Abstract
Background Recent tobacco smoking has been identified as a risk factor for developing tuberculosis, and two studies which have investigated its association with relapse of tuberculosis after completion of treatment had conflicting results (and did not control for confounding). The objective of this study was to investigate risk factors for tuberculosis relapse, with emphasis on smoking.
Methods A cohort of newly diagnosed TB cases was followed up from their discharge after completion of treatment (in 2001–2003) until October 2006 and relapses of tuberculosis ascertained during that period. A case of relapse was defined as a patient who started a second treatment during the follow up.
Results Smoking (OR 2.53, 95% CI 1.23–5.21) and living in an area where the family health program was not implemented (OR 3.61, 95% CI 1.46–8.93) were found to be independently associated with relapse of tuberculosis.
Conclusions Our results establish that smoking is associated with relapse of tuberculosis even after adjustment for the socioeconomic variables. Smoking cessation support should be incorporated in the strategies to improve effectiveness of Tuberculosis Control Programs.
Keywords: Tuberculosis control, successful treatment, relapse, risk factors, smoking
Introduction
Tuberculosis is a significant health problem worldwide; control of tuberculosis relies heavily on identification and successful treatment of cases. Treatment schedules last for 6 months. One of the problems of tuberculosis control is the fact that cases that were successfully treated can relapse, needing a second treatment. Identification of factors that increase the risk of relapse of tuberculosis after cure or completion of treatment is of great interest. The monitoring of the effectiveness of tuberculosis programs is based on the routine classification of the outcome of each tuberculosis treatment into: (i) treatment failure (patient still have positive smear 5 months after start of treatment or later); (ii) death (patient who dies for any reason during the course of treatment); (iii) default (patient interrupts treatment for two consecutive months or more); (iii) cure (patient who is sputum smear negative in two consecutive tests, at least one in the last months of treatment); (iv) treatment completed (a patient who has completed treatment, has no symptoms, but does not meet the criteria for cure because of the absence of a sputum test in the last month of treatment, usually because a sputum test was not conducted or the patient could not produce sputum). Patients classified as cure or completed treatment are considered treatment successes.1
In Recife, Brazil, where this study is conducted, 16% of the people starting treatment against tuberculosis had had at least one previous treatment. Of this, one-third had cured or completed treatment and therefore was a relapse.2 This is typical of many developing countries. So far, factors known to increase the risk of relapse are restricted to deficiencies in treatment (taking the drugs irregularly, taking the wrong drugs, taking the right drugs in the wrong dosage); and (probably as indicators of deficiencies in treatment) patients missing consultations, having adverse events to the drugs used in the treatment of tuberculosis and health services problems like irregular supply of drugs.3 Most exciting, and of potential relevant to relapse, recently tobacco smoking has been identified as a risk factor for developing tuberculosis. The proposed biological mechanism for this is a decreased resistance to Mycobacterium tuberculosis due to functional and morphological changes to macrophages in the alveoli of smokers.4,5 It is therefore plausible that smoking may increase the risk of relapse, by increasing risk of persistence of M. tuberculosis infection after treatment and risk of any residual M. tuberculosis infection leading to disease.5 Two studies have investigated this possibility and had conflicting results.3,6 None controlled for confounding.
This article reports on a study of risk factors for relapse, based on the analysis of cases of relapse in a cohort of cases of tuberculosis that was followed up from start of tuberculosis treatment to expected date for completion of treatment, for a previous study, and then followed up for this analysis for 3–5 years after completion of treatment.
Methods
Study population and design
Site of the study was Recife, a city with a population of one and half million people in the northeast of Brazil. In 2002, the incidence of tuberculosis in Recife was 104 per 100 000 inhabitants7 and treatment consisted of an initial phase of isoniazid, rifampicin and pyrazinamide lasting for 2 months, followed by a second phase with isoniazid and rifampicin only for a period of 4 months.8 This is different from the internationally recommended first-line treatment which uses four drugs in the initial phase, 2 months of ethambutol associated to isoniazid, rifampicin and pyrazinamide.9 The objective of the analysis reported here was to identify risk factors for tuberculosis relapse leading to re-treatment in those who completed their tuberculosis treatment successfully and survived the follow-up period. Methods of the main study (following cases from diagnosis to completion of treatment) were described elsewhere but in short cases of tuberculosis diagnosed from May 2001 to July 2003 who were residents in Recife and aged 13 years or older were recruited into the study, completed a questionnaire on potential risk factors and had blood collected for anti-HIV serology. Immediate outcome of treatment was ascertained from the register of the health unit where each patient was treated and from each patient's medical record, soon after the expected date of end of treatment and again 6 months later, and classified into failure, default, death, confirmed cure or completion of treatment. Results of the initial study of determinants of the outcome of tuberculosis treatment were published elsewhere.10,11 In October 2006, additional information was sought to identify cases from the cohort who relapsed (started re-treatment for tuberculosis after successful completion of treatment); information on patients who died was sought for exclusion from the cohort and for sensitivity analysis.
The study population in this analysis consisted of patients from the original cohort who were discharged from the first treatment after treatment success.1 Exclusion criteria were: not having completed treatment, failure of treatment/having a positive smear at the end of treatment (as these would remain in treatment with a different scheme); having died and having had a previous treatment for tuberculosis (as these were already relapses when recruited into the first study, of determinants of outcome of treatment).
We did not examine separately those with negative sputum at the end of treatment and those with no sputum (cures vs competed treatment) as numbers were not large enough for subgroup analysis; we did not expect the two groups to be different because the main reason for not having a sputum test late in the course of treatment was that the test was not requested (data not shown). A case of relapse was defined as a patient, from the original cohort discharged because of cure or completion of treatment, who was diagnosed with tuberculosis and started a second course of treatment during the follow up period. Patients from the original cohort who survived to the end of the follow up period and did not start a new treatment were the comparison group.
