Abstract
Elite suppressors (ES) are untreated human immunodeficiency virus type 1 (HIV-1)-infected patients who maintain viral loads of <50 copies/ml. The mechanisms involved in this control of viral replication remain unclear. Prior studies suggested that these patients, as well as long-term nonprogressors, are infected with defective HIV-1 variants. Other reports have shown that the HLA-B*27 and -B*57 alleles are overrepresented in these patients, suggesting that host factors play a role in the control of viral replication. In order to distinguish between these hypotheses, we studied differences in viral isolates and immune responses of an HIV-1 transmission pair. While both patients are HLA-B*57 positive, the transmitter progressed to AIDS, whereas the recipient, who is also HLA-B*27 positive, is an ES. Isolates from both patients were replication competent and contained the T242N escape mutation in Gag, which is known to decrease viral fitness. While the acquisition of compensatory mutations occurred in isolates from the progressor, a superior HIV-specific CD8+ T-cell response in the ES appears to have prevented viral replication and thus the evolution toward a more fit variant. In addition, CD8+ T cells in the ES have selected for a rare mutation in an immunodominant HLA-B*27-restricted Gag epitope, which also has a negative impact on fitness. The results strongly suggest that through direct and indirect mechanisms, CD8+ T cells in some ES control HIV-1 isolates are capable of causing profound immunosuppression.
The mechanisms by which a unique subset of human immunodeficiency virus type 1 (HIV-1)-infected individuals called elite suppressors (ES) control viral replication remain largely unknown (16, 43). While several studies suggested that long-term nonprogressors (LTNP) and ES are infected with defective viruses (2, 12, 14, 24, 29, 33, 36, 44, 45, 46), it is clear that host factors such as the expression of protective HLA alleles also play an important role in the control of viremia in LTNP (25, 27) and ES (17, 30, 39, 41). We report here the case of a person with a history of injection drug use who was diagnosed with AIDS 10 years ago when he developed two opportunistic infections, Pneumocystis jirovecii pneumonia and probable central nervous system toxoplasmosis. His viral load was 200,000 copies/ml. He was placed on highly active antiretroviral therapy (HAART) shortly thereafter, and he has maintained undetectable viral loads. His first CD4 count, obtained after he initiated HAART, was 266 cells/μl. His partner (ES9), whose only risk factor was sexual contact with the patient, tested positive for HIV-1 at the same time. However, her CD4 counts have always been >800 cell/μl, and her viral loads have consistently been <50 copies/ml without treatment. We studied viral isolates as well as multiple host factors from both patients. The results have implications for the design of HIV-1 vaccines.
MATERIALS AND METHODS
Viral isolation and sequence analysis.
Isolation and full-length sequence analysis of replication-competent virus from HIV-1 from ES9 and the progressor was performed as previously described (10). Proviral and plasma env (5) and gag (6) clones were amplified as previously described.
Phylogenetic analysis.
Phylogenetic trees and bootstrap values were generated using the maximum parsimony method as implemented in the Mega4 program (http://www.megasoftware.net). Similar results were obtained when the neighbor-joining method was used. Reference sequences for the trees were found using the Los Alamos National Laboratory HIV Sequence Database (http://hiv.lanl.gov) implementation of the BLAST algorithm, which utilizes sequence data from GenBank to find those sequences in the public domain that are most similar to the query. Reference sequences for supplemental trees included B clade sequences from the HIV-1 subtype reference alignments on the Los Alamos National Laboratory server for gag and env.
Site-directed mutagenesis.
To generate the A163S mutant viral vector, the Gag sequence outside the two restriction sites BssHII and SphI was amplified by PCR and then cloned into a Zero Blunt TOPO PCR cloning vector (Invitrogen). In this subcloned vector, the A163S mutation was generated using a QuikChange II XL site-directed mutagenesis kit (Stratagene) with the following pair of primers: 5′-GTAGTAGAAGAGAAGTCTTTCAGCCCAGAAG-3′ and 5′-CTTCTGGGCTGAAAGACTTCTCTTCTACTAC-3′ (the underlined sequences represent mutated bases). Similarly, the N271H mutant viral vector was generated by amplification and subcloning of the Gag sequence outside the two restriction sites SphI and ApaI. The paired primers for mutagenesis are as follows: 5′-GATGGATAATCCTGGGATTACATAAAATAGTAAGAATGTATAGCC-3′ and 5′-GGCTATACATTCTTACTATTTTATGTAATCCCAGGATTATCCATC-3′ (the mutated bases are underlined). All mutated sequences were verified by direct sequencing after site-directed mutagenesis.
Viral fitness assays.
Growth kinetics of the replication virus were performed as described previously (10).
To study the effect of Gag on viral fitness, the gag from replication-competent virus or wild-type versus mutated NL4-3 was amplified by PCR and inserted into a TOPO vector using the Zero Blunt TOPO PCR cloning kit (Invitrogen). The gene was then digested out using Sbf1 and BssHII, whose cut sites were engineered into the primers, and ligated into the previously described pNL4-3-ΔE-GFP NL43dE vector (47) The vector was transformed into maximum-efficiency Stbl-2 cells (Invitrogen) to amplify it and then transfected into 293T cells in conjunction with a vesicular stomatitis virus G env plasmid. After the removal of cell debris by spinning at 300 × g for 10 min and filtering through Steriflip filters (Millipore), viral supernatants were then used for the infection of Jurkat cells via spinoculation at 1,200 × g at 25°C for 2 h, and the fitness of the virus was quantified 48 h later by green fluorescent protein expression of infected cells. The results were normalized by the transfection efficiency.
Immunological assays.
Neutralizing antibody titers were determined as previously described (5). ES9-D1 and ES9-D2 are both proviral env clones; all other clones were amplified from replication-competent virus. Enzyme-linked immunospot (ELISPOT) analysis was performed with gamma interferon (IFN-γ) antibodies from Mabtech according to the manufacturer's instructions. Overlapping peptides spanning the entire HIV-1 genome were obtained from the NIH AIDS Research and Reference Reagent Program. Optimal epitopes were synthesized by the peptide synthesis facility in the Johns Hopkins oncology center core facility. For intracellular staining, the cells were stimulated with different concentrations of wild-type KK10 peptide or the N271 mutant overnight in the presence of brefeldin A and monensin. The cells were then stained with a phycoerythrin-conjugated KK10-HLA-B*27 pentamer (Proimmune) and PC5-conjugated anti-CD8 monoclonal antibody (MAb) (Beckman Coulter) before fixation and permeabilization. The cells were then stained with allophycocyanin-conjugated IFN-γ and fluorescein isothiocyanate-conjugated anti-tumor necrosis factor alpha (TNF-α) (Becton Dickenson). A total of 500,000 events were collected for each sample, and CD8+ pentamer staining cells were analyzed for coexpression of the two cytokines.
