Abstract
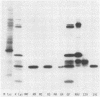
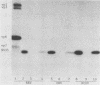
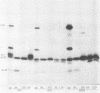
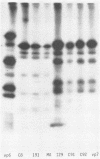
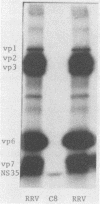
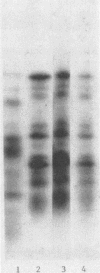
Recent studies using radiolabeled rotavirus lysates have demonstrated a 35-kilodalton viral protein that binds specifically to the surface of MA104 cells (N. Fukuhara, O. Yoshie, S. Kitakoa, and T. Konno, J. Virol. 62:2209-2218, 1988; M. Sabara, J. Gilchrist, G.R. Hudson, and L.A. Babiuk, J. Virol. 53:58-66, 1985). The binding protein was identified as vp7, an outer capsid glycoprotein and the product of rotavirus gene 9. These studies concluded that vp7 mediated viral attachment to MA104 cells and that the binding of a soluble viral protein to a cell monolayer mirrored the attachment of infectious rotavirus to permissive tissue culture cells. In the process of determining which viral protein adheres to the in vivo target cell in rotavirus infection, the mammalian enterocyte, we found that a similar 35-kilodalton rhesus rotavirus (RRV) protein bound to both MA104 cells and murine enterocytes. However, further analysis of this protein by immunoprecipitation, inhibition of glycosylation, and partial proteolysis showed that it was not the RRV gene 9 product, vp7, but the gene 8 product, NS35. Similar results were obtained by using porcine rotavirus (OSU) and bovine rotavirus (NCDV) strains. Binding studies using the in vitro-expressed products of RRV genes 8 and 9 confirmed these results. Since double-shelled virions inhibited the binding of NS35 to cells, we looked for the presence of this protein in preparations of purified virus. Examination of density gradient-purified virus preparations revealed biochemical and immunological evidence that NS35 copurifies in small amounts with double-shelled virions. Thus, these studies clearly demonstrated that when rotavirus proteins are prepared in a soluble form from infected cells, NS35, and not vp7, binds to the surfaces of MA104 cells and murine enterocytes. The observations do not confirm previous experimental results which supported the hypothesis that vp7 was the viral attachment protein. They are consistent with but do not prove the hypothesis that NS35 functions as the rotavirus attachment protein.
Full text
PDF








Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bjerknes M., Cheng H. Methods for the isolation of intact epithelium from the mouse intestine. Anat Rec. 1981 Apr;199(4):565–574. doi: 10.1002/ar.1091990412. [DOI] [PubMed] [Google Scholar]
- Both G. W., Bellamy A. R., Street J. E., Siegman L. J. A general strategy for cloning double-stranded RNA: nucleotide sequence of the Simian-11 rotavirus gene 8. Nucleic Acids Res. 1982 Nov 25;10(22):7075–7088. doi: 10.1093/nar/10.22.7075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bremont M., Cohen J., McCrae M. A. Analysis of the structural polypeptides of a porcine group C rotavirus. J Virol. 1988 Jun;62(6):2183–2185. doi: 10.1128/jvi.62.6.2183-2185.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burns J. W., Greenberg H. B., Shaw R. D., Estes M. K. Functional and topographical analyses of epitopes on the hemagglutinin (VP4) of the simian rotavirus SA11. J Virol. 1988 Jun;62(6):2164–2172. doi: 10.1128/jvi.62.6.2164-2172.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chan W. K., Penaranda M. E., Crawford S. E., Estes M. K. Two glycoproteins are produced from the rotavirus neutralization gene. Virology. 1986 Jun;151(2):243–252. doi: 10.1016/0042-6822(86)90046-2. [DOI] [PubMed] [Google Scholar]
