Abstract
Purpose
To evaluate bone blood flow and bone formation in patients after total hip revision surgery with impacted bone grafting using H2 15O and [18F]fluoride positron emission tomography (PET).
Procedures
To asses bone blood flow and bone metabolism in bone allograft after impaction grafting, four patients treated with total hip revision surgery were enrolled prospectively in this study. Six patients scheduled for primary hip arthroplasties were included as a control group. The study protocol consisted of three H2 15O and [18F]fluoride PET scans in each patient.
Results
Bone blood flow increased significantly compared to the preoperative state in patients treated for primary hip arthroplasty. In patients undergoing revision surgery, bone blood flow was twofold to threefold higher compared to the preoperative state, but did not reach significance. Bone metabolism in patients undergoing revision was threefold higher 2 weeks postoperatively compared to the primary hip group. We found a significant correlation between Ki and bone blood flow.
Conclusions
Allogeneic bone grafts induce a higher rate of local periprosthetic bone formation compared to periprosthetic bone formation after a primary total hip placement. In vivo coupling between bone blood flow and bone metabolism suggests that bone metabolism in allogeneic bone grafts may partly rely on bone blood flow adaptations.
Key words: PET, Hip, Prosthesis, Bone metabolism
Introduction
Despite improved surgical techniques and prosthetic materials, reported failure rates of primary cemented and uncemented total hip prostheses are significant, varying between 7% and 17% [1]. As a result of a loosened prosthesis, acetabular and femoral bone deficiencies may occur because of mechanical wear and osteolysis. During revision surgery, these bone deficiencies may be treated with impacted allografts [2, 3]. Various studies suggest that the results of these operations are encouraging, whereas some authors report survival rates up to 100% after 10.4 years (mean) follow-up [4].
At present, the rate of success is measured by clinical outcome. In practice, routine x-rays at regular intervals after surgery are compared with previous films for signs of bone ingrowth, trabecularization, and cortical remodeling. It is difficult, however, to detect new bone formation (e.g., trabecularization, cortical remodeling) on radiographs and sometimes it is impossible because of metal meshes used for the reconstruction of segmental defects. An alternative is bone scintigraphy, which is useful in assessing loosening, fracture unions, and bone graft incorporation, but which is limited in anatomical precision and accuracy [5]. As both bone blood flow and mineralization of newly formed bone matrix play a crucial role in bone formation and bone graft remodeling, positron emission tomography (PET) may provide a solution since this technique offers a way to assess regional tissue (patho)physiology with a higher resolution than that of bone scintigraphy. In addition, PET allows for the quantification of blood flow and [18F] fluoride ions and thus can be used to study bone formation directly [6–9]. In the process of bone regeneration, osteoblasts produce bone matrix, which is mineralized after an interval of some days. Fluoride ions exchange with hydroxyl groups in the hydroxyapatite crystal of bone (Ca10(PO4)6OH2), which is formed by osteoblasts in the process of mineralization, resulting in fluoroapatite (Ca10(PO4)6F2) [6, 10].
With a better understanding of the biological response of the morselized bone allografts used during surgery, PET may provide a new technique to predict the clinical outcome of the patient. Therefore, the aim of the present study was to evaluate bone blood flow and new bone formation in patients after total hip revision surgery with impacted bone grafting using H2 15O and 18F− PET. In addition, to understand better the biological effect of bone allografts used in revision surgery, these results were compared with those in patients after primary cemented total hip arthroplasty in an attempt to discriminate between the effects of the surgical procedure and those of bone grafting.
