Abstract
Background
Circulating 25 hydroxyvitamin D (25 (OH)D), an accurate measure of vitamin D status, is markedly greater in individuals with increased exposure to ultraviolet B (UVB) light via sunlight or the use of artificial UV light. Aside from the known relationship between vitamin D and bone, vitamin D has also been implicated in immune function and inflammation. Furthermore, a mass of evidence is accumulating that vitamin D deficiency could lead to immune malfunction. Our overall objective was to study the relationship between vitamin D status (as determined by serum 25(OH) D concentrations) and inflammatory markers in healthy women.
Methods
This observational study included 69 healthy women, age 25–82 years. Women with high UVB exposure and women with minimal UVB exposure were specifically recruited to obtain a wide-range of serum 25(OH)D concentrations. Health, sun exposure and habitual dietary intake information were obtained from all subjects. Body composition was determined by dual-energy-x-ray absorptiometry. A fasting blood sample was collected in the morning and analyzed for serum 25(OH)D, parathyroid hormone (iPTH), estradiol (E2), cortisol, and inflammatory markers [tumor necrosis factor -alpha (TNF-α), interleukin-6 and -10 (IL-6, IL-10), and C-reactive protein (CRP)].
Results
Women with regular UVB exposure (Hi-D) had serum 25(OH)D concentrations that were significantly higher (p < 0.0001) and iPTH concentrations that were significantly lower (p < 0.0001) than women without regular UVB exposure (Lo-D). Although IL-6, IL-10, and CRP did not have a statistically significant relationship with 25(OH)D concentrations, linear regression models revealed a significant inverse relationship between serum 25(OH)D and TNF-α concentrations. This relationship remained significant after controlling for potential covariates such as body fat mass, menopausal status, age, or hormonal contraceptive use.
Conclusion
Serum 25(OH)D status is inversely related to TNF-α concentrations in healthy women, which may in part explain this vitamin's role in the prevention and treatment of inflammatory diseases. Results gleaned from this investigation also support the need to re-examine the biological basis for determining optimal vitamin D status.
Background
Circulating 25 hydroxyvitamin D (25(OH)D), an accurate measure of vitamin D status, is markedly increased in individuals who receive regular exposure to ultraviolet B (UVB) light via sunlight or the use of artificial UV light (such as tanning beds) [1-6]. Serum 25(OH)D is hydroxylated in the kidney, as well as in numerous other tissues, to its active form, 1,25-dihydroxyvitamin D (1,25(OH)2D). 1,25(OH)2D binds to nuclear vitamin D receptors in tissues throughout the body. Active vitamin D is responsible for maintaining calcium homeostasis primarily by increasing the efficiency of intestinal calcium absorption and by stimulating the differentiation of bone-resorbing osteoclasts. Furthermore, vitamin D deficiency increases secretion of parathyroid hormone, which accelerates bone breakdown and can lead to decreased bone formation and density [7,8].
There is a growing body of data supporting the contention that desirable serum 25(OH)D concentrations in healthy individuals need to be set higher than the current values to attain the optimal health benefits of vitamin D [8-11], especially the benefits beyond calcium homeostasis [12-14]. For no system does this ring truer than for the influence of vitamin D status on the immune system.
In the last few years, there has been an effort to understand the possible noncalcemic (i.e. non-calcium regulatory) roles of vitamin D, including its role in the immune system [15,16]. Most of the known biological effects of 1,25(OH)2D are mediated through the vitamin D receptor (VDR); and, within the immune system, the VDR is found in significant concentrations in the T lymphocyte and macrophage populations [16]. Moreover, the enzyme responsible for the final and rate-limiting hydroxylation step in the synthesis of active vitamin D, 25(OH)D-1-a-hydroxylase, is expressed by activated macrophages, allowing these phagocytic cells to synthesize and secrete 1,25(OH)2D in a regulated fashion [17]. Additionally, the major 1,25(OH)2D degrading enzyme, 24-hydroxylase, is also expressed in monocytes/macrophages [18]. All of these findings, then, suggest a paracrine role for vitamin D in the immune system [19].
Evidence is accumulating that vitamin D deficiency may lead to immune dysregulation. The relationship between low serum 25(OH)D concentrations and autoimmune disease (especially multiple sclerosis, Type I diabetes and rheumatoid arthritis) has been appreciated for some time [5,20,21]. More recently, studies have shown defective macrophage function, such as impaired chemotaxis, phagocytosis, and increased production of proinflammatory cytokines, in vitamin D-insufficiency [18]. Vitamin D has also been shown to downregulate the expression of monocyte toll-like receptors (TLRs), known inducers of inflammation that can prompt autoimmune disease exacerbation or sepsis [22]. In 2006, a double-blind, randomized, placebo-controlled trial showed that vitamin D supplementation improved cytokine profiles in patients with congestive heart failure [12].
Several provocative reports have been published that also support a role for vitamin D in reducing the risk of certain infectious diseases [23,24], in part through the induction of calthelcidin (also known as hCAP18, LL-37 and FALL-39), an antimicrobial polypeptide [25]. For example, in two seminal papers, Liu et al demonstrated that poor vitamin D status may increase susceptibility to Mycobacterium tuberculosis infection by inefficiently supporting the induction of cathelcidin mRNA in monocytes [26,27].
On balance, the published literature supports the need for further inquiry into vitamin D status and its immune system implications. Thus, our overall objective was to study the relationship between vitamin D status (as determined by serum 25(OH) D concentrations) and inflammatory markers in healthy women. Women with high UVB exposure and women with minimal UVB exposure were specifically recruited to obtain a wide-range of serum 25(OH)D concentrations [1,6,28] We hypothesized that serum 25(OH)D concentrations would be inversely correlated with circulating concentrations of inflammatory markers.
Methods
Subject volunteers
This study used an observational, cross-sectional design to explore the relationship between serum 25(OH)D concentrations and inflammatory marker concentrations in healthy women. Ethical approval for this study was received by the University of Missouri Health Sciences Institutional Review Board (Project number 1069397).
Volunteers were recruited from the University of Missouri-Columbia campus and surrounding community via email notices and flyers posted on campus bulletin boards, and at local tanning salons, fitness and community centers. To be included in the study, volunteers had to be Caucasian females who were at least 25 years of age. High vitamin D women (Hi-D) had to have used a broad-spectrum tanning bed at least once per week for a minimum of four months. Low vitamin D women (Lo-D) had minimal daily sunlight exposure, as assessed by a screening questionnaire, and did not use tanning beds. Volunteers were excluded from the study if they: took a vitamin D supplement other than a regular multivitamin; had a current or previous medical condition or took a medication affecting vitamin D status; had a current or previous medical condition or took a medication affecting immune function; had implanted metal that would interfere with the dual energy x-ray absorptiometry (DXA) scan; were undergoing ultraviolet radiation as medical therapy; exclusively used high-pressure (UVA-only) tanning beds; exercised more than 7 hours per week; were pregnant; or smoked cigarettes.