For analysis of proliferative responses, peripheral blood mononuclear cells (PBMC) were first stained with 5 μm carboxyfluorescein succinimidyl ester (CFSE) at room temperature for 10 min and then stimulated with peptides for 6 days. PBMC were then stained with PC5-conjugated anti-CD8 MAb and KK10-HLA-B*27 pentamer. A total of 500,000 events were collected for each sample, and CD8+ pentamer staining cells were analyzed for the loss of CFSE staining.
For the suppression assay, CD8+ T cells were obtained by positive selection with Miltenyi magnetic beads and were then stained with antibodies to phycoerythrin-conjugated antibodies specific for CD4, CD16, and CD56. Contaminating CD4+ T cells and natural killer (NK) cells were then removed by flow cytometry. NK cells were positively selected using Miltenyi magnetic beads and were then stained with antibodies to fluorescein isothiocyanate-conjugated antibodies specific for CD3. Contaminating lymphocytes were then removed by flow cytometry.
Target cells were obtained by culturing PBMC with 1 μg/ml of phytohemagglutinin for 48 h. The activated cells were depleted of CD8+ T cells with magnetic beads (Invitrogen) for the CD8+ T-cell suppression assay or with MAbs specific for CD8 and CD16 followed by sheep anti-mouse magnetic beads (Invitrogen) for the NK cell suppression assay. The cells were then infected with culture supernatant containing either Ba-L or autologous culture supernatant by spinoculation at 1,200 × g at 25°C for 2 h (2). A total of 105 CD4+ T cells were then incubated with 105 CD8+ T cells or NK cells in RPMI medium with 10% fetal calf serum. Aliquots of culture supernatant were tested for p24 content on days 5 and 7.
Genetic polymorphisms.
The target single-nucleotide polymorphisms (SNPs) were amplified from genomic DNA by PCRs using locus-specific primers as previously described (23). The primers used for detecting the SNPs were as follows: HCP5-F (TAC CCT CAT TGT GTG ACA GCA), HCP5-R (GTC GTG GGA TTT TGC ACT TC), HLA-C-F (AGG GTG GTG CCA AGT ATG AG), HLA-C-R (CTT CTA GAG CCC CGT GGA G), rs9261174-F (CCA ATA CCT TGC TTG CCA TT), rs9261174-R (AGG CCA TAG AGG CAA ACT CA), rs3869068-F (CAA GGA TGT TTC TTG AAT TTT GG), rs3869068-R (CCT GAG CAC TGT ATC TCA TGA CC), rs2074480-F (GAT TTG AAG TCC CAA CAT CCA), rs2074480-R (GTG CAG ACC TAT CAC CCA AAG), rs7758512-F (CAG AAA TCA AAA GGC GGA AG), rs7758512-R (GGC TGA ATT GCT GGA TGA TT), rs9261129-F (GCT AGA GCA GGT AGC CCT GA), rs9261129-R (CTC TGA GAC ACA GGG GTG GT), rs2301753-F (ATC AGC TCC CAG CTG TGC T), rs2301753-R (CTC CCA GTC CAG GTC CAC), rs2074479-F (CCA TAG CCA TCA CGT TCT CA), and rs2074479-R (CCT ACT TTG CGG TGC TTC AT), where F and R indicate forward and reverse primers, respectively. The PCR conditions used included a denaturation step at 94°C for 3 min, followed by 30 cycles of 94°C for 30s, 60°C for 30s, and 68°C for 30s. Polymorphisms were detected by sequence analysis of gel-purified PCR products (Qiagen). All heterozygous SNPs were confirmed by cloning.
The Invitrogen killer immunoglobulin-like receptor (KIR) genotyping SSP kit was used to probe for the KIR3DS1 alleles from genomic DNA according to the manufacturer's instructions.
Nucleotide sequence accession numbers.
The nucleotide sequences determined for this study have been submitted to GenBank under accession numbers EU807759 to EU807838.
RESULTS
Strong evidence for transmission between ES9 and the progressor.
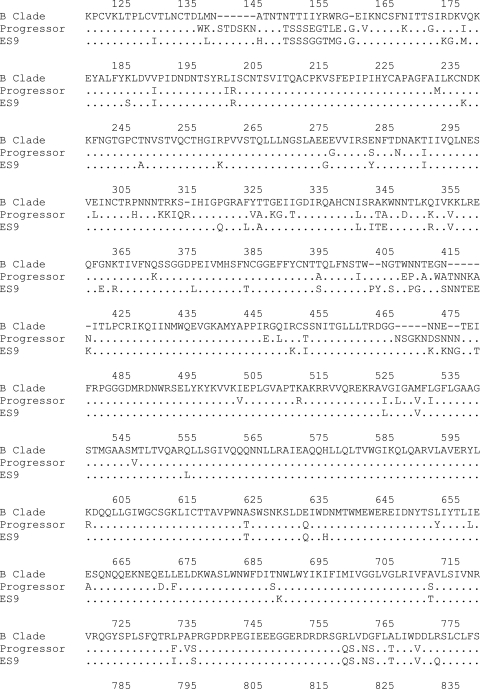
To definitively determine whether virulent virus had in fact been transmitted from the progressor to ES9, we cultured several isolates from the latent reservoirs of both patients. Full-length sequencing and phylogenetic analyses were performed. Figure 1 shows a comparison of the env and gag sequences from these isolates as well as previously obtained proviral and plasma clones from ES9 (5, 6). The trees also include env and gag sequences identified in a search of the Los Alamos HIV database for isolates with the highest degree of homology to the patients' env sequences. The results show that isolates from the transmitter and ES9 are in fact more closely related to each other than to any other isolate in the database, with a high degree of homology (bootstrap values of 99% and 100%). We also compared gag and env sequences from the patient to all clade B isolates (data not shown). We again found that the isolates from the two patients were closer to each other than to any other isolate. Finally, sequence analysis of the tat gene in all isolates from both patients revealed a TAG-to-CAG mutation in the normal Tat stop codon mutation followed immediately by an unusual stop codon. This type of mutation, expected to produce a Tat protein that is 1 amino acid longer than usual (Fig. 2), is present in less than 1% of clade B isolates in the Los Alamos HIV sequence database and is not present in any of the most closely related sequences. Thus, there is strong evidence of transmission of HIV-1 between the two individuals.
FIG. 1.
Phylogenetic analysis of viral sequences obtained from ES9 and the progressor. Phylogenetic trees of env (A) and gag (B) are shown. Sequences amplified from replication-competent virus (circles), provirus (squares), and plasma (triangles) for ES9 (green) and the progressor (blue) are compared to the most homologous clade B env sequences (branches without symbols) in the Los Alamos database. Sequences from clade D (black squares) serve as an outgroup.
