Abstract
Background
Preterm delivery causes substantial neonatal mortality and morbidity. Unrecognized intra-amniotic infections caused by cultivation-resistant microbes may play a role. Molecular methods can detect, characterize and quantify microbes independently of traditional culture techniques. However, molecular studies that define the diversity and abundance of microbes invading the amniotic cavity, and evaluate their clinical significance within a causal framework, are lacking.
Methods and Findings
In parallel with culture, we used broad-range end-point and real-time PCR assays to amplify, identify and quantify ribosomal DNA (rDNA) of bacteria, fungi and archaea from amniotic fluid of 166 women in preterm labor with intact membranes. We sequenced up to 24 rRNA clones per positive specimen and assigned taxonomic designations to approximately the species level. Microbial prevalence, diversity and abundance were correlated with host inflammation and with gestational and neonatal outcomes. Study subjects who delivered at term served as controls. The combined use of molecular and culture methods revealed a greater prevalence (15% of subjects) and diversity (18 taxa) of microbes in amniotic fluid than did culture alone (9.6% of subjects; 11 taxa). The taxa detected only by PCR included a related group of fastidious bacteria, comprised of Sneathia sanguinegens, Leptotrichia amnionii and an unassigned, uncultivated, and previously-uncharacterized bacterium; one or more members of this group were detected in 25% of positive specimens. A positive PCR was associated with histologic chorioamnionitis (adjusted odds ratio [OR] 20; 95% CI, 2.4 to 172), and funisitis (adjusted OR 18; 95% CI, 3.1 to 99). The positive predictive value of PCR for preterm delivery was 100 percent. A temporal association between a positive PCR and delivery was supported by a shortened amniocentesis-to-delivery interval (adjusted hazard ratio 4.6; 95% CI, 2.2 to 9.5). A dose-response association was demonstrated between bacterial rDNA abundance and gestational age at delivery (r2 = 0.42; P<0.002).
Conclusions
The amniotic cavity of women in preterm labor harbors DNA from a greater diversity of microbes than previously suspected, including as-yet uncultivated, previously-uncharacterized taxa. The strength, temporality and gradient with which these microbial sequence types are associated with preterm delivery support a causal relationship.
Introduction
Preterm birth is the leading cause of neonatal mortality worldwide [1], yet its underlying etiologies remain largely unknown [2], [3]. Mortality exhibits an inverse relationship with gestational age, such that early preterm neonates (e.g., less than 32 gestational weeks) account for the vast majority of deaths [4]–[6]. Consequently, the need for insights into factors contributing to early preterm delivery is particularly acute.
A strong body of evidence suggests that occult intra-uterine infection plays a major role in preterm labor and delivery [7]. These infections are thought to escape detection primarily because they are subclinical, but also because they may be caused by cultivation-resistant microbes [8]. Fastidious bacterial taxa, such as mycoplasmas, are among those most commonly implicated in preterm birth [8]–[11], but the specialized techniques required to cultivate them reliably are seldom used in clinical settings [9]–[11]. Other microbial groups, including the majority of species in many ecosystems, cannot be cultivated with current methods [12].
Molecular methods such as polymerase chain reaction (PCR) can detect microbes independently of culture. Moreover, broad-range PCR assays that amplify highly-conserved but phylogenetically-informative gene sequences can identify microbes across broad taxonomic levels, including previously uncharacterized species. By overcoming investigator biases inherent to more specific detection methods, sequencing of broad-range PCR products has emerged as a powerful approach for revealing previously unsuspected microbial diversity in various anatomic niches in human health [13] and disease [14], and for characterizing uncultivated human pathogens [15].
The application of broad-range PCR to amniotic fluid has been limited to date. In particular, molecular investigations that characterize the microbial diversity of the amniotic cavity in a systematic manner, and evaluate findings within a coherent causal framework, are lacking. As an early step in defining the potential role of diverse microbial sequence types, including uncultivated taxa, in preterm delivery, we conducted a broad-range molecular investigation. In parallel with traditional amniotic fluid cultures, we used qualitative and quantitative PCR assays to amplify, identify and quantify ribosomal DNA (rDNA) of bacteria, fungi and archaea from amniotic fluid of patients with spontaneous preterm labor and intact membranes. We examined sequence diversity in samples with detectable rDNA, and correlated findings with pre-specified measures of host inflammation, as well as pregnancy and neonatal outcome. We sought evidence for the types of associations that have been proposed as alternatives to Koch's postulates for inferring causality from molecular data (e.g., associations of space, time and dose) [16]. Here, we report the prevalence, diversity and abundance of microbes in amniotic fluid during preterm labor, and their clinical significance.