Following an initial screening for inclusion and exclusion criteria and after obtaining informed written consent, qualified volunteers were scheduled for testing. Subjects were instructed to refrain from exercise and to fast (water only) for 8 to 10 hours prior to their scheduled morning visit. On testing day, all subjects of childbearing age took a urine pregnancy test to confirm non-pregnant status. All study visits were conducted between late January and early June of 2007, the predicted seasonal nadir of solar UVB-produced serum 25(OH)D concentrations in mid-Missourians [29].
Questionnaires and body composition
Four questionnaires were administered to all subjects: a one-page health history and medical questionnaire developed for this study to collect data on previous health conditions or diseases, menopausal status, current or previous medication use, and exercise habits; a one-page sun exposure questionnaire developed for this study to assess tanning bed use, outdoor sun exposure, and sunscreen use; a Fitzpatrick skin typing questionnaire, a well-established method of determining skin pigmentation and response to UVB exposure and thus potential for the photosynthesis of vitamin D in the skin [30]; and, the 88-question, self-administered Harvard Semi-quantitative Food Frequency Questionnaire, a validated tool to assess habitual dietary intake [31].
Body mass was measured without shoes to the nearest 0.1 kg and height to the nearest 0.5 cm using a medical balance beam scale. Body fat and lean body mass were measured by dual energy x-ray absorptiometry (DXA, Hologic Delphi A bone densitometer, Bedford, MA).
Blood collection
All blood was drawn between the hours of 7:30 am and 11:30 am. Venous blood was collected into vacutainer tubes and allowed to clot at room temperature for 30 minutes. The coagulated blood was centrifuged; the serum was aliquoted into sterile microcentrifuge tubes, and stored at -80°C.
Measurement of serum 25(OH)D
25(OH)D serum concentrations were measured using a 125I radioimmunoassay (RIA) kit (Diasorin, Stillwater, MN, Intra-assay CV = 10.8%). The 25(OH)D RIA is a two-step procedure. First, 25(OH)D and other hydroxylated metabolites are rapidly extracted from serum using acetonitrile. The extracted sample is then assayed using an antibody with specificity to 25(OH)D.
Measurement of parathyroid hormone, estradiol and cortisol
Serum intact-PTH (iPTH) was measured using a commercially-available iPTH (1–84) enzyme-linked immunosorbent assay (ELISA) (ALPCO Diagnositics, Salem, NH, Intra-assay CV = 2.5%). Serum estradiol and cortisol were also measured using commercially available ELISA kits (ALPCO Diagnostics, Salem, NH, Intra-assay CV = 7.7% and 5.8%, respectively).
Measurement of inflammatory markers
Four inflammatory markers were measured: IL-10, C-reactive protein (CRP), IL-6, and TNF-α. IL-10, IL-6, and TNF-α, were measured using commercially available high sensitivity ELISA kits (R&D Systems Inc., Minneapolis, MN, Intra-assay CV = 5.3%, 7.4%, and 7.7%, respectively). An ELISA was also used to measure CRP (R&D Systems Inc., Minneapolis, MN, Intra-assay CV = 5.5%).
Statistical analysis
Unpaired two-tailed t-tests were used to determine differences in subject characteristics and measured outcomes; for data not normally distributed or of unequal variance, a rank-sum test was performed. Linear regression and univariate multiple regression models were used to determine the relationships between serum 25(OH)D and serum inflammatory markers. All statistics were performed using SAS statistical software version 9.1 (SAS Inc, Cary, NC). Statistical significance was accepted when P < 0.05.
Results
Vitamin D status
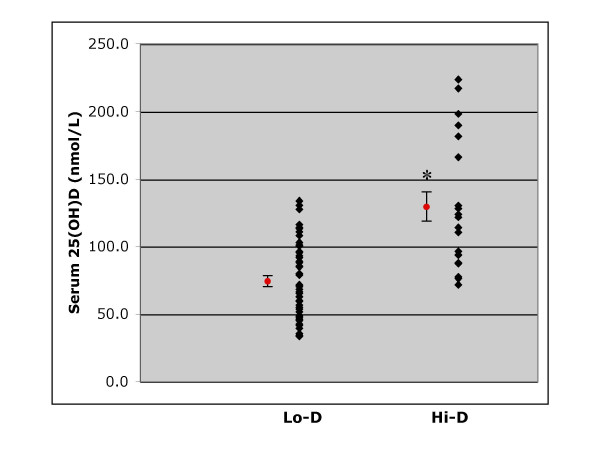
Serum 25(OH)D concentrations of all subjects are presented in Figure 1. Sixty-nine women between the ages of 25 and 82 years participated in the study. Forty-nine of the women were classified as Lo-D and 20 women were classified as Hi-D based on UVB exposure. The mean serum 25(OH)D status (nmol/L) of the Hi-D women (129.6 ± 11.0 nmol/L) was significantly higher than that of the Lo-D women (74.4 ± 4.0 nmol/L) (P < 0.0001).
Figure 1.
Serum 25(OH)D concentrations of Lo-D and Hi-D status women. Mean (± SEM) serum 25(OH)D concentrations of healthy women, age 25–82 years, categorized as low vitamin D status (Lo-D; n = 49) or high vitamin D status (Hi-D; n = 20) based on UVB exposure. Single points for each category are means (± SEMS). *Significantly different from Lo-D, P < 0.0001.
Subject characteristics and serum hormone concentrations
Subject characteristics and serum hormone concentrations by vitamin D status are presented in Table 1. There were no significant differences in age, height, weight, BMI, percent body fat, hormonal contraceptive use or serum estradiol or cortisol concentrations between vitamin D status groups. The mean iPTH concentration of the Hi-D women was significantly lower than that of the Lo-D women (P < 0.0001). Furthermore, there was a significant inverse relationship between 25(OH)D and iPTH concentrations (R2 = 0.2498; P = 0.0001). The skin type of the Hi-D was significantly higher than that of the Lo-D group (P = 0.0031). The Fitzpatrick skin typing method determines skin type based on pigmentation and ability to burn and/or tan with sun exposure (Type I-IV, lighter-darker) [30]. Thus, it is not surprising that the Hi-D women had a higher-level skin type because their skin is capable of tanning; while women with lower-level skin types would not be expected to use a tanning bed since their skin is less able to tan. There were no differences between vitamin D status groups for dietary intakes of energy, macronutrients including omega-3 fatty acids, alcohol or caffeine (data not shown).
