Abstract
Cytotoxic T lymphocytes (CTL) play an important role in recovery from a number of viral infections. They are also implicated in virus-induced immunopathology as best demonstrated in lymphocytic choriomeningitis virus (LCMV) infection of adult immunocompetent mice. In the present study, the structure of the T-cell receptor (TCR) in LCMV-specific CTL in C57BL/6 (B6) mice was investigated. Spleen T cells obtained from LCMV-infected mice were cultured in vitro with virus-infected stimulator cells and then stained with anti-TCR V beta antibodies. A skewing of V beta usage was noticeable in T cells enriched for their reactivity to LCMV, suggesting that particular V segments are important for the recognition of LCMV T-cell epitopes in B6 mice. To gain more detailed information on the structure of the TCR specific for LCMV epitopes, we studied CTL clones. It has been shown that approximately 90% of LCMV-reactive CTL clones generated in H-2b mice are specific for a short peptide fragment of the LCMV glycoprotein, residues 278 to 286, recognized in the context of the class I major histocompatibility complex molecule, Db. Four CTL clones possessing the specificity were randomly selected from a collection of clones, and their TCR genes were isolated by cDNA cloning or by the anchored polymerase chain reaction. All four clones were found to use V alpha gene segments belonging to the V alpha 4 subfamily. By RNA blot analysis, two more clones with the same specificity were also shown to express the V alpha 4 mRNA. In contrast, three different V beta gene segments were used among the four clones examined. J beta 2.1 was used by three of the clones. Although amino acid sequences in the V(D)J junctional regions were dissimilar, aspartic acid was found in the V alpha J alpha and/or V beta D beta J beta junctions of all four of these clones, suggesting that this residue is involved in binding the LCMV fragment. Restricted usage of V alpha and possibly J beta segments in the CTL response to a major T-cell epitope of LCMV raises the possibility that immunopathology in LCMV infection can be treated with antibodies directed against such TCR segments. Thus, similar analysis of the TCR in other virus infections is warranted and may lead to therapeutic strategies for immunopathology due to virus infections.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Acha-Orbea H., Mitchell D. J., Timmermann L., Wraith D. C., Tausch G. S., Waldor M. K., Zamvil S. S., McDevitt H. O., Steinman L. Limited heterogeneity of T cell receptors from lymphocytes mediating autoimmune encephalomyelitis allows specific immune intervention. Cell. 1988 Jul 15;54(2):263–273. doi: 10.1016/0092-8674(88)90558-2. [DOI] [PubMed] [Google Scholar]
- Aebischer T., Oehen S., Hengartner H. Preferential usage of V alpha 4 and V beta 10 T cell receptor genes by lymphocytic choriomeningitis virus glycoprotein-specific H-2Db-restricted cytotoxic T cells. Eur J Immunol. 1990 Mar;20(3):523–531. doi: 10.1002/eji.1830200310. [DOI] [PubMed] [Google Scholar]
- Arden B., Klotz J. L., Siu G., Hood L. E. Diversity and structure of genes of the alpha family of mouse T-cell antigen receptor. 1985 Aug 29-Sep 4Nature. 316(6031):783–787. doi: 10.1038/316783a0. [DOI] [PubMed] [Google Scholar]
- Becker D. M., Pattern P., Chien Y., Yokota T., Eshhar Z., Giedlin M., Gascoigne N. R., Goodnow C., Wolf R., Arai K. Variability and repertoire size of T-cell receptor V alpha gene segments. Nature. 1985 Oct 3;317(6036):430–434. doi: 10.1038/317430a0. [DOI] [PubMed] [Google Scholar]