We identified relapses by searching the Surveillance System for Infectious Diseases (SINAN/MS). SINAN/MS is an electronic system with notifications of infectious diseases managed by the Ministry of Health (MS) in Brazil. The search was done using name and date of birth. In Brazil, tuberculosis treatment is carried out only by the public sector and to receive tuberculosis treatment a patient must be notified, so the notification system should include all diagnosed cases receiving treatment. We identified deaths for exclusion and sensitivity analysis by searching the routine mortality system (SIM/MS), an electronic system with all routine mortality data with cause of death coded to ICD managed by the Ministry of Health in Brazil. SIM/MS was searched based on name and date of birth; and the cause of death was abstracted for all deaths linked to the cohort.
Exposure information had been collected as part of the previous study (follow up from first diagnosis of tuberculosis to discharge) from a standard pre-coded questionnaire in an interview conducted face to face by a trained interviewer and information abstracted from the tuberculosis treatment record and included:
Clinical and epidemiological factors: treatment delay (defined as delay between onset of symptoms and start of treatment (the cut-off point adopted was according to Santos et al.10); Clinical form of tuberculosis (pulmonary or extra pulmonary) and HIV co-infection; history of contact with other cases of tuberculosis: knowing somebody with TB; and living in the same house as a TB patient.
Biological factors: age and sex.
Social factors: employment status of patient and of the head of the household; literacy; income and schooling of the head of the household; access to water supply; ownership of goods (radio, refrigerator, video, washing machine, microwave oven, computer, TV set, air conditioning and a car); number of goods (the number of goods was grouped as follows: first, possession of each of the different goods was counted as 1, independent of the number of each good possessed (for example, owning two cars counted as owning one good); then, the number of goods possessed was summed and grouped into four categories: 0–1; 2–3; 4–6 and 7 or more goods.
Lifestyle: Alcohol consumption, grouped as: does not drink, drinks rarely, drinks at least once a week, drinks every day and once starts drinking finds it difficult to stop. They were aggregated during analysis in two groups: light drinker (none, drinking rarely, weekly) and heavy drinker (drinking every day or finding it difficult to stop once start drinking). Smoking, grouped into whether the person was a smoker at the time of the interview (smokers), had never smoked (never smokers), or used to smoke but stopped, and the date when stopped (ex smokers). For the analysis, because of small numbers and because the hypothesis was that the effect of smoking disappears with time, those who never smoked were grouped with those that had given up smoking one year or more before the interview).
Access to health services: number of health units attended with the complaint eventually diagnosed as tuberculosis before it was diagnosed and treatment started, whether unit attended was in the same neighborhood and in the district of residence of case and whether the Family Health Program (FHP) was implemented in the Health District of residence of case at the time of the start of the treatment. The FHP is a new program in which a team of trained health visitors visit families in the community. Each team is responsible for approximately 3200 residents in a defined geographical area and is linked to a FHP team of clinicians and other health professionals in a supporting health unit. The FHP was being implemented during the study period and some but not all of the study areas were covered.
Statistical analysis
Logistic regression was used to identify risk factors for relapse. Time since end of treatment and ascertainment of re-treatment in October 2006 varied, a variable indicating the year of the end of the first treatment was included in all models. Variables associated with the outcome with a P-value ≤0.25 or with an odds ratio of ≥1.5 were included in a multiple logistic regression analysis which also included the year of entry in the cohort. A step-up procedure was used. Crude and adjusted ORs and 95% CIs were calculated. Since smoking is associated with poverty and alcohol consumption, to increase the confidence that the association between re-treatment and smoking was not a result of confounding by socioeconomic status, additional unplanned analysis were undertaken exploring the effect of each of a large number of social variables collected as part of the first study on the relationship between smoking and relapse: employment status of patient and of the head of the household; literacy; income and schooling of the head of the household; access to water supply; ownership of each of the following goods—radio, refrigerator, video, washing machine, microwave oven, computer, television set, air conditioning, car; number of goods owned; and alcohol consumption. To explore the potential consequences of excluding deaths a sensitivity analysis was conducted modelling the consequences of treating as relapses (i) all deaths coded to tuberculosis and (ii) all deaths. Data were stored by a software program (EPI-INFO version 6.04; Centers for Disease Control, Atlanta, GA) and analysed using STATA 9.0 (Stata-Corp LP, College Station, TX). The study was approved by the Ethical Committee of the Center of Health Sciences of the Federal University of Pernambuco. All patients signed an informed consent form.
Results
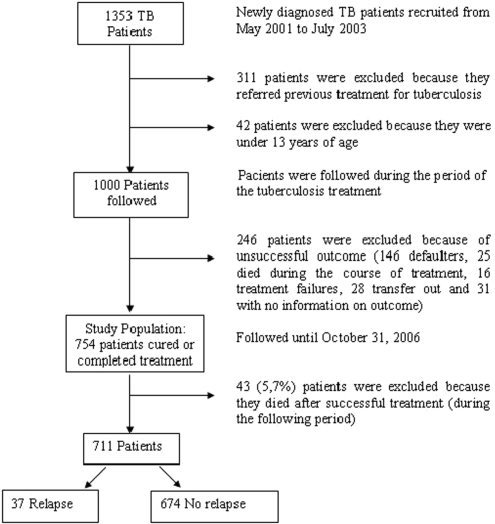
A total of 1353 diagnosed TB patients had been identified for potential selection from the previous study from May 2001 to July 2003. Of these, 311 patients were not eligible for the first study because they referred previous treatment for tuberculosis (and so were already relapses) and 42 patients because they were under 13 years of age. Thus, 1000 patients of potential interest for this analysis were followed during the period of the tuberculosis treatment. From this group, 246 patients were not eligible for this analysis because they had an unsuccessful outcome (146 defaulters, 25 died during the course of treatment, 16 were treatment failures), 28 were excluded because they transferred out to a different tuberculosis treatment unit and 31 because they had no information on outcome of treatment. The study population therefore consisted of 754 patients with successful treatment outcomes (cured or completed treatment), and they were followed until 31 October 2006. During the follow-up period, 43 patients were excluded because they died after completion of treatment; therefore the final group consisted of 711 patients, of which 37 relapsed and 674 had not relapsed by the end of follow up (Figure 1).