Table 1.
Subject characteristics and serum hormone concentrations.
| Characteristic/Hormone | Lo-D (n = 49) | Hi-D (n = 20) | P Value |
| Age (years) | 39.8 ± 1.8 | 41.7 ± 3.5 | 0.5733 |
| Height (m) | 1.70 ± 0.01 | 1.65 ± 0.01 | 0.6894 |
| Weight (kg) | 65.9 ± 1.6 | 67.9 ± 2.7 | 0.3800 |
| Body Mass Index (kg/m2) | 23.8 ± 0.5 | 25.0 ± 1.1 | 0.2488 |
| Body Fat (%) | 30.1 ± 1.0 | 30.6 ± 1.7 | 0.7665 |
| Skin Type | 2.4 ± 0.1 | 3.1 ± 0.2* | 0.0031 |
| Contraceptive Use (%) | 31% | 20% | 0.3780 |
| Estradiol (pg/mL) | 158.0 ± 15.6 | 151.3 ± 15.3 | 0.7974 |
| Cortisol (μg/dL) | 8.4 ± 0.5 | 9.4 ± 1.2 | 0.3129 |
| iPTH (pg/mL) | 48.1 ± 3.1 | 26.2 ± 2.6* | <0.0001 |
Subject characteristics and serum hormone concentrations of healthy women, age 25–82 years, categorized as low vitamin D status (Lo-D) or high vitamin D status (Hi-D) based on UVB exposure. Data are expressed as means ± SEM. *Significantly different from Lo-D, P < 0.05.
Inflammatory marker outcomes
Mean serum TNF-α was significantly lower in the Hi-D than the Lo-D women (1.22 ± 0.11 vs. 0.79 ± 0.11, P = 0.0200. IL-10, CRP and IL-6 did not significantly differ between groups.
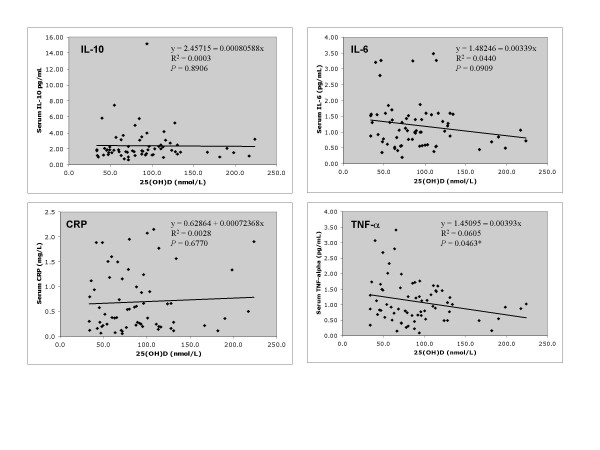
Figure 2 shows the relationships between 25 (OH)D and IL-10, CRP, IL-6, and TNF-α. Serum 25(OH)D concentrations were negatively correlated with TNF-α (R2 = 0.0605, P = 0.0463). Thus, serum 25(OH)D status explained 6.05% of the variation in TNF-α concentrations. IL-10, CRP and IL-6 concentrations were not significantly associated with the concentration of 25(OH)D in serum.
Figure 2.
The relationship between serum 25(OH)D concentrations and inflammatory marker concentrations. The relationship between serum 25(OH)D concentrations and serum IL-10, C-reactive protein (CRP), IL-6 and TNF-a concentrations in healthy women, ages 25–82 years (n = 69). Linear regression equations for each inflammatory marker are shown. * Slope of regression line significantly less than zero, P < 0.05.
When controlling for percent body fat, menopausal status, age, serum estradiol, serum cortisol, and hormonal contraceptive use, a significant relationship (P < 0.05) remained between 25(OH)D and TNF-α. Controlling for percent body fat, menopausal status, age, serum estradiol, serum cortisol, and hormonal contraceptive use did not change the relationship between 25(OH)D concentrations and IL-6, IL-10, and CRP. Analysis of potential covariates revealed a significant positive association between age and IL-6 (R2 = 0.09413, P = 0.0116); and menopausal status and IL-6 (R2 = 0.0764, P = 0.0246).
Discussion
The objective of the present study was to determine the relationship between 25(OH)D concentrations and inflammatory marker concentrations in healthy women. Although IL-6, IL-10, and CRP did not have a statistically significant relationship with 25(OH)D concentrations, linear regression models revealed a significant inverse relationship between serum 25(OH)D and serum TNF-α concentrations. This relationship remained significant after controlling for potential covariates such as body fat mass, menopausal status, age, or hormonal contraceptive use. Hinton et al. found that hormonal contraceptive use was associated with greater TNF-α concentrations in young female athletes [32]. Our data from healthy female non-athletes representing a much wider age range did not reveal such a relationship with TNF-α (P = 0.2336); however, like Hinton et al., there was a significant relationship between hormonal contraceptive and serum cortisol level (P = 0.0030). Interestingly, in our study serum 25(OH)D remained a significant predictor of TNF-α even after controlling for contraceptive use and cortisol concentrations. The lack of significance between serum estradiol and any of the inflammatory markers (data not shown) supports previous research indicating that, in premenopausal women, menstrual phase may affect circulating cytokine concentrations, but the impact is generally not detectable [33].
TNF-α is produced by numerous cell types, including macrophages, monocytes, T-cells, smooth muscle cells, adipocytes, and fibroblasts [34] many of which also have VDR [14,15,35]. Thus, it is difficult to discern the specific mechanisms by which elevations in systemic 25(OH)D attenuate circulating TNF-α concentrations. Nonetheless, our results agree with experimental data showing that vitamin D is capable of suppressing TNF-α production [36-39]. Zhu et al. recently showed that in the colonic tissue of mice with inflammatory bowel disease, 1,25(OH)2D was capable of down-regulating several genes associated with TNFα, including proteins involved in the transcription of TNFα, one of its primary receptors, and TNF-α itself [39].
Human studies of diseased populations have also shown beneficial effects of vitamin D status on TNF-α concentrations. Serum concentrations of TNF-α increased in unsupplemented congestive heart failure patients over a period of 9 months, whereas serum TNF-α concentrations in patients receiving daily supplementation of vitamin D (2000 IU) remained constant [12]. Calcitriol (1,25(OH)2D3) supplementation for 6 months in post-menopausal women with osteoporosis resulted in a significant reduction in serum TNF-α concentrations and an increase in bone mineral density [40]. Additionally, six months of calcitriol supplementation in hemodialysis patients also caused significant decreases in serum TNF-α [41]. Our study is the first to show a significant inverse relationship between serum 25(OH)D and TNF-α concentration in a healthy population.
