Abstract
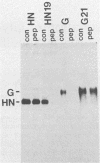
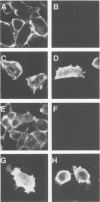
cDNAs encoding the G glycoprotein of respiratory syncytial virus and the hemagglutinin-neuraminidase (HN) glycoprotein of parainfluenza virus type 3 were modified by site-specific mutagenesis and restriction fragment replacement to encode chimeric proteins consisting of the cytoplasmic and transmembrane domains of one protein fused to the ectodomain of the other. In the case of the HN ectodomain attached to the G transmembrane and cytoplasmic domains, cell surface expression of the chimera was reduced. Otherwise, the presence of the heterologous transmembrane and cytoplasmic domains had little effect on the processing of the HN or G ectodomain, as assayed by the acquisition of N-linked and O-linked carbohydrates, transport to the cell surface and, in the case of HN, folding, oligomerization, and hemadsorption activity. These results showed that the synthesis and processing of each ectodomain did not require the homologous transmembrane and cytoplasmic domains. In particular, O glycosylation of the G protein was specified fully by its ectodomain, even though this domain is highly divergent among the respiratory syncytial virus antigenic subgroups. In addition, whereas the cytoplasmic and transmembrane domains of the G protein were relatively highly conserved, they were nonetheless fully replaceable without significantly affecting processing.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aubert J. P., Biserte G., Loucheux-Lefebvre M. H. Carbohydrate-peptide linkage in glycoproteins. Arch Biochem Biophys. 1976 Aug;175(2):410–418. doi: 10.1016/0003-9861(76)90528-2. [DOI] [PubMed] [Google Scholar]
- Briand J. P., Andrews S. P., Jr, Cahill E., Conway N. A., Young J. D. Investigation of the requirements for O-glycosylation by bovine submaxillary gland UDP-N-acetylgalactosamine:polypeptide N-acetylgalactosamine transferase using synthetic peptide substrates. J Biol Chem. 1981 Dec 10;256(23):12205–12207. [PubMed] [Google Scholar]
- Cleveland D. W., Fischer S. G., Kirschner M. W., Laemmli U. K. Peptide mapping by limited proteolysis in sodium dodecyl sulfate and analysis by gel electrophoresis. J Biol Chem. 1977 Feb 10;252(3):1102–1106. [PubMed] [Google Scholar]
- Colman A., Robinson C. Protein import into organelles: hierarchical targeting signals. Cell. 1986 Aug 1;46(3):321–322. doi: 10.1016/0092-8674(86)90650-1. [DOI] [PubMed] [Google Scholar]
- Eckhardt A. E., Timpte C. S., Abernethy J. L., Toumadje A., Johnson W. C., Jr, Hill R. L. Structural properties of porcine submaxillary gland apomucin. J Biol Chem. 1987 Aug 15;262(23):11339–11344. [PubMed] [Google Scholar]
- Elango N., Coligan J. E., Jambou R. C., Venkatesan S. Human parainfluenza type 3 virus hemagglutinin-neuraminidase glycoprotein: nucleotide sequence of mRNA and limited amino acid sequence of the purified protein. J Virol. 1986 Feb;57(2):481–489. doi: 10.1128/jvi.57.2.481-489.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fernie B. F., Dapolito G., Cote P. J., Jr, Gerin J. L. Kinetics of synthesis of respiratory syncytial virus glycoproteins. J Gen Virol. 1985 Sep;66(Pt 9):1983–1990. doi: 10.1099/0022-1317-66-9-1983. [DOI] [PubMed] [Google Scholar]
- Fiat A. M., Jollès J., Aubert J. P., Loucheux-Lefebvre M. H., Jollès P. Localisation and importance of the sugar part of human casein. Eur J Biochem. 1980 Oct;111(2):333–339. doi: 10.1111/j.1432-1033.1980.tb04946.x. [DOI] [PubMed] [Google Scholar]
- Goodwin S. D., Watkins W. M. The peptide moiety of blood-group-specific glycoproteins. Some amino-acid sequences in the regions carrying the carbohydrate chains. Eur J Biochem. 1974 Sep 1;47(2):371–382. doi: 10.1111/j.1432-1033.1974.tb03702.x. [DOI] [PubMed] [Google Scholar]
- Griffiths G., Quinn P., Warren G. Dissection of the Golgi complex. I. Monensin inhibits the transport of viral membrane proteins from medial to trans Golgi cisternae in baby hamster kidney cells infected with Semliki Forest virus. J Cell Biol. 1983 Mar;96(3):835–850. doi: 10.1083/jcb.96.3.835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gruber C., Levine S. Respiratory syncytial virus polypeptides. IV. The oligosaccharides of the glycoproteins. J Gen Virol. 1985 Mar;66(Pt 3):417–432. doi: 10.1099/0022-1317-66-3-417. [DOI] [PubMed] [Google Scholar]
- Hagopian A., Westall F. C., Whitehead J. S., Eylar E. H. Glycosylation of the A1 protein from myelin by a polypeptide N-acetylgalactosaminyltransferase. Identification of the receptor sequence. J Biol Chem. 1971 Apr 25;246(8):2519–2523. [PubMed] [Google Scholar]
- Hill H. D., Jr, Schwyzer M., Steinman H. M., Hill R. L. Ovine submaxillary mucin. Primary structure and peptide substrates of UDP-N-acetylgalactosamine:mucin transferase. J Biol Chem. 1977 Jun 10;252(11):3799–3804. [PubMed] [Google Scholar]