FIG. 2.
Tat sequences from ES9 and the progressor. The amino acid sequences of Tat from isolates obtained from ES9 and the progressor are compared to the consensus clade B sequence. The extra amino acid sequence where the stop codon (asterisk) normally occurs is highlighted.
The actual date of transmission between these individuals is unknown. The progressor has a long history of injection drug use and was sexually active with ES9 for a period of 17 years before the diagnosis of HIV-1 infection was made. Isolates were obtained from the subjects between 5 and 8 years after diagnosis and probably many years after the actual transmission. The virus has thus likely evolved significantly in the progressor since transmission, which potentially explains why the clones from ES9 do not intermingle with clones from the progressor on the phylogenetic trees.
Virus from ES9 is less fit than that from the progressor.
Prior studies suggested that ES may be infected with defective HIV-1 isolates that result in slower disease progression. Isolating replication-competent viruses from ES has proven to be extremely challenging, but after multiple attempts, we were able obtain several isolates from ES9. We compared the replicative capacities of representative isolates from the progressor and ES9: ES9-2, a representative isolate from ES9, was consistently less fit than P-10, a representative isolate from the progressor, in standard viral growth assays (Fig. 3A). To study the role of Gag in this difference, we made recombinant pseudotype viruses containing gag from the two isolates and found that Gag from ES9-2 was significantly less fit than Gag from P-10 (Fig. 3B). Sequence analysis then provided possible explanations for the disparity. All isolates from both patients contained the Gag T242N mutation, which is located within the HLA-B*57-restricted epitope TW10 (positions 240 to 249). This mutation has been shown to significantly affect the fitness of the virus (35), and reversion occurs when this virus is transmitted to HLA-B*57-negative patients (31). Interestingly, a recent study showed that a series of mutations in the cyclophilin A binding region of Gag can restore the fitness of T242N-containing isolates (11). We found that isolates from the progressor but not ES9 had substitutions at two of the three positions (H219Q and M228I) (Fig. 4). In addition, different substitutions were seen in the isolates at Gag163, which is contained within another HLA-B*57-restricted epitope, KF11 (positions 162 to 172). The A163S substitution, seen in all isolates from ES9, is very rare in clade B HIV-1 isolates, as is a third substitution, N271H, which is located in the immunodominant HLA-B*27-restricted Gag epitope KK10 (positions 263 to 272) in isolates from ES9. To determine whether these substitutions had a significant effect on the fitnesses of these viruses, the mutations were independently engineered into the NL4-3 laboratory strain, and pseudotype viruses were generated. As shown in Fig. 3D, the A163S mutation did not significantly affect viral replication in a single-cycle assay, whereas the N271H mutation caused a 40% reduction in infectivity (Fig. 3E). Thus, this rare substitution, along with the uncompensated T242N mutation, likely contributes to the reduced fitness of the isolates from ES9.
FIG. 3.
Fitness of virus from ES9 and the progressor. (A) Growth kinetics of representative replication-competent isolates from ES9 (green) and the progressor (blue) are compared to laboratory strain Ba-L (black). (B and C) Relative fitness of pseudotype virus containing gag from ES9-2 (green) and P-10 (blue) (B), which is based on the green fluorescent protein (GFP) expression seen when the respective pseudotype viruses are used to infect Jurkat cells (C). SSC, side scatter. (D and E) Relative fitness of pseudotype virus containing Gag with the A163S (D) or the N271H (E) mutation (both shown in green) compared to that of wild-type (WT) Gag (black). Error bars represent standard errors of the means from three independent experiments.
FIG. 4.
Sequences of Gag from ES9 and the progressor. Epitopes and compensatory mutations are highlighted.
To determine whether there were any large deletions that could further explain the reduced fitness of isolates from ES9, we sequenced the entire genome of representative isolates from both subjects. As shown in Fig. 5, no such deletions were found.
FIG. 5.
Sequence of the entire genome of representative replication-competent isolates from ES9 and the progressor. Epitopes targeted by ES9 (green) and the progressor (blue) are shaded. LTR, long terminal repeat.
Role of host factors in disease progression.
We examined several host factors that could potentially be contributing to disease progression, including HLA genotypes, genetic factors, titers of neutralizing antibodies (NAb) to autologous Env, T-cell IFN-γ secretion responses to the entire HIV-1 proteome, and the ability of CD8+ T cells and NK cells to inhibit the replication of autologous viral isolates.
A genome-wide analysis recently identified SNPs that are associated with either low viral loads or slow HIV-1 progression in untreated patients (19). We found that neither patient had the protective HCP5 SNP, which is particularly interesting since this SNP is in linkage disequilibrium with HLA-B*5701 (19). This confirms our recent study showing that while this SNP is seen in some HLA-B*5701-positive (HLA-B*5701+) patients, it was not present in a cohort of HLA-B*5703+ ES of African descent (23). ES9 was positive for the protective SNP associated with the HLA-C allele. Interestingly, the progressor but not ES9 was positive for one (rs9261174) of the seven protective SNPs located in or near the ring finger protein 39 and zinc ribbon domain-containing 1 genes that have been associated with slow HIV-1 progression (19). Thus, genetic differences alone probably cannot explain the different outcomes seen in the two subjects. We were also able to culture laboratory and primary HIV-1 isolates in activated CD4+ T cells from ES9, which strongly suggests that the cells from this subject are not intrinsically resistant to viral replication (data not shown).
The donor's HLA alleles were HLA-A*330301, -A*3402, -B*440301, and -B*5703. ES9 had the following alleles: HLA-A*02, -A*30, -B*2703, and -B*5703. It is noteworthy that both patients were HLA-B*57+: this allele is overrepresented in ES (17, 30, 39, 41) but is also present in 10% of patients with progressive disease (39). Additionally, the HLA-B*27 allele is also overrepresented in ES (17, 41) and was present in ES9 but not the donor.
Overall, there was no significant difference in the breadth or width of the IFN-γ response directed to the entire consensus clade B HIV-1 proteome in the two subjects (Table 1). This is consistent with studies showing that the magnitude of HIV-specific IFN-γ responses does not correlate with disease protection (1, 8). Furthermore, a recent study has shown that the pattern of epitope recognition by the immune response is critical and that responses to Gag and Env are associated with low and high levels of viremia, respectively (28). While both patients responded primarily to Gag, the progressor also had responses to multiple epitopes in Pol, and ES9 targeted epitopes in Nef.
TABLE 1.
