Abstract
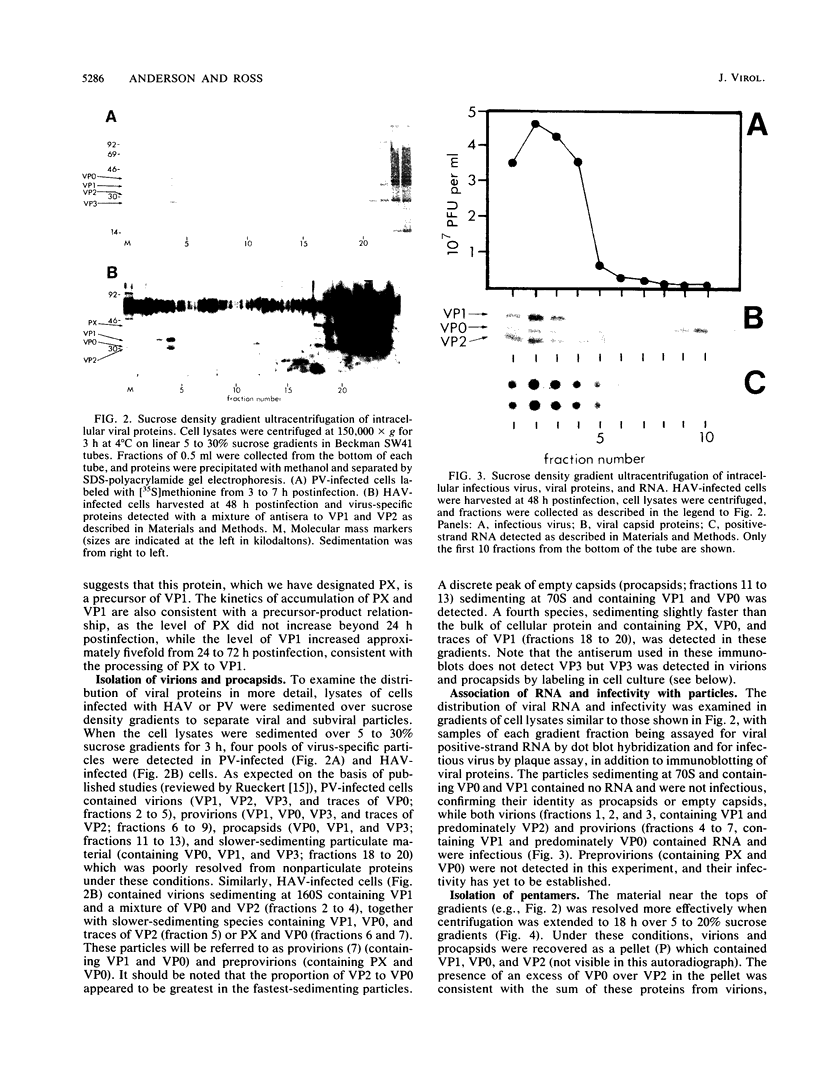
The morphogenesis of hepatitis A virus (HAV) in BS-C-1 cells was examined by immunoblotting with antisera to capsid proteins and labeling of virus-specific proteins with L-[35S]methionine. Antiserum to VP2 detected two virus-specific proteins with apparent molecular masses of 30.6 and 30 kDa, representing VP0 and VP2, while antiserum to VP1 detected proteins with molecular masses of 33 and 40 kDa, representing VP1 and a virus-specific protein which we designated PX, respectively. Sedimentation of cell lysates revealed the presence of virions, procapsids, and pentamers, but particles analogous to the protomers of other picornaviruses were not detected. Although provirions and virions were not found as discrete species in our gradient system, it was evident that the rate of sedimentation was proportional to the relative amounts of VP0 and VP2 in particles, with slower-sedimenting particles (provirions) containing predominantly VP0 rather than VP2. Procapsids contained VP0 in addition to VP1 and VP3. Pentamers also contained VP0, but PX was present rather than VP1. These results suggest that PX is a precursor to VP1 and is most likely 1D2A. Primary cleavage of the viral polyprotein also occurs at the 2A-2B junction in cardioviruses and aphthoviruses, but assembly of pentamers containing 1D2A has not been reported for those viruses. The absence of detectable levels of protomers suggests a high efficiency of pentamer formation, which may be related to the high efficiency of viral RNA encapsidation for HAV (D.A. Anderson, B.C. Ross, and S.A. Locarnini, J. Virol. 62:4201-4206, 1988). The results of this study reveal further unusual aspects of the HAV replicative cycle which distinguish it from other picornaviruses and may contribute to its restricted replication in cell culture.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anderson D. A. Cytopathology, plaque assay, and heat inactivation of hepatitis A virus strain HM175. J Med Virol. 1987 May;22(1):35–44. doi: 10.1002/jmv.1890220106. [DOI] [PubMed] [Google Scholar]
- Anderson D. A., Ross B. C., Locarnini S. A. Restricted replication of hepatitis A virus in cell culture: encapsidation of viral RNA depletes the pool of RNA available for replication. J Virol. 1988 Nov;62(11):4201–4206. doi: 10.1128/jvi.62.11.4201-4206.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clarke B. E., Sangar D. V. Processing and assembly of foot-and-mouth disease virus proteins using subgenomic RNA. J Gen Virol. 1988 Sep;69(Pt 9):2313–2325. doi: 10.1099/0022-1317-69-9-2313. [DOI] [PubMed] [Google Scholar]