Methods
Study population
A retrospective cohort study was conducted by searching our clinical database to identify patients with the diagnosis of spontaneous preterm labor with intact membranes, enrolled at Hutzel Women's Hospital (Detroit, MI) between October 1998 and December 2002, who provided written informed consent and for whom an adequate volume of amniotic fluid was available with which to conduct these research assays after clinically-indicated conventional analyses were completed. Patients were included if they met the following criteria: 1) singleton gestation; 2) gestational age between 18 and 35 weeks; and 3) had an amniocentesis with microbiological studies of amniotic fluid. Patients were excluded from the study if: 1) rupture of the chorioamniotic membranes occurred before amniotic fluid collection; 2) the amniotic fluid was collected transvaginally; 3) delivery occurred elsewhere and/or clinical data were unavailable; and 4) a major fetal congenital anomaly was present.
A total of 166 subjects were included in this study. The pre-specified control group consisted of study subjects who delivered at term. All women provided written informed consent prior to the collection of biological samples. The utilization of samples for research purposes was approved by the Institutional Review Boards of Wayne State University, the National Institute of Child Health and Human Development (NICHD/NIH/DHHS), and Stanford University.
Definitions
Preterm labor was diagnosed by the presence of at least two regular uterine contractions every 10 minutes associated with cervical changes that required hospital admission before 37 weeks of gestation. Preterm delivery was defined as delivery before 37 weeks of gestation. Clinical chorioamnionitis was diagnosed according to criteria previously proposed [17], including maternal temperature of ≥37.8°C and two or more of the following criteria: uterine tenderness, malodorous vaginal discharge, maternal leukocytosis (≥15000 cells/mm3), maternal tachycardia (>100 beats/min) and fetal tachycardia (>160 beats/min). Histologic chorioamnionitis was diagnosed based on the presence of inflammatory cells in the chorionic plate and/or chorioamniotic membranes. Acute funisitis was diagnosed by the presence of neutrophils in the wall of the umbilical vessels and/or Wharton's jelly using criteria previously described [18].
Sampling procedures
Enrolled subjects underwent transabdominal, ultrasound-guided amniocentesis, which is within the standard of care at Hutzel Women's Hospital for evaluating possible microbial invasion of the amniotic cavity of patients with spontaneous preterm labor. Amniotic fluid was immediately transported in a capped sterile syringe to the clinical laboratory where it was cultured for aerobic and anaerobic bacteria, including genital mycoplasmas. White blood cell (WBC) count and Gram stain of amniotic fluid were also performed shortly after collection. Amniotic fluid not required for clinical assessment was centrifuged for 10 minutes at 4°C shortly after the amniocentesis, and the supernatant was aliquoted and stored at −70°C until analysis. Amniotic fluid IL-6 concentrations were determined after delivery for research purposes, and these results were not used in patient management. A flowchart of our overall approach to amniotic fluid analysis is illustrated in Supporting Figure S1; detailed experimental methods, including microbiologic techniques and IL-6 quantitation, appear in Supporting Materials and Methods S1.
Molecular analysis
Microbial genomic DNA was extracted from 200 µl of each amniotic fluid sample. Qualitative analysis was achieved by means of three separate broad-range end-point PCR assays targeting rDNA of either bacteria, fungi, or archaea. Cloned amplicons (up to 24 clones per positive PCR reaction) were bidirectionally sequenced, the sequences aligned, and then subjected to phylogenetic analysis (621 nucleotide positions). Quantitative rDNA analysis was achieved by means of three real-time PCR assays corresponding to each of the three microbial groups targeted by end-point PCR. Methodologic details of the key aspects of our molecular approach (including nucleic acid extraction, PCR assays, phylogenetic analysis, and contamination prevention) appear under Supporting Materials and Methods S1; PCR primer and probe sequences appear in Supporting Table S4.
