Abstract
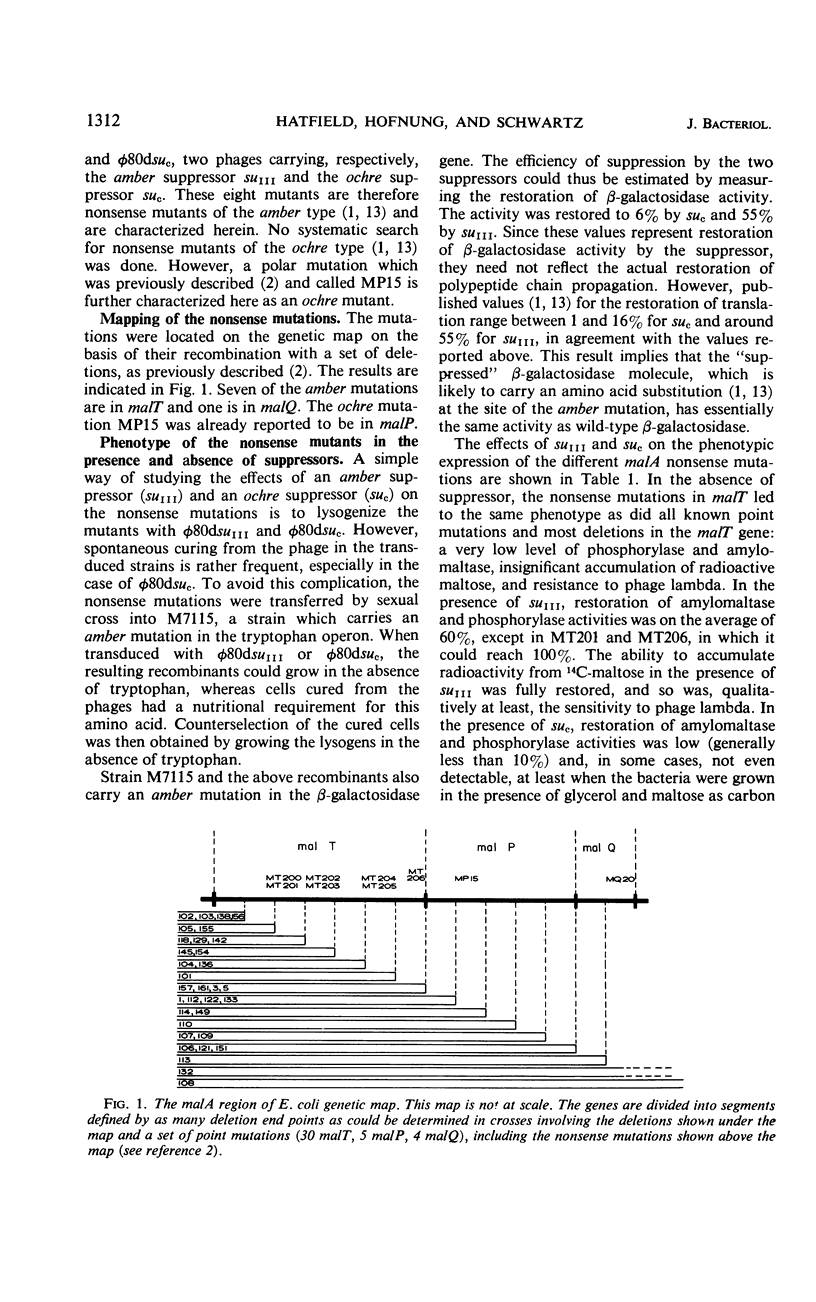
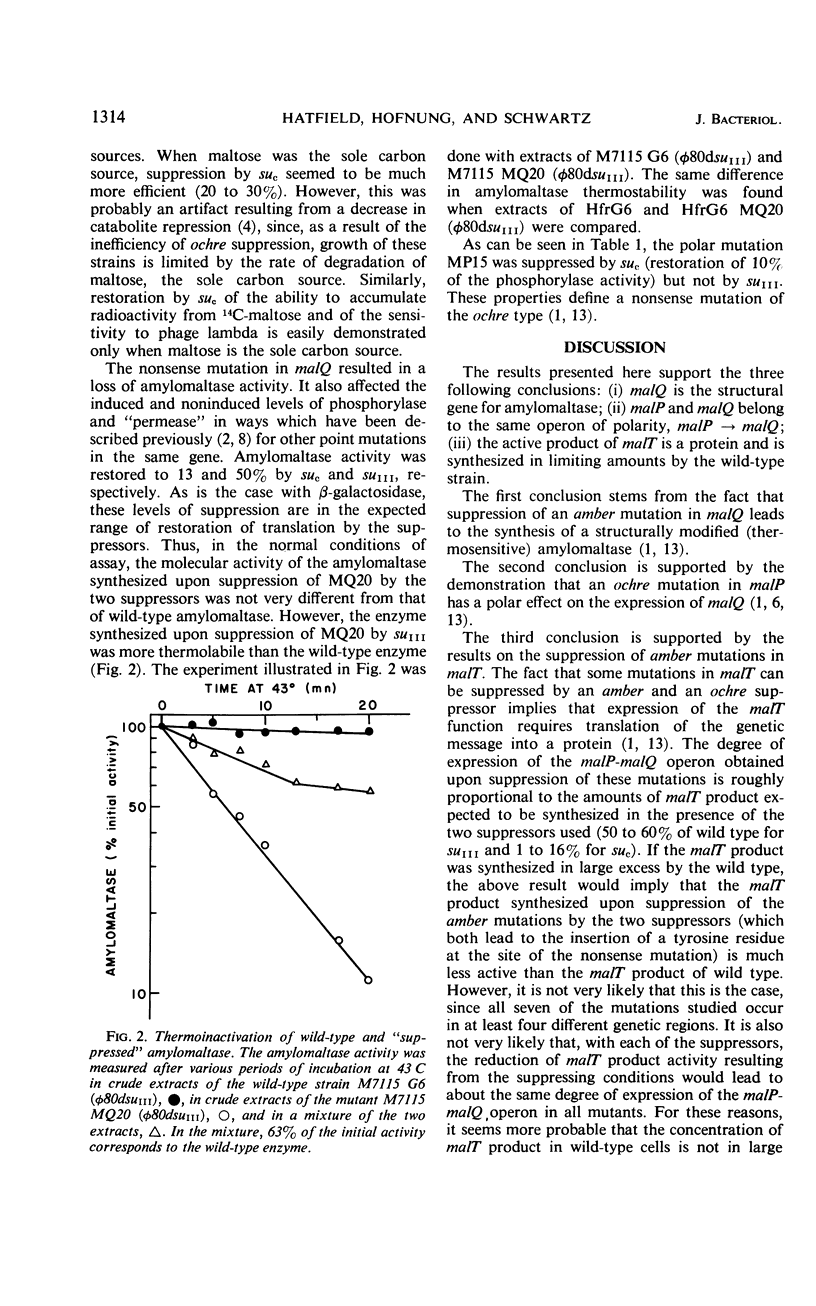
The isolation of one amber mutation in malQ, one ochre mutation in malP, and seven amber mutations in malT is reported. A study of their phenotypic expressions in the presence of the amber suppressor suIII and the ochre suppressor suc suggests that (i) malQ is the structural gene for amylomaltase; (ii) malQ and the structural gene for maltodextrin phosphorylase, malP, belong to the same operon; (iii) the malT product, which promotes the expression of the malP-malQ operon, is a protein synthesized in limiting amounts by the wild-type strain.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Garen A. Sense and nonsense in the genetic code. Three exceptional triplets can serve as both chain-terminating signals and amino acid codons. Science. 1968 Apr 12;160(3824):149–159. doi: 10.1126/science.160.3824.149. [DOI] [PubMed] [Google Scholar]
- Hatfield D., Hofnung M., Schwartz M. Genetic analysis of the maltose A region in Escherichia coli. J Bacteriol. 1969 May;98(2):559–567. doi: 10.1128/jb.98.2.559-567.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henning U., Dennert G., Hertel R., Shipp W. S. Translation of the structural genes of the E. coli pyruvate dehydrogenase complex. Cold Spring Harb Symp Quant Biol. 1966;31:227–234. doi: 10.1101/sqb.1966.031.01.031. [DOI] [PubMed] [Google Scholar]
- MAGASANIK B. Catabolite repression. Cold Spring Harb Symp Quant Biol. 1961;26:249–256. doi: 10.1101/sqb.1961.026.01.031. [DOI] [PubMed] [Google Scholar]
- MONOD J., TORRIANI A. M. De l'amylomaltase d'Escherichia coli. Ann Inst Pasteur (Paris) 1950 Jan;78(1):65–77. [PubMed] [Google Scholar]
- Newton W. A., Beckwith J. R., Zipser D., Brenner S. Nonsense mutants and polarity in the lac operon of Escherichia coli. J Mol Biol. 1965 Nov;14(1):290–296. doi: 10.1016/s0022-2836(65)80250-9. [DOI] [PubMed] [Google Scholar]