Figure 1.
Study flow diagram
The mean age of cases in the analysis was 38.7 in males and 35.6 in females. In this study the rate of relapse was 5% (37/711 patients). Approximately half (48.7%) of the cases of relapse occurred in the first year after discharge.
Tables 1–4 show the results of univariable analysis of the association between relapse and risk factors, adjusted by the year of entry in the cohort. Among biological and lifestyle factors (Table 1), only smoking was associated with relapse (OR 2.34, 95% CI 1.17–4.68). None of the socioeconomic factors was associated with relapse (Table 2). Among the variables related to health services, only not living in an area that receives visits by the FHP was associated with relapse (OR 3.25, 95% CI 1.33–7.94) (Table 3). The only clinical or epidemiological factor associated with relapse was having had a delay of 60 or more days before start of the first treatment of tuberculosis and even this was borderline (OR 2.23, 95% CI 0.99–4.98) (Table 4).
Table 1.
Association between relapse of tuberculosis and biologic and lifestyle characteristics of individuals, OR—crude and adjusted by the year of entry in the cohort, Recife 2006
| Biological factors and lifestyle | Relapse |
Control |
||||||
|---|---|---|---|---|---|---|---|---|
| N | % | N | % | Crude OR (CI) | P | OR (CI) Adjusted | P | |
| Age (years) | 0.154 | 0.401 | ||||||
| 13–29 | 10 | 4.13 | 232 | 95.87 | 1.00 | – | 1.00 | – |
| 30–49 | 22 | 6.96 | 294 | 93.04 | 1.73 (0.81–3.74) | 0.159 | 1.72 (0.80–3.72) | 0.164 |
| ≥50 | 5 | 3.27 | 148 | 96.73 | 0.78 (0.26–2.34) | 0.662 | 0.79 (0.26–2.35) | 0.668 |
| Total | 37 | – | 674 | – | ||||
| Sex | ||||||||
| Female | 14 | 5.56 | 238 | 94.44 | 1.00 | – | 1.00 | – |
| Male | 23 | 5.01 | 436 | 94.99 | 0.89 (0.45–1.77) | 0.755 | 0.88 (0.44–1.76) | 0.732 |
| Total | 37 | – | 674 | – | ||||
| Alcohol consumption | ||||||||
| None or light drinker | 32 | 5.32 | 569 | 94.68 | 1.00 | – | 1.00 | – |
| Heavy drinker | 3 | 4.05 | 71 | 95.95 | 0.75 (0.22–2.52) | 0.643 | 0.76 (0.22–2.54) | 0.653 |
| Total | 35 | – | 640 | – | ||||
| Cigarette smoking | ||||||||
| Never smoking or given up smoking | 23 | 4.16 | 530 | 95.84 | 1.00 | – | 1.00 | – |
| Ever smoking | 14 | 9.21 | 138 | 90.79 | 2.33 (1.17–4.66) | 0.016 | 2.34 (1.17–4.68) | 0.016 |
| Total | 37 | – | 668 | – | ||||
Table 2.
Association between relapse of tuberculosis and socioeconomic characteristics of individuals, OR—crude and adjusted by the year of entry in the cohort, Recife 2006
| Relapse |
Control |
|||||||
|---|---|---|---|---|---|---|---|---|
| Social factors | N | % | N | % | Crude OR (CI) | P | OR (CI) Adjusted | P |
| Literacy | ||||||||
| Yes | 33 | 5.37 | 582 | 94.63 | 1.00 | – | 1.00 | – |
| No | 4 | 4.17 | 92 | 95.83 | 0.76 (0.26–2.21) | 0.624 | 0.75 (0.25–2.17) | 0.596 |
| Total | 37 | – | 674 | – | ||||
| Employment | ||||||||
| Yes | 12 | 5.45 | 208 | 94.55 | 1.00 | – | 1.00 | – |
| No | 25 | 5.11 | 464 | 94.89 | 0.93 (0.46–1.89) | 0.850 | 0.93 (0.45–1.89) | 0.844 |
| Total | 37 | – | 672 | – | ||||
| Number of individuals per household | ||||||||
| 1–4 | 24 | 5.90 | 383 | 94.10 | 1.00 | – | 1.00 | – |
| ≥5 | 13 | 4.44 | 280 | 95.56 | 0.74 (0.37–1.48) | 0.396 | 0.73 (0.37–1.47) | 0.391 |
| Total | 37 | – | 663 | – | ||||
| Income of head of household (MW) | ||||||||
| ≥2 | 5 | 3.60 | 134 | 96.40 | 1.00 | – | 1.00 | – |
| <2 | 31 | 6.14 | 474 | 93.86 | 1.75 (0.67–4.59) | 0.254 | 1.75 (0.67–4.61) | 0.253 |
| Total | 36 | – | 608 | – | ||||
| Employment of head of household | ||||||||
| Yes | 19 | 5.21 | 346 | 94.79 | 1.00 | – | 1.00 | – |
| No | 18 | 5.20 | 328 | 94.80 | 0.99 (0.51–1.93) | 0.998 | 0.99 (0.51–1.92) | 0.986 |