Outcome Measures
In order to assess the clinical significance of the microbes detected with molecular methods, pre-specified outcome variables from four broad categories were measured: 1) markers of intra-amniotic inflammation (including amniotic fluid WBC count [19] and IL-6 concentration [20]); 2) histopathologic evidence of maternal or fetal inflammation (including chorioamnionitis and funisitis); 3) pregnancy outcomes (including gestational age at delivery, and amniocentesis-to-delivery interval); and 4) neonatal outcome. Neonatal outcome was assessed by measuring composite neonatal morbidity and mortality, defined as the presence of one or more of the following: bronchopulmonary dysplasia, respiratory distress syndrome, necrotizing enterocolitis, intraventricular hemorrhage≥grade III, and sepsis, which were diagnosed according to previously described criteria [21], as well as respiratory failure requiring mechanical ventilation, and neonatal death.
Statistical analysis
Statistical analyses were performed using ‘R’ (open source, www.r-project.org) version 2.4.1, including the ‘Epi’ and ‘Survival’ packages. For gestational outcomes, patients with an episode of spontaneous preterm labor who delivered at term served as controls. Positive predictive values were estimated by means of contingency table analysis. Differences in the median between groups were computed using the Mann Whitney U test when two groups were compared and the Kruskal-Wallis analysis of variance test when more than two groups were compared. Unadjusted and adjusted odds ratios (OR) for binary outcomes were estimated by simple and multiple logistic regression modeling, respectively. Correlation coefficients for continuous outcomes were estimated by means of least squares linear regression modeling. Time-to-event outcomes were modeled by Kaplan-Meier survival methods; differences between survival curves were evaluated by the Mantel-Haenszel log-rank test, and proportional hazard ratios were estimated by the method of Cox. For all analyses, a two-tailed P value <0.05 was considered significant. Additional details of the statistical methods used, including the calculation of false discovery rates for P values corresponding to multiple pairwise comparisons, appear under Supporting Materials and Methods S1.
Results
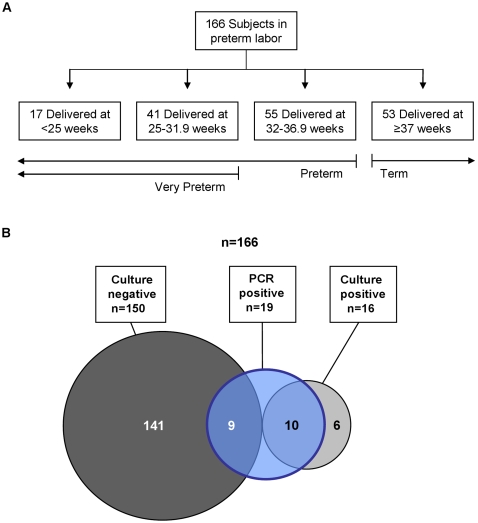
Of 166 subjects, 53 (32%) delivered at term and 113 (68%) delivered preterm (Figure 1A). Ten patients (6%) had clinical chorioamnionitis. Baseline characteristics appear in Supporting Table S1.
Figure 1. Gestational Outcomes and Amniotic Fluid Findings.
Panel A shows outcomes of enrolled subjects. Panel B summarizes results of culture and PCR analysis of amniotic fluid. PCR refers to end-point PCR for bacteria, fungi and archaea. Culture refers to the use of routine cultivation methods for bacteria (aerobic, anaerobic and genital mycoplasmas) and fungi. Circle areas are not to scale.
Microbial prevalence and diversity
Microbial invasion of the amniotic cavity – as defined by either a positive end-point PCR or culture of amniotic fluid – was found in 15% (25/166) of patients. Of these, bacteria were cultivated from 15 and a fungus from 1, whereas bacterial rDNA was amplified from 17 and fungal rDNA from 2. Six culture-positive samples were negative by PCR, and nine PCR-positive samples were negative by culture (Figure 1B). No archaea were detected. Viruses were not targeted by molecular methods; however, culture for cytomegalovirus (CMV) was performed on 79 samples. Of these, 2.5% (2/79) were positive; both subjects delivered full-term neonates.