Whole-proteome screen with PBMC from the two subjectsa
| Sequence | Positions in HXB2 | HIV-1 protein | IFN-γ response magnitude (SFC/106 PBMC) | Sequence polymorphism(s)
|
|
|---|---|---|---|---|---|
| Progressor | ES9 | ||||
| ES9 | |||||
| ASRELERFAVNPGLL | 37-51 | Gag | 65 | ||
| KAFSPEVIPMF | 162-172 | Gag | 600 | A163N | A163S |
| PVGEIYKRWIILGLN | 257-271 | Gag | 75 | N271H | |
| IYKRWIILGLNKIVR | 261-275 | Gag | 3,075 | N271H | |
| FRDYVDRFYKTLRAE | 293-307 | Gag | 70 | ||
| YKTLRAEQASQEVKN | 301-315 | Gag | 130 | ||
| DCKTILKALGPAATL | 329-343 | Gag | 55 | ||
| KEGHIAKNCRAPRKK | 397-411 | Gag | 130 | K411R | K411R |
| IAKNCRAPRKKGCWK | 401-415 | Gag | 170 | K411R | K411R |
| QEEEEVGFPVRPQVP | 61-75 | Nef | 75 | 65-E insertion | |
| YKAAVDLSHFLKEKG | 81-95 | Nef | 410 | A83G, V85L | A83G, V85L |
| TQGYFPDWQNYTPGP | 117-131 | Nef | 75 | ||
| LSPRTLNAW* | 147-155 | Gag | <30 | ||
| KSFSPEVIPMF* | 162-172 | Gag | 520 | ||
| TSNLQEQIAW* | 240-249 | Gag | <30 | ||
| Progressor | |||||
| QMVHQAISPRTLNAW | 141-155 | Gag | 110 | A146P, I147L | A146P, I147L |
| IVRMYSPTSILDIRQ | 273-287 | Gag | 60 | R286K | R286K |
| RQANFLGKIWPSHKG | 429-443 | Gag | 75 | H441Y | |
| FLGKIWPSHKGRPGN | 433-447 | Gag | 80 | H441Y | |
| WIILGLNKIVRMYSP | 265-279 | Gag | 60 | N271H | |
| FRDYVDRFYKTLRAE | 293-307 | Gag | 70 | ||
| EAVQKIATESIVIWG | 525-539 | Pol | 165 | I530V, A531T, V536I | |
| KIATESIVIWGKTPK | 529-543 | Pol | 135 | K540R, T541I | I530V, A531T, V536I, T541I |
| IVGAETFYVDGAANR | 589-603 | Pol | 385 | V590I | V590I |
| ETFYVDGAANRETKL | 593-607 | Pol | 295 | K606R | |
| QKQITKIQNFRVYYR | 929-943 | Pol | 70 | ||
| LEWKFDSRLAFHHMA | 181-195 | Nef | 55 | E182V, K184R, M194V | E182V, K184R, M194V |
| LSPRTLNAW* | 147-155 | Gag | <30 | ||
| KNFSPEVIPMF* | 162-172 | Gag | <30 | ||
| TSNLQEQITW* | 240-249 | Gag | <30 | ||
Epitopes that elicited an IFN-γ response are shown with the magnitude of the response (spot-forming cells [SFC] per million PBMC). Substitutions observed in the targeted epitopes are noted. An asterisk indicates responses to autologous HLA-B*57-restricted Gag peptides.
In order to determine whether virologic escape in the HLA-B*57-restricted epitopes was the cause of the difference in outcome seen, we examined the responses to autologous peptides corresponding to escape mutations seen in isolates from the two subjects. Three of the four known HLA-B*57-restricted epitopes in Gag contained substitutions in both patients, IW9 (positions 147 to 155), KF11 (positions 162 to 172), and TW10 (positions 240 to 249). CD8+ T cells from ES9 continued to recognize the KF11 epitope with the A163 mutation (6); however, no responses to the other mutated HLA-B*57-restricted Gag epitopes, IW9 and TW10, were detected in either subject.
The N271H substitution in the KK10 epitope is a partial escape mutation.
Considering that the N271H mutation in the immunodominant HLA-B*27-restricted epitope KK10 had a negative impact on fitness, we performed studies to determine whether this rare substitution was an escape mutation. Our initial ELISPOT assay revealed that the mutant elicited a lower level of IFN-γ secretion response from CD8+ T cells at all peptide concentrations (Fig. 6A). It has recently been shown that the presence of CD8+ T cells that secrete multiple cytokines is a correlate of immune protection in HIV-1-infected patients (3, 9); it has also been shown that CD8+ T cells that secrete IFN-γ and TNF-α are more likely to be cytotoxic than cells that secrete IFN-γ alone (32). Considering this, we looked at the abilities of wild-type and mutant KK10 peptides to elicit the secretion of both cytokines from ES9's CD8+ T cells. As shown in Fig. 6B, the mutant peptide activated markedly fewer polyfunctional KK10-specific CD8+ T cells than did the wild-type peptide. Studies by Migueles and colleagues have shown that CD8+ T-cell proliferation in response to HIV-specific antigens occurs in ES but not in patients with progressive disease (38). We therefore looked at the ability of the mutant and wild-type peptides to elicit the proliferation of KK10-specific CD8+ T cells. At a relatively high concentration, both peptides induced robust clonal expansion (Fig. 6C) and proliferation as assessed by CFSE dilution (data not shown), but at lower concentrations, the wild-type peptide induced greater levels of both proliferation (Fig. 6C) and clonal expansion (Fig. 6D). It thus appears that the N271H mutation confers partial escape from KK10-specific CD8+ T cells.
FIG. 6.
Responses to wild-type and mutant KK10 epitopes. (A) CD8+ IFN-γ responses to the wild-type KK10 epitope (black) versus peptide with the N271H mutation detected by ELISPOT (green). SFC, spot-forming cells. (B) Percentage of KK10-positive cells that secrete both IFN-γ and TNF-α in response to wild-type and mutant peptide. (C) Percentage of CD8+ T cells that are KK10 positive after 6 days of stimulation of PBMC with wild-type and mutant peptide at either 0.01 or 1.0 μg/ml. (D) Percentage of KK10 cells that have proliferated after stimulation with 0.01 μg/ml of wild-type or mutant peptide. FITC, fluorescein isothiocyanate.
Functional HIV-1-specific CD8+ T-cell responses are present in ES9 but not the progressor.