| Total | 37 | – | 674 | – | ||||
| Schooling of the head of household | ||||||||
| 9 or more years | 3 | 2.78 | 105 | 97.22 | 1.00 | – | 1.00 | – |
| Up to 8 years | 31 | 5.58 | 525 | 94.42 | 2.06 (0.62–6.88) | 0.237 | 2.06 (0.61–6.89) | 0.239 |
| Total | 34 | – | 630 | – | ||||
| Piped water | ||||||||
| Yes | 35 | 5.29 | 627 | 94.71 | 1.00 | – | 1.00 | – |
| No | 2 | 4.44 | 43 | 95.56 | 0.83 (0.19–3.58) | 0.806 | 0.85 (0.20–3.69) | 0.835 |
| Total | 37 | – | 670 | – | ||||
| Ownership of washing machine | ||||||||
| Yes | 7 | 4.86 | 137 | 95.14 | 1.00 | – | 1.00 | – |
| No | 29 | 5.17 | 532 | 94.83 | 1.06 (0.45–2.48) | 0.881 | 1.06 (0.45–2.47) | 0.890 |
| Total | 36 | – | 669 | – | ||||
| Ownership of microwave oven | ||||||||
| Yes | 3 | 3.95 | 73 | 96.05 | 1.00 | – | 1.00 | – |
| No | 33 | 5.29 | 591 | 94.71 | 1.36 (0.41–4.54) | 0.619 | 1.35 (0.40–4.53) | 0.627 |
| Total | 36 | – | 664 | – | ||||
| Ownership of computer | ||||||||
| Yes | 2 | 3.03 | 64 | 96.97 | 1.00 | – | 1.00 | – |
| No | 35 | 5.52 | 599 | 94.48 | 1.87 (0.44–7.95) | 0.397 | 1.86 (0.43–7.94) | 0.399 |
| Total | 37 | – | 663 | – | ||||
| Ownership of a car | ||||||||
| Yes | 8 | 5.88 | 128 | 94.12 | 1.00 | – | 1.00 | – |
| No | 29 | 5.07 | 543 | 94.93 | 0.85 (0.38–1.91) | 0.702 | 0.84 (0.37–1.89) | 0.685 |
| Total | 37 | – | 671 | – | ||||
| Ownership of air conditioning | ||||||||
| Yes | 2 | 2.44 | 80 | 97.56 | 1.00 | – | 1.00 | – |
| No | 35 | 5.58 | 592 | 94.42 | 2.36 (0.56–10.0) | 0.243 | 2.32 (0.54–9.85) | 0.255 |
| Total | 37 | – | 672 | – | ||||
| Number of goods possessed | 0.312 | 0.592 | ||||||
| 7–9 goods | 3 | 4.69 | 61 | – | 1.00 | – | 1.00 | |
| 4–6 goods | 8 | 3.77 | 204 | 0.744 | 0.80 (0.20–3.10) | 0.750 | 0.80 (0.20–3.12) | 0.750 |
| 2–3 goods | 19 | 5.23 | 344 | 0.855 | 1.12 (0.32–3.91) | 0.858 | 1.12 (0.32–3.92) | 0.858 |
| 0–1 goods | 6 | 10.53 | 51 | 0.234 | 2.39 (0.57–10.04) | 0.243 | 2.36 (0.56–9.97) | 0.243 |
| Total | 36 | – | 660 | – | ||||
Table 3.
Association between relapse of tuberculosis and health services variables, OR—crude and adjusted by the year of entry in the cohort, Recife 2006
| Relapse |
Control |
|||||||
|---|---|---|---|---|---|---|---|---|
| Access to health services factors | N | % | N | % | Crude OR (CI) | P | OR (CI) Adjusted | P |
| Number of health units | ||||||||
| Only 1 | 5 | 3.73 | 129 | 96.27 | 1.00 | – | 1.00 | – |
| ≥2 | 31 | 5.50 | 533 | 94.50 | 1.50 (0.57–3.93) | 0.409 | 1.47 (0.56–3.88) | 0.428 |
| Total | 36 | – | 662 | – | ||||
| HS in same district of residence | ||||||||
| Yes | 23 | 5.18 | 421 | 94.82 | 1.00 | – | 1.00 | – |
| No | 14 | 5.24 | 253 | 94.76 | 1.01 (0.51–2.00) | 0.971 | 1.02 (0.52–2.04) | 0.935 |
| Total | 37 | – | 674 | – | ||||
| HS in neighborhood of residence | ||||||||
| Yes | 7 | 7.78 | 83 | 92.22 | 1.00 | – | 1.00 | – |
| No | 30 | 4.83 | 591 | 95.17 | 0.60 (0.25–1.41) | 0.244 | 0.60 (0.25–1.41) | 0.244 |
| Total | 37 | – | 674 | – | ||||
| Residence in areas of FHP visits | ||||||||
| Yes | 6 | 2.32 | 253 | 97.68 | 1.00 | – | 1.00 | – |
| No | 30 | 6.99 | 399 | 93.01 | 3.17 (1.30–7.72) | 0.011 | 3.25 (1.33–7.94) | 0.010 |
| Total | 36 | – | 652 | – | ||||
| Health District of treatment | 0.412 | 0,615 | ||||||
| HD IV | 6 | 3.51 | 165 | 96.49 | 1.00 | – | 1.00 | – |
| HD I | 0 | 0 | 59 | 100.0 | – | – | – | – |
| HD II | 7 | 4.55 | 147 | 95.45 | 1.31 (0.43–3.98) | 0.635 | 1.35 (0.42–4.29) | 0.609 |
| HD III | 11 | 8.21 | 123 | 91.79 | 2.46 (0.88–6.83) | 0.084 | 2.43 (0.87–6.82) | 0.091 |
| HD V | 11 | 6.81 | 149 | 93.13 | 2.03 (0.73–5.62) | 0.173 | 2.08 (0.74–5.77) | 0.160 |
| HD VI | 2 | 6.06 | 31 | 93.94 | 1.77 (0.34–9.19) | 0.495 | 1.86 (0.35–9.79) | 0.461 |
| Total | 37 | – | 674 | – | ||||
Table 4.