TNF-α concentrations are increased in several disease states such multiple sclerosis (MS), inflammatory bowel disease (IBD), rheumatoid arthritis (RA), heart disease, and osteoporosis; and are often correlated with clinical impairment [42,43]. Therefore, attenuating the concentrations of circulating TNF-α has the potential to positively impact the risk for or treatment of such conditions. Our data suggest that serum 25(OH)D status explains ~6% of the variation in TNF-α concentrations in healthy women, thus a mild relationship.
Even a slight drop in circulating TNF-α due to improved vitamin D status may have clinical significance. MS patients with < 2 active brain lesions visible on magnetic resonance imagery were shown to have serum TNF-α concentrations that were slightly but significantly (1.6 pg/mL) less than those with ≥ 2 active brain lesions [44]. Patients with active ulcerative colitis were found to have 41% greater mean TNF-α concentrations than those with inactive disease (9.46 and 5.54 pg/mL, respectively); while, those with active Crohn's disease had TNF-α concentrations that were only 18% greater than patients with inactive Crohn's (14.0 and 11.5 pg/mL, respectively) [45].
Increases in circulating TNF-α concentrations have been associated with heart disease progression. Koller-Strametz reported that TNF-α concentrations were 3.2 ± 0.2 pg/mL in patients with New York Heart Association (NYHA) function class II, 4.0 ± 0.3 pg/mL in NYHA function class III patients, and 5.3 ± 0.9 pg/mL in NYHA function class IV patients [46].
Anti-TNF-α medications are efficacious in the management of IBD [47]. Martinez-Borra et al. found that patients with lower TNF-α concentrations (14 ± 25 pg/mL) prior to treatment with the anti-TNF drug, infliximab, responded to the treatment, whereas non-responders had significantly higher baseline serum concentrations (201 ± 362 pg/mL) [48]. Therefore, it is possible vitamin D supplementation may be a viable adjunct to anti-TNF therapy.
Human studies involving diseased populations have shown positive relationships between 25(OH)D concentrations and IL-10 [12]. Despite this evidence, in the present study, serum IL-10 was not significantly correlated with serum 25(OH)D, suggesting that in healthy adults, vitamin D status does not affect IL-10 secretion into systemic circulation.
Similarly, serum 25(OH)D and serum CRP were not correlated in the present study. As a non-specific inflammatory marker of general wellness, CRP increases with mild chronic infection, aging, and tissue damage [49]. Research in diseased populations, such as diabetes [50], arthritis [51,52], prolonged chronic illness [53], and clinical vitamin D deficiency (25(OH)D <27.5 nmol/L) [54] have demonstrated negative associations between vitamin D status and CRP concentrations. Nevertheless, intervention studies of healthy post-menopausal women [55] and patients with congestive heart failure [12] failed to see changes in CRP concentrations after vitamin D supplementation.
Although the result of the linear regression analysis was not statistically significant, there appears to be a slight tendency towards an inverse relationship between 25(OH)D concentrations and serum IL-6 (P = 0.0909). Several in vitro studies have shown that 1,25(OH)2D and several of its analogs are capable of inhibiting the production of IL-6 in various cell types [38,56-59]; while most published in vivo studies have failed to show an effect of vitamin D status on circulating IL-6 concentrations in humans [12,52,60,61]. One report, however, involving hemodialysis patients with elevated parathyroid hormone (PTH) demonstrated that both oral and intravenous 1,25(OH)2D supplementation were capable of significantly decreasing serum IL-6 concentrations following 6 months of treatment [62]. It has been well documented that PTH induces the production of IL-6 by osteoblasts [63,64], thus, it is likely that the effects of vitamin D supplementation on serum IL-6 in this population were mediated primarily through the inverse relationship between 25(OH)D and PTH. In our study, there was no relationship between intact PTH and IL-6 concentrations (P = 0.8039). The significant relationship found between age and IL-6 (P = 0.0116) in this study was anticipated due to several reports showing that circulating IL-6 concentrations increase with advancing age [65-68]. Further, IL-6 has been implicated in age-associated diseases (such as lymphoproliferative disorders, multiple myeloma, osteoporosis, and Alzheimer's disease) and frailty; and, it is postulated that certain clinically important late-life changes are due to an inappropriate presence of IL-6. Therefore, our results indicating a trend for a negative relationship between vitamin D status and IL-6 concentrations warrants further investigation. The lowering of circulating IL-6 through the improvement of vitamin D nutriture may have the potential to decrease disability and mortality in older populations in addition to helping maintain muscle strength and bone health.
The range of serum 25 (OH)D concentrations observed in our healthy female subjects are in accordance with the overwhelming number of reports documenting the prevalence of vitamin D deficiency and insufficiency in the general population [69-75]. In recent years, mounting data has highlighted the need to re-examine vitamin D status and recommendations [76]. Bischoff-Ferrari et al. summarized results from randomized controlled trials, prospective and cross-sectional epidemiologic studies, strong mechanistic evidence, and dose-response relationships to determine an optimal serum 25(OH)D concentration [77]. They showed that for all endpoints (bone mineral density, lower-extremity function, dental health, and risk of falls, fractures, and colorectal cancer), optimal 25(OH)D status began at 75 nmol/L. Our study demonstrates that like these other health outcomes, circulating TNF-α concentrations continue to be associated with serum 25(OH)D concentrations above this point, in a manner consistent with decreased disease risk/progression (i.e. lower TNF-α concentrations).
The primary limitation of this study was sample size. Women who were regularly exposed to UVB light and qualified to participate based on our inclusion and exclusion criteria were far more difficult to recruit than women with minimal UVB exposure. Additionally, women who tan regularly are inherently different from non-tanners. Frequent tanning bed use is associated with high risk behaviours, including frequent dieting, laxative use or vomiting to control weight, cigarette smoking, binge drinking, and recreational drug use. [78]. In light of this, the present study was designed to control or account for these behaviors through subject inclusion/exclusion criteria and inclusion of pertinent questionnaire data in the multiple regression analysis.
Conclusion
Serum TNF-α concentrations are negatively correlated with vitamin D status in healthy women. This study is the first known report to show this inverse relationship in a non-diseased population. Results gleaned from this investigation also support the need to re-examine the biological basis for determining optimal vitamin D status. More studies are needed to fully characterize the relationship between vitamin D and TNF-α relationship; but if proven effective, vitamin D therapy may show promise as adjunct to anti-TNF therapy in inflammatory disease states.