- Cleveland D. W., Fischer S. G., Kirschner M. W., Laemmli U. K. Peptide mapping by limited proteolysis in sodium dodecyl sulfate and analysis by gel electrophoresis. J Biol Chem. 1977 Feb 10;252(3):1102–1106. [PubMed] [Google Scholar]
- Dyall-Smith M. L., Elleman T. C., Hoyne P. A., Holmes I. H., Azad A. A. Cloning and sequence of UK bovine rotavirus gene segment 7: marked sequence homology with simian rotavirus gene segment 8. Nucleic Acids Res. 1983 May 25;11(10):3351–3362. doi: 10.1093/nar/11.10.3351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Espejo R. T., López S., Arias C. Structural polypeptides of simian rotavirus SA11 and the effect of trypsin. J Virol. 1981 Jan;37(1):156–160. doi: 10.1128/jvi.37.1.156-160.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Estes M. K., Graham D. Y., Mason B. B. Proteolytic enhancement of rotavirus infectivity: molecular mechanisms. J Virol. 1981 Sep;39(3):879–888. doi: 10.1128/jvi.39.3.879-888.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Estes M. K., Graham D. Y., Ramig R. F., Ericson B. L. Heterogeneity in the structural glycoprotein (VP7) of simian rotavirus SA11. Virology. 1982 Oct 15;122(1):8–14. doi: 10.1016/0042-6822(82)90372-5. [DOI] [PubMed] [Google Scholar]
- Flores J., Perez-Schael I., Gonzalez M., Garcia D., Perez M., Daoud N., Cunto W., Chanock R. M., Kapikian A. Z. Protection against severe rotavirus diarrhoea by rhesus rotavirus vaccine in Venezuelan infants. Lancet. 1987 Apr 18;1(8538):882–884. doi: 10.1016/s0140-6736(87)92858-3. [DOI] [PubMed] [Google Scholar]
- Fukuhara N., Yoshie O., Kitaoka S., Konno T. Role of VP3 in human rotavirus internalization after target cell attachment via VP7. J Virol. 1988 Jul;62(7):2209–2218. doi: 10.1128/jvi.62.7.2209-2218.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greenberg H. B., Flores J., Kalica A. R., Wyatt R. G., Jones R. Gene coding assignments for growth restriction, neutralization and subgroup specificities of the W and DS-1 strains of human rotavirus. J Gen Virol. 1983 Feb;64(Pt 2):313–320. doi: 10.1099/0022-1317-64-2-313. [DOI] [PubMed] [Google Scholar]
- Greenberg H. B., Vo P. T., Jones R. Cultivation and characterization of three strains of murine rotavirus. J Virol. 1986 Feb;57(2):585–590. doi: 10.1128/jvi.57.2.585-590.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalica A. R., Flores J., Greenberg H. B. Identification of the rotaviral gene that codes for hemagglutination and protease-enhanced plaque formation. Virology. 1983 Feb;125(1):194–205. doi: 10.1016/0042-6822(83)90073-9. [DOI] [PubMed] [Google Scholar]
- Kouvelos K., Petric M., Middleton P. J. Comparison of bovine, simian and human rotavirus structural glycoproteins. J Gen Virol. 1984 Jul;65(Pt 7):1211–1214. doi: 10.1099/0022-1317-65-7-1211. [DOI] [PubMed] [Google Scholar]
- Kouvelos K., Petric M., Middleton P. J. Oligosaccharide composition of calf rotavirus. J Gen Virol. 1984 Jul;65(Pt 7):1159–1164. doi: 10.1099/0022-1317-65-7-1159. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lee P. W., Hayes E. C., Joklik W. K. Protein sigma 1 is the reovirus cell attachment protein. Virology. 1981 Jan 15;108(1):156–163. doi: 10.1016/0042-6822(81)90535-3. [DOI] [PubMed] [Google Scholar]