In contrast to the proliferative response elicited from ES9 in response to wild-type and mutant KK10 peptides, epitopes that elicited IFN-γ secretion from the progressor's CD8+ T cells did not induce any detectable CD8+ T-cell proliferation (data not shown). This difference in CD8+ T-cell function may potentially explain the difference in outcomes seen in the subjects (38), although we cannot definitively rule out the possibility that the diminished CD8+ proliferative response was a consequence rather than a cause of disease progression. A recent study has shown that unstimulated CD8+ T cells from ES but not patients with progressive disease are able to inhibit the replication of a laboratory HIV-1 isolate (42). We thus compared the abilities of unstimulated CD8+ T cells from the progressor and ES9 to inhibit the replication of laboratory strain Ba-L as well as isolate P-10 from the progressor. As shown in Fig. 7A, CD8+ T cells from ES9 dramatically inhibited the replication of both Ba-L and P-10, while CD8+ T cells from the progressor had no significant effect on either isolate. The inhibitory response was abrogated when CD8+ T cells from ES9 were placed in transwells, suggesting that the effect was not mediated by cytokine secretion alone (data not shown). We were not able to perform this assay with isolate ES9-2 because of its poor replicative capacity.
FIG. 7.
Immune responses of ES9 and the progressor (P). (A and B) Replication of either Ba-L or P-10 in the presence or absence of autologous CD8+ T cells (A) or NK cells (B) from both patients. Error bars represent standard errors of the means from three independent experiments. (C) Titers of NAb to env clones obtained from ES9 (green) and the progressor (blue). IC50, 50% inhibitory concentration.
NK cells expressing certain KIR alleles have been shown to inhibit HIV-1 replication (4). Neither subject was positive for the KIR3DS1 allele, which, in conjunction with HLA-Bw4-80I alleles including HLA-B*57, has been associated with slow HIV-1 disease progression (34), and we found that NK cells from both patients did not significantly inhibit HIV-1 replication (Fig. 7B). This finding corroborates a recent study that compared NK cell-mediated inhibition in subjects with and those without the KIR3DS1 allele (4).
Recent studies have found that ES do not have high titers of NAb to Env from laboratory strains (15, 41). Because the sequence of Env varies significantly from that of primary isolates, we cloned this gene from provirus and cultured virus in both patients and looked at the abilities of plasma from both patients to neutralize cell entry of the expressed protein. The titer of NAb to autologous Env ranged from <1:20 to 1:125 in the progressor and ES9 (Fig. 6C). These titers are relatively low and are similar to the titers to autologous virus in ES and patients on HAART that we have recently reported (5). It is thus unlikely that NAb can explain the control of viremia in ES9.
DISCUSSION
In summary, we provide the first documentation of transmission of HIV-1 from a patient who developed AIDS to a patient who has maintained undetectable viral loads. This is the best evidence to date that the immune responses of some ES are capable of controlling viral isolates that are virulent enough in vivo to cause severe immunosuppression.
This case is particularly interesting because both patients are HLA-B*5703+. In addition, the ES also has the B*27 allele, which has also been associated with protective immune responses. Escape mutations in HLA-B*57-restricted epitopes were present in both patients, which suggests that these mutations by themselves do not explain the difference in outcomes seen in these patients. This confirms prior studies that showed that HLA-B*57+ progressors are not more likely to likely to have to escape mutations than ES (37) or LTNP (40). While we have demonstrated that virus with escape mutations can be found in the plasma of HLA-B*57+ ES, we found that ES9 was unique in that she was the only ES who had escape mutations present in every proviral gag clone (6). This strongly suggests that she was infected with an isolate that contained these mutations. The fact that the progressor is also HLA-B*57+ and has similar escape mutations makes this hypothesis all the more likely. Recent studies have suggested that recently infected patients have lower viral loads when they are infected with virus containing escape mutations (13, 20). The measurable benefit was most clear in recipients who do not have alleles that are associated with good outcomes. However, it has been shown that in the case of HLA-B*57, the escape mutations eventually revert to the wild type if the selecting allele is not present (31), and thus, it will be interesting to see if there is a long-term benefit in these individuals.
ES9 is HLA-B*5703+, and thus, the escape mutations that were transmitted to her did not revert over time. It is likely that transmission occurred early in the course of the progressor's infection, and the transmitted virus contained the initial T242N escape mutation, which has been shown to develop shortly after infection in HLA-B*57+ patients (31). The transmitted virus probably did not contain compensatory mutations and thus was somewhat attenuated (11). CD8+ T cells in ES9, which are capable of directly suppressing the replication of the transmitted isolate, probably prevented the virus from developing compensatory mutations. In addition, cytotoxic T lymphocytes specific for the KK10 epitope have selected for another mutation (N271H) that has a negative impact on viral fitness. This rare mutation is particularly interesting; while it appears to confer partial escape from KK10-specific CD8+ T cells, a de novo polyfunctional CD8+ T-cell-specific CD8+ T-cell response has developed, and this probably contributes to the control of the attenuated virus. This mutation thus differs from the classic KK10 epitope mutations (R264K/T), which are associated with complete virologic escape and disease progression (18, 21, 22, 26). The impact of this N271H mutation needs to be further confirmed in large HLA-matched cohorts.
A recent study has shown that isolates from HLA-B*57+ LTNP are generally less replication competent than isolates from HLA-B*57 progressors (40). Our results suggest that in some cases, the lower replication capacity of HIV-1 isolates in LTNP and ES may be the result, rather than the cause, of suppressed evolution: a qualitatively superior HIV-1-specific immune response that limits viral replication will prevent evolution toward greater fitness. This could explain why some HLA-B*57 ES control viral replication even though they are infected with fully replication-competent virus that lacks the T242N mutation (7, 10). In sum, we conclude that the immune system of ES9 is controlling viral replication by at least two different mechanisms: there is a direct inhibition of viral replication by polyfunctional HIV-1-specific CD8+ T cells that proliferate in response to autologous viral peptides, and there is selection for and maintenance of escape mutations that have a negative impact on viral fitness. Vaccines that elicit CD8+ T cells with both properties may be very effective at controlling HIV-1 replication.
Acknowledgments
This work was supported by NIH grants K08 AI51191 and R56 AI73185-01A1 and the Howard Hughes Medical Institute.
We thank Ferdynand Kos of the Johns Hopkins Human Immunology core facility for flow cytometry analysis and Hao Zhang of the Johns Hopkins School of Public Health for sorting.
Footnotes
Published ahead of print on 21 May 2008.