Materials and Methods
Study Design
Six patients scheduled for primary hip arthroplasties (group A: six females; mean age 76 ± 4) and four patients treated with total hip revision surgery (group B: two males, two females; mean age 76 ± 5) using impacted bone allografts were enrolled prospectively to quantitatively assess the process of bone graft remodeling and new bone formation. The diagnosis for all six primary total hip arthroplasties was primary arthrosis. The indication for all four acetabular and three femoral component revisions was aseptic loosening. In all patients, cemented total hip prostheses were used. The study was approved by the medical ethics committee of the VU University Medical Centre (VUmc). All patients gave written informed consent after being fully informed about the purpose of the study and any potential risks. The study protocol consisted of three repeat H2 15O and 18F− PET scans in each patient performed at approximately 1 day before operation and at 2 and 12 weeks postoperatively.
Surgical Procedure
In all patients, a cemented Exeter™ (Howmedica, London, UK) total hip prosthesis in a posterolateral approach was used. The technique used for impacted bone allografts has been described in detail previously [2, 3]. First, segmental defects were closed with wire meshes after the removal of scar tissue, cement, and fibrous interface. Next, deep frozen bone graft chips of the VUmc bone bank (cancellous, diameter of approximately 8 mm) were inserted and impacted.
Postoperative Management
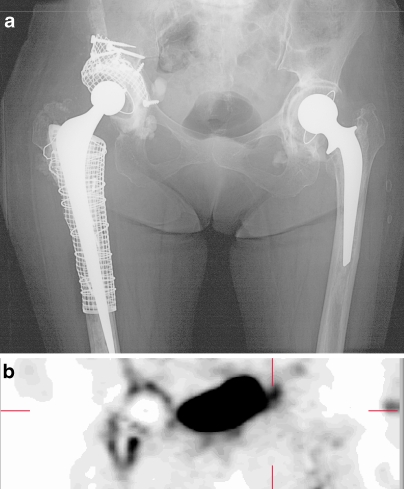
Postoperative management included systemic antibiotics during the first 24 h, followed by anticoagulation therapy for 3 months. Mobilization of patients started with bed exercises on the first postoperative day up to a period of 3 weeks, followed by partial mobilization with limited weight bearing on the operated side. Patients were then seen in the outpatient clinic at 6 weeks, 3 months, 6 months, and 1 year after surgery. Routine x-rays were performed at regular intervals after surgery and compared with previous films for signs of bone ingrowth, trabecularization, cortical remodeling, and migration. (Fig. 1a)
Fig. 1.
a Postoperative AP radiograph showing a patient treated with hip revision arthroplasty using impaction grafting and reconstruction of segmental defects of the right hip. b Coronal 18F− PET image of the same patient as illustrated in a. Increased uptake is present in the area surrounding the femoral and acetabular prosthesis.
Data Acquisition
Dynamic H2 15O and 18F− scans were performed using an ECAT Exact HR+ scanner (Siemens/CTI, Knoxville, USA), which records 63 continuous planes in an axial field of view of 15.5 cm. First, a 10-min transmission scan of the hip region was performed, using three rotating 68Ge sources. After bolus injection (15 s; 10 ml/min) using an automated injector (Medrad, Pittsburgh, USA) of 1,000 MBq H2 15O in an antecubital vein, a dynamic emission scan (in 2-D mode) was started. The scanning sequence consisted of the following frames: 12 × 5, 12 × 10, 6 × 20, and 10 × 30 s (total = 10 min). During the H2 15O scan, arterial blood was withdrawn continuously (5 ml/min) from the distal radial artery using an online blood sampler (Veenstra, Joure, The Netherlands). 18F− PET acquisition started 10 min after the H2 15O scan to allow for decay of H2 15O activity. A 60-min dynamic emission scan was started after i.v. injection of 100 MBq 18F− using the following scanning sequence: 6 × 5, 6 × 10, 3 × 20, 5 × 30, 5 × 60, 8 × 150, and 6 × 300 s. To acquire the input function, i.e., the time course of H2 15O and 18F− in arterial plasma, arterial blood was withdrawn continuously from the radial artery at a rate of 5 ml/min during the first 10 min and at a rate of 2.5 ml/min thereafter. At set times, continuous withdrawal was interrupted to collect manual samples. For these samples, both whole blood and plasma concentrations were measured using a cross-calibrated well-counter.