Association between relapse of tuberculosis and clinical and epidemiological characteristics of individuals, OR—crude and adjusted by the year of entry in the cohort, Recife 2006
| Relapse |
Control |
|||||||
|---|---|---|---|---|---|---|---|---|
| Clinical and epidemiological factors | N | % | N | % | Crude OR (CI) | P | OR (CI) Adjusted | P |
| Knows someone with tuberculosis | ||||||||
| No | 23 | 5.64 | 385 | 94.36 | 1.00 | – | 1.00 | – |
| Yes | 14 | 4.75 | 281 | 95.25 | 0.83 (0.42–1.64) | 0.602 | 0.83 (0.42–1.65) | 0.606 |
| Total | 37 | – | 666 | – | ||||
| Household contact with a case of tuberculosis | ||||||||
| No | 32 | 5.22 | 581 | 94.78 | 1.00 | – | 1.00 | – |
| Yes | 05 | 5.10 | 93 | 94.90 | 0.97 (0.37–2.57) | 0.961 | 0.98 (0.37–2.59) | 0.974 |
| Total | 37 | – | 674 | – | ||||
| Clinical form of tuberculosis | ||||||||
| Pulmonary | 31 | 5.12 | 574 | 94.88 | 1.00 | – | 1.00 | – |
| Extra pulmonary | 05 | 4.81 | 99 | 95.19 | 0.93 (0.35–2.46) | 0.892 | 0.93 (0.35–2.45) | 0.889 |
| Total | 36 | – | 673 | – | ||||
| Treatment delay | ||||||||
| ≤60 days | 8 | 3.01 | 258 | 96.99 | 1.00 | – | 1.00 | – |
| >60 days | 28 | 6.57 | 398 | 93.43 | 2.27 (1.01–5.05) | 0.045 | 2.23 (0.99–4.98) | 0.050 |
| Total | 36 | – | 656 | – | ||||
| HIV co-infection | 0.962 | 0.972 | ||||||
| No | 1 | 7.14 | 13 | 92.86 | 1.00 | – | 1.00 | – |
| Yes | 12 | 5.38 | 211 | 94.62 | 0.74 (0.09–6.13) | 0.780 | 0.76 (0.09–6.40) | 0.807 |
| Not known | 24 | 5.37 | 423 | 94.63 | 0.73 (0.09–5.87) | 0.774 | 0.76 (0.09–6.15) | 0.804 |
| Total | 37 | – | 647 | – | ||||
Table 5 shows the results of the multivariable analysis. The following variable remained in the final model: smoking (OR 2.53; 95% CI 1.23–5.21) and living in an area in which the FHP was not implemented (OR 3.61; 95% CI 1.46–8.93).
Table 5.
Multivariate analysis of the association between relapse of tuberculosis and characteristics of individuals, OR—crude and adjusted for each other and by the year of entry in the cohort, Recife 2006
| OR and 95% CI, adjusted by year of entry in the cohort | P | OR and 95% CI, adjusted by year of entry in the cohort, and by each other | P | paf | |
|---|---|---|---|---|---|
| Cigarette smoking | |||||
| Never smoking or given up smoking | 1.00 | – | 1.00 | – | |
| Ever smoking | 2.34 (1.17–4.68) | 0.016 | 2.53 (1.23–5.21) | 0.011 | 0.228 |
| Residence in areas of FHP visits | |||||
| Yes | 1.00 | – | 1.00 | – | |
| No | 3.25 (1.33–7.94) | 0.010 | 3.61 (1.46–8.93) | 0.005 | 0.585 |
The association between relapse and smoking was adjusted for all socioeconomic variables and alcohol consumption to exclude the possibility of confounding by these variables. There was no important change in the magnitude of this association when these variables were included in the model; the change in the value of the crude and adjusted OR was less than 10%; when adjustment was made for income, the CI was wider and close to 1 but this was associated with a small change in the value of the OR (from 2.33 to 2.03). To evaluate the potential effect of misclassification of relapse in patients who died we repeated the analysis considering the six deaths with tuberculosis as the cause and considering all 43 deaths as relapses. The inclusion of these cases did not affect substantially our results, as only minor changes of the ORs were observed: The OR was 2.05 (95% CI 1.06–3.96) when six deaths were treated as relapses and 1.97 (95% CI 1.19–3.25) when all deaths were treated as relapses.
Discussion
Smoking and living in an area where the FHP was not implemented were found to be independently associated with relapse of tuberculosis.