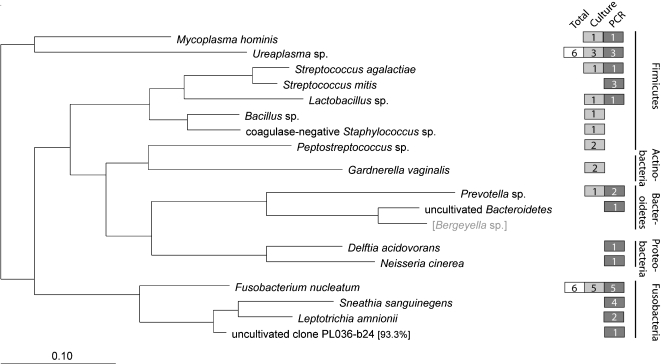
Among the 25 patients whose amniotic fluid tested positive by culture or end-point PCR, 17 bacterial species (belonging to 5 phyla) and 1 fungal species were identified. Of the 17 bacterial taxa identified, 6 were detected by both culture and PCR (Mycoplasma hominis, Ureaplasma sp., Streptococcus agalactiae, Lactobacillus sp., Prevotella sp., and Fusobacterium nucleatum), 4 were detected by culture only (coagulase-negative Staphylococcus sp., Bacillus sp. not anthracis, Peptostreptococcus sp., and Gardnerella vaginalis), and 7 were detected by PCR only (Streptococcus mitis, uncultivated Bacteroidetes bacterium, Delftia acidovorans, Neisseria cinerea, Sneathia (formerly Leptotrichia) sanguinegens, Leptotrichia amnionii, and one phylotype (clone PL036-b24) with <95% rDNA sequence identity to its closest Genbank relative that appeared to represent a novel species) (Figure 2). The single fungal species identified was Candida albicans. The fastidious nature of many taxa detected only by PCR (e.g. S. sanguinegens and L. amnionii) supported the underlying premises of this study. Characteristics of individual subjects testing positive by PCR or culture, including GenBank accession numbers of rDNA sequences, are detailed in Supporting Table S2. We next investigated the relevance of molecular detection of these diverse taxa in the population as a whole by examining associations with host inflammation, and seeking evidence for categorical, temporal, and dose-response associations with preterm delivery.
Figure 2. Microbial Diversity.
Phylogeny of the 17 bacterial taxa identified in this study, based on a maximum likelihood algorithm. Colored boxes indicate the number of subjects who were positive for a given taxon by culture (gray) or PCR (blue) (some samples were polymicrobial). For most individual taxa, the larger of the two numbers in the corresponding gray or blue box represents the total number of positive subjects; for taxa where neither method detected all positive subjects, the total number is shown in the white box. A 99% sequence similarity cutoff threshold was used for phylotype assignment, which was based on 621 unambiguous nucleotide positions. Bergeyella sp. (bracketed and in gray type) is included as a reference species only and was not detected in the study population. A single fungal species, Candida albicans, was detected by culture in 1 subject and by PCR in 2 (data not shown). GenBank accession numbers of bacterial and fungal rDNA sequences from this study appear in Supporting Table S2.
Association of intra-amniotic microbial DNA with host (maternal and fetal) inflammation
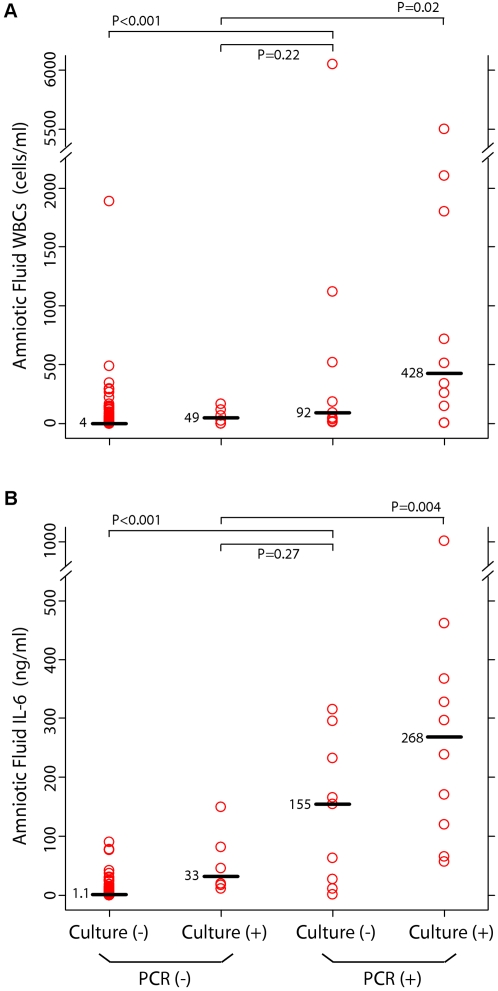
A host inflammatory response has been implicated in the pathogenesis of preterm delivery [8]. In order to evaluate the association between microbial DNA and intra-amniotic inflammation, results from end-point PCR and culture were correlated with amniotic fluid WBC count and IL-6 concentration, which typically are elevated in the setting of infection [19], [21], [22]. Figure 3 shows that the median amniotic fluid WBC count and IL-6 concentration in the PCR-positive / culture-negative (P+/C−) group were higher than the median concentrations of each marker in the P−/C− group (P<0.001 for WBC and for IL-6), and equivalent to the median concentrations of each respective marker in the P−/C+ group (P = 0.22 for WBC; P = 0.27 for IL-6). These results indicate that the inflammatory response in the amniotic cavity of patients who are positive only by PCR is as robust as the inflammatory response in those who are positive only by culture. In addition, the median WBC count and IL-6 concentration in the P+/C+ group were higher than the median concentrations of each respective marker in the P−/C+ group (for WBC, P = 0.02, false discovery rate 152/1000; for IL-6, P = 0.004, false discovery rate 45/1000) (Figure 3), suggesting that a positive PCR result may be clinically significant even in the setting of a positive culture.
