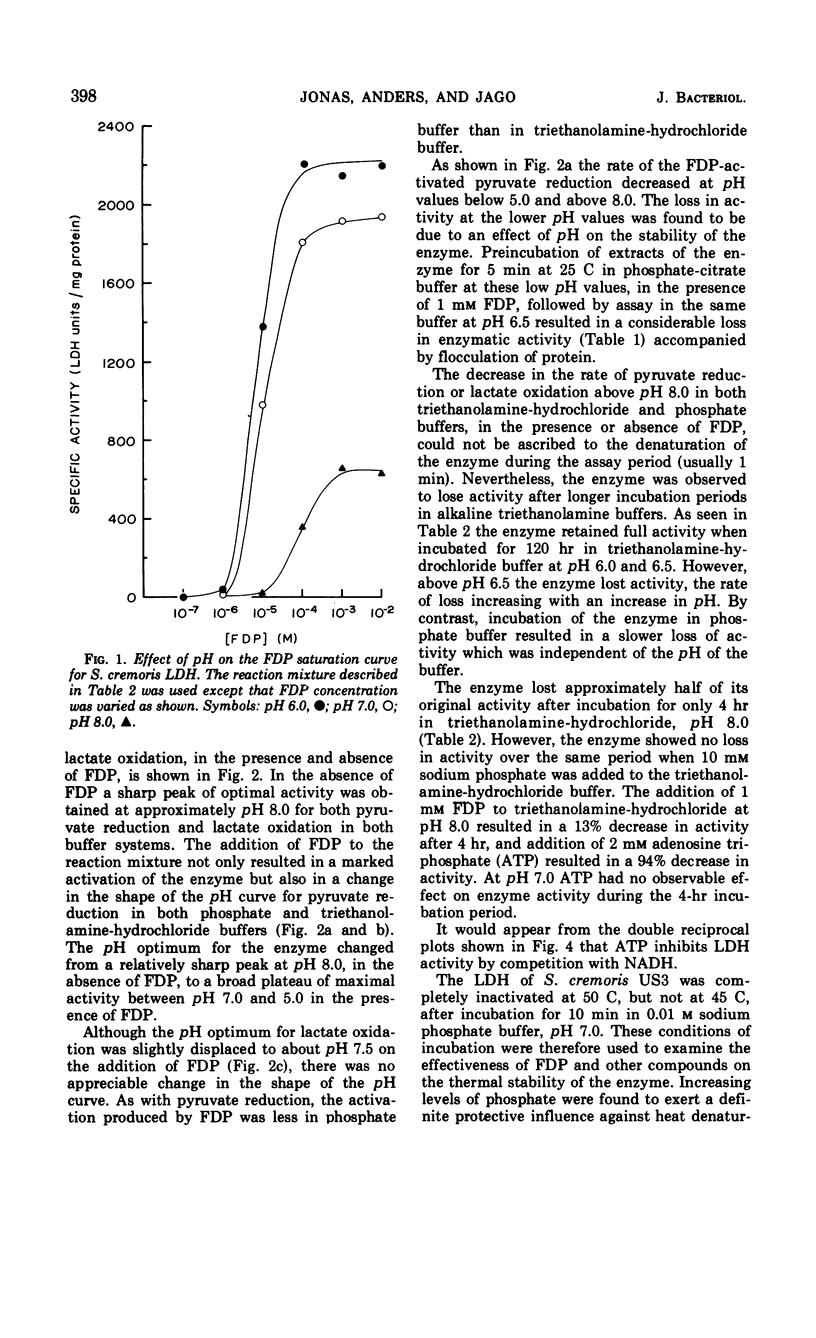
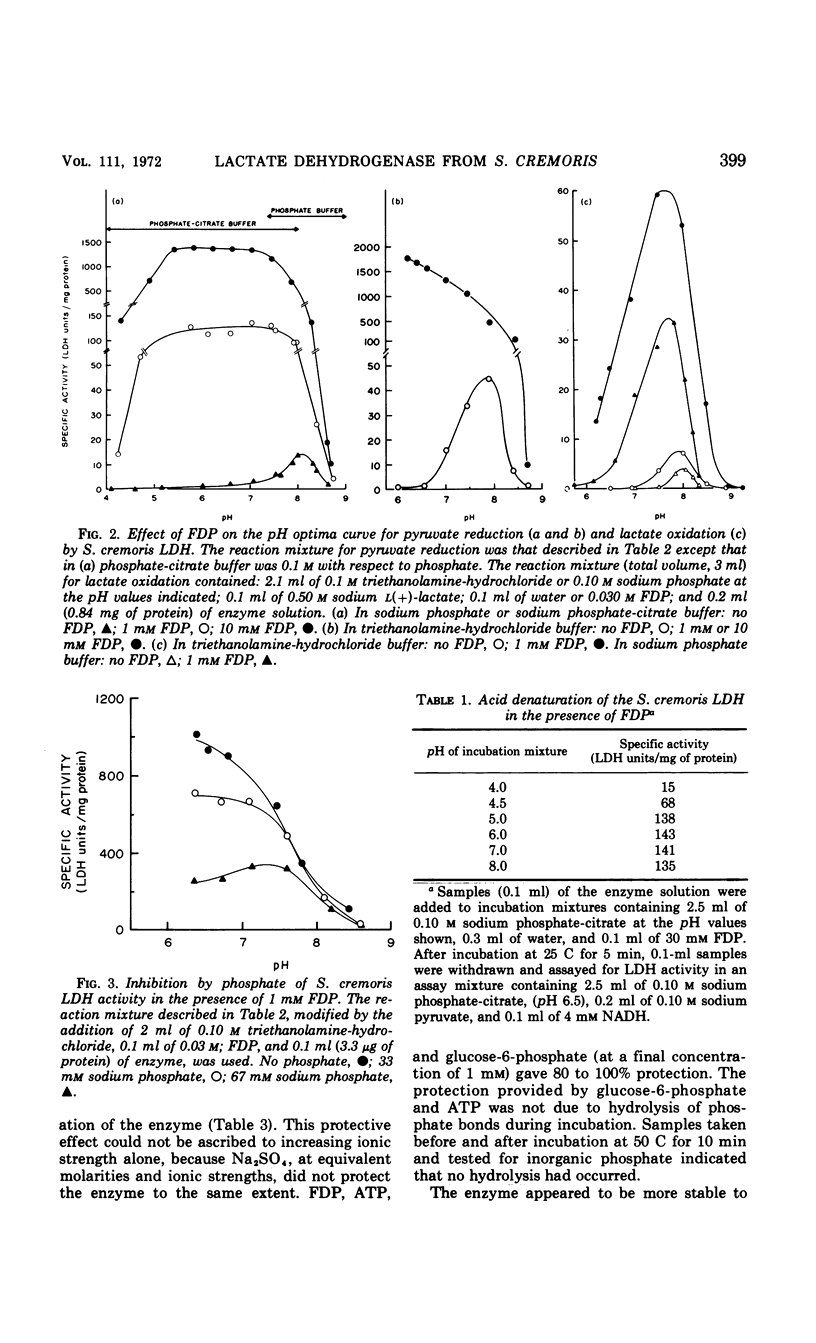
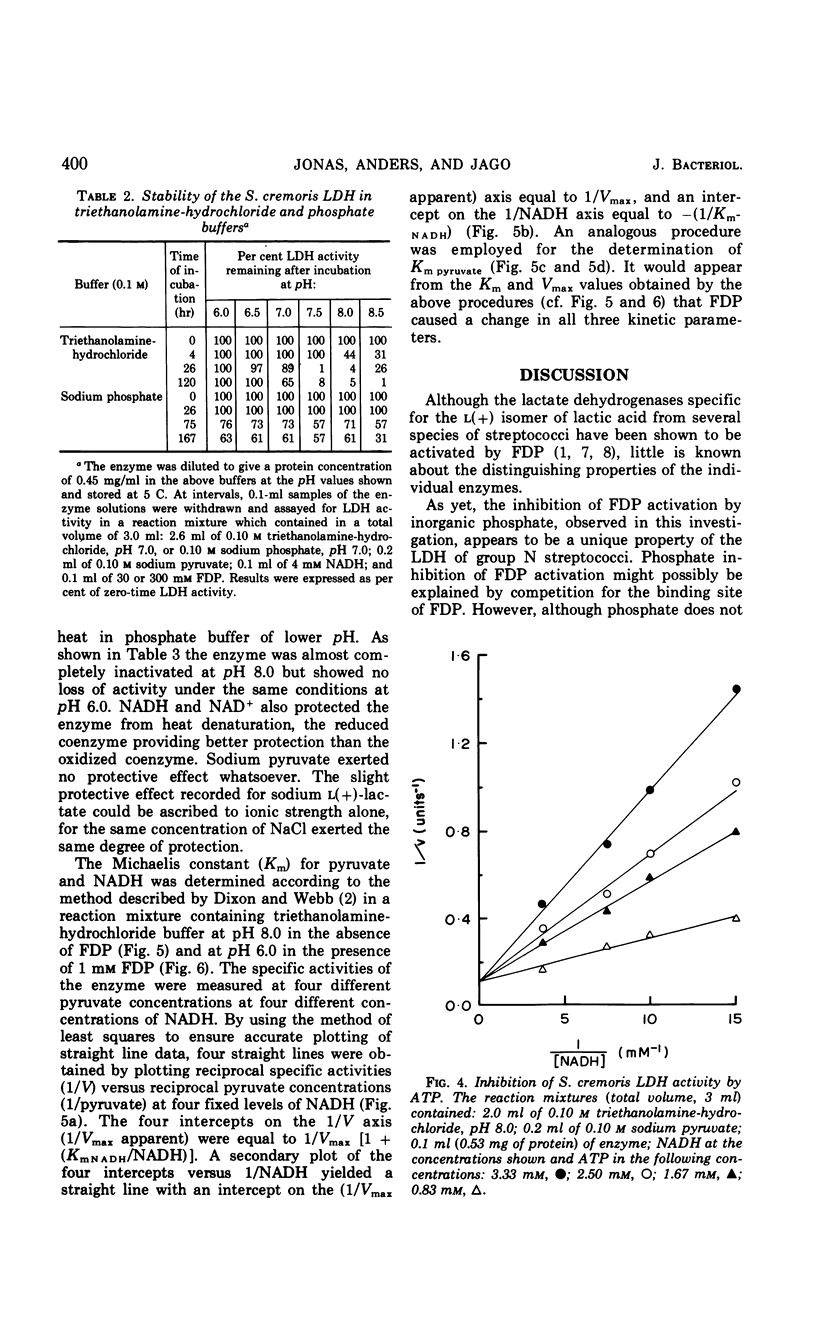
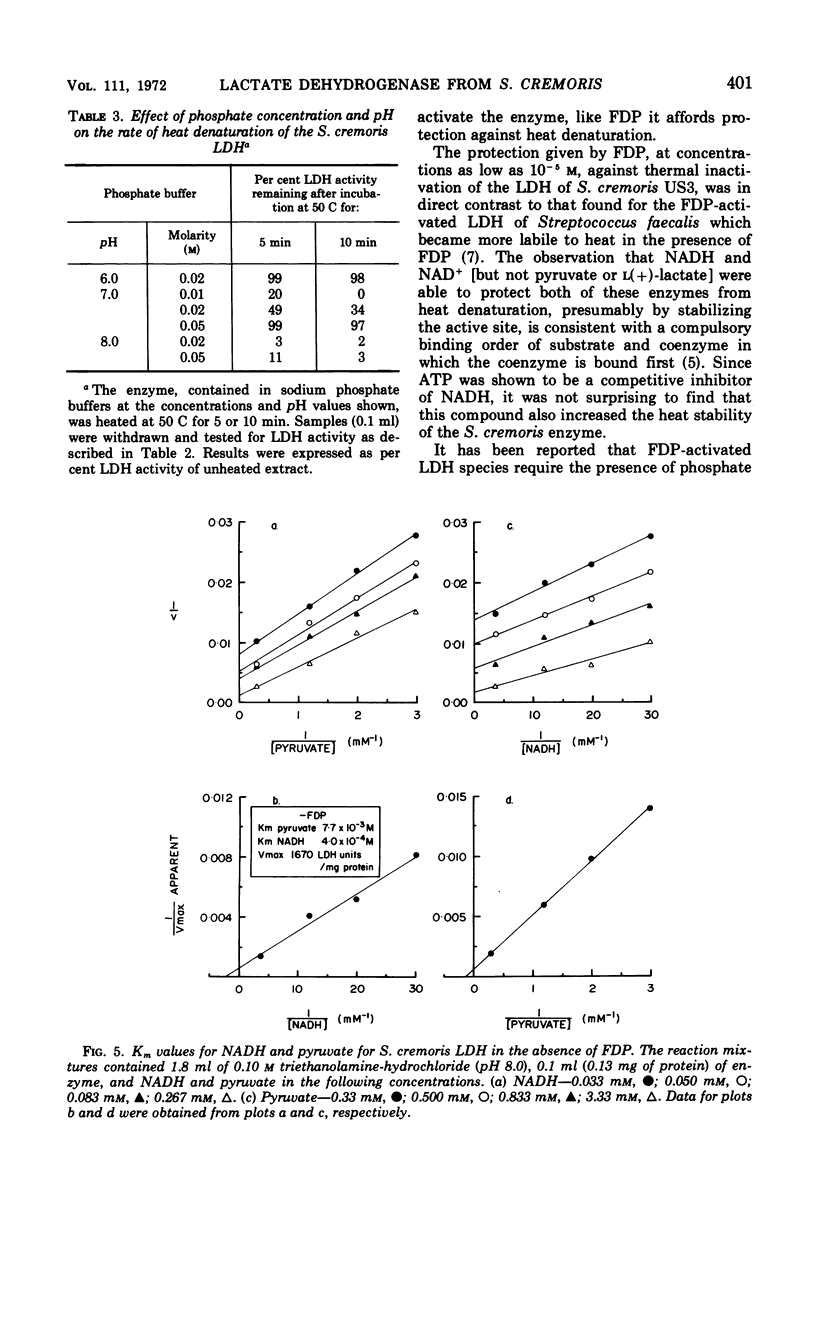
Abstract
Studies with partially purified extracts of the nicotinamide adenine dinucleotide-linked l(+)-lactate dehydrogenase of Streptococcus cremoris US3 showed that fructose-1,6-diphosphate (FDP) was essential for the catalytic reduction of pyruvate in the pH range 5.0 to 7.0, outside of which the organism does not grow. In the absence of FDP, enzyme activity was observed only in the region of pH 8.0. The optimal pH for the oxidation of