The incidence of relapse in this cohort of patients (5.2%) was lower than that found by Thomas et al. in a study carried out in South India (12.3%).6 This difference might be related to some extent to differences between the two populations (for example in degree of under nutrition), but we believe that the two most likely explanations are active ascertainment in the Indian study and better treatment adherence in Brazil. We expand on these below. First, in the Indian case study finding was active with field workers visiting the study subjects at 6, 12 and 18 months after completion of treatment, and will have identified cases that might have progressed to cure without diagnosis, and cases that would have been diagnosed much later, whereas in the Brazilian study relapses were ascertained as they presented to health services with symptoms. Second, the quality of the tuberculosis control program in Brazil is probably better than in India. The quality of the programme is essential in assuring adherence to treatment, and low adherence is a well-established determinant of relapse. The tuberculosis control programme has been improving in Brazil, with a marked decrease in the percentage of patients notified with relapse in the city of Recife as well as in Brazil, from 2001 to 2005.7
In our study the bulk (64.8%) of the relapses occurred during the first year of follow-up. This finding is similar to that of Nogueira et al. and Oliveira and Moreira who found, respectively, percentages of 65% and 61.9% year after completion of treatment.12,13
In this study, as expected, neither sex nor age nor social factors were risk factors for relapse of tuberculosis. Similar results were found by Oliveira and Moreira, and Ormerod and Prescott.3,14
Tuberculosis has been characterized as a disease of poverty, but this is an aspect of developing tuberculosis, not of relapse. Measures of low socioeconomic status, like low family income, illiteracy and low social class have been found to be associated with an increased risk of developing tuberculosis.15 Although socioeconomic variables have never been shown to be associated with an increase in tuberculosis relapse, inadequate treatment has, and it is possible that poverty may lead to inadequate treatment in some circumstances. Even in countries where the tuberculosis control program is well structured and diagnosis and treatment are provided with no costs to the patient, cases may not be able to pay the indirect costs of treatment, e.g. for transport to the health units.16 In a study carried out in Salvador, Northeast of Brazil, the costs to families because of loss of income and expenses due to the disease were very high, on average 33% of the family income.17 In Recife, where this study was conducted, low socioeconomic status, measured as illiteracy or low income of the head of household increased the risk of all three negative outcomes of tuberculosis treatment—dropout, treatment failure and death.11 However, the present study in the same population found no association of relapse to poverty. This may be because of low power, or may indicate the relative magnitude of biological mechanisms on relapse.
The present study found no association between alcohol consumption and relapse, which differs from the study of Selassie et al. which described an increase of almost 4-fold in the risk of relapse in the group of alcoholics.18 However, Selassie et al. did not control for smoking, and if alcohol and tobacco consumption are associated in that setting, the association measured may be reflecting an unmeasured effect of smoking. In relation to the role of alcohol consumption and smoking in the risk of developing tuberculosis (rather than relapsing), Ruffino-Neto and Ruffino,19 several decades ago, investigated the synergism between these two factors and found that smoking was associated with tuberculosis only in the group of drinkers (alcohol), whereas Brown and Campbell, on examining confounding between the two variables, concluded that alcohol had a stronger association with tuberculosis.20 Due to these findings, smoking was adjusted for alcohol consumption in the present study, but there was no confounding effect between alcohol consumption and smoking on the risk of relapse (data not shown). In addition, as smoking is more frequent among the poor, the association between smoking and relapse was adjusted for each of the socioeconomic variables collected previously and there was only a very small change in the value of the OR. Thomas et al., who also described an association between smoking and relapse, did not control for the socioeconomic factors.6
Despite the fact that in the second half of the last century several studies pointed to an association between smoking and developing tuberculosis,19,21,22 there was a gap of decades until scientific interest in this topic was renewed. More recently, studies have investigated and found evidence of smoking as a risk factor for developing tuberculosis, independently, or interacting with alcohol consumption, and evidence of progression from latent tuberculosis infection to disease.4,5 The association between smoking and death from tuberculosis remains controversial; while in the review of Chiang et al. smoking was associated with increased TB mortality,23 Bates et al. described no clear effect of smoking on the case fatality rate in those with active TB.24
Smoking was found to be significantly associated with relapse in the present study (OR 2.34; 95% CI 1.17–4.68) corroborating the finding of Thomas et al. who described a similar association (OR 3.1, 95% CI 1.6–6.0).6 Several studies highlight the association between smoking and the development of tuberculosis4,5,23–25 and the mechanism proposed to explain this association is the neutralisation of the tumour necrosis factor α (TNF-α) in the pulmonary macrophages by substances of the tobacco, leaving the patient more susceptible to a progressive development of disease from latent tuberculosis infection.5 The reduction in the TNF-α in this group may be explained by the high level of iron in the bronchoalveolar macrophages of smokers.26 This mechanism may also be behind the development of relapse.
Tobacco consumption has been increasing mainly in developing countries. In Brazil, one of the greatest producers and exporters of tobacco, the National Program of Control of Smoking, of the Ministry of Health, has been acting through the National Institute of Cancer (INCA), and there has been some important achievements as a decrease of nearly 50% in the prevalence of smokers between 1989 and 2003.27 Nevertheless, it is still important to incorporate in the strategies of the Tuberculosis Control Program the evidence that smoking increases the risk of relapse. This knowledge may become one more tool in the control of the disease.
Concerning the factors related to health services, the risk of relapse was significantly lower among patients living in areas where the FHP was implemented. The Ministry of Health has defined a policy of decentralization and has transferred to the local authorities the administration of the health services. This policy which aims to facilitate the access to health services and includes periodical visits of trained and reasonably well-paid health agents to all families has been implemented with good results.28 The Tuberculosis Control Plan emphasizes the performance of the health agents and FHP teams as a tool to increase the therapeutic adherence and prevent patients from interrupting treatment.8,29 During the period from 2000 and 2006, the proportion of health services that implemented supervised treatment, whether or not using the FHP, increased from 7 to 81%.30 In Recife, as in Brazil as a whole, there has been a marked decrease in the percentage of patients notified with relapse from 2001 to 2005.7 It was not possible to establish the degree of adherence in this study, as there is no record of treatment adherence. However, a robust explanation for the lower rate of relapse in patients who received visits from the agent from the FHP is that the visits lead to better adherence to treatment during the 6 months long therapeutic regimen. A closer monitoring of tuberculosis patients even after discharge would have increased detection of relapsed patients and tend to decrease the magnitude of the association.
Since Brazil does not follow the internationally recommended first-line treatment,9 it is of course not possible to evaluate if the type of treatment affects the frequency of relapse in this study, although the proportion of cases relapsing is not out of line with other countries. Concerning the association between FHP and smoking with relapse, as all patients studied were treated with the same drug regimen, this would not distort the association between these factors and relapse, but of course to generalize these findings to other treatment regimens the study must be repeated in the appropriate settings.