- Cohen J. I., Ticehurst J. R., Purcell R. H., Buckler-White A., Baroudy B. M. Complete nucleotide sequence of wild-type hepatitis A virus: comparison with different strains of hepatitis A virus and other picornaviruses. J Virol. 1987 Jan;61(1):50–59. doi: 10.1128/jvi.61.1.50-59.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Chastonay J., Siegl G. Replicative events in hepatitis A virus-infected MRC-5 cells. Virology. 1987 Apr;157(2):268–275. doi: 10.1016/0042-6822(87)90269-8. [DOI] [PubMed] [Google Scholar]
- Fernandez-Tomas C. B., Baltimore D. Morphogenesis of poliovirus. II. Demonstration of a new intermediate, the proviron. J Virol. 1973 Nov;12(5):1122–1130. doi: 10.1128/jvi.12.5.1122-1130.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gauss-Müller V., Deinhardt F. Effect of hepatitis A virus infection on cell metabolism in vitro. Proc Soc Exp Biol Med. 1984 Jan;175(1):10–15. doi: 10.3181/00379727-175-41757. [DOI] [PubMed] [Google Scholar]
- Harmon S. A., Summers D. F., Ehrenfeld E. Detection of hepatitis A virus RNA and capsid antigen in individual cells. Virus Res. 1989 Apr;12(4):361–369. doi: 10.1016/0168-1702(89)90093-2. [DOI] [PubMed] [Google Scholar]
- Jackson R. J. A detailed kinetic analysis of the in vitro synthesis and processing of encephalomyocarditis virus products. Virology. 1986 Feb;149(1):114–127. doi: 10.1016/0042-6822(86)90092-9. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Provost P. J., Hilleman M. R. Propagation of human hepatitis A virus in cell culture in vitro. Proc Soc Exp Biol Med. 1979 Feb;160(2):213–221. doi: 10.3181/00379727-160-40422. [DOI] [PubMed] [Google Scholar]
- Rueckert R. R., Wimmer E. Systematic nomenclature of picornavirus proteins. J Virol. 1984 Jun;50(3):957–959. doi: 10.1128/jvi.50.3.957-959.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siegl G., Weitz M., Kronauer G. Stability of hepatitis A virus. Intervirology. 1984;22(4):218–226. doi: 10.1159/000149554. [DOI] [PubMed] [Google Scholar]
- Siegl G., deChastonay J., Kronauer G. Propagation and assay of hepatitis A virus in vitro. J Virol Methods. 1984 Aug;9(1):53–67. doi: 10.1016/0166-0934(84)90083-1. [DOI] [PubMed] [Google Scholar]
- Vakharia V. N., Devaney M. A., Moore D. M., Dunn J. J., Grubman M. J. Proteolytic processing of foot-and-mouth disease virus polyproteins expressed in a cell-free system from clone-derived transcripts. J Virol. 1987 Oct;61(10):3199–3207. doi: 10.1128/jvi.61.10.3199-3207.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vallbracht A., Hofmann L., Wurster K. G., Flehmig B. Persistent infection of human fibroblasts by hepatitis A virus. J Gen Virol. 1984 Mar;65(Pt 3):609–615. doi: 10.1099/0022-1317-65-3-609. [DOI] [PubMed] [Google Scholar]
- Wheeler C. M., Fields H. A., Schable C. A., Meinke W. J., Maynard J. E. Adsorption, purification, and growth characteristics of hepatitis A virus strain HAS-15 propagated in fetal rhesus monkey kidney cells. J Clin Microbiol. 1986 Mar;23(3):434–440. doi: 10.1128/jcm.23.3.434-440.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]