Abstract
Background and purpose:
The effects of dofetilide were studied in monkeys and dogs. Pharmacokinetic data were generated together with the monitoring of cardiovascular changes in order to compare effects relative to human exposure.
Experimental approach:
Beagle dogs and cynomolgus monkeys were telemetered to collect arterial blood pressure, heart rate and ECG for 6 h after selected oral doses of dofetilide. Pharmacokinetic parameters were determined for each dose.
Key results:
Dogs: increases in the QTc interval reached 56 ms in dogs dosed with 0.3 mg kg−1 of dofetilide. Premature ventricular contractions and right bundle branch block were evident at this dose, without changes in cardiovascular parameters. The mean Cmax values were 3.35 and 60.15 ng mL−1 at doses of 0.03 and 0.3 mg kg−1, respectively. Monkeys: increases in QTc intervals reached 40–50 ms after 0.03 mg kg−1. T-wave changes were observed after 0.03 mg kg−1 without changes in cardiovascular parameters. The mean Cmax values following oral doses of 0.01 and 0.03 mg kg−1 were 0.919 ng mL−1 and 1.85 ng mL−1, respectively.
Conclusions and implications:
Despite dofetilide exposure comparable to that in humans, QTc responses in dogs were greater than those reported in humans. A comparable human dose used in the monkey achieved only half of the exposure but was associated with twofold greater increases in QTc. Our data support the view that safety risk assessments of new drugs in animal models should ensure that the clinical therapeutic range of exposure is achieved and any untoward effects interpreted accordingly.
Keywords: dofetilide, telemetry, safety pharmacology, cardiovascular, pharmacokinetics, arrhythmias, beagle dogs, cynomolgus monkeys, QTc prolongation, torsades de pointes
Introduction
In the past two decades, a number of patients have exhibited severe cardiac anomalies produced by non-cardiovascular (CV) drugs (Crumb and Cavero, 1999). These drugs were not expected to produce life-threatening situations based on known mechanisms of action. Such drugs belonged to a wide variety of pharmacological classes: psychotherapeutics (sertindole, droperidol, fluoxetine (Belardinelli et al., 2003; Olsen, 2005)), antimalarial agents (chloroquinine), antibiotics (erythromycin, clarithromycin, sporofloxin (Tschida et al., 1996; Schaffer et al., 2002; Stanat et al., 2003)), fluoroquinolones (Demolis et al., 2000; Noel et al., 2004), antifungal agents (ketoconazole, intraconazole (Cubeddu, 2003)), tricyclic antidepressants (desipramine, doxepin, fluoxetine, imipramine, sertraline (Cubeddu, 2003)) and selective H1 receptor antagonists (astemizole, terfenadine (Pratt et al., 1996)). In some cases, these agents are believed to have contributed to the occurrence of sudden cardiac death associated with the prolongation of ventricular repolarization manifested as lengthening of the QT interval of the ECG, and the development of polymorphic ventricular tachycardia, such as torsades de pointes (TdP) (Crumb and Cavero, 1999; Fermini and Fossa, 2003).
The QT interval of the ECG is a frequently used surrogate variable to gauge the effects of a test substance on ventricular repolarization. The duration of the QT interval is a measure of ventricular repolarization and its prolongation has become associated with the risk of sudden cardiac death. Additional CV parameters of interest in risk assessments include the following: arterial blood pressure, heart rate, PR interval, QRS duration, the presence of U-waves and arrhythmias (Smith et al., 1992). However, not all drugs that prolong the QT interval are associated with an increased risk for TdP. Additionally, there are reports that could not demonstrate a correlation between the incidence of drug-induced TdP and the magnitude of the increase in the QT interval (Belardinelli et al., 2003). Evidence of QT interval prolongation during preclinical drug development is the subject of increased regulatory review and is considered a significant risk factor in predicting human safety of new drug chemical entities (Fermini and Fossa, 2003). The Food and Drug Administration has issued guidance documents (ICH guidelines S7A and S7B) that recommend in vivo/in vitro electrophysiology studies in intact animal and tissue/cellular models for the investigation of potential ventricular repolarization effects of drugs to induce cardiac arrhythmias (Anon, 2001, 2005). In these studies, QT interval duration (collected in vivo) is used as an indicator for any ventricular repolarization effects that a potential drug may possess.
We report here the CV effects of a Class III anti-arrhythmic, dofetilide, a drug derived from D-sotalol, the non-β-adrenoceptor-blocking moiety of racemic sotalol. Dofetilide is known to cause QT prolongation and TdP in humans or surrogate arrhythmias in animal studies and experimental cardiac preparations (Belardinelli et al., 2003). Animal studies have demonstrated that dofetilide selectively inhibits the time-dependent outward K+ current (Ikr) in the myocardium and increases the ventricular effective refractory period and action potential duration without affecting the fast inward Na+ current (Smith et al., 1992). We have studied the effects of dofetilide in two animal species commonly employed in preclinical drug development: cynomolgus monkeys and beagle dogs. In combination with our pharmacokinetic (PK) data, the CV changes that occur following doses of dofetilide, chosen in relation to reported exposures achieved in humans, are described.
