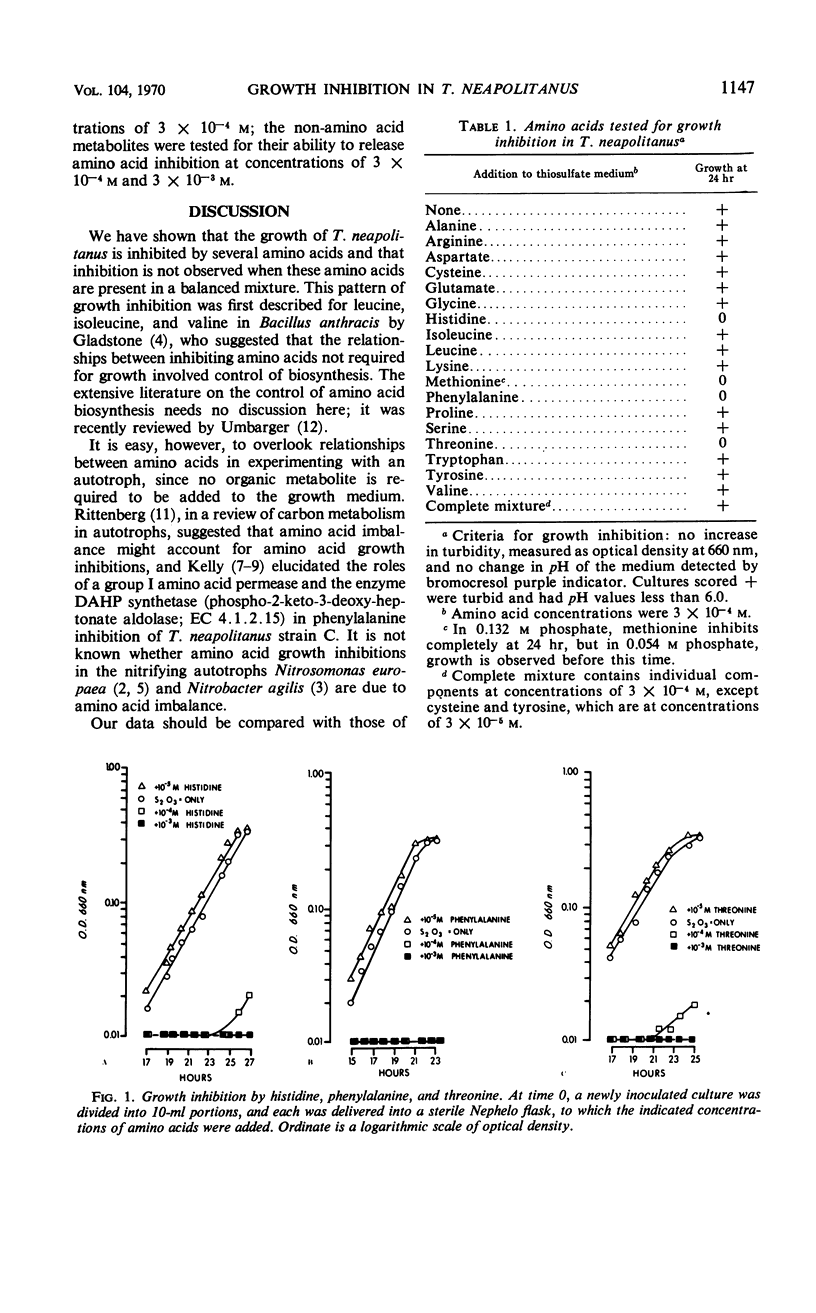
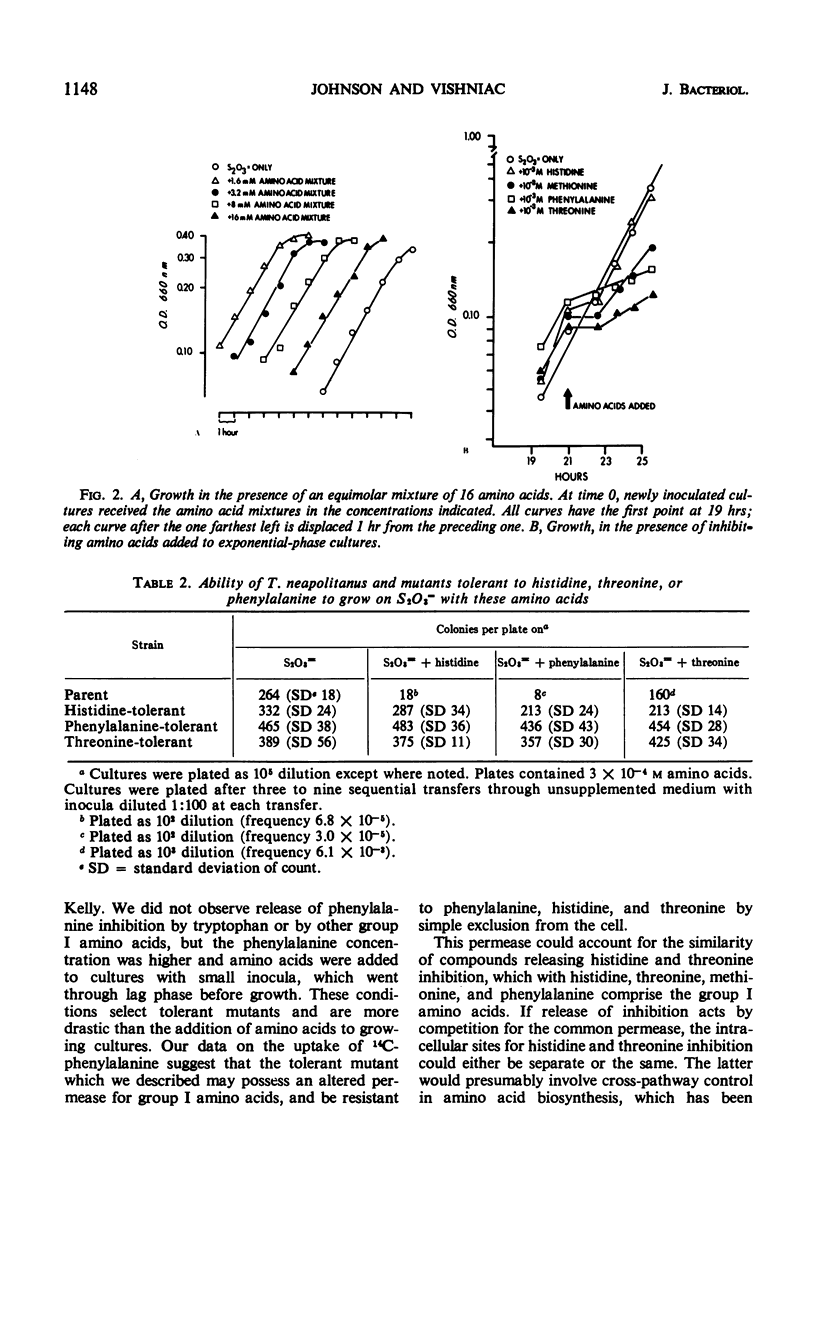
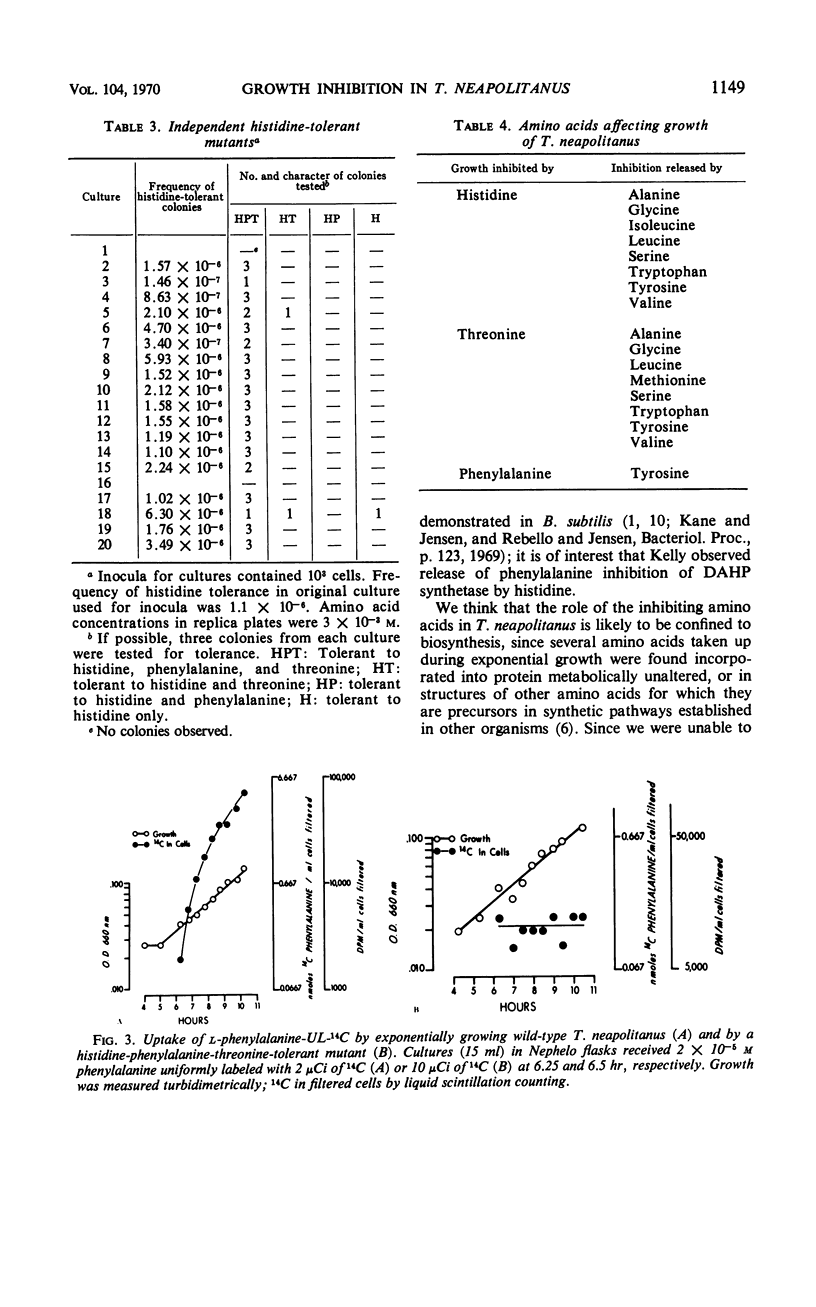
Thiobacillus neapolitanus, a strict chemoautotroph, is sensitive to the addition of 10−4m methionine, histidine, threonine, or phenylalanine to the thiosulfate medium on which it grows. When histidine, threonine, or phenylalanine are added at the time of inoculation, spontaneous mutants tolerant to the three amino acids are selected. These mutants appear to result from a single genetic change; of 18 independently isolated histidine-tolerant mutants, all are also tolerant to phenylalanine and threonine. The uptake of 14C-phenylalanine into exponentially growing cells of one such mutant is negligible in contrast with the uptake observed in the phenylalanine-sensitive parent. The addition of methionine to the medium slows growth, but spontaneous mutants are not selected. Inhibition of growth by these amino acids is observed only under conditions of amino acid imbalance; the addition of an equimolar mixture of 16 amino acids, in which each component is present at a concentration of 10−3m, causes no inhibition. Histidine and threonine inhibition may be released by equimolar amounts of any one of seven amino acids: serine, alanine, glycine, leucine, valine, tryptophan, or tyrosine; histidine inhibition is also released by isoleucine, and threonine