A limitation of this study was that we did not use the CAGE standard questionnaire to ascertain alcoholism, and therefore can only speak of alcohol consumption. Another limitation is that it is not possible to differentiate relapse due to reactivation of the disease from tuberculosis resulting form a new infection, as genotyping was not used to identify the strains of M. tuberculosis. This is not unusual: Cox et al.31 reported that only one, among the 16 studies included in their systematic review, differentiated true relapse from reinfection with a different strain of M. tuberculosis.
Ascertainment of outcomes relied on a routine surveillance system and studies relying on passive detection of relapse are more likely to under-report. However, we expect that in Brazil, the risk relapse without diagnoses is low because the National Program for Tuberculosis Control (PNCT) has standard procedures for investigation and diagnosis in the whole country and the standardized treatment regimen is only delivered after the case is notified to SINAN. The potential risk of differential detection of relapse for different groups, e.g. by SES, is minimized by the partnership of the PNCT with the Community Outreach Program (PACS) and FHP which widens the actions and coverage of the PNCT, especially in areas of low SES.28,29
A total of 43 patients were excluded because they died after completion of the successful treatment, and this could generate survival bias. Tuberculosis was pointed as basic cause of death in 6 of the 43 (data from mortality information system of the Ministry of Health—SIM/MS). However, sensitivity analysis confirmed that the results are not affected by including deaths coded to tuberculosis and other deaths, suggesting that any survival bias must be very small.
It is possible that other factors might be associated with relapse, including short temporary interruptions of treatment, diabetes and malnutrition. Information on these were not collected. They may have a significant impact on risk of relapse but we have no reason to think that they would be associated with smoking.
The sample size- the small number of relapses—may be a limitation of this study. Some associations, especially those related to socioeconomic conditions and alcohol consumption may have been present and not have been identified because of the power of the study. However, this does not question the validity of the associations found.
Our findings are relevant for Tuberculosis Control Programs. Since just to cure patients may not be enough to achieve the control of the disease (as cured patients can relapse), it is important to incorporate in the strategies of the Tuberculosis Control Program strategies to reduce the risk of relapse, including smoke cessation support. Finally, the effect of home visits of the FHP confirms that in successful treatments of tuberculosis, close monitoring of adherence during the 6 months of the treatment may help prevent relapses.
Acknowledgements
The authors wish to thank the Brazilian National Research Council (Conselho Nacional de Desenvolvimento Científico e Tecnológico—CNPq), British Council and REDE-TB do Brasil for funding this study. The authors were partially supported by CNPq (scholarship 305947/2006-0 to M.F.P.M., 300917/2006-6 to R.A.A.X. and 134361/2005-9 to J.D.L.B.).
Conflict of interest: None declared.
KEY MESSAGES.
Smoking was independently associated with relapse of tuberculosis after successful tuberculosis treatment even when adjusted for socioeconomic factors and alcohol consumption.
Our findings provide evidence for incorporating smoking cessation support to the strategies to improve effectiveness of tuberculosis control programs.
Living in an area where the FHP (which provides home visits for routine health supervision) was not implemented, increased the risk of relapse of tuberculosis, suggesting close monitoring of adherence during the 6 months of the treatment may not only increase successful treatment but also decrease tuberculosis relapses.
References
- 1.World Health Organization. The International Union Against Tuberculosis and Lung Disease, The Royal Netherlands Tuberculosis Association. Revised international definitions in tuberculosis control. Int J Tuberc Lung Dis. 2001;5:213–15. [PubMed] [Google Scholar]
- 2.Campos HS, Albuquerque MFM, Campelo ARL, et al. O retratamento da tuberculose no município do Recife, 1997: uma abordagem epidemiológica. J Brás Pneumol. 2000;26:235–40. [Google Scholar]
- 3.Oliveira HB, Moreira Filho DC. Recidiva em tuberculose e seus fatores de risco. Rev Panam Salud Pub. 2000;7:232–41. doi: 10.1590/s1020-49892000000400004. [DOI] [PubMed] [Google Scholar]
- 4.Maurya V, Vijayan VK, Shah A. Smoking and tuberculosis: an association overlooked. Int J Tuberc Lung Dis. 2002;6:942–51. [PubMed] [Google Scholar]
- 5.Davies PDO, Yew WW, Ganguly D, et al. Smoking and tuberculosis: the epidemiological association and immunopathogenesis. Trans R Soc Trop Med Hyg. 2006;100:291–98. doi: 10.1016/j.trstmh.2005.06.034. [DOI] [PubMed] [Google Scholar]
- 6.Thomas A, Gopi PG, Santha T, et al. Predictors of relapse among pulmonary tuberculosis patients treated in a DOTS programme in South India. Int J Tuberc Lung Dis. 2005;9:556–61. [PubMed] [Google Scholar]
- 7.Governo do Estado de Pernambuco, Secretaria Estadual de Saúde, Programa de Controle da Tuberculose [homepage on the Internet]. Indicadores e informações em saúde SINAN/MS/SUS. Brasília, DF: Ministério da Saúde; 2002. [cited 2007 Feb 19] Available from: www.saude.gov.br.