Materials and methods
All animal procedures complied with the NIH guidelines. Six male beagle dogs (9–12 kg) and six male cynomolgus monkeys (4–6 kg) were instrumented with radiotelemetry transmitters for collection of arterial blood pressure, heart rate and Lead II ECGs.
Telemetry instrumentation
Anaesthesia was induced by intravenous administration of propofol (5–7 mg kg−1) to dogs or ketaset (0.1 mL kg−1) to monkeys. A cuffed endotracheal tube was inserted into the trachea connected to a semi-closed loop anaesthetic machine (Surgivet, Waukesha, WI, USA). Balanced gaseous anaesthesia (medical grade oxygen and approximately 2–4% isoflurane) was used throughout the procedure (2–3 h) to maintain a surgical plane of anaesthesia.
Once anaesthetized, each animal was shaved and surgically scrubbed, encompassing surgical sites of the right inguinal area, chest and right lateral abdomen. Throughout the surgical procedure, the animal was appropriately monitored (Pro-pac10, Beaverton, OR, USA) for continuous assessment of vital signs. An incision was made along the right medial thigh and the femoral artery exposed by blunt dissection. Another incision was made in the lateral lumbar area cranial to the iliac crest for a tunnelling needle to be passed subcutaneously from the incision on the medial thigh to the lumbar incision. A blood pressure catheter was passed through the needle from the lumbar incision and the needle removed. The femoral artery was ligated distally and incised to insert a catheter that was advanced until the tip resided in the femoral artery/abdominal aorta. It was then secured by ligation. The incision in the lumbar area was enlarged and a subcutaneous pocket created in the left dorsal lumbar area to hold the transmitter body. The transmitter (Data Sciences International, St Paul, MN, USA) was inserted into the subcutaneous pocket and a suture used to hold the transmitter body. The two ECG leads (positive and negative) were positioned using a trocar to tunnel each lead to the appropriate region (base of the right side of the neck for the negative lead and the 5th–6th intercostal space on the left side of the thoracic cage for the positive lead). Approximately 1 cm of the silastic insulator was removed from each lead and the exposed wire coiled into a tight loop. This loop of wire was then placed and secured in place using non-absorbable suture material. The incision was closed using nylon suture, absorbable sutures or sterile staples. Postoperative recovery lasted 2 weeks during which supplemental analgesics/antibiotics were administered (at least 7 days). Once the postoperative recovery period was complete, the animal was examined for study acceptability by the staff veterinarian and the implanted transmitter signal verified.
Study design
The study was divided into two phases. The first CV phase used two separate crossover designs consisting of six male beagle dogs and six male cynomolgus monkeys instrumented with radiotelemetry transmitters being given a single oral (gavage) dose of either the vehicle (0.4% aqueous methylcellulose) or one of two dose levels of dofetilide: 0.03 or 0.3 mg kg−1 (dog); 0.01 or 0.03 mg kg−1 (monkey). At least 72 h elapsed between the doses to ensure that each animal received each dose with an adequate washout period. ECGs, heart rate and arterial blood pressure data were recorded using a DSI/Notocord data acquizition system (Paris, France) 1 h prior to and then continuously for at least 6 h following dofetilide/vehicle administration. A blood sample for determination of dofetilide plasma concentrations was collected from all dogs (via jugular venepuncture) and monkeys (via femoral artery/vein or saphenous vein) on each of the three dose days at approximately 6 h post-dose.
The second phase of each study consisted of an abbreviated PK investigation. Blood samples were collected from dogs (via jugular venepuncture) and monkeys (via femoral or saphenous vein) at 0, 0.5, 1, 2, 4, 6 and 8 h post-dose. Monkeys (three per group) were administered 0.01 and 0.03 mg kg−1 dofetilide and dogs (three per group) were dosed with 0.03 or 0.3 mg kg−1 of dofetilide. The animals in the PK phase of the investigation were a subset of the animals used during the CV studies. Blood samples were collected and the plasma assayed for dofetilide concentrations using a liquid chromatography with a standard tandem mass spectrometry assay. The PK phase plasma concentration–time data were evaluated and Cmax (maximum observed concentration), Tmax (time of Cmax) and AUC (area under the curve) were identified for each dose level. Cardiovascular parameters were not collected during the PK phase of the investigation.
ECG analysis
An ECG waveform morphology assessment, for the entire monitoring period, was completed for each dog and monkey by a safety pharmacologist or veterinarian cardiologist. Standard ECG intervals (PR, RR, QRS and QT) were automatically measured by the Notocord data acquizition system and reported as 10 min averages. The signals were collected with Data Sciences International Systems hardware. PR and QT manual overreads were conducted as appropriate. The manual measurements were completed from 50 mm s−1 tracings at 30 min intervals. A mean of the three waveforms per time point are reported. The QT intervals were corrected for heart rate (QTc) values using the formula by Funck-Bretano and Jaillon (1993).