- Johnson P. R., Spriggs M. K., Olmsted R. A., Collins P. L. The G glycoprotein of human respiratory syncytial viruses of subgroups A and B: extensive sequence divergence between antigenically related proteins. Proc Natl Acad Sci U S A. 1987 Aug;84(16):5625–5629. doi: 10.1073/pnas.84.16.5625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalderon D., Roberts B. L., Richardson W. D., Smith A. E. A short amino acid sequence able to specify nuclear location. Cell. 1984 Dec;39(3 Pt 2):499–509. doi: 10.1016/0092-8674(84)90457-4. [DOI] [PubMed] [Google Scholar]
- Kornfeld R., Kornfeld S. Assembly of asparagine-linked oligosaccharides. Annu Rev Biochem. 1985;54:631–664. doi: 10.1146/annurev.bi.54.070185.003215. [DOI] [PubMed] [Google Scholar]
- Lazarovits J., Roth M. A single amino acid change in the cytoplasmic domain allows the influenza virus hemagglutinin to be endocytosed through coated pits. Cell. 1988 Jun 3;53(5):743–752. doi: 10.1016/0092-8674(88)90092-x. [DOI] [PubMed] [Google Scholar]
- Morrison T. G., Ward L. J. Intracellular processing of the vesicular stomatitis virus glycoprotein and the Newcastle disease virus hemagglutinin-neuraminidase glycoprotein. Virus Res. 1984;1(3):225–239. doi: 10.1016/0168-1702(84)90041-8. [DOI] [PubMed] [Google Scholar]
- Mottet G., Portner A., Roux L. Drastic immunoreactivity changes between the immature and mature forms of the Sendai virus HN and F0 glycoproteins. J Virol. 1986 Jul;59(1):132–141. doi: 10.1128/jvi.59.1.132-141.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Munro S., Pelham H. R. A C-terminal signal prevents secretion of luminal ER proteins. Cell. 1987 Mar 13;48(5):899–907. doi: 10.1016/0092-8674(87)90086-9. [DOI] [PubMed] [Google Scholar]
- Olmsted R. A., Murphy B. R., Lawrence L. A., Elango N., Moss B., Collins P. L. Processing, surface expression, and immunogenicity of carboxy-terminally truncated mutants of G protein of human respiratory syncytial virus. J Virol. 1989 Jan;63(1):411–420. doi: 10.1128/jvi.63.1.411-420.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Päbo S., Bhat B. M., Wold W. S., Peterson P. A. A short sequence in the COOH-terminus makes an adenovirus membrane glycoprotein a resident of the endoplasmic reticulum. Cell. 1987 Jul 17;50(2):311–317. doi: 10.1016/0092-8674(87)90226-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rose J. K., Doms R. W. Regulation of protein export from the endoplasmic reticulum. Annu Rev Cell Biol. 1988;4:257–288. doi: 10.1146/annurev.cb.04.110188.001353. [DOI] [PubMed] [Google Scholar]
- Rothman J. E. Protein sorting by selective retention in the endoplasmic reticulum and Golgi stack. Cell. 1987 Aug 14;50(4):521–522. doi: 10.1016/0092-8674(87)90024-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Satake M., Coligan J. E., Elango N., Norrby E., Venkatesan S. Respiratory syncytial virus envelope glycoprotein (G) has a novel structure. Nucleic Acids Res. 1985 Nov 11;13(21):7795–7812. doi: 10.1093/nar/13.21.7795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tartakoff A. M. Perturbation of vesicular traffic with the carboxylic ionophore monensin. Cell. 1983 Apr;32(4):1026–1028. doi: 10.1016/0092-8674(83)90286-6. [DOI] [PubMed] [Google Scholar]
- Thompson S. D., Laver W. G., Murti K. G., Portner A. Isolation of a biologically active soluble form of the hemagglutinin-neuraminidase protein of Sendai virus. J Virol. 1988 Dec;62(12):4653–4660. doi: 10.1128/jvi.62.12.4653-4660.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vidal S., Mottet G., Kolakofsky D., Roux L. Addition of high-mannose sugars must precede disulfide bond formation for proper folding of Sendai virus glycoproteins. J Virol. 1989 Feb;63(2):892–900. doi: 10.1128/jvi.63.2.892-900.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wertz G. W., Collins P. L., Huang Y., Gruber C., Levine S., Ball L. A. Nucleotide sequence of the G protein gene of human respiratory syncytial virus reveals an unusual type of viral membrane protein. Proc Natl Acad Sci U S A. 1985 Jun;82(12):4075–4079. doi: 10.1073/pnas.82.12.4075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wertz G. W., Krieger M., Ball L. A. Structure and cell surface maturation of the attachment glycoprotein of human respiratory syncytial virus in a cell line deficient in O glycosylation. J Virol. 1989 Nov;63(11):4767–4776. doi: 10.1128/jvi.63.11.4767-4776.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young J. D., Tsuchiya D., Sandlin D. E., Holroyde M. J. Enzymic O-glycosylation of synthetic peptides from sequences in basic myelin protein. Biochemistry. 1979 Oct 2;18(20):4444–4448. doi: 10.1021/bi00587a026. [DOI] [PubMed] [Google Scholar]
- Zoller M. J., Smith M. Oligonucleotide-directed mutagenesis: a simple method using two oligonucleotide primers and a single-stranded DNA template. DNA. 1984 Dec;3(6):479–488. doi: 10.1089/dna.1.1984.3.479. [DOI] [PubMed] [Google Scholar]