Abstract
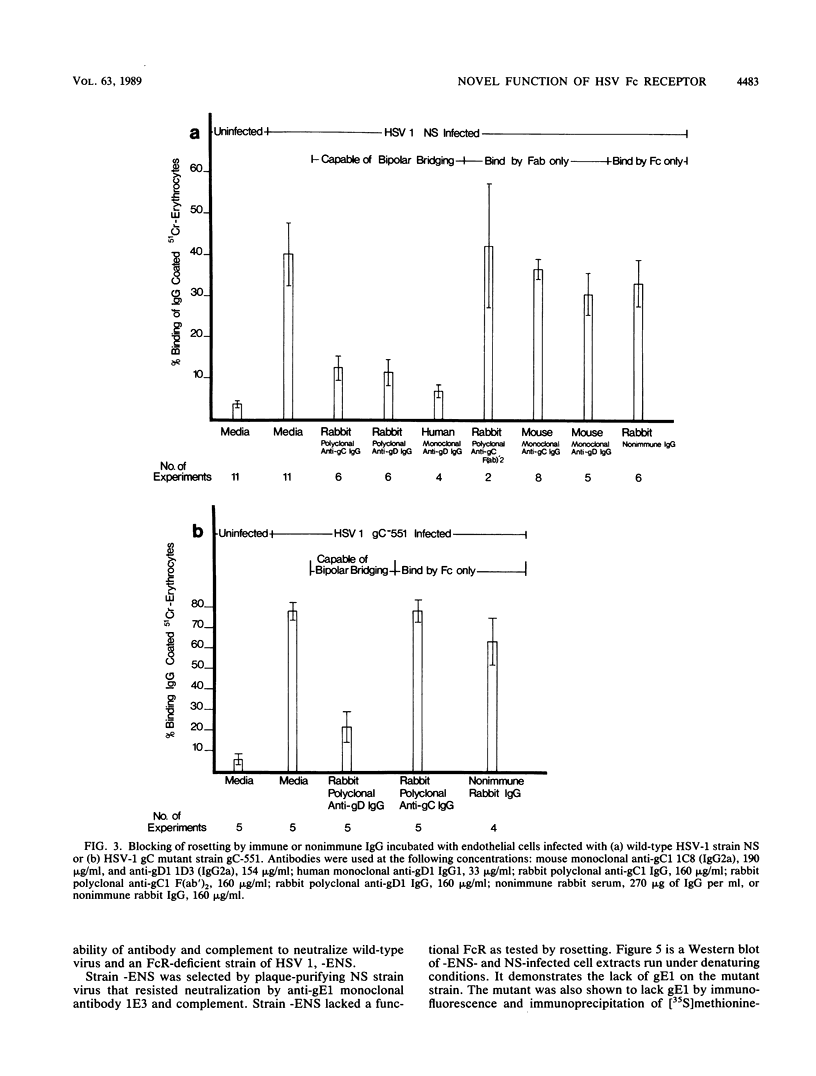
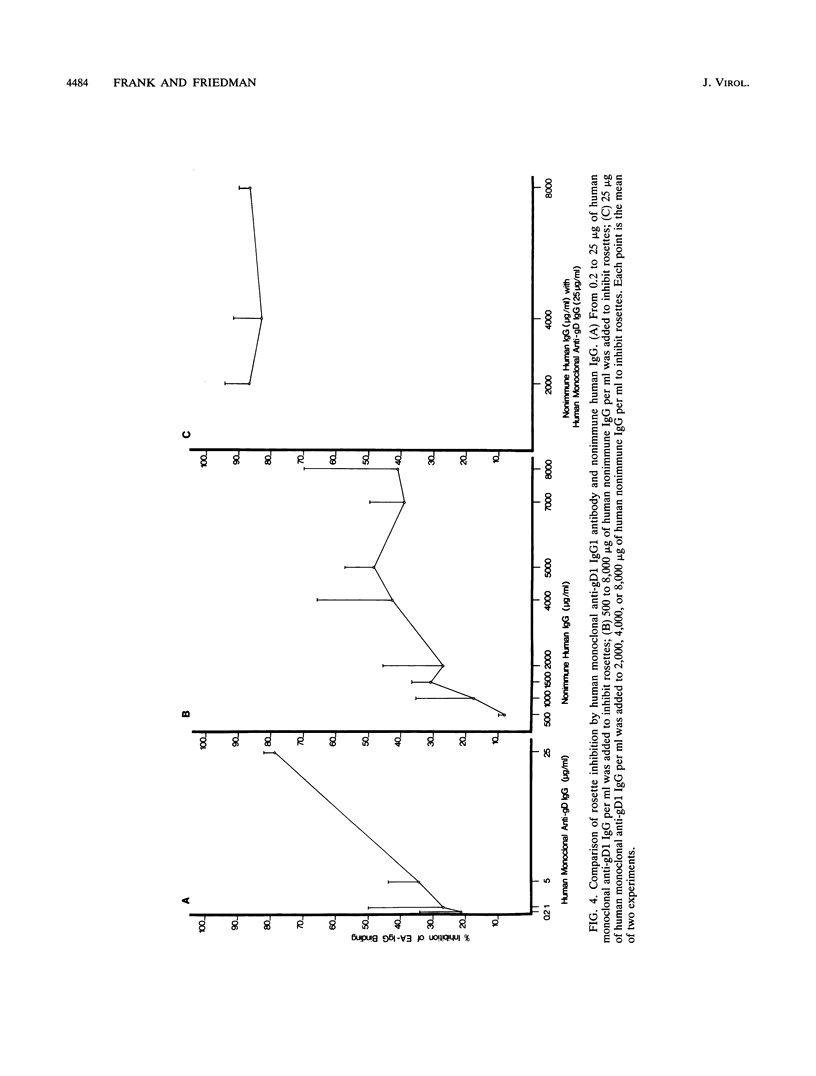
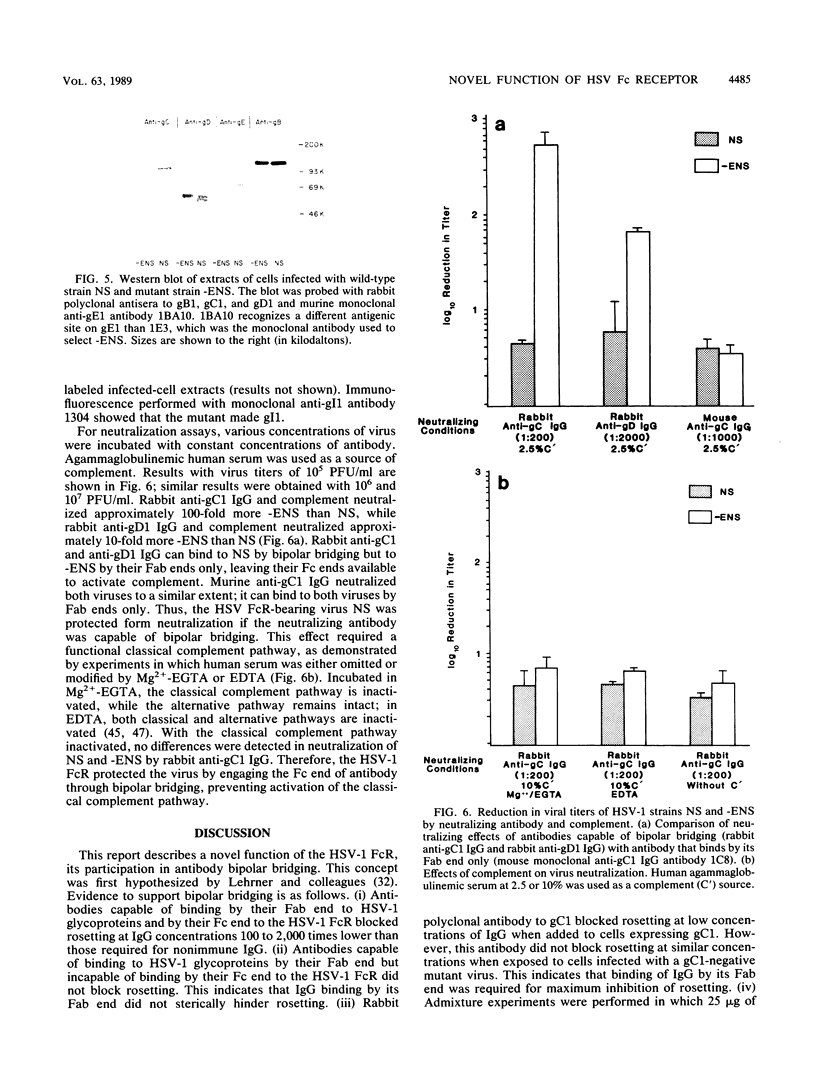
We describe a novel function of the Fc receptor of herpes simplex virus type 1 (HSV-1), its ability to participate in antibody bipolar bridging. This refers to the binding of a single immunoglobulin G (IgG) molecule by its Fab end to its antigenic target and by its Fc end to an Fc receptor (FcR). We demonstrate that various immune IgG antibodies, including polyclonal rabbit antibodies to HSV-1 glycoproteins gC1 and gD1 and monoclonal human antibody to gD1 blocked rosetting of IgG-coated erythrocytes at IgG concentrations 100- to 2,000-fold lower than required for rosette inhibition with nonimmune IgG. Steric hindrance did not account for the observed differences between immune and nonimmune IgG since rabbit anti-gC1 F(ab')2 fragments did not block rosetting. Murine anti-gC1 or anti-gD1 IgG, a species of IgG incapable of binding by its Fc end to the HSV-1 FcR, also did not block rosetting. When cells were infected with a gC1-deficient mutant, anti-gC1 IgG inhibited rosetting to the same extent as nonimmune IgG. This indicates that binding by the Fab end of the IgG molecule was required for maximum inhibition of rosetting. Bipolar bridging was shown to occur even when small concentrations of immune IgG were present in physiologic concentrations of nonimmune IgG. The biologic relevance of antibody bipolar bridging was evaluated by comparing antibody- and complement-dependent virus neutralization of an FcR-negative mutant and its parent HSV-1 strain. By engaging the Fc end of antiviral IgG, the parent strain resisted neutralization mediated by the classical complement pathway. These observations provide insight into the role of the HSV-1 FcR in pathogenesis and may help explain the function of FcR detected on other microorganisms.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adler R., Glorioso J. C., Cossman J., Levine M. Possible role of Fc receptors on cells infected and transformed by herpesvirus: escape from immune cytolysis. Infect Immun. 1978 Aug;21(2):442–447. doi: 10.1128/iai.21.2.442-447.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baucke R. B., Spear P. G. Membrane proteins specified by herpes simplex viruses. V. Identification of an Fc-binding glycoprotein. J Virol. 1979 Dec;32(3):779–789. doi: 10.1128/jvi.32.3.779-789.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benichou G., Voisin G. A. Antibody bipolar bridging: isotype-dependent signals given to guinea pig alveolar macrophages by anti-MHC alloantibodies. Cell Immunol. 1987 May;106(2):304–317. doi: 10.1016/0008-8749(87)90174-2. [DOI] [PubMed] [Google Scholar]