REFERENCES
- 1.Addo, M. M., X. G. Yu, A. Rathod, D. Cohen, R. L. Eldridge, D. Strick, M. N. Johnston, C. Corcoran, A. G. Wurcel, C. A. Fitzpatrick, M. E. Feeney, W. R. Rodriguez, N. Basgoz, R. Draenert, D. R. Stone, C. Brander, P. J. R. Goulder, E. S. Rosenberg, M. Altfeld, and B. D. Walker. 2003. Comprehensive epitope analysis of human immunodeficiency virus type 1 (HIV-1)-specific T-cell responses directed against the entire expressed HIV-1 genome demonstrate broadly directed responses, but no correlation to viral load. J. Virol. 772081-2092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Alexander, L., E. Weiskopf, T. C. Greenough, N. C. Gaddis, M. R. Auerbach, M. H. Malim, S. J. O'Brien, B. D. Walker, J. L. Sullivan, and R. C. Desrosiers. 2000. Unusual polymorphisms in human immunodeficiency virus type 1 associated with nonprogressive infection. J. Virol. 744361-4376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Almeida, J. R., D. A. Price, L. Papagno, Z. A. Arkoub, D. Sauce, E. Bornstein, T. E. Asher, A. Samri, A. Schnuriger, I. Theodorou, D. Costagliola, C. Rouzioux, H. Agut, A. G. Marcelin, D. Douek, B. Autran, and V. Appay. 2007. Superior control of HIV-1 replication by CD8+ T cells is reflected by their avidity, polyfunctionality, and clonal turnover. J. Exp. Med. 2042473-2485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Alter, G., M. P. Martin, N. Teigen, W. H. Carr, T. J. Suscovich, A. Schneidewind, H. Streeck, M. Waring, A. Meier, C. Brander, J. D. Lifson, T. M. Allen, M. Carrington, and M. Altfeld. 2007. Differential natural killer cell-mediated inhibition of HIV-1 replication based on distinct KIR/HLA subtypes. J. Exp. Med. 2043027-3036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bailey, J. R., K. G. Lassen, H. C. Yang, T. C. Quinn, S. C. Ray, J. N. Blankson, and R. F. Siliciano. 2006. Neutralizing antibodies do not mediate suppression of human immunodeficiency virus type 1 in elite suppressors or selection of plasma virus variants in patients on highly active antiretroviral therapy. J. Virol. 804758-4770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bailey, J. R., T. M. Williams, R. F. Siliciano, and J. N. Blankson. 2006. Maintenance of viral suppression in HIV-1-infected HLA-B*57+ elite suppressors despite CTL escape mutations. J. Exp. Med. 2031357-1369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bailey, J. R., H. Zhang, B. W. Wegweiser, H. C. Yang, L. Herrera, A. Ahonkhai, T. M. Williams, R. F. Siliciano, and J. N. Blankson. 2007. Evolution of HIV-1 in an HLA-B*57-positive patient during virologic escape. J. Infect. Dis. 19650-55. [DOI] [PubMed] [Google Scholar]
- 8.Betts, M. R., D. R. Ambrozak, D. C. Douek, S. Bonhoeffer, J. M. Brenchley, J. P. Casazza, R. A. Koup, and L. J. Picker. 2001. Analysis of total human immunodeficiency virus (HIV)-specific CD4+ and CD8+ T-cell responses: relationship to viral load in untreated HIV infection. J. Virol. 7511983-11991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Betts, M. R., M. C. Nason, S. M. West, S. C. De Rosa, S. A. Migueles, J. Abraham, M. M. Lederman, J. M. Benito, P. A. Goepfert, M. Connors, M. Roederer, and R. A. Koup. 2006. HIV nonprogressors preferentially maintain highly functional HIV-specific CD8+ T cells. Blood 1074781-4789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Blankson, J. N., J. R. Bailey, S. Thayil, H. C. Yang, K. Lassen, J. Lai, S. K. Gandhi, J. D. Siliciano, T. M. Williams, and R. F. Siliciano. 2007. Isolation and characterization of replication-competent human immunodeficiency virus type 1 from a subset of elite suppressors. J. Virol. 812508-2518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Brockman, M. A., A. Schneidewind, M. Lahaie, A. Schmidt, T. Miura, I. Desouza, F. Ryvkin, C. A. Derdeyn, S. Allen, E. Hunter, J. Mulenga, P. A. Goepfert, B. D. Walker, and T. M. Allen. 2007. Escape and compensation from early HLA-B57-mediated cytotoxic T-lymphocyte pressure on human immunodeficiency virus type 1 Gag alter capsid interactions with cyclophilin A. J. Virol. 8112608-12618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Calugi, G., F. Montella, C. Favalli, and A. Benedetto. 2006. Entire genome of a strain of human immunodeficiency virus type 1 with a deletion of nef that was recovered 20 years after primary infection: large pool of proviruses with deletions of env. J. Virol. 8011892-11896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Chopera, D. R., Z. Woodman, K. Mlisana, M. Mlotshwa, D. P. Martin, C. Seoighe, F. Treurnicht, D. A. de Rosa, W. Hide, S. A. Karim, C. M. Gray, C. Williamson, and the CAPRISA 002 Study Team. 2008. Transmission of HIV-1 CTL escape variants provides HLA-mismatched recipients with a survival advantage. PLoS Pathog. 4e1000033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Deacon, N. J., A. Tsykin, A. Solomon, K. Smith, M. Ludford-Menting, D. J. Hooker, D. A. McPhee, A. L. Greenway, A. Ellett, C. Chatfield, V. A. Lawson, S. Crowe, A. Maerz, S. Sonza, J. Learmont, J. S. Sullivan, A. Cunningham, D. Dwyer, D. Dowton, and J. Mills. 1995. Genomic structure of an attenuated quasi species of HIV-1 from a blood transfusion donor and recipients. Science 270988-991. [DOI] [PubMed] [Google Scholar]
- 15.Deeks, S. G., B. Schweighardt, T. Wrin, J. Galovich, R. Hoh, E. Sinclair, P. Hunt, J. M. McCune, J. N. Martin, C. J. Petropoulos, and F. M. Hecht. 2006. Neutralizing antibody responses against autologous and heterologous viruses in acute versus chronic human immunodeficiency virus (HIV) infection: evidence for a constraint on the ability of HIV to completely evade neutralizing antibody responses. J. Virol. 806155-6164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Deeks, S. G., and B. D. Walker. 2007. Human immunodeficiency virus controllers: mechanisms of durable virus control in the absence of antiretroviral therapy. Immunity 27406-416. [DOI] [PubMed] [Google Scholar]
