Abstract
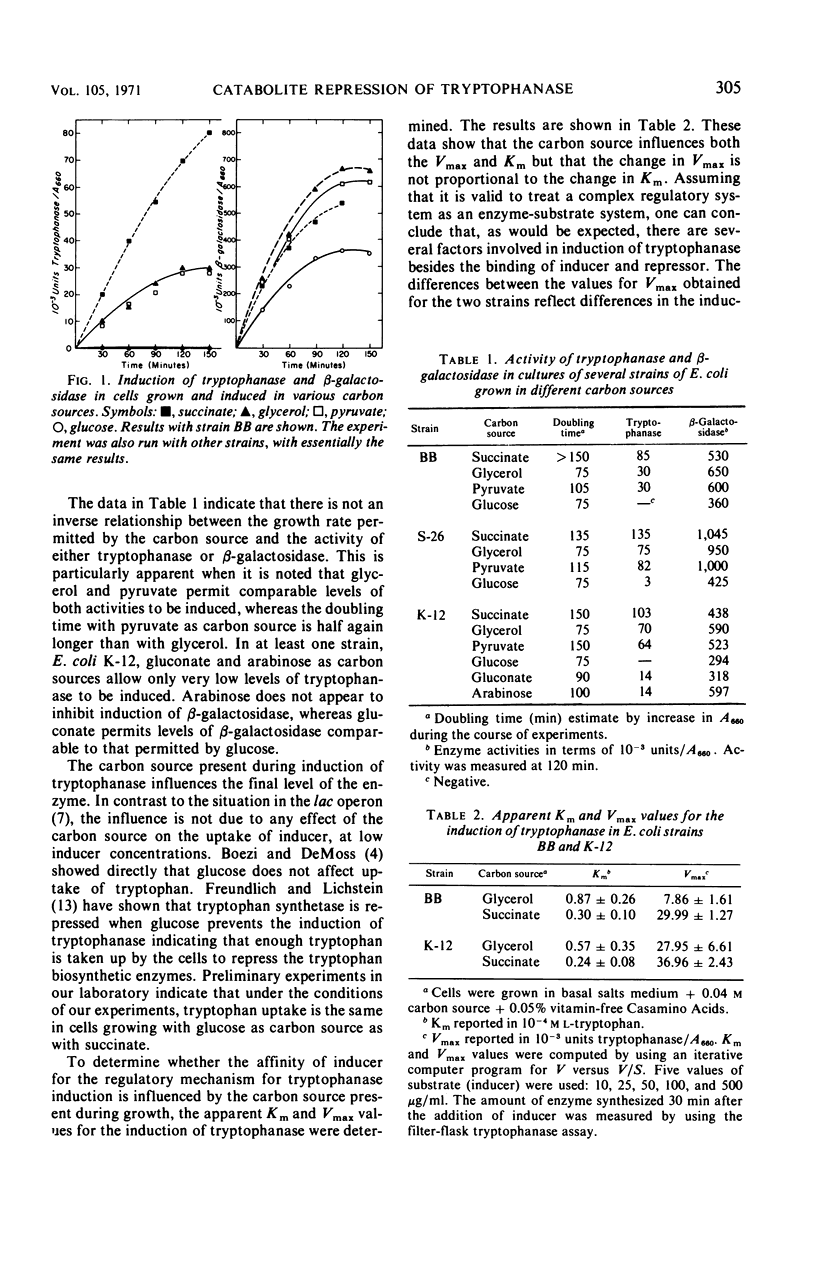
Catabolite repression of tryptophanase was studied in detail under various conditions in several strains of Escherichia coli and was compared with catabolite repression of β-glactosidase. Induction of tryptophanase and β-galactosidase in cultures grown with various carbon sources including succinate, glycerol, pyruvate, glucose, gluconate, and arabinose is affected differently by the various carbon sources. The extent of induction does not seem to be related to the growth rate of the culture permitted by the carbon source during the course of the experiment. In cultures grown with glycerol as carbon source, preinduced for β-galactosidase or tryptophanase and made permeable by ethylenediaminetetraacetic acid (EDTA) treatment, catabolite repression of tryptophanase was not affected markedly by the addition of cAMP (3′,5′-cyclic adenosine monophosphate). Catabolite repression by glucose was only partially relieved by the addition of cAMP. In contrast, under the same conditions, cAMP completely relieved catabolite repression of β-galactosidase by either pyruvate or glucose. Under conditions of limited oxygen, induction of tryptophanase is sensitive to catabolite repression; under the same conditions, β-galactosidase induction is not sensitive to catabolite repression. Induction of tryptophanase in cells grown with succinate as carbon source is sensitive to catabolite repression by glycerol and pyruvate as well as by glucose. Studies with a glycerol kinaseless mutant indicate that glycerol must be metabolized before it can cause catabolite repression. The EDTA treatment used to make the cells permeable to cAMP was found to affect subsequent growth and induction of either β-galactosidase or tryptophanase much more adversely in E. coli strain BB than in E. coli strain K-12. Inducation of tryptophanase was reduced by the EDTA treatment significantly more than induction of β-galactosidase in both strains. Addition of 2.5 × 10−3m cAMP appeared partially to reverse the inhibitory effect of the EDTA treatment on enzyme induction but did not restore normal growth.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aboud M., Burger M. The effect of catabolite repression and of cyclic 3',5' adenosine monophosphate on the translation of the lactose messenger RNA in Escherichia coli. Biochem Biophys Res Commun. 1970 Mar 27;38(6):1023–1032. doi: 10.1016/0006-291x(70)90342-6. [DOI] [PubMed] [Google Scholar]
- Anderson R. L., Wood W. A. Carbohydrate metabolism in microorganisms. Annu Rev Microbiol. 1969;23:539–578. doi: 10.1146/annurev.mi.23.100169.002543. [DOI] [PubMed] [Google Scholar]