- Bennett W. S., Huber R. Structural and functional aspects of domain motions in proteins. CRC Crit Rev Biochem. 1984;15(4):291–384. doi: 10.3109/10409238409117796. [DOI] [PubMed] [Google Scholar]
- Burova L. A., Christensen P., Grubb R., Jonsson A., Samuelsson G., Schalén C., Svensson M. L. Changes in virulence, M protein and IgG Fc receptor activity in a type 12 group A streptococcal strain during mouse passages. Acta Pathol Microbiol Scand B. 1980 Aug;88(4):199–205. doi: 10.1111/j.1699-0463.1980.tb02629.x. [DOI] [PubMed] [Google Scholar]
- Christensen P., Johansson B. G., Kronvall G. Interaction of streptococci with the Fc fragment of IgG. Acta Pathol Microbiol Scand C. 1976 Apr;84(2):73–76. doi: 10.1111/j.1699-0463.1976.tb00001.x. [DOI] [PubMed] [Google Scholar]
- Cines D. B., Lyss A. P., Bina M., Corkey R., Kefalides N. A., Friedman H. M. Fc and C3 receptors induced by herpes simplex virus on cultured human endothelial cells. J Clin Invest. 1982 Jan;69(1):123–128. doi: 10.1172/JCI110422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen G. H., Dietzschold B., Ponce de Leon M., Long D., Golub E., Varrichio A., Pereira L., Eisenberg R. J. Localization and synthesis of an antigenic determinant of herpes simplex virus glycoprotein D that stimulates the production of neutralizing antibody. J Virol. 1984 Jan;49(1):102–108. doi: 10.1128/jvi.49.1.102-108.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen G. H., Isola V. J., Kuhns J., Berman P. W., Eisenberg R. J. Localization of discontinuous epitopes of herpes simplex virus glycoprotein D: use of a nondenaturing ("native" gel) system of polyacrylamide gel electrophoresis coupled with Western blotting. J Virol. 1986 Oct;60(1):157–166. doi: 10.1128/jvi.60.1.157-166.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colman P. M., Laver W. G., Varghese J. N., Baker A. T., Tulloch P. A., Air G. M., Webster R. G. Three-dimensional structure of a complex of antibody with influenza virus neuraminidase. 1987 Mar 26-Apr 1Nature. 326(6111):358–363. doi: 10.1038/326358a0. [DOI] [PubMed] [Google Scholar]