- 17.Emu, B., E. Sinclair, H. Hatano, A. Ferre, B. Shacklett, J. N. Martin, J. M. McCune, and S. G. Deeks. 2008. HLA class I-restricted T-cell responses may contribute to the control of human immunodeficiency virus type 1 infection, but such responses are not always necessary for long-term virus control. J. Virol. 825398-5407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Feeney, M. E., Y. Tang, K. A. Roosevelt, A. J. Leslie, K. McIntosh, N. Karthas, B. D. Walker, and P. J. Goulder. 2004. Immune escape precedes breakthrough human immunodeficiency virus type 1 viremia and broadening of the cytotoxic T-lymphocyte response in an HLA-B27-positive long-term-nonprogressing child. J. Virol. 788927-8930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Fellay, J., K. V. Shianna, D. Ge, S. Colombo, B. Ledergerber, M. Weale, K. Zhang, C. Gumbs, A. Castagna, A. Cossarizza, A. Cozzi-Lepri, A. De Luca, P. Easterbrook, P. Francioli, S. Mallal, J. Martinez-Picado, J. M. Miro, N. Obel, J. P. Smith, J. Wyniger, P. Descombes, S. E. Antonarakis, N. L. Letvin, A. J. McMichael, B. F. Haynes, A. Telenti, and D. B. Goldstein. 2007. A whole-genome association study of major determinants for host control of HIV-1. Science 317944-947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Goepfert, P. A., W. Lumm, P. Farmer, P. Matthews, A. Prendergast, J. M. Carlson, C. A. Derdeyn, J. Tang, R. A. Kaslow, A. Bansal, K. Yusim, D. Heckerman, J. Mulenga, S. Allen, P. J. Goulder, and E. Hunter. 2008. Transmission of HIV-1 Gag immune escape mutations is associated with reduced viral load in linked recipients. J. Exp. Med. 2051009-1017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Goulder, P. J., C. Brander, Y. Tang, C. Tremblay, R. A. Colbert, M. M. Addo, E. S. Rosenberg, T. Nguyen, R. Allen, A. Trocha, M. Altfeld, S. He, M. Bunce, R. Funkhouser, S. I. Pelton, S. K. Burchett, K. McIntosh, B. T. Korber, and B. D. Walker. 2001. Evolution and transmission of stable CTL escape mutations in HIV infection. Nature 412334-338. [DOI] [PubMed] [Google Scholar]
- 22.Goulder, P. J., R. E. Phillips, R. A. Colbert, S. McAdam, G. Ogg, M. A. Nowak, P. Giangrande, G. Luzzi, B. Morgan, A. Edwards, A. J. McMichael, and S. Rowland-Jones. 1997. Late escape from an immunodominant cytotoxic T-lymphocyte response associated with progression to AIDS. Nat. Med. 3212-217. [DOI] [PubMed] [Google Scholar]
- 23.Han, Y., J. Lai, P. Barditch-Crovo, J. E. Gallant, T. M. Williams, R. F. Siliciano, and J. N. Blankson. 2008. The role of protective HCP5 and HLA-C associated polymorphisms in the control of HIV-1 replication in a subset of elite suppressors. AIDS 22541-544. [DOI] [PubMed] [Google Scholar]
- 24.Iversen, A. K., E. G. Shpaer, A. G. Rodrigo, M. S. Hirsch, B. D. Walker, H. W. Sheppard, T. C. Merigan, and J. I. Mullins. 1995. Persistence of attenuated rev genes in a human immunodeficiency virus type 1-infected asymptomatic individual. J. Virol. 695743-5753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kaslow, R. A., M. Carrington, R. Apple, L. Park, A. Munoz, A. J. Saah, J. J. Goedert, C. Winkler, S. J. O'Brien, C. Rinaldo, R. Detels, W. Blattner, J. Phair, H. Erlich, and D. L. Mann. 1996. Influence of combinations of human major histocompatibility complex genes on the course of HIV-1 infection. Nat. Med. 2405-411. [DOI] [PubMed] [Google Scholar]
- 26.Kelleher, A. D., C. Long, E. C. Holmes, R. L. Allen, J. Wilson, C. Conlon, C. Workman, S. Shaunak, K. Olson, P. Goulder, C. Brander, G. Ogg, J. S. Sullivan, W. Dyer, I. Jones, A. J. McMichael, S. Rowland-Jones, and R. E. Phillips. 2001. Clustered mutations in HIV-1 gag are consistently required for escape from HLA-B27-restricted cytotoxic T lymphocyte responses. J. Exp. Med. 193375-386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kiepiela, P., A. J. Leslie, I. Honeyborne, D. Ramduth, C. Thobakgale, S. Chetty, P. Rathnavalu, C. Moore, K. J. Pfafferott, L. Hilton, P. Zimbwa, S. Moore, T. Allen, C. Brander, M. M. Addo, M. Altfeld, I. James, S. Mallal, M. Bunce, L. D. Barber, J. Szinger, C. Day, P. Klenerman, J. Mullins, B. Korber, H. M. Coovadia, B. D. Walker, and P. J. Goulder. 2004. Dominant influence of HLA-B in mediating the potential co-evolution of HIV and HLA. Nature 432769-775. [DOI] [PubMed] [Google Scholar]
- 28.Kiepiela, P., K. Ngumbela, C. Thobakgale, D. Ramduth, I. Honeyborne, E. Moodley, S. Reddy, C. de Pierres, Z. Mncube, N. Mkhwanazi, K. Bishop, M. van der Stok, K. Nair, N. Khan, H. Crawford, R. Payne, A. Leslie, J. Prado, A. Prendergast, J. Frater, N. McCarthy, C. Brander, G. H. Learn, D. Nickle, C. Rousseau, H. Coovadia, J. I. Mullins, D. Heckerman, B. D. Walker, and P. Goulder. 2007. CD8+ T-cell responses to different HIV proteins have discordant associations with viral load. Nat. Med. 1346-53. [DOI] [PubMed] [Google Scholar]
- 29.Kirchhoff, F., T. C. Greenough, D. B. Brettler, J. L. Sullivan, and R. C. Desrosiers. 1995. Brief report: absence of intact nef sequences in a long-term survivor with nonprogressive HIV-1 infection. N. Engl. J. Med. 332228-232. [DOI] [PubMed] [Google Scholar]
- 30.Lambotte, O., F. Boufassa, Y. Madec, A. Nguyen, C. Goujard, L. Meyer, C. Rouzioux, A. Venet, J. F. Delfraissy, and the SEROCO-HEMOCO Study Group. 2005. HIV controllers: a homogeneous group of HIV-1-infected patients with spontaneous control of viral replication. Clin. Infect. Dis. 411053-1056. [DOI] [PubMed] [Google Scholar]