Statistical analysis
The systemic blood pressures and heart rate data collected via radiotelemetry were averaged across consecutive 10 min time intervals during the 6 h post-dosing period for each animal. A baseline of a stable 10 min period was selected prior to the start of dosing. The baseline measurement for each animal was subtracted on each dosing day from the animal's post-dosing 10 min averages. Dose-level averages (and accompanying s.d.) of these baseline-adjusted 10 min averages were calculated across study animals. Repeated measures ANOVA were applied to these baseline-adjusted averages. Pairwise t-tests (Scheffe, 1959) were applied within the ANOVA to identify the presence of significant differences of the dose levels relative to vehicle. These t-tests were performed at a 0.05/2=0.025 significance level, so that the overall error rate across the comparisons in a given interval was not higher than 0.05. ECG interval data (reported at 30 min intervals) were analysed using repeated-measures ANOVA techniques similar to the systemic blood pressure and heart rate data described above.
Toxicokinetic analysis
In each species, a mean plasma concentration for each dose level of dofetilide was calculated. On a subsequent study day (PK phase), three animals per dose group were administered 0.03 or 0.3 mg kg−1 (dogs) and 0.01 or 0.03 mg kg−1 (monkey) of dofetilide. Blood samples (targeting 1.5 mL per sample) were collected from a femoral or saphenous vein (monkey) or jugular vein (dog) at 0, 0.5, 1, 2, 4, 6 (dog and monkey) and 8 h (dog only) post-dose. Plasma was obtained by centrifugation, frozen and subsequently analysed for dofetilide concentrations.
Reagents
Dofetilide was provided by the Schering-Plough Corporation. Its chemical formula is C19H27N3O5S2 (Rasmussen et al., 1992).
Results
Beagle dog
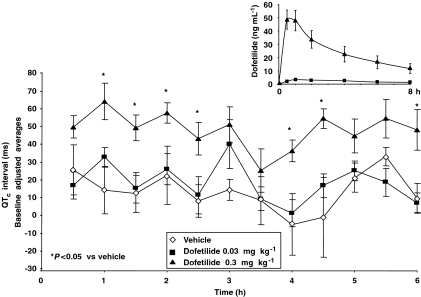
As anticipated, the QTc intervals of the ECG were increased (by 17–56 ms) in dogs dosed with 0.3 mg kg−1 of dofetilide, throughout the post-dose period (1–6 h). Although individual dogs dosed with 0.03 mg kg−1 of dofetilide showed some increase in QTc (ranging from 1 to 52 ms) intervals, the group did not exhibit consistent and robust increases in QTc prolongation during the study compared with vehicle-dosed animals (Figure 1). This inconsistency may have been a result of varying individual exposures in this cohort of animals. Changes in ECG morphology were not observed during the pre-dose period or for the vehicle-dosed animals. ECG morphology changes were noted in dogs administered 0.3 mg kg−1 (that is, mean plasma concentrations of dofetilide ranged from 16.5 to 48.7 ng mL−1) from 30 min post-dose and persisted for the 6 h post-dose monitoring period. These changes consisted primarily of premature ventricular contractions, T-wave anomalies (Dart–Dome complexes) and right bundle branch block. No other dofetilide-related changes were identified in the other ECG intervals (PR, QRS and RR) or haemodynamic measures (systolic, diastolic, mean arterial pressure or heart rate; see Table 1).
Figure 1.
Baseline adjusted QTc interval averages in beagle dogs. Comparison of baseline adjusted QTc intervals (±s.e.mean) for dogs given 0.03 or 0.3 mg kg−1 dofetilide. Inset: dofetilide plasma levels from subset of dogs encompassing the cardiovascular monitoring period.
Table 1.