Data Analysis
Dynamic scan data were corrected for decay, dead time, scatter, randoms, and (measured) photon attenuation and reconstructed as 128 × 128 matrices using filtered back projection with a Hanning filter (cut-off 0.5 cycles/pixel). To enhance the anatomical orientation and to facilitate correct positioning of the regions of interest (ROI), the last ten frames of the [18F−]fluoride scan were summed (20–60 min p.i.) (Fig. 1b). ROI were drawn around the endoprosthesis in an axial plane. These ROI were then grouped to form one single ROI, which was copied, mirrored, and positioned onto the contralateral femur or acetabulum at the same level on both [18F–]fluoride and H2 15O dynamic scans. In a similar fashion, ROI were defined around the allogeneic cancellous grafts (within the acetabulum and femoral cavity) of the acetabulum or femur of the operated side. Next, both ROI were subtracted from each other, resulting in tissue time–activity curves of allogeneic cancellous grafts and cortical bone.
Blood flow was estimated by fitting H2 15O tissue time–activity curves to a single tissue compartment model using standard nonlinear regression techniques [11, 12]. These time–activity curves were fitted both with and without including an arterial blood volume (V a) component. In these fits, the delay and dispersion of the online input function were individually fixed to the values obtained from a fit of the total count rate of the scanner, which included these parameters [11].
Fluoride kinetics were estimated by fitting tissue time–activity curves to a two-tissue compartment model. The two-tissue compartments consisted of the extravascular compartment and bone mineral. Four different rate constants (k
1, k
2, k
3, k
4) describe the exchange of fluoride between plasma and tissue and between the two-tissue compartments. The net clearance of fluoride from plasma to bone mineral is described by the parameter K
i. K
i is calculated with the following equation: 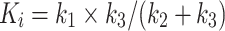 . The presence of an arterial blood volume (V
a) and the fourth kinetic parameter (k
4) in the fluoride model was evaluated using standard criteria [13, 14].
. The presence of an arterial blood volume (V
a) and the fourth kinetic parameter (k
4) in the fluoride model was evaluated using standard criteria [13, 14].
Absolute values of blood flow and bone metabolism (K i) at the operated side were compared to those of the contralateral hip region and to the preoperative values using the nonparametric Friedman test or Wilcoxon matched pairs test. A p value of <0.05 was considered to be significant. Results are expressed as mean values with their standard deviation. PET parameters K i and blood flow were correlated using linear regression.
Results
In five out of 30 scanning sequences, the H2 15O scan could not be performed because of problems related to the production of H2 15O and, in one occasion, to hardware failure of the PET scanner. No artifacts because of the implant materials were observed in the reconstructed images.
Clinical Outcome
Clinical outcome was good for all patients. In all cases, routine x-rays showed signs of bone ingrowth, trabecularization, and cortical remodeling. All patients were mobilized using a standardized protocol and visited our outpatient clinic as described previously.