- Daniels C. A., Borsos T., Rapp H. J., Snyderman R., Notkins A. L. Neutralization of sensitized virus by purified components of complement. Proc Natl Acad Sci U S A. 1970 Mar;65(3):528–535. doi: 10.1073/pnas.65.3.528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Daëron M., Voisin G. A. H-2 antigens, on mast cell membrane, as target antigens for anaphylactic degranulation. Cell Immunol. 1978 May;37(2):467–472. doi: 10.1016/0008-8749(78)90214-9. [DOI] [PubMed] [Google Scholar]
- De Miranda-Santos I. K., Campos-Neto A. Receptor for immunoglobulin Fc on pathogenic but not on nonpathogenic protozoa of the Trypanosomatidae. J Exp Med. 1981 Dec 1;154(6):1732–1742. doi: 10.1084/jem.154.6.1732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deisenhofer J., Jones T. A., Huber R., Sjödahl J., Sjöquist J. Crystallization, crystal structure analysis and atomic model of the complex formed by a human Fc fragment and fragment B of protein A from Staphylococcus aureus. Hoppe Seylers Z Physiol Chem. 1978 Aug;359(8):975–985. doi: 10.1515/bchm2.1978.359.2.975. [DOI] [PubMed] [Google Scholar]
- Dossett J. H., Kronvall G., Williams R. C., Jr, Quie P. G. Antiphagocytic effects of staphylococcal protein A. J Immunol. 1969 Dec;103(6):1405–1410. [PubMed] [Google Scholar]
- Dowler K. W., Veltri R. W. In vitro neutralization of HSV-2: inhibition by binding of normal IgG and purified Fc to virion Fc receptor (FcR). J Med Virol. 1984;13(3):251–259. doi: 10.1002/jmv.1890130307. [DOI] [PubMed] [Google Scholar]
- Eisenberg R. J., Ponce de Leon M., Friedman H. M., Fries L. F., Frank M. M., Hastings J. C., Cohen G. H. Complement component C3b binds directly to purified glycoprotein C of herpes simplex virus types 1 and 2. Microb Pathog. 1987 Dec;3(6):423–435. doi: 10.1016/0882-4010(87)90012-x. [DOI] [PubMed] [Google Scholar]
- Ellerson J. R., Yasmeen D., Painter R. H., Dorrington K. J. A fragment corresponding to the C(H)2 region of immunoglobulin G (IgG) with complement fixing activity. FEBS Lett. 1972 Aug 15;24(3):318–322. doi: 10.1016/0014-5793(72)80381-8. [DOI] [PubMed] [Google Scholar]
- Forsgren A., Sjöquist J. "Protein A" from S. aureus. I. Pseudo-immune reaction with human gamma-globulin. J Immunol. 1966 Dec;97(6):822–827. [PubMed] [Google Scholar]
