Abstract
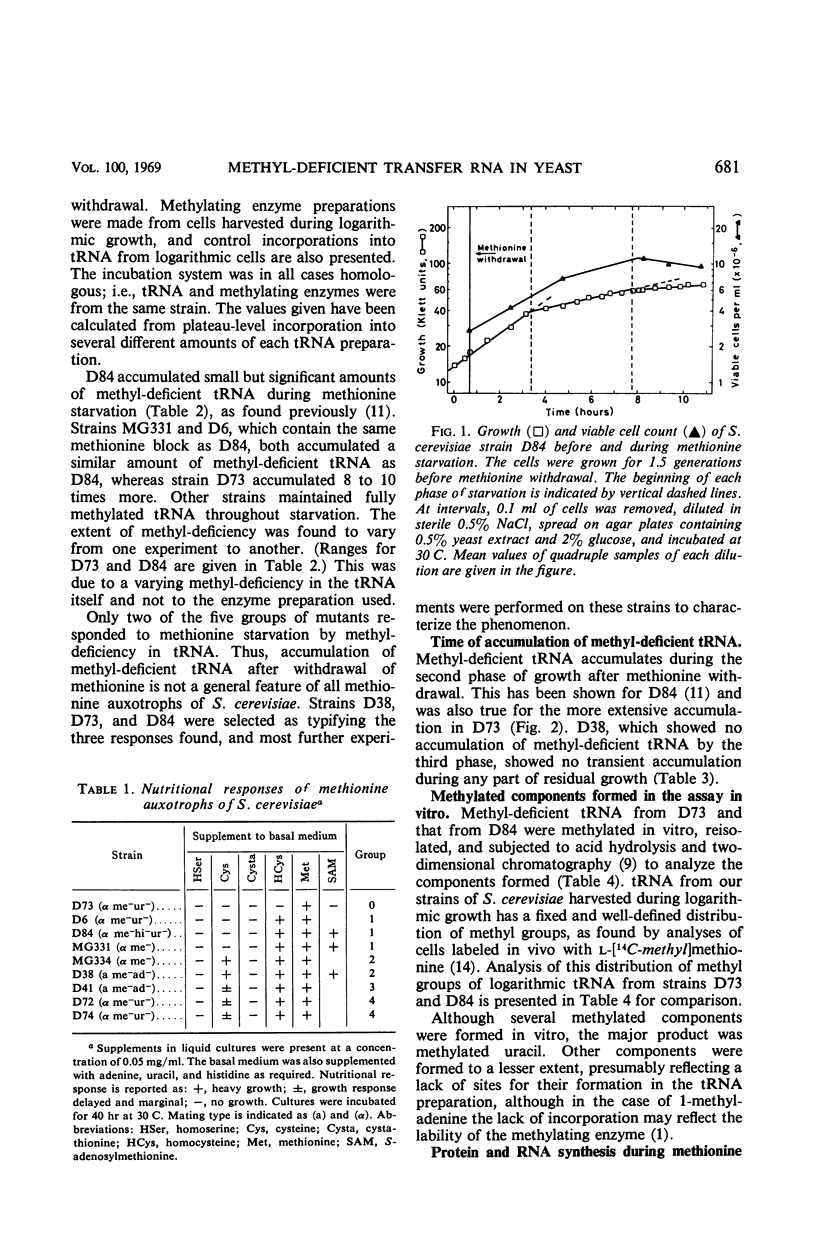
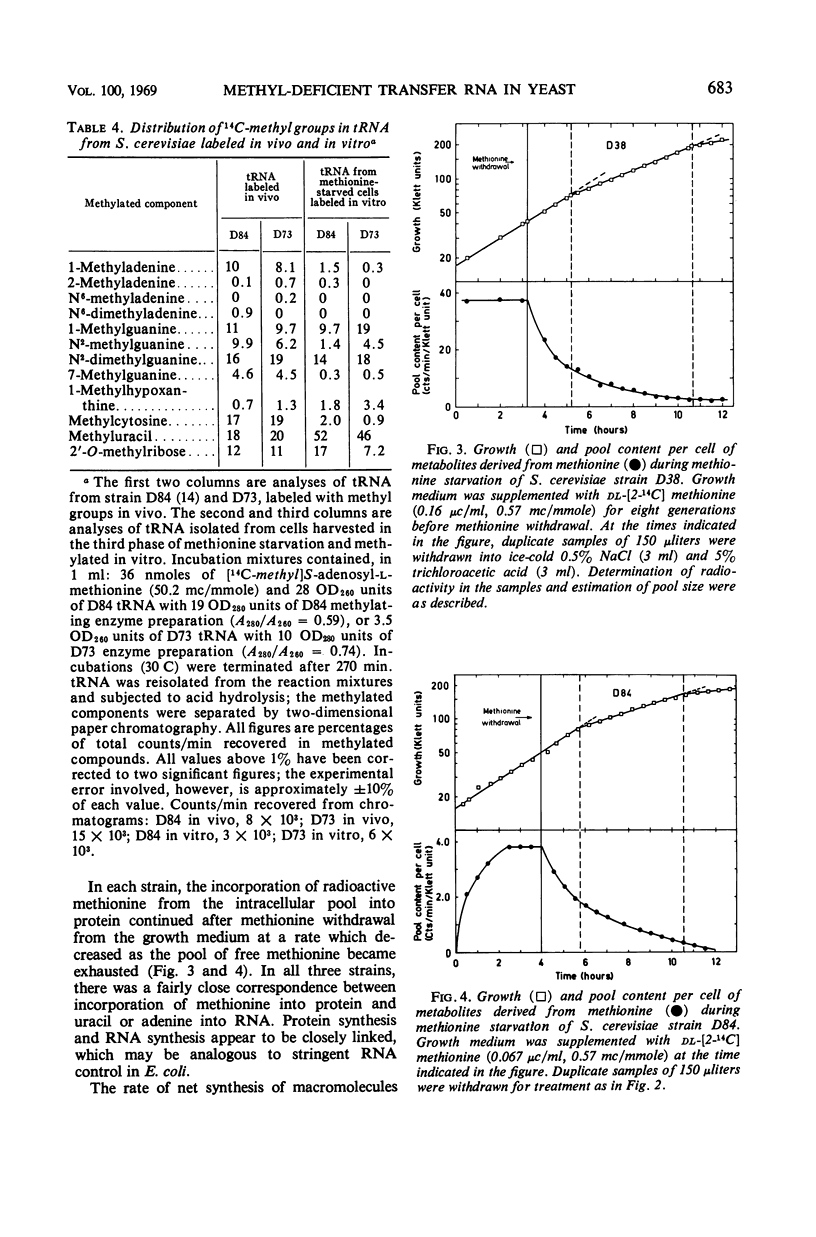
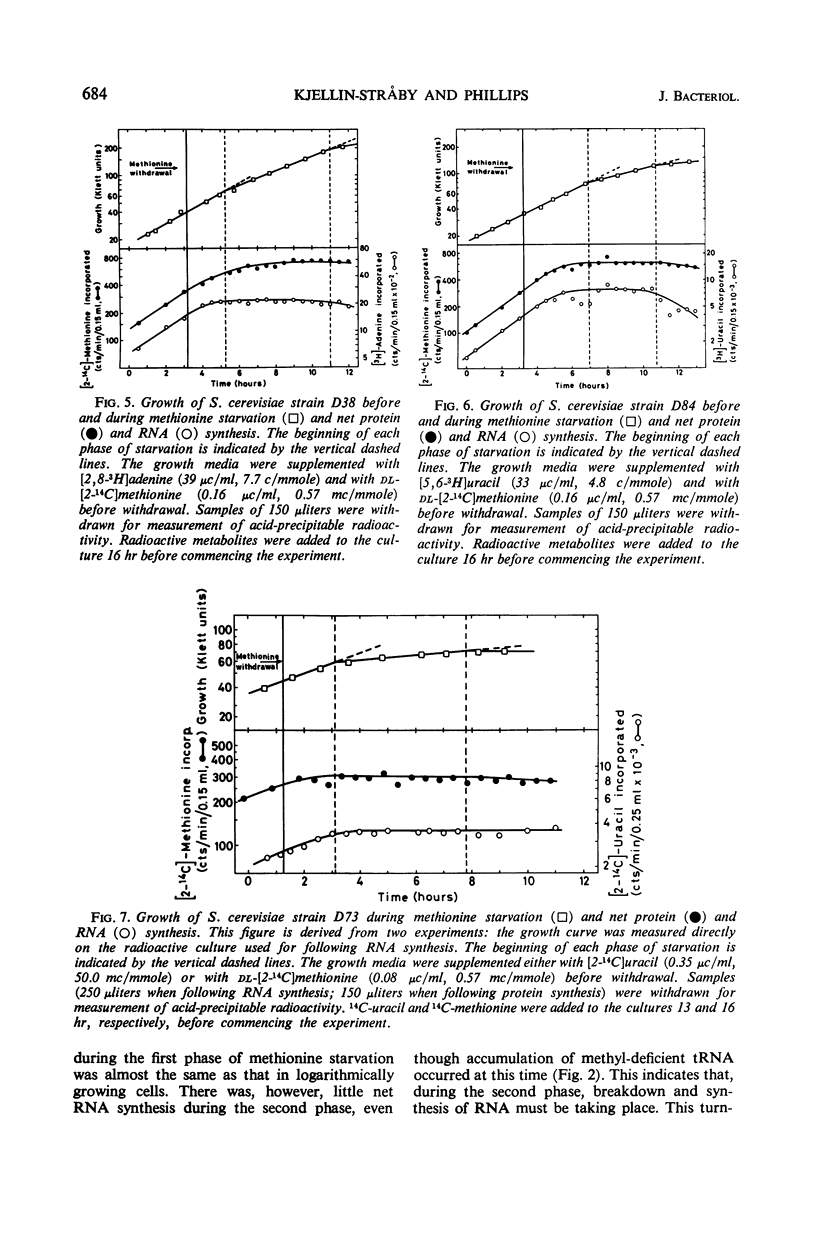
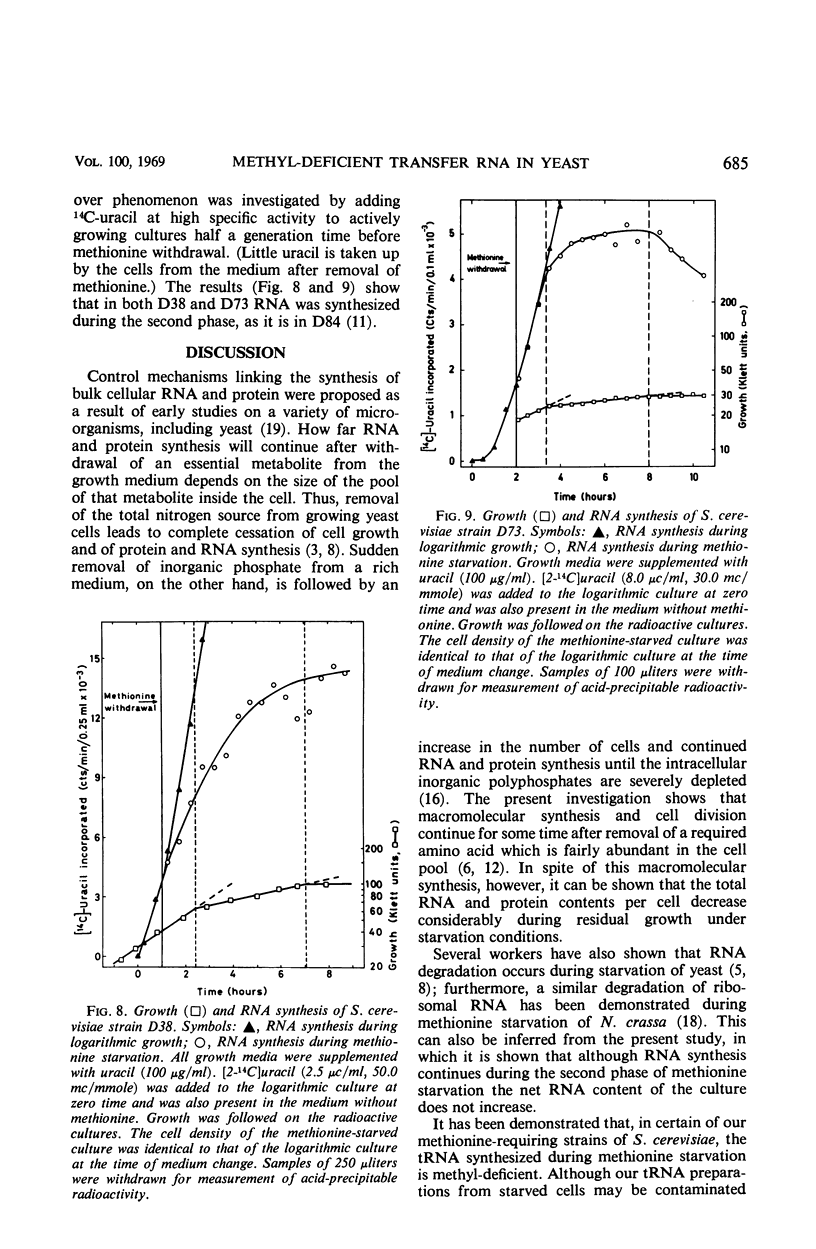
Haploid methionine auxotrophs of Saccharomyces cerevisiae continue to multiply for several hours after withdrawal of a required amino acid from the medium. Macro-molecular synthesis continues during this period of residual growth, although the net ribonucleic acid (RNA) and protein content is constant during the later part of this period. In this study, growth after withdrawal of methionine was in some cases accompanied by accumulation of transfer RNA (tRNA), which was shown by methylation in vitro to be deficient in methyl groups. This phenomenon was shown by only four of nine methionine auxotrophs tested, but no evidence could be found that these four strains had “relaxed” control of RNA synthesis. The nine methionine-requiring strains represent mutations in five different positions in the methionine biosynthesis pathway, and only mutants blocked at two of these five positions accumulated methyl-deficient tRNA. This accumulation therefore appears to be correlated with the position of the strain's block in the pathway of methionine biosynthesis.