- Behlke M. A., Spinella D. G., Chou H. S., Sha W., Hartl D. L., Loh D. Y. T-cell receptor beta-chain expression: dependence on relatively few variable region genes. Science. 1985 Aug 9;229(4713):566–570. doi: 10.1126/science.3875151. [DOI] [PubMed] [Google Scholar]
- Bill J., Kanagawa O., Woodland D. L., Palmer E. The MHC molecule I-E is necessary but not sufficient for the clonal deletion of V beta 11-bearing T cells. J Exp Med. 1989 Apr 1;169(4):1405–1419. doi: 10.1084/jem.169.4.1405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bjorkman P. J., Saper M. A., Samraoui B., Bennett W. S., Strominger J. L., Wiley D. C. Structure of the human class I histocompatibility antigen, HLA-A2. Nature. 1987 Oct 8;329(6139):506–512. doi: 10.1038/329506a0. [DOI] [PubMed] [Google Scholar]
- Bjorkman P. J., Saper M. A., Samraoui B., Bennett W. S., Strominger J. L., Wiley D. C. The foreign antigen binding site and T cell recognition regions of class I histocompatibility antigens. Nature. 1987 Oct 8;329(6139):512–518. doi: 10.1038/329512a0. [DOI] [PubMed] [Google Scholar]
- Blüthmann H., Kisielow P., Uematsu Y., Malissen M., Krimpenfort P., Berns A., von Boehmer H., Steinmetz M. T-cell-specific deletion of T-cell receptor transgenes allows functional rearrangement of endogenous alpha- and beta-genes. Nature. 1988 Jul 14;334(6178):156–159. doi: 10.1038/334156a0. [DOI] [PubMed] [Google Scholar]
- Buchmeier M. J., Welsh R. M., Dutko F. J., Oldstone M. B. The virology and immunobiology of lymphocytic choriomeningitis virus infection. Adv Immunol. 1980;30:275–331. doi: 10.1016/s0065-2776(08)60197-2. [DOI] [PubMed] [Google Scholar]
- Byrne J. A., Ahmed R., Oldstone M. B. Biology of cloned cytotoxic T lymphocytes specific for lymphocytic choriomeningitis virus. I. Generation and recognition of virus strains and H-2b mutants. J Immunol. 1984 Jul;133(1):433–439. [PubMed] [Google Scholar]
- Chien Y. H., Gascoigne N. R., Kavaler J., Lee N. E., Davis M. M. Somatic recombination in a murine T-cell receptor gene. Nature. 1984 May 24;309(5966):322–326. doi: 10.1038/309322a0. [DOI] [PubMed] [Google Scholar]
- Chien Y., Becker D. M., Lindsten T., Okamura M., Cohen D. I., Davis M. M. A third type of murine T-cell receptor gene. Nature. 1984 Nov 1;312(5989):31–35. doi: 10.1038/312031a0. [DOI] [PubMed] [Google Scholar]
- Chothia C., Boswell D. R., Lesk A. M. The outline structure of the T-cell alpha beta receptor. EMBO J. 1988 Dec 1;7(12):3745–3755. doi: 10.1002/j.1460-2075.1988.tb03258.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis M. M., Bjorkman P. J. T-cell antigen receptor genes and T-cell recognition. Nature. 1988 Aug 4;334(6181):395–402. doi: 10.1038/334395a0. [DOI] [PubMed] [Google Scholar]
- Dutko F. J., Oldstone M. B. Genomic and biological variation among commonly used lymphocytic choriomeningitis virus strains. J Gen Virol. 1983 Aug;64(Pt 8):1689–1698. doi: 10.1099/0022-1317-64-8-1689. [DOI] [PubMed] [Google Scholar]
- Engel I., Hedrick S. M. Site-directed mutations in the VDJ junctional region of a T cell receptor beta chain cause changes in antigenic peptide recognition. Cell. 1988 Aug 12;54(4):473–484. doi: 10.1016/0092-8674(88)90068-2. [DOI] [PubMed] [Google Scholar]
- Fink P. J., Matis L. A., McElligott D. L., Bookman M., Hedrick S. M. Correlations between T-cell specificity and the structure of the antigen receptor. Nature. 1986 May 15;321(6067):219–226. doi: 10.1038/321219a0. [DOI] [PubMed] [Google Scholar]