Haemodynamic and ECG interval data at baseline and at Tmax following dofetilide administration to the dog and monkey
|
Haemodynamic and ECG parameters at baseline and at ∼Tmax (dog) | ||||||||
|---|---|---|---|---|---|---|---|---|
| Dose (mg kg−1) |
Systolic (mm Hg) |
Diastolic (mm Hg) |
MAP (mm Hg) |
HR (b.p.m.) |
||||
| Baseline | Tmax | Baseline | Tmax | Baseline | Tmax | Baseline | Tmax | |
| 0 | 158±8 | 154±5 | 82±12 | 81±9 | 110±11 | 109±8 | 104±30 | 112±30 |
| 0.03 | 154±12 | 154±12 | 80±12 | 80±7 | 108±11 | 108±8 | 106±14 | 109±13 |
| 0.30 | 154±15 | 144±12 | 78±11 | 75±10 | 105±13 | 99±11 | 91±10 | 91±11 |
| Dose (mg kg−1) |
RR (ms) |
PR (ms) |
QRS (ms) |
QT (ms) |
||||
| |
Baseline |
Tmax |
Baseline |
Tmax |
Baseline |
Tmax |
Baseline |
Tmax |
| 0 | 700±146 | 627±157 | 98±15 | 105±10 | 34±3 | 34±3 | 210±16 | 213±28 |
| 0.03 | 666±120 | 627±68 | 98±11 | 105±5 | 33±3 | 34±4 | 212±12 | 222±11 |
| 0.3 | 755±82 | 788±115 | 103±6 | 116±8 | 32±5 | 32±5 | 211±9 | 258±11* |
|
Haemodynamic and ECG parameters at baseline and at ∼Tmax (monkey) | ||||||||
| Dose (mg kg−1) |
Systolic (mm Hg) |
Diastolic (mm Hg) |
MAP (mm Hg) |
HR (b.p.m.) |
||||
| |
Baseline |
Tmax |
Baseline |
Tmax |
Baseline |
Tmax |
Baseline |
Tmax |
| 0 | 131±30 | 128±26 | 81±17 | 81±16 | 105±22 | 104±20 | 142±33 | 99±19 |
| 0.01 | 130±38 | 126.±29 | 81±23 | 78±16 | 105±29 | 101±22 | 145±32 | 139±34 |
| 0.03 | 133±26 | 122±24 | 83±16 | 77±20 | 107±20 | 99±19 | 144±28 | 145±20 |
| Dose (mg kg−1) |
RR (ms) |
PR (ms) |
QRS (ms) |
QT (ms) |
||||
| |
Baseline |
Tmax |
Baseline |
Tmax |
Baseline |
Tmax |
Baseline |
Tmax |
| 0 | 455±104 | 408±44 | 96±11 | 94±10 | 32±4 | 31±4 | 241±36 | 215±13 |
| 0.01 | 449±98 | 425±100 | 94±7 | 93±12 | 32±5 | 32±5 | 238±33 | 223±46 |
| 0.03 | 437±77 | 424±56 | 93±9 | 93±7 | 32±5 | 31±4 | 238±24 | 250±35** |
Abbreviations: Tmax, time of Cmax; HR, heart rate; MAP, mean arterial pressure.
Dog: Tmax ∼1.3 h post-dose after 0.03 mg kg−1 and 0.75 h post-dose after 0.3 mg kg−1 in this investigation. Data are mean and s.e.mean. *Statistically significant (P⩽0.025) difference compared with control.
Monkey: Tmax ∼1 h post-dose after 0.03 mg kg−1 and ∼2 h post-dose after 0.01 mg kg−1 in this investigation. Data are mean and s.e.mean. **Statistically significant (P⩽0.025) difference compared with control.
Plasma analysis
Blood samples were collected from each dog on its respective day of dosing, at 6 h post-dosing. The mean concentration and coefficient of variation (%CV=s.d./mean × 100) of dofetilide at this time was 1.59 (±0.03) and 23.1 (±3.68) ng mL−1, after a dose of 0.03 and 0.3 mg kg−1, respectively, confirming exposure in this group of animals.
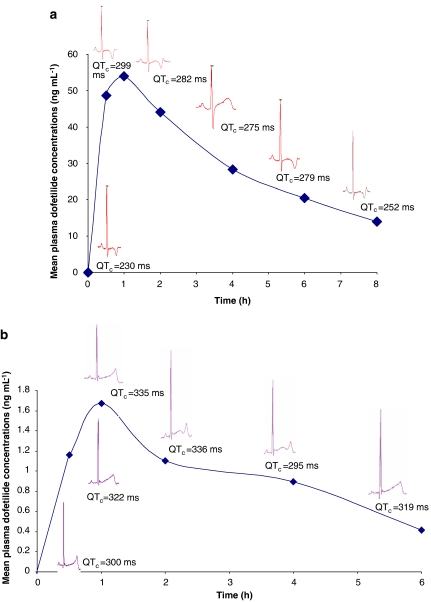
Following the abbreviated PK phase of the study, the mean Cmax values were 3.35 and 60.15 ng mL−1 at doses of 0.03 and 0.3 mg kg−1, respectively. The 0–8 h mean AUC values were determined to be 11.8 and 261.9 h ng mL−1 after the 0.03 and 0.3 mg kg−1 doses, respectively. Increases in QTc were directly related to the dofetilide dose and plasma concentrations. An example of serial plasma concentrations from one dog dosed with 0.3 mg kg−1 and its representative Lead II ECG waveforms plotted at the corresponding time points is shown in Figure 3a.
Figure 3.
(a, b) Dofetilide concentrations (ng mL−1) with superimposed examples of QT interval prolongation recorded during the cardiovascular phase of the investigation in dog, 0.3 mg kg−1 (a) and monkey, 0.03 mg kg−1 (b).