Abbreviations
1,25(OH)2D: 1,25-dihydroxyvitamin D; 25(OH)D: 25-hydroxyvitamin D; CRP: C-reactive protein; DXA: dual-energy x-ray absorptiometry; Hi-D: high vitamin D status; Lo-D: low vitamin D status; IL-6: interleukin-6; IL-10: interleukin 10; TNF-α: tumor necrosis factor-alpha.
Competing interests
The authors declare that they have no competing interests.
Authors' contributions
CAP developed the project idea and study design; obtained IRB approval; and wrote the manuscript. MEH, the graduate student under CAP's mentorship, coordinated the project including subject recruitment, testing, sample collection and analyses. Both authors contributed to the final edits of the manuscript.
Acknowledgments
Acknowledgements
This work was supported by the University Of Missouri Department Of Nutritional Sciences and the University of Missouri Research Council (grant #C2250048). The authors would like to thank the Schade family for their generous support of MEH through the establishment of the Maxine Seabaugh Schade Graduate Fellowship. The authors also wish to thank Laura Hillman for her assistance with the 25(OH)D assay and Dr. Mark Ellersieck for his assistance with the statistical analysis
Contributor Information
Catherine A Peterson, Email: petersonca@missouri.edu.
Mary E Heffernan, Email: heffernanm@missouri.edu.
References
- Grant WB, Garland CF, Holick MF. Comparisons of estimated economic burdens due to insufficient solar ultraviolet irradiance and vitamin D and excess solar UV irradiance for the United States. Photochem Photobiol. 2005;81:1276–1286. doi: 10.1562/2005-01-24-RA-424. [DOI] [PubMed] [Google Scholar]
- Grant WB, Garland CF. The association of solar ultraviolet B (UVB) with reducing risk of cancer: multifactorial ecologic analysis of geographic variation in age-adjusted cancer mortality rates. Anticancer Res. 2006;26:2687–2699. [PubMed] [Google Scholar]
- Gronowitz E, Larko O, Gilljam M, Hollsing A, Lindblad A, Mellstrom D, Strandvik B. Ultraviolet B radiation improves serum levels of vitamin D in patients with cystic fibrosis. Acta Paediatr. 2005;94:547–552. doi: 10.1080/08035250410025276. [DOI] [PubMed] [Google Scholar]
- Koutkia P, Lu Z, Chen TC, Holick MF. Treatment of vitamin D deficiency due to Crohn's disease with tanning bed ultraviolet B radiation. Gastroenterology. 2001;121:1485–1488. doi: 10.1053/gast.2001.29686. [DOI] [PubMed] [Google Scholar]
- Ponsonby AL, Lucas RM, vanderMei IA. UVR, vitamin D and three autoimmune diseases--multiple sclerosis, type 1 diabetes, rheumatoid arthritis. Photochem Photobiol. 2005;81:1267–1275. doi: 10.1562/2005-02-15-IR-441. [DOI] [PubMed] [Google Scholar]
- Tangpricha V, Turner A, Spina C, Decastro S, Chen TC, Holick MF. Tanning is associated with optimal vitamin D status (serum 25-hydroxyvitamin D concentration) and higher bone mineral density. Am J Clin Nutr. 2004;80:1645–1649. doi: 10.1093/ajcn/80.6.1645. [DOI] [PubMed] [Google Scholar]
- Holick MF. The role of vitamin D for bone health and fracture prevention. Curr Osteoporos Rep. 2006;4:96–102. doi: 10.1007/BF02686953. [DOI] [PubMed] [Google Scholar]
- Heaney RP. The Vitamin D requirement in health and disease. J Steroid Biochem Mol Biol. 2005;97:13–19. doi: 10.1016/j.jsbmb.2005.06.020. [DOI] [PubMed] [Google Scholar]
- Holick MF. High prevalence of vitamin D inadequacy and implications for health. Mayo Clin Proc. 2006;81:353–373. doi: 10.4065/81.3.353. [DOI] [PubMed] [Google Scholar]
- Holick MF. Resurrection of vitamin D deficiency and rickets. J Clin Invest. 2006;116:2062–2072. doi: 10.1172/JCI29449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chapuy MC, Preziosi P, Maamer M, Arnaud S, Galan P, Hercberg S, Meunier PJ. Prevalence of vitamin D insufficiency in an adult normal population. Osteoporos Int. 1997;7:439–443. doi: 10.1007/s001980050030. [DOI] [PubMed] [Google Scholar]
- Schleithoff SS, Zittermann A, Tenderich G, Berthold HK, Stehle P, Koerfer R. Vitamin D supplementation improves cytokine profiles in patients with congestive heart failure: a double-blind, randomized, placebo-controlled trial. Am J Clin Nutr. 2006;83:754–759. doi: 10.1093/ajcn/83.4.754. [DOI] [PubMed] [Google Scholar]
- Holick MF. Vitamin D: its role in cancer prevention and treatment. Prog Biophys Mol Biol. 2006;92:49–59. doi: 10.1016/j.pbiomolbio.2006.02.014. [DOI] [PubMed] [Google Scholar]
- Cantorna MT. Vitamin D and autoimmunity: is vitamin D status an environmental factor affecting autoimmune disease prevalence? Proc Soc Exp Biol Med. 2000;223:230–233. doi: 10.1046/j.1525-1373.2000.22333.x. [DOI] [PubMed] [Google Scholar]
- Cantorna MT, Zhu Y, Froicu M, Wittke A. Vitamin D status, 1,25-dihydroxyvitamin D3, and the immune system. Am J Clin Nutr. 2004;80:1717S–1720S. doi: 10.1093/ajcn/80.6.1717S. [DOI] [PubMed] [Google Scholar]
- Deluca HF, Cantorna MT. Vitamin D: its role and uses in immunology. FASEB J. 2001;15:2579–2585. doi: 10.1096/fj.01-0433rev. [DOI] [PubMed] [Google Scholar]
- Mathieu C, Adorini L. The coming of age of 1,25-dihydroxyvitamin D(3) analogs as immunomodulatory agents. Trends Mol Med. 2002;8:174–179. doi: 10.1016/S1471-4914(02)02294-3. [DOI] [PubMed] [Google Scholar]