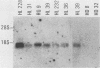
- Furutani M., Yanagi Y., Fujisawa I., Nakayama T., Kishimoto H., Kuida K., Asano Y., Tada T. Post-transcriptional allelic exclusion of two functionally rearranged T cell receptor alpha genes. Int Immunol. 1989;1(3):281–288. doi: 10.1093/intimm/1.3.281. [DOI] [PubMed] [Google Scholar]
- Gubler U., Hoffman B. J. A simple and very efficient method for generating cDNA libraries. Gene. 1983 Nov;25(2-3):263–269. doi: 10.1016/0378-1119(83)90230-5. [DOI] [PubMed] [Google Scholar]
- Hedrick S. M., Cohen D. I., Nielsen E. A., Davis M. M. Isolation of cDNA clones encoding T cell-specific membrane-associated proteins. Nature. 1984 Mar 8;308(5955):149–153. doi: 10.1038/308149a0. [DOI] [PubMed] [Google Scholar]
- Hedrick S. M., Engel I., McElligott D. L., Fink P. J., Hsu M. L., Hansburg D., Matis L. A. Selection of amino acid sequences in the beta chain of the T cell antigen receptor. Science. 1988 Mar 25;239(4847):1541–1544. doi: 10.1126/science.2832942. [DOI] [PubMed] [Google Scholar]
- Hochgeschwender U., Simon H. G., Weltzien H. U., Bartels F., Becker A., Epplen J. T. Dominance of one T-cell receptor in the H-2Kb/TNP response. Nature. 1987 Mar 19;326(6110):307–309. doi: 10.1038/326307a0. [DOI] [PubMed] [Google Scholar]
- Iwamoto A., Ohashi P. S., Pircher H., Walker C. L., Michalopoulos E. E., Rupp F., Hengartner H., Mak T. W. T cell receptor variable gene usage in a specific cytotoxic T cell response. Primary structure of the antigen-MHC receptor of four hapten-specific cytotoxic T cell clones. J Exp Med. 1987 Mar 1;165(3):591–600. doi: 10.1084/jem.165.3.591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joly E., Salvato M., Whitton J. L., Oldstone M. B. Polymorphism of cytotoxic T-lymphocyte clones that recognize a defined nine-amino-acid immunodominant domain of lymphocytic choriomeningitis virus glycoprotein. J Virol. 1989 May;63(5):1845–1851. doi: 10.1128/jvi.63.5.1845-1851.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanagawa O., Palmer E., Bill J. The T cell receptor V beta 6 domain imparts reactivity to the Mls-1a antigen. Cell Immunol. 1989 Apr 1;119(2):412–426. doi: 10.1016/0008-8749(89)90255-4. [DOI] [PubMed] [Google Scholar]
- Land H., Grez M., Hauser H., Lindenmaier W., Schütz G. 5'-Terminal sequences of eucaryotic mRNA can be cloned with high efficiency. Nucleic Acids Res. 1981 May 25;9(10):2251–2266. doi: 10.1093/nar/9.10.2251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loh E. Y., Elliott J. F., Cwirla S., Lanier L. L., Davis M. M. Polymerase chain reaction with single-sided specificity: analysis of T cell receptor delta chain. Science. 1989 Jan 13;243(4888):217–220. doi: 10.1126/science.2463672. [DOI] [PubMed] [Google Scholar]
- Nikolić-Zugić J., Bevan M. J. Thymocytes expressing CD8 differentiate into CD4+ cells following intrathymic injection. Proc Natl Acad Sci U S A. 1988 Nov;85(22):8633–8637. doi: 10.1073/pnas.85.22.8633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oldstone M. B., Whitton J. L., Lewicki H., Tishon A. Fine dissection of a nine amino acid glycoprotein epitope, a major determinant recognized by lymphocytic choriomeningitis virus-specific class I-restricted H-2Db cytotoxic T lymphocytes. J Exp Med. 1988 Aug 1;168(2):559–570. doi: 10.1084/jem.168.2.559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parekh B. S., Buchmeier M. J. Proteins of lymphocytic choriomeningitis virus: antigenic topography of the viral glycoproteins. Virology. 1986 Sep;153(2):168–178. doi: 10.1016/0042-6822(86)90020-6. [DOI] [PubMed] [Google Scholar]