Cynomolgus monkey
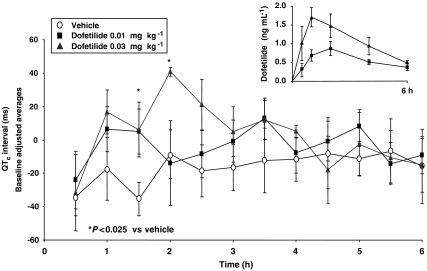
Dofetilide-related increases in QTc (40–50 ms) were observed in monkeys dosed with 0.03 mg kg−1 at 1.5–2 h post-dose and with 0.01 mg kg−1 at 1.5 h post-dose (40 ms) compared with vehicle treatment (Figure 2). T-wave morphology changes were observed only after administration of 0.03 mg kg−1. There were no changes in other CV monitored parameters (systolic, diastolic, mean arterial pressure; heart rate (Table 1); or other ECG intervals: PR, QRS or RR; (Figure 3b)) throughout the 6 h observation period.
Figure 2.
Baseline adjusted QTc interval averages in cynomolgus monkeys. Comparison of baseline adjusted QTc intervals (±s.e.mean) for cynomolgus monkeys given 0.01 or 0.03 mg kg−1 dofetilide. Inset: dofetilide plasma levels from subset of monkeys encompassing the cardiovascular monitoring period.
Plasma analysis
During the CV phase, a single blood sample was obtained in this series of dosed monkeys. Mean (%CV) dofetilide plasma concentrations (6 h) following administration of 0.01 and 0.03 mg kg−1 dofetilide were 0.284 (42) and 0.505 (20) ng mL−1 for the 0.01 and 0.03 mg kg−1 dose groups, respectively, confirming exposure in this group of animals. From the abbreviated PK phase of the study with a subset of the monkeys from the CV phase of the study, the mean Cmax, Tmax and AUC (0–6 h) values for the monkeys dosed with dofetilide were 0.919 (33) ng mL−1, 2.33 h and 3.38 h ng mL−1, respectively (for 0.01 mg kg−1 dofetilide), and 1.85 (15) ng mL−1, 0.833 (35) h and 6.37 (25) h ng mL−1, respectively (for 0.03 mg kg−1 dofetilide).
Discussion
The pharmaceutical industry is experiencing increasing pressure from regulatory agencies to identify, in the preclinical stages, any potential CV risks associated with new drug entities. In clinical situations there are numerous documented case reports in which patients have exhibited adverse events following administration of non-CV drugs that were not expected to produce life-threatening situations (Crumb and Cavero, 1999). In response, the Food and Drug Administration has adopted the following guidance documents:
International Conference on Harmonization guideline on safety pharmacology (S7A; Anon, 2001) and the nonclinical evaluation of the potential for delayed ventricular repolarization (QT interval prolongation) by human pharmaceuticals (S7B; Anon, 2005).
These guidelines require investigative studies to evaluate the acute risk of new drug candidates. These studies are recommended to be conducted in appropriate laboratory species. The pharmaceutical industry typically uses the beagle dog or cynomolgus monkey for these studies (Friedrichs et al., 2005). Cardiovascular evaluation routinely includes a review of arterial pressure and heart rate as well as ECG morphology and ECG interval duration (PR, RR, QRS and QT) following acute administration of the test article. Dofetilide, either by the oral or intravenous route of administration, has been shown to prolong the QT and QTc intervals in phase I clinical trials, as well as other studies in humans (Allen et al., 2000). Dofetilide administered via the intravenous route in humans results in a hysteresis loop between dofetilide plasma concentrations and QTc interval (Rasmussen et al., 1992), whereas oral administration of dofetilide results in dose-proportional linear relationships (Rasmussen et al., 1992; LeCoz et al., 1995).The results of our studies confirm some characteristics reported in humans: oral administration of dofetilide results in a linear relationship between plasma concentration and effect.
Independent of the route of administration, dofetilide therapy causes prolongation of the QTc interval, which, when combined with precipitating factors, can increase the risk of initiating TdP in humans. In our investigation, we used dofetilide, also known to prolong QT intervals in dogs and cynomolgus monkeys (Belardinelli et al., 2003; Watkins et al., 2003), to evaluate differences in ECG parameters at selected exposure levels.
ECG morphology changes after 0.3 mg kg−1 in the dog induced a variety of conduction disturbances: secondary atrioventricular block (Mobitz), premature ventricular contractions, T-wave anomalies (Dart–Dome complexes) and right bundle branch block. The range of plasma concentrations of dofetilide was 20–60 ng mL−1. In contrast, changes in ECG morphology were not observed in dogs administered 0.03 mg kg−1 of dofetilide in the presence of plasma concentrations of approximately 1–3 ng mL−1.