Blood Flow
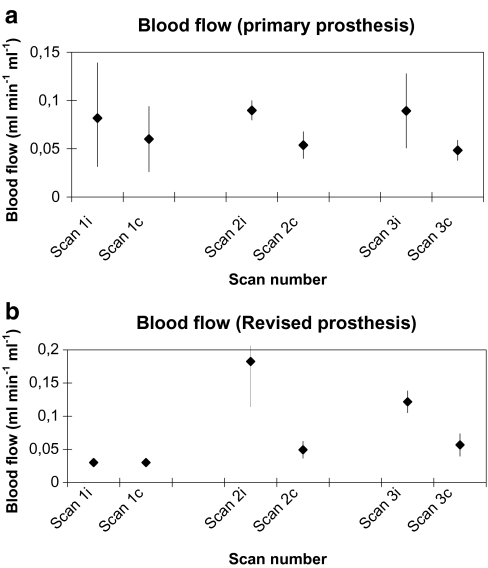
In the primary group (Fig. 2a), preoperative mean bone blood flow was 0.082 ± 0.050 ml·min−1·ml−1 in the hip region where a hip prosthesis had to be inserted and 0.060 ± 0.033 ml·min−1·ml−1 in the contralateral hip (p = 0.22; group A; scan 1i vs. scan 1c). Two weeks after surgery, blood flow was 0.090 ± 0.010 ml·min−1·ml−1 around the prosthesis and 0.054 ± 0.014 ml·min−1·ml−1 at the contralateral side (p = 0.11; group A; scan 2i vs. scan 2c). Three months after surgery, bone blood flow was significantly higher around the prosthesis with 0.089 ± 0.038 ml·min−1·ml−1 compared to 0.048 ± 0.010 ml·min−1·ml−1 at the contralateral side (p = 0.043; group A; scan 3i vs. scan 3c). In the group of patients undergoing a revision (Fig. 2b), bone blood flow around the prosthesis was twofold to threefold higher compared to the contralateral side, but differences did not reach significance (group B; scan 2i vs. scan 2c and scan 3c).
Fig. 2.
a Bone blood flow and 95% confidence intervals in the group of patients undergoing primary hip arthroplasty preoperatively (scan 1) and 2 weeks (scan 2) and 12 weeks (scan 3) after surgery. i ipsilateral, c contralateral. b Bone blood flow and 95% confidence intervals in the group of patients undergoing hip revision arthroplasty preoperatively (scan 1) and 2 weeks (scan 2) and 12 weeks (scan 3) after surgery. i ipsilateral, c contralateral.
Patients undergoing a revision had a higher bone blood flow at the prosthetic side of 0.183 ml·min−1·ml−1 compared to the primary group who had a mean blood flow of 0.090 ml·min−1·ml−1 (p = 0.05) 2 weeks postoperatively (group A vs. B; scan 2i). At the two other time points (preoperatively and 3 months postoperatively), bone blood flow was not significantly different between primary and revision groups.
Bone Metabolism
Only in ten out of 104 comparisons the four-parameter model was preferred for the description of fluoride kinetics, according to standard criteria [13, 14]. Therefore, a three-parameter model (i.e., k 4 was set to 0) was used for the analysis of fluoride kinetics. In Fig. 3, a summary of the K i values is presented as measured in both operated and nonoperated hips of patients undergoing primary total hip arthroplasty or hip revision arthroplasty.
Fig. 3.
a K i of 18F− and 95% confidence intervals in the group of patients undergoing primary hip arthroplasty preoperatively (scan 1) and 2 weeks (scan 2) and 12 weeks (scan 3) after surgery. i ipsilateral, c contralateral. b K i of 18F− and 95% confidence intervals in the group of patients undergoing hip revision arthroplasty preoperatively (scan 1) and 2 weeks (scan 2) and 12 weeks (scan 3) after surgery. i ipsilateral, c contralateral.
In patients undergoing a primary total hip prosthesis (Fig. 3a), the periprosthetic K i increased nonsignificantly from 0.0046 ± 0.0023 to 0.015 ± 0.010 min−1 at 2 weeks (p = 0.08) and significantly to 0.020 ± 0.010 min−1 at 12 weeks (p = 0.043; group A; scan 1i, 2i, and 3i). In patients undergoing a primary total hip prosthesis implantation, bone metabolism in the contralateral hip region increased from 0.0059 ± 0.0051 to 0.010 ± 0.0035 min−1 at 2 weeks (p = 0.08) and to 0.010 ± 0.0036 min−1 at 12 weeks (p = 0.043; group A; scan1c vs. 2c vs. 3c). In patients undergoing hip revision surgery (Fig. 3b), K i increased from 0.030 ± 0.024 min−1 preoperatively to 0.046 ± 0.024 min−1 at 2 weeks after surgery (p = 0.18; group B; scan1c vs. 2c). At 12 weeks after revision surgery, the K i returned to 0.028 ± 0.015 min−1, not different from the preoperative K i (p = 0.59; group B; scan1c vs. 3c).