- Overbergh L, Decallonne B, Valckx D, Verstuyf A, Depovere J, Laureys J, Rutgeerts O, Saint-Arnaud R, Bouillon R, Mathieu C. Identification and immune regulation of 25-hydroxyvitamin D-1-alpha-hydroxylase in murine macrophages. Clin Exp Immunol. 2000;120:139–146. doi: 10.1046/j.1365-2249.2000.01204.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- vanEtten E, Mathieu C. Immunoregulation by 1,25-dihydroxyvitamin D3: basic concepts. J Steroid Biochem Mol Biol. 2005;97:93–101. doi: 10.1016/j.jsbmb.2005.06.002. [DOI] [PubMed] [Google Scholar]
- Cutolo M, Otsa K, Uprus M, Paolino S, Seriolo B. Vitamin D in rheumatoid arthritis. Autoimmun Rev. 2007;7:59–64. doi: 10.1016/j.autrev.2007.07.001. [DOI] [PubMed] [Google Scholar]
- Young AR, Walker SL. UV radiation, vitamin D and human health: an unfolding controversy introduction. Photochem Photobiol. 2005;81:1243–1245. doi: 10.1562/2005-10-16-RA-716. [DOI] [PubMed] [Google Scholar]
- Sadeghi K, Wessner B, Laggner U, Ploder M, Tamandl D, Friedl J, Zugel U, Steinmeyer A, Pollak A, Roth E, Boltz-Nitulescu G, Spittler A. Vitamin D3 down-regulates monocyte TLR expression and triggers hyporesponsiveness to pathogen-associated molecular patterns. Eur J Immunol. 2006;36:361–370. doi: 10.1002/eji.200425995. [DOI] [PubMed] [Google Scholar]
- Aloia JF, Li-Ng M. Re: epidemic influenza and vitamin D. Epidemiol Infect. 2007;135:1095–1096. doi: 10.1017/S0950268807008308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zasloff M. Fighting infections with vitamin D. Nat Med. 2006;12:388–390. doi: 10.1038/nm0406-388. [DOI] [PubMed] [Google Scholar]
- Grant WB. Hypothesis--ultraviolet-B irradiance and vitamin D reduce the risk of viral infections and thus their sequelae, including autoimmune diseases and some cancers. Photochem Photobiol. 2008;84:356–365. doi: 10.1111/j.1751-1097.2007.00266.x. [DOI] [PubMed] [Google Scholar]
- Liu PT, Stenger S, Li H, Wenzel L, Tan BH, Krutzik SR, Ochoa MT, Schauber J, Wu K, Meinken C, Kamen DL, Wagner M, Bals R, Steinmeyer A, Zugel U, Gallo RL, Eisenberg D, Hewison M, Hollis BW, Adams JS, Bloom BR, Modlin RL. Toll-like receptor triggering of a vitamin D-mediated human antimicrobial response. Science. 2006;311:1770–1773. doi: 10.1126/science.1123933. [DOI] [PubMed] [Google Scholar]
- Liu PT, Stenger S, Tang DH, Modlin RL. Cutting edge: vitamin D-mediated human antimicrobial activity against Mycobacterium tuberculosis is dependent on the induction of cathelicidin. J Immunol. 2007;179:2060–2063. doi: 10.4049/jimmunol.179.4.2060. [DOI] [PubMed] [Google Scholar]
- Rajakumar K, Greenspan SL, Thomas SB, Holick MF. SOLAR ultraviolet radiation and vitamin D: a historical perspective. Am J Public Health. 2007;97:1746–1754. doi: 10.2105/AJPH.2006.091736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Webb AR, Engelsen O. Calculated ultraviolet exposure levels for a healthy vitamin D status. Photochem Photobiol. 2006;82:1697–1703. doi: 10.1562/2005-09-01-RA-670. [DOI] [PubMed] [Google Scholar]
- Fitzpatrick TB. The validity and practicality of sun-reactive skin types I through VI. Arch Dermatol. 1988;124:869–871. doi: 10.1001/archderm.124.6.869. [DOI] [PubMed] [Google Scholar]
- Salvini S, Hunter DJ, Sampson L, Stampfer MJ, Colditz GA, Rosner B, Willett WC. Food-based validation of a dietary questionnaire: the effects of week-to-week variation in food consumption. Int J Epidemiol. 1989;18:858–867. doi: 10.1093/ije/18.4.858. [DOI] [PubMed] [Google Scholar]
- Hinton PS, Rector.R.S. Peppers JE, Imhoff RD, Hillman LS. Serum markers of inflammation and endothelial function are elevated by hormonal contraceptive use but not exercise-associated menstrual disorders in physically active young women. J Sports Sci Med. 2006;5:235–242. [PMC free article] [PubMed] [Google Scholar]
- Banks RE. Measurement of cytokines in clinical samples using immunoassays: problems and pitfalls. Crit Rev Clin Lab Sci. 2000;37:131–182. doi: 10.1080/10408360091174187. [DOI] [PubMed] [Google Scholar]
- Popa C, Netea MG, van Riel PL, van der Meer JW, Stalenhoef AF. The role of TNF-alpha in chronic inflammatory conditions, intermediary metabolism, and cardiovascular risk. J Lipid Res. 2007;48:751–762. doi: 10.1194/jlr.R600021-JLR200. [DOI] [PubMed] [Google Scholar]
- Cantorna MT, Mahon BD. D-hormone and the immune system. J Rheumatol Suppl. 2005;76:11–20. [PubMed] [Google Scholar]
- Cantorna MT, Woodward WD, Hayes CE, Deluca HF. 1,25-dihydroxyvitamin D3 is a positive regulator for the two anti-encephalitogenic cytokines TGF-beta 1 and IL-4. J Immunol. 1998;160:5314–5319. [PubMed] [Google Scholar]
- Cohen ML, Douvdevani A, Chaimovitz C, Shany S. Regulation of TNF-alpha by 1alpha,25-dihydroxyvitamin D3 in human macrophages from CAPD patients. Kidney Int. 2001;59:69–75. doi: 10.1046/j.1523-1755.2001.00467.x. [DOI] [PubMed] [Google Scholar]
- Evans KN, Nguyen L, Chan J, Innes BA, Bulmer JN, Kilby MD, Hewison M. Effects of 25-hydroxyvitamin D3 and 1,25-dihydroxyvitamin D3 on cytokine production by human decidual cells. Biol Reprod. 2006;75:816–822. doi: 10.1095/biolreprod.106.054056. [DOI] [PubMed] [Google Scholar]
- Zhu Y, Mahon BD, Froicu M, Cantorna MT. Calcium and 1 alpha,25-dihydroxyvitamin D3 target the TNF-alpha pathway to suppress experimental inflammatory bowel disease. Eur J Immunol. 2005;35:217–224. doi: 10.1002/eji.200425491. [DOI] [PubMed] [Google Scholar]