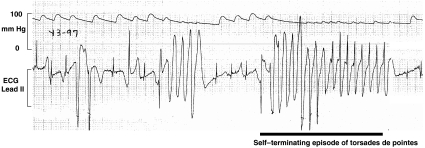
QT prolongation was noted in cynomolgus monkeys following 0.03 mg kg−1 dofetilide, although the major change in ECG morphology was T-wave prolongation. Higher doses of dofetilide in pilot studies (0.1 and 0.2 mg kg−1) have been shown to produce premature ventricular depolarizations and episodes of TdP that were self-terminating (Watkins et al., 2003) (Figure 4).
Figure 4.
Example of dofetilide-induced torsades de pointes in the cynomolgus monkey after a dose of 0.2 mg kg−1. The upper trace depicts arterial blood pressure and the lower trace shows Lead II ECG and an example of a short episode of self-terminating torsades de pointes (bar below ECG trace), which returned to sinus rhythm shortly thereafter. Note the ECG irregularities preceding the torsades de pointes.
The incidence of ECG morphology changes in dogs administered 0.3 mg kg−1 has not been reported in other species. Our PK data may explain the profound ECG morphology alterations observed in dogs given 0.3 mg kg−1. A 20-fold increase in Cmax (60 ng mL−1) and a 15-fold increase in AUC (262 h ng mL−1) was achieved in dogs after 0.3 mg kg−1 compared with the Cmax achieved by dogs given 0.03 mg kg−1 (3 ng mL−1), and devoid of ECG morphology changes, that is, a 10-fold increase in dose, resulting in greater than dose-proportional (20- and 15-fold) increase in Cmax and AUC, respectively. Comparison of PK parameters for selected species (Table 2) indicates that the PK of high doses of dofetilide administered to dogs is quite different from that reported in the other species.
Table 2.
Mean comparative pharmacokinetic parameters in beagle dogs, cynomolgus monkeys and humans
| Species | Dose | Cmax | Tmax | AUC | Max. Δin QTc | References | |
|---|---|---|---|---|---|---|---|
| (mg kg−1) | (mg) | (ng mL−1) | (h) | (ng mL−1) | (ms) | ||
| Monkey | 0.01 | 0.05 | 0.919 | 2.33 | 3.38 | 35 | Present study |
| 0.03 | 0.13 | 1.85 | 0.833 | 6.37 | 50 | Present study | |
| Dog | 0.03 | 0.3 | 3.35 | 1.33 | 17.84 | 44 | Present study |
| 0.3 | 3.5 | 60.15 | 0.75 | 261.89 | 64 | Present study | |
| Human | 0.001a | 0.7 | — | — | — | No Effect | Tham et al. (1993) |
| 0.002a | 0.15 | — | — | — | No Effect | Tham et al. (1993) | |
| 0.005a | 0.37 | — | 2 | — | 29 | Tham et al. (1993) | |
| 0.006b | 0.5 | 1.9 | 2.6 | 19.4 | 61 | LeCoz et al. (1995) | |
| 0.006c | 0.5 | 2.05 | 1.8 | 21.3 | 14.5–20.7 | Abel et al. (2000) | |
| 0.02d | 1.0e | 1.7 | 2.29 | 8.61 | — | Allen et al. (2000) | |
| 0.022d | 1.5f | 2.26 | 2.13 | 10.81 | 17–19 | Allen et al. (2000) | |
| 0.04d | 2.5g | 3.55 | 2.29 | 18.89 | — | Allen et al. (2000) |
Abbreviations: AUC, area under the curve; Cmax, maximum observed concentration; Tmax, time of Cmax; QTc, QT interval corrected for heart rate.
Estimate from 10 male subjects with a mean body weight of 74 kg.
Estimate from 10 healthy male subjects with a mean body weight of 58–86 kg.
Estimate from 20 healthy male subjects with a mean body weight of 79 kg.
Estimate from 25 male subjects with a mean body weight of 68 kg.
Based on 330 μg three times daily.
Based on 500 μg three times daily.
Based on 830 μg three times daily.
Humans administered dofetilide (oral administration) typically exhibit a linear, dose-proportional increase in Cmax and AUC (LeCoz et al., 1995; Abel et al., 2000; Allen et al., 2000). However, we found that in dogs given high doses of dofetilide, the relationship between dose and AUC is nonlinear. Nonlinear PK has been described for other drugs at high concentrations and is typically the result of saturating a metabolic carrier-mediated system in the body or individual organs (Schoenwald, 2002). The transition from first-order to nonlinear PK could have led to increased concentrations of dofetilide. As the chemical structure of dofetilide contains a tertiary amine with a pKa of 8.0, it is considered moderately lipophilic (Walker et al., 1996a). This differs from the parent entity, sotalol, which is one of the more hydrophilic compounds of this class (Walker et al., 1996b). Dofetilide's lipophilicity, a primary determinant of myocardial uptake (Lullmann et al., 1979), coupled with the saturation kinetics, may have resulted in greater dofetilide concentrations reaching the heart to produce Ikr blockade, which affects ventricular repolarization and thereby induces myocardial electrical instability. Although speculative, this hypothesis may explain the higher prevalence of arrhythmias in these dogs, which would not otherwise be expected at these doses. However, tissue concentrations of dofetilide achieved in the heart were not measured in our investigation.