inhibition by methionine. None of the inhibiting amino acids inhibits oxidation of thiosulfate in cell suspensions. A group of hexoses, pentoses, and Krebs cycle intermediates were tested for inhibition of growth or release of inhibition by histidine, phenylalanine, or threonine, but no effects, either inhibition or relief of inhibition, were found.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Chapman L. F., Nester E. W. Common element in the repression control of enzymes of histidine and aromatic amino acid biosynthesis in Bacillus subtilus. J Bacteriol. 1968 Nov;96(5):1658–1663. doi: 10.1128/jb.96.5.1658-1663.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark C., Schmidt E. L. Growth response of Nitrosomonas europaea to amino acids. J Bacteriol. 1967 Apr;93(4):1302–1308. doi: 10.1128/jb.93.4.1302-1308.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Delwiche C. C., Finstein M. S. Carbon and Energy Sources for the Nitrifying Autotroph Nitrobacter. J Bacteriol. 1965 Jul;90(1):102–107. doi: 10.1128/jb.90.1.102-107.1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kelly D. P. Influence of amino acids and organic antimetabolites on growth and biosynthesis of the chemoautotroph Thiobacillus neapolitanus strain C. Arch Mikrobiol. 1967 Feb 20;56(2):91–105. doi: 10.1007/BF00408761. [DOI] [PubMed] [Google Scholar]