- Inanir A, Ozoran K, Tutkak H, Mermerci B. The effects of calcitriol therapy on serum interleukin-1, interleukin-6 and tumour necrosis factor-alpha concentrations in post-menopausal patients with osteoporosis. J Int Med Res. 2004;32:570–582. doi: 10.1177/147323000403200602. [DOI] [PubMed] [Google Scholar]
- Borazan A, Ustun H, Cefle A, Sekitmez N, Yilmaz A. Comparative efficacy of oral and intravenous calcitriol treatment in haemodialysis patients: effects on serum biochemistry and cytokine levels. J Int Med Res. 2003;31:489–496. doi: 10.1177/147323000303100604. [DOI] [PubMed] [Google Scholar]
- Kieseier BC, Giovannoni G, Hartung HP. Immunological surrogate markers of disease activity in multiple sclerosis. Electroencephalogr Clin Neurophysiol Suppl. 1999;50:570–583. [PubMed] [Google Scholar]
- Prince HE. Biomarkers for diagnosing and monitoring autoimmune diseases. Biomarkers. 2005;10 Suppl 1:S44–S49. doi: 10.1080/13547500500214194. [DOI] [PubMed] [Google Scholar]
- Giovannoni G, Miller DH, Losseff NA, Sailer M, Lewellyn-Smith N, Thompson AJ, Thompson EJ. Serum inflammatory markers and clinical/MRI markers of disease progression in multiple sclerosis. J Neurol. 2001;248:487–495. doi: 10.1007/s004150170158. [DOI] [PubMed] [Google Scholar]
- Komatsu M, Kobayashi D, Saito K, Furuya D, Yagihashi A, Araake H, Tsuji N, Sakamaki S, Niitsu Y, Watanabe N. Tumor necrosis factor-alpha in serum of patients with inflammatory bowel disease as measured by a highly sensitive immuno-PCR. Clin Chem. 2001;47:1297–1301. [PubMed] [Google Scholar]
- Koller-Strametz J, Pacher R, Frey B, Kos T, Woloszczuk W, Stanek B. Circulating tumor necrosis factor-alpha levels in chronic heart failure: relation to its soluble receptor II, interleukin-6, and neurohumoral variables. J Heart Lung Transplant. 1998;17:356–362. [PubMed] [Google Scholar]
- Kam LY, Targan SR. Cytokine-based therapies in inflammatory bowel disease. Curr Opin Gastroenterol. 1999;15:302–307. doi: 10.1097/00001574-199907000-00005. [DOI] [PubMed] [Google Scholar]
- Martinez-Borra J, Lopez-Larrea C, Gonzalez S, Fuentes D, Dieguez A, Deschamps EM, Perez-Pariente JM, Lopez-Vazquez A, de FR, Rodrigo L. High serum tumor necrosis factor-alpha levels are associated with lack of response to infliximab in fistulizing Crohn's disease. Am J Gastroenterol. 2002;97:2350–2356. doi: 10.1111/j.1572-0241.2002.05990.x. [DOI] [PubMed] [Google Scholar]
- Kao PC, Shiesh SC, Wu TJ. Serum C-reactive protein as a marker for wellness assessment. Ann Clin Lab Sci. 2006;36:163–169. [PubMed] [Google Scholar]
- Targher G, Bertolini L, Padovani R, Zenari L, Scala L, Cigolini M, Arcaro G. Serum 25-hydroxyvitamin D3 concentrations and carotid artery intima-media thickness among type 2 diabetic patients. Clin Endocrinol (Oxf) 2006;65:593–597. doi: 10.1111/j.1365-2265.2006.02633.x. [DOI] [PubMed] [Google Scholar]
- Patel S, Farragher T, Berry J, Bunn D, Silman A, Symmons D. Association between serum vitamin D metabolite levels and disease activity in patients with early inflammatory polyarthritis. Arthritis Rheum. 2007;56:2143–2149. doi: 10.1002/art.22722. [DOI] [PubMed] [Google Scholar]
- Oelzner P, Franke S, Muller A, Hein G, Stein G. Relationship between soluble markers of immune activation and bone turnover in post-menopausal women with rheumatoid arthritis. Rheumatology (Oxford) 1999;38:841–847. doi: 10.1093/rheumatology/38.9.841. [DOI] [PubMed] [Google Scholar]
- Vanden Berghe G, VanRoosbroeck D, Vanhove P, Wouters PJ, DePourcq L, Bouillon R. Bone turnover in prolonged critical illness: effect of vitamin D. J Clin Endocrinol Metab. 2003;88:4623–4632. doi: 10.1210/jc.2003-030358. [DOI] [PubMed] [Google Scholar]
- Timms PM, Mannan N, Hitman GA, Noonan K, Mills PG, Syndercombe-Court. Aganna E, Price CP, Boucher BJ. Circulating MMP9, vitamin D and variation in the TIMP-1 response with VDR genotype: mechanisms for inflammatory damage in chronic disorders? QJM. 2002;95:787–796. doi: 10.1093/qjmed/95.12.787. [DOI] [PubMed] [Google Scholar]
- Pittas AG, Harris SS, Stark PC, Dawson-Hughes B. The effects of calcium and vitamin D supplementation on blood glucose and markers of inflammation in nondiabetic adults. Diabetes Care. 2007;30:980–986. doi: 10.2337/dc06-1994. [DOI] [PubMed] [Google Scholar]
- Muller K, Heilmann C, Poulsen LK, Barington T, Bendtzen K. The role of monocytes and T cells in 1,25-dihydroxyvitamin D3 mediated inhibition of B cell function in vitro. Immunopharmacology. 1991;21:121–128. doi: 10.1016/0162-3109(91)90015-Q. [DOI] [PubMed] [Google Scholar]
- Equils O, Naiki Y, Shapiro AM, Michelsen K, Lu D, Adams J, Jordan S. 1,25-Dihydroxyvitamin D inhibits lipopolysaccharide-induced immune activation in human endothelial cells. Clin Exp Immunol. 2006;143:58–64. doi: 10.1111/j.1365-2249.2005.02961.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Komine M, Watabe Y, Shimaoka S, Sato F, Kake K, Nishina H, Ohtsuki M, Nakagawa H, Tamaki K. The action of a novel vitamin D3 analogue, OCT, on immunomodulatory function of keratinocytes and lymphocytes. Arch Dermatol Res. 1999;291:500–506. doi: 10.1007/s004030050444. [DOI] [PubMed] [Google Scholar]