- Friedman H. M., Cohen G. H., Eisenberg R. J., Seidel C. A., Cines D. B. Glycoprotein C of herpes simplex virus 1 acts as a receptor for the C3b complement component on infected cells. Nature. 1984 Jun 14;309(5969):633–635. doi: 10.1038/309633a0. [DOI] [PubMed] [Google Scholar]
- Friedman H. M., Glorioso J. C., Cohen G. H., Hastings J. C., Harris S. L., Eisenberg R. J. Binding of complement component C3b to glycoprotein gC of herpes simplex virus type 1: mapping of gC-binding sites and demonstration of conserved C3b binding in low-passage clinical isolates. J Virol. 1986 Nov;60(2):470–475. doi: 10.1128/jvi.60.2.470-475.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayashida I., Nagafuchi S., Hayashi Y., Kino Y., Mori R., Oda H., Ohtomo N., Tashiro A. Mechanism of antibody-mediated protection against herpes simplex virus infection in athymic nude mice: requirement of Fc portion of antibody. Microbiol Immunol. 1982;26(6):497–509. doi: 10.1111/j.1348-0421.1982.tb00203.x. [DOI] [PubMed] [Google Scholar]
- Jennings S. R., Lippe P. A., Pauza K. J., Spear P. G., Pereira L., Tevethia S. S. Kinetics of expression of herpes simplex virus type 1-specific glycoprotein species on the surfaces of infected murine, simian, and human cells: flow cytometric analysis. J Virol. 1987 Jan;61(1):104–112. doi: 10.1128/jvi.61.1.104-112.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johansson P. J., Myhre E. B., Blomberg J. Specificity of Fc receptors induced by herpes simplex virus type 1: comparison of immunoglobulin G from different animal species. J Virol. 1985 Nov;56(2):489–494. doi: 10.1128/jvi.56.2.489-494.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johansson P. J., Schröder A. K., Nardella F. A., Mannik M., Christensen P. Interaction between herpes simplex type 1-induced Fc receptor and human and rabbit immunoglobulin G (IgG) domains. Immunology. 1986 Jun;58(2):251–255. [PMC free article] [PubMed] [Google Scholar]
- Johnson D. C., Feenstra V. Identification of a novel herpes simplex virus type 1-induced glycoprotein which complexes with gE and binds immunoglobulin. J Virol. 1987 Jul;61(7):2208–2216. doi: 10.1128/jvi.61.7.2208-2216.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson D. C., Frame M. C., Ligas M. W., Cross A. M., Stow N. D. Herpes simplex virus immunoglobulin G Fc receptor activity depends on a complex of two viral glycoproteins, gE and gI. J Virol. 1988 Apr;62(4):1347–1354. doi: 10.1128/jvi.62.4.1347-1354.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keller R., Peitchel R., Goldman J. N., Goldman M. An IgG-Fc receptor induced in cytomegalovirus-infected human fibroblasts. J Immunol. 1976 Mar;116(3):772–777. [PubMed] [Google Scholar]