Monkeys administered dofetilide at a dose of 0.03 mg kg−1 experienced increases in QTc intervals of 40–50 ms. In general, plasma concentrations above 1 ng mL−1 were associated with QTc increases of 10–50 ms. In contrast, a comparable human dose of 0.04 mg kg−1 produces Cmax and AUC values (Allen et al., 2000) that are 2–3 times that achieved in the monkey. Paradoxically, the increases in QTc interval were twofold greater in the monkey than those reported in man. Others have reported higher maximum changes in QTc in man (Tham et al., 1993; LeCoz et al., 1995). Likewise, a dose of 0.03 mg kg−1 in select dogs (that is, dogs that responded to this dose) was associated with a maximum QTc interval increase of 44 ms but devoid of conduction disturbances. Possessing a comparable PK profile to humans at the lowest dose tested in our dogs, increases in QTc intervals in select dogs are 1.5- to 2-fold greater than the values reported in humans.
Thus, despite dofetilide exposure comparable to that in humans, the QTc response exhibited in the dog is greater than that experienced in humans, whereas a comparable human dose in the monkey is associated with exposures that are half that of humans, but associated with QTc interval increases that are twofold greater.
The results of this investigation indicate that using relevant therapeutic doses of dofetilide in both the dog and monkey produce CV ‘responses' that may not be completely aligned to the effects seen in man. The availability of human data for dofetilide allowed for a retrospective evaluation to take place. Although it is imperative that a safety risk assessment is generated from the appropriate species with supporting PK, the information must be put into perspective, or investigators could identify untoward events that may not necessarily be relevant to the therapeutic range of the drug in humans.
Acknowledgments
We thank Ms K Pula, Mr R Tedesco and Dr R Watkins for their contributions to this paper.
Abbreviations
- AUC
area under the curve
- Cmax
maximum observed concentration
- PK
pharmacokinetic
- QTc
QT interval corrected for heart rate
- TdP
torsades de pointes
- Tmax
time of Cmax
Conflict of interest
The authors state no conflict of interest.
References
- Abel S, Nichols DJ, Brearley CJ, Eve MD. Effect of cimedtidine and ranitidine on pharmacokinetics and pharmacodynamics of a single dose of dofetilide. Br J Clin Pharmacol. 2000;49:64–71. doi: 10.1046/j.1365-2125.2000.00114.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allen MJ, Nichols NJ, Oliver SD. The pharmacokinetics and pharmacodynamics of oral dofetilide after twice daily and three times daily dosing. Br J Clin Pharmacol. 2000;50:247–253. doi: 10.1046/j.1365-2125.2000.00243.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anon ICH S7A Safety pharmacology studies for human pharmaceuticals, implemented by MHLW, CPMP, FDA. Federal Register. 2001;66:36791–36792. [PubMed] [Google Scholar]
- Anon ICHS7B Guidance on safety pharmacology studies for assessing the potential for delayed ventricular repolarization (QT interval prolongation) by human pharmaceuticals. 2005. [PubMed]
- Belardinelli L, Antzelevitch C, Vos MA. Assessing predictors of drug-induced torsade de pointes. Trends Pharmacol Sci. 2003;24:619–625. doi: 10.1016/j.tips.2003.10.002. [DOI] [PubMed] [Google Scholar]
- Cubeddu LX. QT prolongation and fatal arrhythmias: a review of clinical implications and effects of drugs. Am J Ther. 2003;10:452–457. doi: 10.1097/00045391-200311000-00013. [DOI] [PubMed] [Google Scholar]
- Crumb W, Cavero I. QT Interval prolongation by non-cardiovascular drugs: issues and solutions for novel drug development. PSTT. 1999;2:270–280. doi: 10.1016/s1461-5347(99)00172-8. [DOI] [PubMed] [Google Scholar]
- Demolis JL, Kubitza D, Tenneze L, Funck-Bretano C. Effect of a single oral dose of moxifloxacin (400 and 800 mg) on ventricular repolarization in healthy subjects. Clin Pharmacol Ther. 2000;68:658–666. doi: 10.1067/mcp.2000.111482. [DOI] [PubMed] [Google Scholar]
- Fermini B, Fossa AA. The impact of drug-induced QT interval prolongation on drug discovery and development. Nat Rev. 2003;2:439–447. doi: 10.1038/nrd1108. [DOI] [PubMed] [Google Scholar]