- Kelly D. P. Regulation of chemoautotrophic metabolism. 3. DAHP synthetase in Thiobacillus neapolitanus. Arch Mikrobiol. 1969;69(4):360–369. doi: 10.1007/BF00408576. [DOI] [PubMed] [Google Scholar]
- Kelly D. P. Regulation of chemoautotrophic metabolism. I. Toxicity of phenylalanine to thiobacilli. Arch Mikrobiol. 1969;69(4):330–342. doi: 10.1007/BF00408574. [DOI] [PubMed] [Google Scholar]
- Kelly D. P. Regulation of chemoautotrophic metabolism. II. Competition between amino acids for incorporation into Thiobacillus. Arch Mikrobiol. 1969;69(4):343–359. [PubMed] [Google Scholar]
- Nester E. W. Cross pathway regulation: effect of histidine on the synthesis and activity of enzymes of aromatic acid biosynthesis in Bacillus subtilis. J Bacteriol. 1968 Nov;96(5):1649–1657. doi: 10.1128/jb.96.5.1649-1657.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Umbarger H. E. Regulation of amino acid metabolism. Annu Rev Biochem. 1969;38:323–370. doi: 10.1146/annurev.bi.38.070169.001543. [DOI] [PubMed] [Google Scholar]
- VISHNIAC W., SANTER M. The thiobacilli. Bacteriol Rev. 1957 Sep;21(3):195–213. doi: 10.1128/br.21.3.195-213.1957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- VISHNIAC W., TRUDINGER P. A. Symposium on autotrophy. V. Carbon dioxide fixation and substrate oxidation in the chemosynthetic sulfur and hydrogen bacteria. Bacteriol Rev. 1962 Jun;26:168–175. [PMC free article] [PubMed] [Google Scholar]