- Klein M., Haeffner-Cavaillon N., Isenman D. E., Rivat C., Navia M. A., Davies D. R., Dorrington K. J. Expression of biological effector functions by immunoglobulin G molecules lacking the hinge region. Proc Natl Acad Sci U S A. 1981 Jan;78(1):524–528. doi: 10.1073/pnas.78.1.524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurlander R. J. Blockade of Fc receptor-mediated binding to U-937 cells by murine monoclonal antibodies directed against a variety of surface antigens. J Immunol. 1983 Jul;131(1):140–147. [PubMed] [Google Scholar]
- Lehner T., Wilton J. M., Shillitoe E. J. Immunological basis for latency, recurrences and putative oncogenicity of herpes simplex virus. Lancet. 1975 Jul 12;2(7924):60–62. doi: 10.1016/s0140-6736(75)90499-7. [DOI] [PubMed] [Google Scholar]
- Leung-Tack J., Neveu T., Lefroit-Joliy M., Voisin G. A. Alloantibody bipolar bridging; a new mechanism of cell surface activation. Immunol Lett. 1982 Jul;5(1):23–28. doi: 10.1016/0165-2478(82)90086-4. [DOI] [PubMed] [Google Scholar]
- Longnecker R., Chatterjee S., Whitley R. J., Roizman B. Identification of a herpes simplex virus 1 glycoprotein gene within a gene cluster dispensable for growth in cell culture. Proc Natl Acad Sci U S A. 1987 Jun;84(12):4303–4307. doi: 10.1073/pnas.84.12.4303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Longnecker R., Roizman B. Generation of an inverting herpes simplex virus 1 mutant lacking the L-S junction a sequences, an origin of DNA synthesis, and several genes including those specifying glycoprotein E and the alpha 47 gene. J Virol. 1986 May;58(2):583–591. doi: 10.1128/jvi.58.2.583-591.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maciag T., Hoover G. A., Stemerman M. B., Weinstein R. Serial propagation of human endothelial cells in vitro. J Cell Biol. 1981 Nov;91(2 Pt 1):420–426. doi: 10.1083/jcb.91.2.420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marlin S. D., Holland T. C., Levine M., Glorioso J. C. Epitopes of herpes simplex virus type 1 glycoprotein gC are clustered in two distinct antigenic sites. J Virol. 1985 Jan;53(1):128–136. doi: 10.1128/jvi.53.1.128-136.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McGeoch D. J., Dolan A., Donald S., Rixon F. J. Sequence determination and genetic content of the short unique region in the genome of herpes simplex virus type 1. J Mol Biol. 1985 Jan 5;181(1):1–13. doi: 10.1016/0022-2836(85)90320-1. [DOI] [PubMed] [Google Scholar]