lactate was approximately 8.0 in the presence and absence of FDP. The FDP-activated enzyme was markedly inhibited by inorganic phosphate. The enzyme lost activity on standing at 5 C in alkaline triethanolamine, was quite stable at pH 6.0 to 6.5, and underwent irreversible denaturation below pH 5.0. Inorganic phosphate or FDP increased the stability of the enzyme in alkaline buffers. Some distinguishing properties of individual lactate dehydrogenases, activated by FDP, are discussed.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Jago G. R., Nichol L. W., O'Dea K., Sawyer W. H. Physicochemical studies on the lactate dehydrogenase of Streptococcus cremoris US3: the effects of modifiers. Biochim Biophys Acta. 1971 Nov 13;250(2):271–285. doi: 10.1016/0005-2744(71)90184-7. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Mou L., Mulvena D. P., Jonas H. A., Jago G. R. Purification and properties of nicotinamide adenine dinucleotide-dependent D- and L- lactate dehydrogenases in a group N streptococcus. J Bacteriol. 1972 Aug;111(2):392–396. doi: 10.1128/jb.111.2.392-396.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WALTER C., FRIEDEN E. THE PREVALENCE AND SIGNIFICANCE OF THE PRODUCT INHIBITION OF ENZYMES. Adv Enzymol Relat Areas Mol Biol. 1963;25:167–274. doi: 10.1002/9780470122709.ch4. [DOI] [PubMed] [Google Scholar]
- WOLIN M. J. FRUCTOSE-1,6-DIPHOSPHATE REQUIREMENT OF STREPTOCOCCAL LACTIC DEHYDROGENASES. Science. 1964 Nov 6;146(3645):775–777. doi: 10.1126/science.146.3645.775. [DOI] [PubMed] [Google Scholar]
- Wittenberger C. L., Angelo N. Purificationa and properties of a fructose-1,6-diphosphate-activated lactate dehydrogenase from Streptococcus faecalis. J Bacteriol. 1970 Mar;101(3):717–724. doi: 10.1128/jb.101.3.717-724.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]