- Pircher H., Michalopoulos E. E., Iwamoto A., Ohashi P. S., Baenziger J., Hengartner H., Zinkernagel R. M., Mak T. W. Molecular analysis of the antigen receptor of virus-specific cytotoxic T cells and identification of a new V alpha family. Eur J Immunol. 1987 Dec;17(12):1843–1846. doi: 10.1002/eji.1830171226. [DOI] [PubMed] [Google Scholar]
- Riviere Y., Southern P. J., Ahmed R., Oldstone M. B. Biology of cloned cytotoxic T lymphocytes specific for lymphocytic choriomeningitis virus. V. Recognition is restricted to gene products encoded by the viral S RNA segment. J Immunol. 1986 Jan;136(1):304–307. [PubMed] [Google Scholar]
- Rodewald H. R., Koszinowski U. H., Eichmann K., Melchers I. Predominant utilization of V beta 8+ T cell receptor genes in the H-2Ld-restricted cytotoxic T cell response against the immediate-early protein pp89 of the murine cytomegalovirus. J Immunol. 1989 Dec 15;143(12):4238–4243. [PubMed] [Google Scholar]
- Roehm N., Herron L., Cambier J., DiGuisto D., Haskins K., Kappler J., Marrack P. The major histocompatibility complex-restricted antigen receptor on T cells: distribution on thymus and peripheral T cells. Cell. 1984 Sep;38(2):577–584. doi: 10.1016/0092-8674(84)90512-9. [DOI] [PubMed] [Google Scholar]
- Rupp F., Brecher J., Giedlin M. A., Mosmann T., Zinkernagel R. M., Hengartner H., Joho R. H. T-cell antigen receptors with identical variable regions but different diversity and joining region gene segments have distinct specificities but cross-reactive idiotypes. Proc Natl Acad Sci U S A. 1987 Jan;84(1):219–222. doi: 10.1073/pnas.84.1.219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saiki R. K., Gelfand D. H., Stoffel S., Scharf S. J., Higuchi R., Horn G. T., Mullis K. B., Erlich H. A. Primer-directed enzymatic amplification of DNA with a thermostable DNA polymerase. Science. 1988 Jan 29;239(4839):487–491. doi: 10.1126/science.2448875. [DOI] [PubMed] [Google Scholar]
- Saito H., Kranz D. M., Takagaki Y., Hayday A. C., Eisen H. N., Tonegawa S. A third rearranged and expressed gene in a clone of cytotoxic T lymphocytes. Nature. 1984 Nov 1;312(5989):36–40. doi: 10.1038/312036a0. [DOI] [PubMed] [Google Scholar]
- Saito H., Kranz D. M., Takagaki Y., Hayday A. C., Eisen H. N., Tonegawa S. Complete primary structure of a heterodimeric T-cell receptor deduced from cDNA sequences. 1984 Jun 28-Jul 4Nature. 309(5971):757–762. doi: 10.1038/309757a0. [DOI] [PubMed] [Google Scholar]
- Sakai K., Sinha A. A., Mitchell D. J., Zamvil S. S., Rothbard J. B., McDevitt H. O., Steinman L. Involvement of distinct murine T-cell receptors in the autoimmune encephalitogenic response to nested epitopes of myelin basic protein. Proc Natl Acad Sci U S A. 1988 Nov;85(22):8608–8612. doi: 10.1073/pnas.85.22.8608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schulz M., Aichele P., Vollenweider M., Bobe F. W., Cardinaux F., Hengartner H., Zinkernagel R. M. Major histocompatibility complex--dependent T cell epitopes of lymphocytic choriomeningitis virus nucleoprotein and their protective capacity against viral disease. Eur J Immunol. 1989 Sep;19(9):1657–1667. doi: 10.1002/eji.1830190921. [DOI] [PubMed] [Google Scholar]