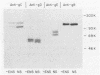
- McKendall R. R. IgG-mediated viral clearance in experimental infection with herpes simplex virus type 1: role for neutralization and Fc-dependent functions but not C' cytolysis and C5 chemotaxis. J Infect Dis. 1985 Mar;151(3):464–470. doi: 10.1093/infdis/151.3.464. [DOI] [PubMed] [Google Scholar]
- McNearney T. A., Odell C., Holers V. M., Spear P. G., Atkinson J. P. Herpes simplex virus glycoproteins gC-1 and gC-2 bind to the third component of complement and provide protection against complement-mediated neutralization of viral infectivity. J Exp Med. 1987 Nov 1;166(5):1525–1535. doi: 10.1084/jem.166.5.1525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neidhardt H., Schröder C. H., Kaerner H. C. Herpes simplex virus type 1 glycoprotein E is not indispensable for viral infectivity. J Virol. 1987 Feb;61(2):600–603. doi: 10.1128/jvi.61.2.600-603.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oakes J. E., Lausch R. N. Role of Fc fragments in antibody-mediated recovery from ocular and subcutaneous herpes simplex virus infections. Infect Immun. 1981 Jul;33(1):109–114. doi: 10.1128/iai.33.1.109-114.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Para M. F., Baucke R. B., Spear P. G. Glycoprotein gE of herpes simplex virus type 1: effects of anti-gE on virion infectivity and on virus-induced fc-binding receptors. J Virol. 1982 Jan;41(1):129–136. doi: 10.1128/jvi.41.1.129-136.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Para M. F., Goldstein L., Spear P. G. Similarities and differences in the Fc-binding glycoprotein (gE) of herpes simplex virus types 1 and 2 and tentative mapping of the viral gene for this glycoprotein. J Virol. 1982 Jan;41(1):137–144. doi: 10.1128/jvi.41.1.137-144.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Platts-Mills T. A., Ishizaka K. Activation of the alternate pathway of human complements by rabbit cells. J Immunol. 1974 Jul;113(1):348–358. [PubMed] [Google Scholar]