Bone metabolism (K
i) in the periprosthetic region in patients undergoing revision was threefold higher 2 weeks postoperatively compared to the primary hip group (0.046 ± 0.024 vs. 0.015 ± 0.010 min−1, respectively; p = 0.036; group A vs. group B; scan 2c). Hence, aside from the operation effects, impaction bone grafting induced additional fluoride incorporation within 2 weeks postoperatively compared to patients who received a hip prosthesis without bone grafting. However, at 3 months, a nonsignificant difference in bone metabolism was seen between patients with (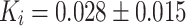 ) and without (
) and without (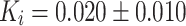 ) bone grafts (p = 0.556; group A vs. group B; scan 3c).
) bone grafts (p = 0.556; group A vs. group B; scan 3c).
Correlation between Fluoride Metabolism and Bone Blood Flow
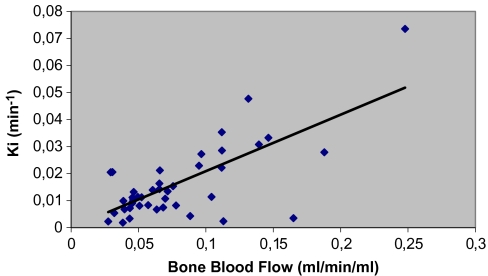
Linear regression analysis showed a significant correlation between K i and bone blood flow in the same region with a correlation coefficient of 0.72 (p < 0.001), as shown in Fig. 4.
Fig. 4.
Correlation between K i of 18F− and bone blood flow (BF). The straight line represents the regression line, which is given by K i = 0.209 × BF − 1.16; r = 0.72.
Discussion
We found a significant increase in periprosthetic bone metabolism in patients in whom allogeneic bone grafts were used compared to patients where no bone grafts were used. Hence, the addition of bone graft induces higher osteoblastic activity than surgery or surgical trauma to bone alone. Furthermore, increased uptake of [18F−]fluoride was found in the contralateral hip of patients receiving a primary hip prosthesis, indicating increased bone formation at the nonoperated side. Periprosthetic bone blood flow was increased at 2 weeks and 3 months after surgery. Finally, periprosthetic [18F−]fluoride uptake was correlated with bone blood flow, suggesting that bone formation and bone blood flow are coupled. Unfortunately, in five out of 30 scanning sequences, the H2 15O scan could not be performed. Furthermore, the limited number of patients may be an important factor for some of the nonsignificant findings.
[18F−]fluoride has been applied for the evaluation of bone metabolism and neoplastic diseases [6, 8, 15–22]. In the present study, this technique was applied to evaluate bone metabolism of morselized allografts in patients treated with total hip revision arthroplasty and primary total hip arthroplasties. Patients receiving allogeneic bone grafts had higher [18F−]fluoride influx compared to patients undergoing implantation of a total hip prostheses. In an earlier study where a simplified Patlak analysis was used for the calculation of [18F−] fluoride kinetics, a threefold to fourfold higher [18F−]fluoride uptake was found 1 week after surgery compared to the contralateral bone in the same patient [8]. As the main interest of the present study was bone formation and effects of allogeneic bone grafts, a control group undergoing primary total hip implantation was used to account for local surgical trauma. Two weeks after surgery, patients receiving bone grafts had a threefold higher [18F−]fluoride uptake compared to patients receiving total hip implantation. Hence, these results suggest that allogeneic bone grafts induce rapid new bone formation independent of local surgical trauma. In the present analysis, a two-tissue compartmental model together with nonlinear regression was used to calculate the net rate of uptake K i of 18F−. This was based on a study of Piert et al. [23] who demonstrated that the K i obtained from nonlinear regression has been shown to correlate better with histomorphometric parameters of bone formation than the Patlak-derived K i. Increased [18F−]fluoride uptake was found within 2 weeks after surgery. Whether this rapid increased uptake of [18F−]fluoride is the result of new bone formation from the patient’s own osteoblasts or from osteoblasts originating from the allograft remains uncertain. It is interesting to note that an earlier study has shown new bone formation and osteogenic activity from bone cells in bone allografts [24]. The osteoinductive potential is, however, more likely to result from matrix-associated bone morphogenetic proteins. It is interesting to note that also the contralateral femur of patients receiving a primary hip arthroplasty showed an increased level of bone formation. This increase of bone formation at the contralateral side could be induced by increased exercise or walking by the patients after surgery compared to the exercise level before surgery. On the other hand, circulating mediators influencing bone formation may also have contributed to the rise of 18F− uptake in the contralateral hip.