- Mackow E. R., Barnett J. W., Chan H., Greenberg H. B. The rhesus rotavirus outer capsid protein VP4 functions as a hemagglutinin and is antigenically conserved when expressed by a baculovirus recombinant. J Virol. 1989 Apr;63(4):1661–1668. doi: 10.1128/jvi.63.4.1661-1668.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mason B. B., Graham D. Y., Estes M. K. In vitro transcription and translation of simian rotavirus SA11 gene products. J Virol. 1980 Mar;33(3):1111–1121. doi: 10.1128/jvi.33.3.1111-1121.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsuno S., Inouye S. Purification of an outer capsid glycoprotein of neonatal calf diarrhea virus and preparation of its antisera. Infect Immun. 1983 Jan;39(1):155–158. doi: 10.1128/iai.39.1.155-158.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCrae M. A., Faulkner-Valle G. P. Molecular biology of rotaviruses. I. Characterization of basic growth parameters and pattern of macromolecular synthesis. J Virol. 1981 Aug;39(2):490–496. doi: 10.1128/jvi.39.2.490-496.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patton J. T., Gallegos C. O. Structure and protein composition of the rotavirus replicase particle. Virology. 1988 Oct;166(2):358–365. doi: 10.1016/0042-6822(88)90506-5. [DOI] [PubMed] [Google Scholar]
- Petrie B. L., Graham D. Y., Hanssen H., Estes M. K. Localization of rotavirus antigens in infected cells by ultrastructural immunocytochemistry. J Gen Virol. 1982 Dec;63(2):457–467. doi: 10.1099/0022-1317-63-2-457. [DOI] [PubMed] [Google Scholar]
- Sabara M., Babiuk L. A., Gilchrist J., Misra V. Effect of tunicamycin on rotavirus assembly and infectivity. J Virol. 1982 Sep;43(3):1082–1090. doi: 10.1128/jvi.43.3.1082-1090.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sabara M., Gilchrist J. E., Hudson G. R., Babiuk L. A. Preliminary characterization of an epitope involved in neutralization and cell attachment that is located on the major bovine rotavirus glycoprotein. J Virol. 1985 Jan;53(1):58–66. doi: 10.1128/jvi.53.1.58-66.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sato T., Suzuki H., Kitaoka S., Konno T., Ishida N. Patterns of polypeptide synthesis in human rotavirus infected cells. Arch Virol. 1986;90(1-2):29–40. doi: 10.1007/BF01314142. [DOI] [PubMed] [Google Scholar]
- Shaw R. D., Stoner-Ma D. L., Estes M. K., Greenberg H. B. Specific enzyme-linked immunoassay for rotavirus serotypes 1 and 3. J Clin Microbiol. 1985 Aug;22(2):286–291. doi: 10.1128/jcm.22.2.286-291.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shaw R. D., Vo P. T., Offit P. A., Coulson B. S., Greenberg H. B. Antigenic mapping of the surface proteins of rhesus rotavirus. Virology. 1986 Dec;155(2):434–451. doi: 10.1016/0042-6822(86)90205-9. [DOI] [PubMed] [Google Scholar]
- Smith M. L., Lazdins I., Holmes I. H. Coding assignments of double-stranded RNA segments of SA 11 rotavirus established by in vitro translation. J Virol. 1980 Mar;33(3):976–982. doi: 10.1128/jvi.33.3.976-982.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Svensson L., Sheshberadaran H., Vene S., Norrby E., Grandien M., Wadell G. Serum antibody responses to individual viral polypeptides in human rotavirus infections. J Gen Virol. 1987 Mar;68(Pt 3):643–651. doi: 10.1099/0022-1317-68-3-643. [DOI] [PubMed] [Google Scholar]
- Weiner H. L., Drayna D., Averill D. R., Jr, Fields B. N. Molecular basis of reovirus virulence: role of the S1 gene. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5744–5748. doi: 10.1073/pnas.74.12.5744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolf J. L., Cukor G., Blacklow N. R., Dambrauskas R., Trier J. S. Susceptibility of mice to rotavirus infection: effects of age and administration of corticosteroids. Infect Immun. 1981 Aug;33(2):565–574. doi: 10.1128/iai.33.2.565-574.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]