- Riachy R, Vandewalle B, Belaich S, Kerr-Conte J, Gmyr V, Zerimech F, d'Herbomez M, Lefebvre J, Pattou F. Beneficial effect of 1,25 dihydroxyvitamin D3 on cytokine-treated human pancreatic islets. J Endocrinol. 2001;169:161–168. doi: 10.1677/joe.0.1690161. [DOI] [PubMed] [Google Scholar]
- Gannage-Yared MH, Azoury M, Mansour I, Baddoura R, Halaby G, Naaman R. Effects of a short-term calcium and vitamin D treatment on serum cytokines, bone markers, insulin and lipid concentrations in healthy post-menopausal women. J Endocrinol Invest. 2003;26:748–753. doi: 10.1007/BF03347358. [DOI] [PubMed] [Google Scholar]
- Puts MT, Visser M, Twisk JW, Deeg DJ, Lips P. Endocrine and inflammatory markers as predictors of frailty. Clin Endocrinol (Oxf) 2005;63:403–411. doi: 10.1111/j.1365-2265.2005.02355.x. [DOI] [PubMed] [Google Scholar]
- Turk S, Akbulut M, Yildiz A, Gurbilek M, Gonen S, Tombul Z, Yeksan M. Comparative effect of oral pulse and intravenous calcitriol treatment in hemodialysis patients: the effect on serum IL-1 and IL-6 levels and bone mineral density. Nephron. 2002;90:188–194. doi: 10.1159/000049041. [DOI] [PubMed] [Google Scholar]
- Greenfield EM, Shaw SM, Gornik SA, Banks MA. Adenyl cyclase and interleukin 6 are downstream effectors of parathyroid hormone resulting in stimulation of bone resorption. J Clin Invest. 1995;96:1238–1244. doi: 10.1172/JCI118157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li NH, Ouchi Y, Okamoto Y, Masuyama A, Kaneki M, Futami A, Hosoi T, Nakamura T, Orimo H. Effect of parathyroid hormone on release of interleukin 1 and interleukin 6 from cultured mouse osteoblastic cells. Biochem Biophys Res Commun. 1991;179:236–242. doi: 10.1016/0006-291X(91)91360-O. [DOI] [PubMed] [Google Scholar]
- Ershler WB, Keller ET. Age-associated increased interleukin-6 gene expression, late-life diseases, and frailty. Annu Rev Med. 2000;51:245–270. doi: 10.1146/annurev.med.51.1.245. [DOI] [PubMed] [Google Scholar]
- Cappola AR, Xue QL, Ferrucci L, Guralnik JM, Volpato S, Fried LP. Insulin-like growth factor I and interleukin-6 contribute synergistically to disability and mortality in older women. J Clin Endocrinol Metab. 2003;88:2019–2025. doi: 10.1210/jc.2002-021694. [DOI] [PubMed] [Google Scholar]
- Cohen HJ, Pieper CF, Harris T, Rao KM, Currie MS. The association of plasma IL-6 levels with functional disability in community-dwelling elderly. J Gerontol A Biol Sci Med Sci. 1997;52:M201–M208. doi: 10.1093/gerona/52a.4.m201. [DOI] [PubMed] [Google Scholar]
- Ferrucci L, Harris TB, Guralnik JM, Tracy RP, Corti MC, Cohen HJ, Penninx B, Pahor M, Wallace R, Havlik RJ. Serum IL-6 level and the development of disability in older persons. J Am Geriatr Soc. 1999;47:639–646. doi: 10.1111/j.1532-5415.1999.tb01583.x. [DOI] [PubMed] [Google Scholar]
- Hanley DA, Davison KS. Vitamin D insufficiency in North America. J Nutr. 2005;135:332–337. doi: 10.1093/jn/135.2.332. [DOI] [PubMed] [Google Scholar]
- Hypponen E, Power C. Hypovitaminosis D in British adults at age 45 y: nationwide cohort study of dietary and lifestyle predictors. Am J Clin Nutr. 2007;85:860–868. doi: 10.1093/ajcn/85.3.860. [DOI] [PubMed] [Google Scholar]
- Rockell JE, Skeaff CM, Williams SM, Green TJ. Serum 25-hydroxyvitamin D concentrations of New Zealanders aged 15 years and older. Osteoporos Int. 2006;17:1382–1389. doi: 10.1007/s00198-006-0118-x. [DOI] [PubMed] [Google Scholar]
- Stein EM, Laing EM, Hall DB, Hausman DB, Kimlin MG, Johnson MA, Modlesky CM, Wilson AR, Lewis RD. Serum 25-hydroxyvitamin D concentrations in girls aged 4-8 y living in the southeastern United States. Am J Clin Nutr. 2006;83:75–81. doi: 10.1093/ajcn/83.1.75. [DOI] [PubMed] [Google Scholar]
- Vieth R, Cole DE, Hawker GA, Trang HM, Rubin LA. Wintertime vitamin D insufficiency is common in young Canadian women, and their vitamin D intake does not prevent it. Eur J Clin Nutr. 2001;55:1091–1097. doi: 10.1038/sj.ejcn.1601275. [DOI] [PubMed] [Google Scholar]
- Visser M, Deeg DJ, Puts MT, Seidell JC, Lips P. Low serum concentrations of 25-hydroxyvitamin D in older persons and the risk of nursing home admission. Am J Clin Nutr. 2006;84:616–622. doi: 10.1093/ajcn/84.3.616. [DOI] [PubMed] [Google Scholar]
- Zadshir A, Tareen N, Pan D, Norris K, Martins D. The prevalence of hypovitaminosis D among US adults: data from the NHANES III. Ethn Dis. 2005;15:S5–97–101. [PubMed] [Google Scholar]
- Vieth R, Bischoff-Ferrari H, Boucher BJ, Dawson-Hughes B, Garland CF, Heaney RP, Holick MF, Hollis BW, Lamberg-Allardt C, McGrath JJ, Norman AW, Scragg R, Whiting SJ, Willett WC, Zittermann A. The urgent need to recommend an intake of vitamin D that is effective. Am J Clin Nutr. 2007;85:649–650. doi: 10.1093/ajcn/85.3.649. [DOI] [PubMed] [Google Scholar]
- Bischoff-Ferrari HA, Giovannucci E, Willett WC, Dietrich T, Dawson-Hughes B. Estimation of optimal serum concentrations of 25-hydroxyvitamin D for multiple health outcomes. Am J Clin Nutr. 2006;84:18–28. doi: 10.1093/ajcn/84.1.18. [DOI] [PubMed] [Google Scholar]
- O'Riordan DL, Field AE, Geller AC, Brooks DR, Aweh G, Colditz GA, Frazier AL. Frequent tanning bed use, weight concerns, and other health risk behaviors in adolescent females (United States) Cancer Causes Control. 2006;17:679–686. doi: 10.1007/s10552-005-0453-9. [DOI] [PubMed] [Google Scholar]