Compared to the contralateral femur, a doubling of blood flow within 2 weeks after surgery was found. In addition, at 3 months, increased bone blood flow values were found compared to the contralateral femur. These findings are similar to findings in earlier reports and are most likely the result of surgical trauma, subsequent neovascularization, and proliferation of bone grafts [8, 25, 26]. When comparing blood flow between patients treated with a primary and revision arthroplasties 2 weeks postoperatively, however, a clear trend of increased flow in the group with revision arthroplasty was seen. Lack of statistical significance may be explained by the relatively low number of patients. This marked increase in blood flow at 2 weeks after the use of bone grafts may suggest that morselized bone allografts have an osteogenic effect causing an increase in neovascularization and subsequent osteoblast activity. This osteogenic effect of allografts has previously been suggested [8], but without verification using a control group (primary arthroplasty). Three months after surgery, blood flow stabilized at approximately 150% of the preoperative flow value. Blood flow was correlated to some extent with K i, representing the net [18F−]fluoride uptake. Approximately 50% of K i variability could be explained by the variability in bone blood flow. In an earlier study, a similar result was found, showing a coupling of porcine blood flow and metabolism in high-turnover bone disease [23]. Other factors, such as bone morphogenetic proteins, may also have an effect on the alterations in bone metabolism, as 50% of the variation in bone metabolism could not be explained by variation in bone blood flow [19].
Conclusion
Increased bone blood flow indicates early neovascularization within 2 weeks after implantation of allogeneic bone grafts. Allogeneic bone grafts induce a higher rate of local periprosthetic bone formation compared to periprosthetic bone formation after a primary total hip placement. PET provides a way to assess the metabolic activity in bone grafts and thus may provide an important tool for clinicians to assess in vivo bone graft behavior, which relates to clinical outcome. However, because of the limited number of patients currently included, further studies are needed to assess the clinical value of PET imaging in patients treated with revision hip surgery.