- Schwartz M. Expression phénotypique et localisation génétique de mutations affectant le métabolisme du maltose chez Escherichia coli K 12. Ann Inst Pasteur (Paris) 1967 Jun;112(6):673–698. [PubMed] [Google Scholar]
- Schwartz M., Hofnung M. La maltodextrine phosphorylase d'Escherichia coli. Eur J Biochem. 1967 Sep;2(2):132–145. doi: 10.1111/j.1432-1033.1967.tb00117.x. [DOI] [PubMed] [Google Scholar]
- Schwartz M. Location of the maltose A and B loci on the genetic map of Escherichia coli. J Bacteriol. 1966 Oct;92(4):1083–1089. doi: 10.1128/jb.92.4.1083-1089.1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwartz M. Sur l'existence chez Escherichia coli K 12 d'une régulation commune à la biosynthèse des récepteurs du bactériophage et au métabolisme du maltose. Ann Inst Pasteur (Paris) 1967 Nov;113(5):685–704. [PubMed] [Google Scholar]
- Sheppard D. E., Englesberg E. Further evidence for positive control of the L-arabinose system by gene araC. J Mol Biol. 1967 May 14;25(3):443–454. doi: 10.1016/0022-2836(67)90197-0. [DOI] [PubMed] [Google Scholar]
- Signer E. R. Interaction of prophages at the att80 site with the chromosome of Escherichia coli. J Mol Biol. 1966 Jan;15(1):243–255. doi: 10.1016/s0022-2836(66)80224-3. [DOI] [PubMed] [Google Scholar]
- Stretton A. O., Kaplan S., Brenner S. Nonsense codons. Cold Spring Harb Symp Quant Biol. 1966;31:173–179. doi: 10.1101/sqb.1966.031.01.025. [DOI] [PubMed] [Google Scholar]
- Taylor A. L., Trotter C. D. Revised linkage map of Escherichia coli. Bacteriol Rev. 1967 Dec;31(4):332–353. doi: 10.1128/br.31.4.332-353.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WIESMEYER H., COHN M. The characterization of the pathway of maltose utilization by Escherichia coli. III. Adescription of the concentrating mechanism. Biochim Biophys Acta. 1960 Apr 22;39:440–447. doi: 10.1016/0006-3002(60)90196-7. [DOI] [PubMed] [Google Scholar]