- BURNS R. O., DEMOSS R. D. Properties of tryptophanase from Escherichia coli. Biochim Biophys Acta. 1962 Dec 4;65:233–244. doi: 10.1016/0006-3002(62)91042-9. [DOI] [PubMed] [Google Scholar]
- Bilezikian J. P., Kaempfer R. O., Magasanik B. Mechanism of tryptophanase induction in Escherichia coli. J Mol Biol. 1967 Aug 14;27(3):495–506. doi: 10.1016/0022-2836(67)90054-x. [DOI] [PubMed] [Google Scholar]
- COHN M., HORIBATA K. Inhibition by glucose of the induced synthesis of the beta-galactoside-enzyme system of Escherichia coli. Analysis of maintenance. J Bacteriol. 1959 Nov;78:601–612. doi: 10.1128/jb.78.5.601-612.1959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chambers D. A., Zubay G. The stimulatory effect of cyclic adenosine 3'5'-monophosphate on DNA-directed synthesis of beta-galactosidase in a cell-free system. Proc Natl Acad Sci U S A. 1969 May;63(1):118–122. doi: 10.1073/pnas.63.1.118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cozzarelli N. R., Freedberg W. B., Lin E. C. Genetic control of L-alpha-glycerophosphate system in Escherichia coli. J Mol Biol. 1968 Feb 14;31(3):371–387. doi: 10.1016/0022-2836(68)90415-4. [DOI] [PubMed] [Google Scholar]
- De Crombrugghe B., Perlman R. L., Varmus H. E., Pastan I. Regulation of inducible enzyme synthesis in Escherichia coli by cyclic adenosine 3', 5'-monophosphate. J Biol Chem. 1969 Nov 10;244(21):5828–5835. [PubMed] [Google Scholar]
- DeMoss R. D., Moser K. Tryptophanase in diverse bacterial species. J Bacteriol. 1969 Apr;98(1):167–171. doi: 10.1128/jb.98.1.167-171.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dobrogosz W. J. Corepressor system for catabolite repression of the lac operon in Escherichia coli. J Bacteriol. 1969 Mar;97(3):1083–1092. doi: 10.1128/jb.97.3.1083-1092.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eisenberg R. C., Dobrogosz W. J. Gluconate metabolism in Escherichia coli. J Bacteriol. 1967 Mar;93(3):941–949. doi: 10.1128/jb.93.3.941-949.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- FREUNDLICH M., LICHSTEIN H. C. Inhibitory effect of glucose on tryptophanase. J Bacteriol. 1960 Nov;80:633–638. doi: 10.1128/jb.80.5.633-638.1960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- GARTNER T. K., RILEY M. ISOLATION OF MUTANTS AFFECTING TRYPTOPHANASE PRODUCTION IN ESCHERICHIA COLI. J Bacteriol. 1965 Feb;89:313–318. doi: 10.1128/jb.89.2.313-318.1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoch J. A., DeMoss R. D. Physiological role of tryptophanase in control of tryptophan biosynthesis in Bacillus alvei. J Bacteriol. 1966 Feb;91(2):667–672. doi: 10.1128/jb.91.2.667-672.1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leive L. Studies on the permeability change produced in coliform bacteria by ethylenediaminetetraacetate. J Biol Chem. 1968 May 10;243(9):2373–2380. [PubMed] [Google Scholar]