- Sherman D. H., Hochman P. S., Dick R., Tizard R., Ramachandran K. L., Flavell R. A., Huber B. T. Molecular analysis of antigen recognition by insulin-specific T-cell hybridomas from B6 wild-type and bm12 mutant mice. Mol Cell Biol. 1987 May;7(5):1865–1872. doi: 10.1128/mcb.7.5.1865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spinella D. G., Hansen T. H., Walsh W. D., Behlke M. A., Tillinghast J. P., Chou H. S., Whiteley P. J., Kapp J. A., Pierce C. W., Shevach E. M. Receptor diversity of insulin-specific T cell lines from C57BL (H-2b) mice. J Immunol. 1987 Jun 1;138(11):3991–3995. [PubMed] [Google Scholar]
- Staerz U. D., Rammensee H. G., Benedetto J. D., Bevan M. J. Characterization of a murine monoclonal antibody specific for an allotypic determinant on T cell antigen receptor. J Immunol. 1985 Jun;134(6):3994–4000. [PubMed] [Google Scholar]
- Tan K. N., Datlof B. M., Gilmore J. A., Kronman A. C., Lee J. H., Maxam A. M., Rao A. The T cell receptor V alpha 3 gene segment is associated with reactivity to p-azobenzenearsonate. Cell. 1988 Jul 15;54(2):247–261. doi: 10.1016/0092-8674(88)90557-0. [DOI] [PubMed] [Google Scholar]
- Townsend A., Bodmer H. Antigen recognition by class I-restricted T lymphocytes. Annu Rev Immunol. 1989;7:601–624. doi: 10.1146/annurev.iy.07.040189.003125. [DOI] [PubMed] [Google Scholar]
- Urban J. L., Kumar V., Kono D. H., Gomez C., Horvath S. J., Clayton J., Ando D. G., Sercarz E. E., Hood L. Restricted use of T cell receptor V genes in murine autoimmune encephalomyelitis raises possibilities for antibody therapy. Cell. 1988 Aug 12;54(4):577–592. doi: 10.1016/0092-8674(88)90079-7. [DOI] [PubMed] [Google Scholar]
- Whitton J. L., Gebhard J. R., Lewicki H., Tishon A., Oldstone M. B. Molecular definition of a major cytotoxic T-lymphocyte epitope in the glycoprotein of lymphocytic choriomeningitis virus. J Virol. 1988 Mar;62(3):687–695. doi: 10.1128/jvi.62.3.687-695.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whitton J. L., Tishon A., Lewicki H., Gebhard J., Cook T., Salvato M., Joly E., Oldstone M. B. Molecular analyses of a five-amino-acid cytotoxic T-lymphocyte (CTL) epitope: an immunodominant region which induces nonreciprocal CTL cross-reactivity. J Virol. 1989 Oct;63(10):4303–4310. doi: 10.1128/jvi.63.10.4303-4310.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winoto A., Urban J. L., Lan N. C., Goverman J., Hood L., Hansburg D. Predominant use of a V alpha gene segment in mouse T-cell receptors for cytochrome c. Nature. 1986 Dec 18;324(6098):679–682. doi: 10.1038/324679a0. [DOI] [PubMed] [Google Scholar]
- Wysocki L. J., Sato V. L. "Panning" for lymphocytes: a method for cell selection. Proc Natl Acad Sci U S A. 1978 Jun;75(6):2844–2848. doi: 10.1073/pnas.75.6.2844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yanagi Y., Yoshikai Y., Leggett K., Clark S. P., Aleksander I., Mak T. W. A human T cell-specific cDNA clone encodes a protein having extensive homology to immunoglobulin chains. Nature. 1984 Mar 8;308(5955):145–149. doi: 10.1038/308145a0. [DOI] [PubMed] [Google Scholar]
- Zinkernagel R. M., Doherty P. C. MHC-restricted cytotoxic T cells: studies on the biological role of polymorphic major transplantation antigens determining T-cell restriction-specificity, function, and responsiveness. Adv Immunol. 1979;27:51–177. doi: 10.1016/s0065-2776(08)60262-x. [DOI] [PubMed] [Google Scholar]
- Zinkernagel R. M., Doherty P. C. Restriction of in vitro T cell-mediated cytotoxicity in lymphocytic choriomeningitis within a syngeneic or semiallogeneic system. Nature. 1974 Apr 19;248(5450):701–702. doi: 10.1038/248701a0. [DOI] [PubMed] [Google Scholar]