- Friedrichs GS, Patmore L, Bass A. Non-clinical evaluation of ventricular repolarization (ICH S7B): results of an interim survey of international pharmaceutical companies. J Pharmacol Toxicol Methods. 2005;52:6–11. doi: 10.1016/j.vascn.2005.05.001. [DOI] [PubMed] [Google Scholar]
- Funck-Bretano C, Jaillon P. Rate-corrected QT interval, techniques and limitations. Am J Cardiol. 1993;72:17B–22B. doi: 10.1016/0002-9149(93)90035-b. [DOI] [PubMed] [Google Scholar]
- LeCoz F, Funck-Brentano C, Morell T, Ghadanfar MM, Jaillan P. Pharmacokinetics and pharmacodynamics modeling of the effects of oral and intravenous administration of dofetilide on ventricular repolarization. Clin Pharmacol Ther. 1995;57:533–542. doi: 10.1016/0009-9236(95)90038-1. [DOI] [PubMed] [Google Scholar]
- Lullmann H, Timmermans PB, Ziegler A. Accumulation of drugs by resting or beating cardiac tissue. Eur J Pharmacol. 1979;60:277–285. doi: 10.1016/0014-2999(79)90231-0. [DOI] [PubMed] [Google Scholar]
- Noel TM, Olsen KM, McCartan MA, Speidal KM, Miller MA, Levit AV. Pharmacologic predictors of QTc prolongation and proarrhythmia in the adult medical intensive care unit. Crit Care Med. 2004;32 Suppl:154A. [Google Scholar]
- Olsen KM. Pharmacologic agents associated with QT interval prolongation. J Family Pract. 2005. pp. S8–S14. [PubMed]
- Pratt CM, Ruberg S, Morganroth J, McNutt S, Ruskin J, Moye L. Dose–response relation between terfenadine (seldane) and the QTc. Distinguishing a drug effect from spontaneous variability. Am Heart J. 1996;131:472–480. doi: 10.1016/s0002-8703(96)90525-6. [DOI] [PubMed] [Google Scholar]
- Rasmussen HS, Allen MJ, Blackburn KJ, Buteus GS, Dalrymple HW. Dofetilide, a novel class III antiarrhythmic agent. J Cardiovasc Pharmacol. 1992;20 Suppl 2:S96–S105. [PubMed] [Google Scholar]
- Schaffer D, Singer S, Korvick J, Honig P. Concomitant risk factors in reports of torsades de pointes associated with macrolide use. Review of the United States Food and Drug Administration Adverse Event Reporting System. Clin Infect Dis. 2002;35:197–200. doi: 10.1086/340861. [DOI] [PubMed] [Google Scholar]
- Scheffe H. The Analysis of Variance. John Wiley & Sons Inc.: New York; 1959. [Google Scholar]
- Schoenwald RD. Pharmacokinetics in Drug Discovery and Development 2002CRC Press; 5–10.Boca Raton, FL, USA, pp [Google Scholar]
- Smith DA, Rasmussen HS, Stopher DA, Walker DK. Pharmacokinetics and metabolism of dofetilide in mouse, rat, dog and man. Xenobiotica. 1992;22:709–719. doi: 10.3109/00498259209053133. [DOI] [PubMed] [Google Scholar]
- Stanat SJ, Carlton CG, Crumb WJ, Jr, Agrawal KC, Clarkson CW. Characterization of the inhibitory effects of erythromycin and clarithromycin on the HERG potassium channel. Mol Cell Biochem. 2003;254:1–7. doi: 10.1023/a:1027309703313. [DOI] [PubMed] [Google Scholar]
- Tham TCK, MacLennan BA, Burke MT, Harron DWG. Pharmacodynamics and pharmacokinetics of the class III antiarrhythmic agent dofetilide (UK-68798) in humans. J Cardiovasc Pharmacol. 1993;21:507–512. doi: 10.1097/00005344-199303000-00024. [DOI] [PubMed] [Google Scholar]
- Tschida SJ, Guay DR, Straka RJ, Hoey LL, Johanning R, Vance-Bryan K. QTc-interval prolongation associated with slow intravenous erythromycin lacrobionate infusions in critically ill patients: a prospective evaluation and review of literature. Pharmacotherapy. 1996;16:663–674. [PubMed] [Google Scholar]
- Walker DK, Alabaster CT, Congreve GS, Hargreaves MB, Hyland R, Jones BC, et al. Significance of metabolism in the disposition and action of the antidysrhythmic drug, dofetilide. Drug Metab Disp. 1996a;24:447–455. [PubMed] [Google Scholar]
- Walker DK, Beaumont KC, Stopher DA, Smith DA. Pharmacokinetics of a series of bis (methane sulphonamide-alkyl) amines in the beagle dog. Xenobiotica. 1996b;26:1101–1111. doi: 10.3109/00498259609167425. [DOI] [PubMed] [Google Scholar]
- Watkins R, Pula K, Chintala M, Bass AS.(Abstract) Dofetilide prolongs QT interval and induces torsades de pointes in conscious telemetered monkeys 2003. Cardiovascular Risk Assessment Workshop. International Life Sciences Institute Health, Environmental Sciences Institute, Arlington, VA June