Acknowledgements
The authors would like to thank Gert Leurtsema for his contributions to this manuscript, as well as the production of the applied PET tracers.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Footnotes
Grant Support: The Dutch Health Care Insurance Board (00155)
References
- 1.Malchau H, Herberts P, Eisler T, Garellick G, Soderman P. The Swedish total hip replacement register. J Bone Joint Surg Am. 2002;84(A Suppl 2):2–20. doi: 10.2106/00004623-200200002-00002. [DOI] [PubMed] [Google Scholar]
- 2.Slooff TJJM, Buma P, Gardeniers JWM, Schreurs BW, Schimmel JW, Huiskes R. Revision of the acetabular component. In: Callaghan JJ, editor. The adult hip. Philladelphia: Lippincott-Raven; 1989. pp. 1449–1459. [Google Scholar]
- 3.Mikhail WEM, Ling RSM, Weidenhielm LRA, Gie GA. Revision of the femoral component: impaction grafting. In: Callaghan JJ, editor. The adult hip. Philadelphia: Lippincott-Raven; 1998. pp. 1527–1536. [Google Scholar]
- 4.Schreurs BW, Arts JJ, Verdonschot N, Buma P, Slooff TJ, Gardeniers JW. Femoral component revision with use of impaction bone-grafting and a cemented polished stem. J Bone Joint Surg Am. 2005;87(11):2499–2507. doi: 10.2106/JBJS.D.02547. [DOI] [PubMed] [Google Scholar]
- 5.Temmerman OP, Raijmakers PG, Berkhof J, Hoekstra OS, Teule GJ, Heyligers IC. Accuracy of diagnostic imaging techniques in the diagnosis of aseptic loosening of the femoral component of a hip prosthesis: a meta-analysis. J Bone Joint Surg Br. 2005;87(6):781–785. doi: 10.1302/0301-620X.87B6.15625. [DOI] [PubMed] [Google Scholar]
- 6.Hawkins RA, Hoh CK, Phelps ME. PET imaging of the skeletal system. In: Sandler MP, Coleman RE, Wackers FJTh, editors. Diagnostic nuclear medicine. Baltimore: Williams & Wilkins; 1996. p. 725. [Google Scholar]
- 7.Lammertsma AA, Frackowiak RS. Positron emission tomography. Crit Rev Biomed Eng. 1985;13(2):125–169. [PubMed] [Google Scholar]
- 8.Sorensen J, Ullmark G, Langstrom B, Nilsson O. Rapid bone and blood flow formation in impacted morselized allografts: positron emission tomography (PET) studies on allografts in 5 femoral component revisions of total hip arthroplasty. Acta Orthop Scand. 2003;74(6):633–643. doi: 10.1080/00016470310018126. [DOI] [PubMed] [Google Scholar]
- 9.Pedersen NW, Kiaer T, Kristensen KD, Starklint H. Intraosseous pressure, oxygenation, and histology in arthrosis and osteonecrosis of the hip. Acta Orthop Scand. 1989;60(4):415–417. doi: 10.3109/17453678909149309. [DOI] [PubMed] [Google Scholar]
- 10.Hawkins RA, Choi Y, Huang SC, Hoh CK, Dahlbom M, Schiepers C, Satyamurthy N, Barrio JR, Phelps ME. Evaluation of the skeletal kinetics of fluorine-18-gluoride ion with PET. J Nucl Med. 1992;33(5):633–642. [PubMed] [Google Scholar]
- 11.Lammertsma AA, Cunningham VJ, Deiber MP, Heather JD, Bloomfield PM, Nutt J, Frackowiak RS, Jones T. Combination of dynamic and integral methods for generating reproducible functional CBF images. J Cereb Blood Flow Metab. 1990;10(5):675–686. doi: 10.1038/jcbfm.1990.121. [DOI] [PubMed] [Google Scholar]
- 12.Kety SS. The theory and applications of the exchange of inert gas at the lungs and tissues. Pharmacol Rev. 1951;3(1):1–41. [PubMed] [Google Scholar]
- 13.Akaike H. New look at statistical model identification. IEEE Trans Automat Cont. 1974;19(6):716–723. doi: 10.1109/TAC.1974.1100705. [DOI] [Google Scholar]
- 14.Schwarz G. Estimating dimensions of a model. Ann Stat. 1978;6(2):461–464. doi: 10.1214/aos/1176344136. [DOI] [Google Scholar]