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Björk G. R., Svensson I. Studies on microbial RNA. Fractionation of tRNA methylases from Saccharomyces cerevisiae. Eur J Biochem. 1969 Jun;9(2):207–215. doi: 10.1111/j.1432-1033.1969.tb00596.x. [DOI] [PubMed] [Google Scholar]
- Edlin G., Broda P. Physiology and genetics of the "ribonucleic acid control" locus in escherichia coli. Bacteriol Rev. 1968 Sep;32(3):206–226. doi: 10.1128/br.32.3.206-226.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HALVORSON H., FRY W., SCHWEMMIN D. A study of the properties of the free amino acid pool and enzyme synthesis in yeast. J Gen Physiol. 1955 Mar 20;38(4):549–573. doi: 10.1085/jgp.38.4.549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HURWITZ J., GOLD M., ANDERS M. THE ENZYMATIC METHYLATION OF RIBONUCLEIC ACID AND DEOXYRIBONUCLEIC ACID. IV. THE PROPERTIES OF THE SOLUBLE RIBONUCLEIC ACID-METHYLATING ENZYMES. J Biol Chem. 1964 Oct;239:3474–3482. [PubMed] [Google Scholar]
- IMAHORI K., KUDO Y. CHARACTERIZATION OF RIBOSOMAL PARTICLES AND RIBONUCLEIC ACIDS OF PROLIFERATING AND STARVING CELLS OF YEAST. Biochim Biophys Acta. 1963 Dec 20;76:525–533. [PubMed] [Google Scholar]