- MAKMAN R. S., SUTHERLAND E. W. ADENOSINE 3',5'-PHOSPHATE IN ESCHERICHIA COLI. J Biol Chem. 1965 Mar;240:1309–1314. [PubMed] [Google Scholar]
- MCFALL E., MANDELSTAM J. SPECIFIC METABOLIC REPRESSION OF THREE INDUCED ENZYMES IN ESCHERICHIA COLI. Biochem J. 1963 Nov;89:391–398. doi: 10.1042/bj0890391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moses V., Prevost C. Catabolite repression of beta-galactosidase synthesis in Escherichia coli. Biochem J. 1966 Aug;100(2):336–353. doi: 10.1042/bj1000336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okinaka R. T., Dobrogosz W. J. Catabolite repression and pyruvate metabolism in Escherichia coli. J Bacteriol. 1967 May;93(5):1644–1650. doi: 10.1128/jb.93.5.1644-1650.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paigen K. Phenomenon of transient repression in Escherichia coli. J Bacteriol. 1966 Mar;91(3):1201–1209. doi: 10.1128/jb.91.3.1201-1209.1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pastan I., Perlman R. L. Stimulation of tryptophanase synthesis in Escherichia coli by cyclic 3',5'-adenosine monophosphate. J Biol Chem. 1969 Apr 25;244(8):2226–2232. [PubMed] [Google Scholar]
- Perlman R. L., De Crombrugghe B., Pastan I. Cyclic AMP regulates catabolite and transient repression in E. coli. Nature. 1969 Aug 23;223(5208):810–812. doi: 10.1038/223810a0. [DOI] [PubMed] [Google Scholar]
- Perlman R. L., Pastan I. Regulation of beta-galactosidase synthesis in Escherichia coli by cyclic adenosine 3',5'-monophosphate. J Biol Chem. 1968 Oct 25;243(20):5420–5427. [PubMed] [Google Scholar]
- Perlman R., Pastan I. Cyclic 3'5-AMP: stimulation of beta-galactosidase and tryptophanase induction in E. coli. Biochem Biophys Res Commun. 1968 Mar 27;30(6):656–664. doi: 10.1016/0006-291x(68)90563-9. [DOI] [PubMed] [Google Scholar]
- Rickenberg H. V., Hsie A. W., Janecek J. The CR mutation and catabolite repression in Escherichia coli. Biochem Biophys Res Commun. 1968 May 23;31(4):603–608. doi: 10.1016/0006-291x(68)90521-4. [DOI] [PubMed] [Google Scholar]
- Tyler B., Loomis W. F., Jr, Magasanik B. Transient repression of the lac operon. J Bacteriol. 1967 Dec;94(6):2001–2011. doi: 10.1128/jb.94.6.2001-2011.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tyler B., Magasanik B. Molecular basis of transient repression of beta-galactosidase in Escherichia coli. J Bacteriol. 1969 Feb;97(2):550–556. doi: 10.1128/jb.97.2.550-556.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tyler B., Wishnow R., Loomis W. F., Jr, Magasanik B. Catabolite repression gene of Escherichia coli. J Bacteriol. 1969 Nov;100(2):809–816. doi: 10.1128/jb.100.2.809-816.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]