- Rahman A. A., Teschner M., Sethi K. K., Brandis H. Appearance of IgG (Fc) receptor(s) on cultured human fibroblasts infected with human cytomegalovirus. J Immunol. 1976 Jul;117(1):253–258. [PubMed] [Google Scholar]
- Rawls W. E., Iwamoto K., Adam E., Melnick J. L. Measurement of antibodies to herpesvirus types 1 and 2 in human sera. J Immunol. 1970 Mar;104(3):599–606. [PubMed] [Google Scholar]
- Schalén C., Truedsson L., Christensen K. K., Christensen P. Blocking of antibody complement-dependent effector functions by streptococcal IgG Fc-receptor and staphylococcal protein A. Acta Pathol Microbiol Immunol Scand B. 1985 Dec;93(6):395–400. doi: 10.1111/j.1699-0463.1985.tb02908.x. [DOI] [PubMed] [Google Scholar]
- Seigneurin J. M., Desgranges C., Seigneurin D., Paire J., Renversez J. C., Jacquemont B., Micouin C. Herpes simplex virus glycoprotein D: human monoclonal antibody produced by bone marrow cell line. Science. 1983 Jul 8;221(4606):173–175. doi: 10.1126/science.6304881. [DOI] [PubMed] [Google Scholar]
- Tarleton R. L., Kemp W. M. Demonstration of IgG-Fc and C3 receptors on adult Schistosoma mansoni. J Immunol. 1981 Jan;126(1):379–384. [PubMed] [Google Scholar]
- Torpier G., Capron A., Ouaissi M. A. Receptor for IgG(Fc) and human beta2-microglobulin on S. mansoni schistosomula. Nature. 1979 Mar 29;278(5703):447–449. doi: 10.1038/278447a0. [DOI] [PubMed] [Google Scholar]
- WATKINS J. F. ADSORPTION OF SENSITIZED SHEEP ERYTHROCYTES TO HELA CELLS INFECTED WITH HERPES SIMPLEX VIRUS. Nature. 1964 Jun 27;202:1364–1365. doi: 10.1038/2021364a0. [DOI] [PubMed] [Google Scholar]
- Wallis C., Melnick J. L. Herpesvirus neutralization: the role of complement. J Immunol. 1971 Nov;107(5):1235–1242. [PubMed] [Google Scholar]
- Westmoreland D., St Jeor S., Rapp F. The development by cytomegalovirus-infected cells of binding affinity for normal human immunoglobulin. J Immunol. 1976 Jun;116(6):1566–1570. [PubMed] [Google Scholar]
- Yoshino K., Hashimoto M., Shinkai K. Studies on the neutralization of herpes simplex virus. VIII. Significance of viral sensitization for inactivation by complement. Microbiol Immunol. 1977;21(4):231–241. doi: 10.1111/j.1348-0421.1977.tb00284.x. [DOI] [PubMed] [Google Scholar]
- Zweerink H. J., Corey L. Virus-specific antibodies in sera from patients with genital herpes simplex virus infection. Infect Immun. 1982 Aug;37(2):413–421. doi: 10.1128/iai.37.2.413-421.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]