- Isaksson L. A., Phillips J. H. Studies on microbial RNA. V. A comparison of the in vivo methylated components of ribosomal RNA from Escherichia coli and Saccharomyces cerevisiae. Biochim Biophys Acta. 1968 Jan 29;155(1):63–71. [PubMed] [Google Scholar]
- Kjellin-Straby K., Boman H. G. Studies on microbial RNA, 3. Formation of submethylated sRNA in Saccharomyces cerevisiae. Proc Natl Acad Sci U S A. 1965 Jun;53(6):1346–1352. doi: 10.1073/pnas.53.6.1346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kjellin-Stråby K. Influence of methionine pool composition on the formation of methyl-deficient transfer ribonucleic acid in Saccharomyces cerevisiae. J Bacteriol. 1969 Nov;100(2):687–694. doi: 10.1128/jb.100.2.687-694.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kjellin-Stråby K., Phillips J. H. Studies on microbial ribonucleic acid. VI. Appearance of methyl-deficient transfer ribonucleic acid during logarithmic growth of Saccharomyces cerevisiae. J Bacteriol. 1968 Sep;96(3):760–767. doi: 10.1128/jb.96.3.760-767.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morris D. W., Kjeldgaard N. O. Evidence for the non-co-ordinate regulation of ribonucleic acid synthesis in stringent strains of Escherichia coli. J Mol Biol. 1968 Jan 14;31(1):145–148. doi: 10.1016/0022-2836(68)90064-8. [DOI] [PubMed] [Google Scholar]