- 15.Berding G, Burchert W, van den Hoff J, Pytlik C, Neukam FW, Meyer GJ, Gratz KF, Hundeshagen H. Evaluation of the incorporation of bone grafts used in maxillofacial surgery with [18F]fluoride ion and dynamic positron emission tomography. Eur J Nucl Med. 1995;22(10):1133–1140. doi: 10.1007/BF00800595. [DOI] [PubMed] [Google Scholar]
- 16.Berding G, Kirchhoff TD, Burchert W, von der Hoff J, Zeidler H, Hundeshagen H, Knapp WH. [18F]Fluoride PET indicates reduced bone formation in severe glucocorticoid-induced osteoporosis. Nuklearmedizin. 1998;37(2):76–79. [PubMed] [Google Scholar]
- 17.Hoh CK, Hawkins RA, Dahlbom M, Glaspy JA, Seeger LL, Choi Y, Schiepers CW, Huang SC, Satyamurthy N, Barrio JR. Whole body skeletal imaging with [18F]Fluoride ion and PET. J Comput Assist Tomogr. 1993;17(1):34–41. doi: 10.1097/00004728-199301000-00005. [DOI] [PubMed] [Google Scholar]
- 18.Messa C, Goodman WG, Hoh CK, Choi Y, Nissenson AR, Salusky IB, Phelps ME, Hawkins RA. Bone metabolic activity measured with positron emission tomography and [18F]Fluoride ion in renal osteodystrophy: correlation with bone histomorphometry. J Clin Endocrinol Metab. 1993;77(4):949–955. doi: 10.1210/jc.77.4.949. [DOI] [PubMed] [Google Scholar]
- 19.Piert M, Winter E, Becker GA, Bilger K, Machulla H, Muller-Schauenburg W, Bares R, Becker HD. Allogenic bone graft viability after hip revision arthroplasty assessed by dynamic [18F]Fluoride ion positron emission tomography. Eur J Nucl Med. 1999;26(6):615–624. doi: 10.1007/s002590050429. [DOI] [PubMed] [Google Scholar]
- 20.Piert M, Machulla HJ, Jahn M, Stahlschmidt A, Becker GA, Zittel TT. Coupling of porcine bone blood flow and metabolism in high-turnover bone disease measured by [(15)O]H(2)O and [(18)F]fluoride ion positron emission tomography. Eur J Nucl Med Mol Imaging. 2002;29(7):907–914. doi: 10.1007/s00259-002-0797-2. [DOI] [PubMed] [Google Scholar]
- 21.Schiepers C, Nuyts J, Bormans G, Dequeker J, Bouillon R, Mortelmans L, Verbruggen A, De Roo M. Fluoride kinetics of the axial skeleton measured in vivo with fluorine-18-fluoride PET. J Nucl Med. 1997;38(12):1970–1976. [PubMed] [Google Scholar]
- 22.Schiepers C, Broos P, Miserez M, Bormans G, De Roo M. Measurement of skeletal flow with positron emission tomography and 18F-fluoride in femoral head osteonecrosis. Arch Orthop Trauma Surg. 1998;118(3):131–135. doi: 10.1007/s004020050332. [DOI] [PubMed] [Google Scholar]
- 23.Piert M, Zittel TT, Becker GA, Jahn M, Stahlschmidt A, Maier G, Machulla HJ, Bares R. Assessment of porcine bone metabolism by dynamic [18F]Fluoride ion PET: correlation with bone histomorphometry. J Nucl Med. 2001;42(7):1091–1100. [PubMed] [Google Scholar]
- 24.Heyligers IC, Klein-Nulend J. Detection of living cells in non-processed but deep-frozen bone allografts. Cell Tissue Bank. 2005;6(1):25–31. doi: 10.1007/s10561-005-1089-4. [DOI] [PubMed] [Google Scholar]
- 25.Ashcroft GP, Evans NT, Roeda D, Dodd M, Mallard JR, Porter RW, Smith FW. Measurement of blood flow in tibial fracture patients using positron emission tomography. J Bone Joint Surg Br. 1992;74(5):673–677. doi: 10.1302/0301-620X.74B5.1527111. [DOI] [PubMed] [Google Scholar]
- 26.Schliephake H, Berding G, Knapp WH, Sewilam S. Monitoring of graft perfusion and osteoblast activity in revascularised fibula segments using [18F]-positron emission tomography. Int J Oral Maxillofac Surg. 1992;28(5):349–355. doi: 10.1016/S0901-5027(99)80081-2. [DOI] [PubMed] [Google Scholar]