- 8.Ministério da Saúde. Secretaria de Políticas de Saúde. Brasília, DF: Ministério da Saúde; 2002. Departamento de Atenção Básica. Manual técnico para o controle da tuberculose. Cadernos de Atenção Básica, n° 6. [Google Scholar]
- 9.Migliori GB, Hopewell PC, Blasi F, et al. Improving the TB case management: The International Standards for Tuberculosis care. Eur Respir J. 2006;28:687–90. doi: 10.1183/09031936.06.00097506. [DOI] [PubMed] [Google Scholar]
- 10.Santos MAPS, Albuquerque MFPM, Ximenes RAA, et al. Risk factors for treatment delay in pulmonary tuberculosis in Recife, Brazil. BMC Public Health. 2005;5:25. doi: 10.1186/1471-2458-5-25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Albuquerque MFPM, Ximenes RAA, Silva NL, et al. Factors associated with treatment failure, dropout, and death in a cohort of tuberculosis patients in Recife, Pernambuco State, Brazil. Cad Saúde Pública. 2007;23:105–14. doi: 10.1590/s0102-311x2007000700008. [DOI] [PubMed] [Google Scholar]
- 12.Nogueira PA, Belluomini M, Almeida MMB, et al. Resumos do IV Congresso Paulista de Saúde Pública; 1993. Algumas características dos reingressantes ao Sistema Experimental de Vigilância, segundo tipo de alta anterior. July 10–14, São Paulo, Brazil. [Google Scholar]
- 13.Oliveira HB, Moreira Filho DC. Abandono de tratamento e recidiva da tuberculose: aspectos de episódios prévios, Campinas, SP, Brasil, 1993–1994. Rev Saúde Pública. 2000;34:437–43. doi: 10.1590/s0034-89102000000500002. [DOI] [PubMed] [Google Scholar]
- 14.Ormerod LP, Prescott RJ. Inter-relations between relapses, drug regimens and compliance with treatment in tuberculosis. Respir Med. 1991;85:239–42. doi: 10.1016/s0954-6111(06)80087-9. [DOI] [PubMed] [Google Scholar]
- 15.Menezes AMB, Costa JD, Gonçalves H, et al. Incidência e fatores de risco para tuberculose em Pelotas, uma cidade do Sul do Brasil. Rev Bras Epidemiol. 1998;1:50–60. [Google Scholar]
- 16.Tackling poverty in tuberculosis control. Lancet. 2063. Vol. 366. 2005. Editorial. [DOI] [PubMed] [Google Scholar]
- 17.Costa JG, Santos AC, Rodrigues LC, et al. Tuberculosis in Salvador, Brazil: costs to health system and families. Rev Saúde Pública. 2005;39:122–28. doi: 10.1590/s0034-89102005000100016. [DOI] [PubMed] [Google Scholar]
- 18.Selassie AW, Pozsik C, Wilson D, et al. Why pulmonary tuberculosis recurs: a population-based epidemiological study. Ann Epidemiol. 2005;15:519–25. doi: 10.1016/j.annepidem.2005.03.002. [DOI] [PubMed] [Google Scholar]
- 19.Ruffino-Netto A, Caron-Ruffino M. Interação de fatores risco em tuberculose. Rev Saúde Pública. 1979;3:119–22. doi: 10.1590/s0034-89101979000200008. [DOI] [PubMed] [Google Scholar]
- 20.Brown KE, Campbell AH. Tobacco, alcohol and tuberculosis. Br J Dis Chest. 1961;55:150–58. [Google Scholar]
- 21.Lowe CR. An association between smoking and respiratory tuberculosis. Br Med J. 1956;2:1081–83. doi: 10.1136/bmj.2.5001.1081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Doll R, Peto R, Wheatley K, et al. Mortality in relation to smoking: 40 years’ observations on male British doctors. Br Med J. 1994;309:901–11. doi: 10.1136/bmj.309.6959.901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chiang CY, Slama K, Enarson DA. Associations between tobacco and tuberculosis. Int J Tuberc Lung Dis. 2007;11:258–62. [PubMed] [Google Scholar]
- 24.Bates MN, Khalakdina A, Pai M, et al. Risk of tuberculosis from exposure to tobacco smoke: a systematic review and meta-analysis. Arch Intern Med. 2007;167:335–42. doi: 10.1001/archinte.167.4.335. [DOI] [PubMed] [Google Scholar]
- 25.Alcaide J, Altet MN, Plans P, et al. Cigarette smoking as a risk factor for tuberculosis in young adults: a case-control study. Int J Tuberc Lung Dis. 1996;77:112–16. doi: 10.1016/s0962-8479(96)90024-6. [DOI] [PubMed] [Google Scholar]
- 26.Boelaert JR, Gomes MS, Gordeuk VR. Smoking, iron and TB. Lancet. 2003;362:1243–44. doi: 10.1016/S0140-6736(03)14529-1. [DOI] [PubMed] [Google Scholar]
- 27.Temporão JG. Saúde Pública e controle do tabagismo no Brasil. Cad Saúde Pública. 2005;21:670–71. doi: 10.1590/s0102-311x2005000300001. [DOI] [PubMed] [Google Scholar]
- 28.Cavalcante MGS, Samico I, Frias PG, et al. Análise de implantação das áreas estratégicas da atenção básica nas equipes de Saúde da Família em município de uma Região Metropolitana do Nordeste Brasileiro. Rev Bras Saúde Matern Infant. 2006;6:437–45. [Google Scholar]
- 29.Ruffino-Netto A, Villa TCS. Tuberculosis treatment. DOTS implementation in some regions of Brazil. Background and regional features. Ribeirão Preto, SP: Millennium Instiitute–Rede TB; 2000. [Google Scholar]
- 30.Santos J. Resposta brasileira ao controle da tuberculose. Rev Saúde Pública. 2007;41(Suppl 1):89–94. doi: 10.1590/s0034-89102007000800012. [DOI] [PubMed] [Google Scholar]
- 31.Cox HS, Morrow M, Deutschmann PW. Long term efficacy of DOTS regimens for tuberculosis: systematic review. Br Med J. 2008;336:484–47. doi: 10.1136/bmj.39463.640787.BE. [DOI] [PMC free article] [PubMed] [Google Scholar]