- Phillips J. H., Kjellin-Stråby K. Studies on microbial ribonucleic acid. IV. Two mutants of Saccharomyces cerevisiae lacking N-2-dimethylguanine in soluble ribonucleic acid. J Mol Biol. 1967 Jun 28;26(3):509–518. doi: 10.1016/0022-2836(67)90318-x. [DOI] [PubMed] [Google Scholar]
- SCHMIDT G., SERAIDARIAN K., GREENBAUM L. M., HICKEY M. D., THANNHAUSER S. J. The effects of certain nutritional conditions on the formation of purines and of ribonucleic acid in baker's yeast. Biochim Biophys Acta. 1956 Apr;20(1):135–149. doi: 10.1016/0006-3002(56)90272-4. [DOI] [PubMed] [Google Scholar]
- Sarkar N., Comb D. G. The predominant site of in vitro uracil methylation of methyl-deficient transfer RNA. J Mol Biol. 1966 Jun;17(2):541–545. doi: 10.1016/s0022-2836(66)80165-1. [DOI] [PubMed] [Google Scholar]
- Shugart L., Chastain B. H., Novelli G. D., Stulberg M. P. Restoration of aminoacylation activity of undermethylated transfer RNA by in vitro methylation. Biochem Biophys Res Commun. 1968 May 10;31(3):404–409. doi: 10.1016/0006-291x(68)90490-7. [DOI] [PubMed] [Google Scholar]
- YCAS M., BRAWERMAN G. Interrelations between nucleic acid and protein biosynthesis in microorganisms. Arch Biochem Biophys. 1957 May;68(1):118–129. doi: 10.1016/0003-9861(57)90332-6. [DOI] [PubMed] [Google Scholar]