- 31.Leslie, A. J., K. J. Pfafferott, P. Chetty, R. Draenert, M. M. Addo, M. Feeney, Y. Tang, E. C. Holmes, T. Allen, J. G. Prado, M. Altfeld, C. Brander, C. Dixon, D. Ramduth, P. Jeena, S. A. Thomas, A. St. John, T. A. Roach, B. Kupfer, G. Luzzi, A. Edwards, G. Taylor, H. Lyall, G. Tudor-Williams, V. Novelli, J. Martinez-Picado, P. Kiepiela, B. D. Walker, and P. J. Goulder. 2004. HIV evolution: CTL escape mutation and reversion after transmission. Nat. Med. 10282-289. [DOI] [PubMed] [Google Scholar]
- 32.Lichterfeld, M., X. G. Yu, M. T. Waring, S. K. Mui, M. N. Johnston, D. Cohen, M. M. Addo, J. Zaunders, G. Alter, E. Pae, D. Strick, T. M. Allen, E. S. Rosenberg, B. D. Walker, and M. Altfeld. 2004. HIV-1-specific cytotoxicity is preferentially mediated by a subset of CD8(+) T cells producing both interferon-gamma and tumor necrosis factor-alpha. Blood 104487-494. [DOI] [PubMed] [Google Scholar]
- 33.Mariani, R., F. Kirchhoff, T. C. Greenough, J. L. Sullivan, R. C. Desrosiers, and J. Skowronski. 1996. High frequency of defective nef alleles in a long-term survivor with nonprogressive human immunodeficiency virus type 1 infection. J. Virol. 707752-7764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Martin, M. P., X. Gao, J. H. Lee, G. W. Nelson, R. Detels, J. J. Goedert, S. Buchbinder, K. Hoots, D. Vlahov, J. Trowsdale, M. Wilson, S. J. O'Brien, and M. Carrington. 2002. Epistatic interaction between KIR3DS1 and HLA-B delays the progression to AIDS. Nat. Genet. 31429-434. [DOI] [PubMed] [Google Scholar]
- 35.Martinez-Picado, J., J. G. Prado, E. E. Fry, K. Pfafferott, A. Leslie, S. Chetty, C. Thobakgale, I. Honeyborne, H. Crawford, P. Matthews, T. Pillay, C. Rousseau, J. I. Mullins, C. Brander, B. D. Walker, D. I. Stuart, P. Kiepiela, and P. Goulder. 2006. Fitness cost of escape mutations in p24 Gag in association with control of human immunodeficiency virus type 1. J. Virol. 803617-3623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Michael, N. L., G. Chang, L. A. d'Arcy, P. K. Ehrenberg, R. Mariani, M. P. Busch, D. L. Birx, and D. H. Schwartz. 1995. Defective accessory genes in a human immunodeficiency virus type 1-infected long-term survivor lacking recoverable virus. J. Virol. 694228-4236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Migueles, S. A., A. C. Laborico, H. Imamichi, W. L. Shupert, C. Royce, M. McLaughlin, L. Ehler, J. Metcalf, S. Liu, C. W. Hallahan, and M. Connors. 2003. The differential ability of HLA B*5701+ long-term nonprogressors and progressors to restrict human immunodeficiency virus replication is not caused by loss of recognition of autologous viral gag sequences. J. Virol. 776889-6898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Migueles, S. A., A. C. Laborico, W. L. Shupert, M. S. Sabbaghian, R. Rabin, C. W. Hallahan, D. Van Baarle, S. Kostense, F. Miedema, M. McLaughlin, L. Ehler, J. Metcalf, S. Liu, and M. Connors. 2002. HIV-specific CD8+ T cell proliferation is coupled to perforin expression and is maintained in nonprogressors. Nat. Immunol. 31061-1068. [DOI] [PubMed] [Google Scholar]
- 39.Migueles, S. A., M. S. Sabbaghian, W. L. Shupert, M. P. Bettinotti, F. M. Marincola, L. Martino, C. W. Hallahan, S. M. Selig, D. Schwartz, J. Sullivan, and M. Connors. 2000. HLA B*5701 is highly associated with restriction of virus replication in a subgroup of HIV-infected long term nonprogressors. Proc. Natl. Acad. Sci. USA 972709-2714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Navis, M., I. Schellens, D. van Baarle, J. Borghans, P. van Swieten, F. Miedema, N. Kootstra, and H. Schuitemaker. 2007. Viral replication capacity as a correlate of HLA B57/B5801-associated nonprogressive HIV-1 infection. J. Immunol. 1793133-3143. [DOI] [PubMed] [Google Scholar]
- 41.Pereyra, F., M. M. Addo, D. E. Kaufmann, Y. Liu, T. Miura, A. Rathod, B. Baker, A. Trocha, R. Rosenberg, E. Mackey, P. Ueda, Z. Lu, D. Cohen, T. Wrin, C. J. Petropoulos, E. S. Rosenberg, and B. D. Walker. 2008. Genetic and immunologic heterogeneity among persons who control HIV infection in the absence of therapy. J. Infect. Dis. 197563-571. [DOI] [PubMed] [Google Scholar]
- 42.Saez-Cirion, A., C. Lacabaratz, O. Lambotte, P. Versmisse, A. Urrutia, F. Boufassa, F. Barre-Sinoussi, J. F. Delfraissy, M. Sinet, G. Pancino, A. Venet, and the Agence Nationale de Recherches sur le Sida EP36 HIV Controllers Study Group. 2007. HIV controllers exhibit potent CD8 T cell capacity to suppress HIV infection ex vivo and peculiar cytotoxic T lymphocyte activation phenotype. Proc. Natl. Acad. Sci. USA 1046776-6781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Saez-Cirion, A., G. Pancino, M. Sinet, A. Venet, O. Lambotte, and the ANRS EP36 HIV Controllers Study Group. 2007. HIV controllers: how do they tame the virus? Trends Immunol. 28532-540. [DOI] [PubMed] [Google Scholar]
- 44.Salvi, R., A. R. Garbuglia, A. Di Caro, S. Pulciani, F. Montella, and A. Benedetto. 1998. Grossly defective nef gene sequences in a human immunodeficiency virus type 1-seropositive long-term nonprogressor. J. Virol. 723646-3657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Wang, B., M. Mikhail, W. B. Dyer, J. J. Zaunders, A. D. Kelleher, and N. K. Saksena. 2003. First demonstration of a lack of viral sequence evolution in a nonprogressor, defining replication-incompetent HIV-1 infection. Virology 312135-150. [DOI] [PubMed] [Google Scholar]
- 46.Yamada, T., and A. Iwamoto. 2000. Comparison of proviral accessory genes between long-term nonprogressors and progressors of human immunodeficiency virus type 1 infection. Arch. Virol. 1451021-1027. [DOI] [PubMed] [Google Scholar]
- 47.Zhang, H., Y. Zhou, C. Alcock, T. Kiefer, D. Monie, J. Siliciano, Q. Li, P. Pham, J. Cofrancesco, D. Persaud, and R. F. Siliciano. 2004. Novel single-cell-level phenotypic assay for residual drug susceptibility and reduced replication capacity of drug-resistant human immunodeficiency virus type 1. J. Virol. 781718-1729. [DOI] [PMC free article] [PubMed] [Google Scholar]