Figure 3. Correlation of PCR and Culture Results with Markers of Infection in Amniotic Fluid.
Amniotic fluid concentrations of white blood cells (WBCs) (Panel A) and interleukin-6 (IL-6) (Panel B) as a function of PCR and culture results. Each circle represents one subject; each horizontal bar and adjacent number indicates the median value for that group. P values were calculated by means of the Mann-Whitney U test. PCR refers to end-point PCR for bacteria, fungi and archaea. Culture refers to the use of routine cultivation methods for bacteria (aerobic, anaerobic and genital mycoplasmas) and fungi.
To evaluate associations of microbial DNA with inflammation of maternal and fetal tissues, results from end-point PCR and culture were correlated with histologic chorioamnionitis, which has been previously associated with preterm delivery [23], and with funisitis. Table 1 summarizes results from simple and multiple logistic regression modeling. After controlling for other covariates exhibiting statistically significant associations (see Table 1), the adjusted ORs of PCR and of culture for histological chorioamnionitis were 20 (95% CI, 2.4 to 172) and 8.6 (95% CI, 1.0 to 76), respectively; the adjusted ORs of PCR and of culture for funisitis were 18 (95% CI, 3.1 to 99) and 5.8 (95% CI, 1.1 to 32), respectively.
Table 1. Factors Associated with Histologic Chorioamnionitis, Funisitis, and Neonatal Morbidity and Mortality.
| Variable | Histologic Chorioamnionitis* (n = 57) | Funisitis† (n = 48) | Composite Neonatal Morbidity and Mortality‡ (n = 63) | |||
| Univariate | Multivariate | Univariate | Multivariate | Univariate | Multivariate | |
| Positive amniotic fluid PCR | 37 (4.8–288) | 20 (2.4–172) | 24 (5.2–109) | 18 (3.1–99) | 16.2 (3.6–73) | 5.2 (0.84–32) |
| Positive amniotic fluid culture | 31 (4.0–243) | 8.6 (1.0–76) | 20 (4.2–91) | 5.8 (1.1–32) | 23 (2.9–179) | 7.1 (0.66–76) |
| Maternal age | 0.99 (0.93–1.0) | 0.97 (0.90–1.0) | 0.95 (0.89–1.0) | 0.9 (0.84–0.98) | 1.0 (0.97–1.1) | NA |
| Gestational age at amniocentesis | 0.87 (0.80–0.94) | 0.89 (0.81–0.97) | 0.9 (0.81–0.96) | 0.89 (0.83–0.98) | 0.84 (0.78–92) | 0.80 (0.72–0.88) |
| Cervical dilatation | 1.1 (0.91–1.4) | NA | 1.1 (0.90–1.4) | NA | 1.4 (1.1–1.7) | 1.6 (1.2–2.1) |
Results are reported as odds ratios (95% confidence interval).
The table includes variables that exhibited a statistically significant association with one or more of the three measured outcomes. For each outcome, the multivariate model was comprised of those variables exhibiting a significant association in the univariate model. Other variables that were subject to univariate analysis but did not exhibit a significant association with any of these outcomes were: African-American race, nulliparity, previous preterm delivery, cigarette smoking, and recreational drug use.
Diagnosed based on the presence of inflammatory cells in the chorionic plate and/or chorioamniotic membranes.
Defined as the presence of neutrophils in the wall of the umbilical vessels and/or Wharton's jelly.
Defined as the presence of any one or more of the following: bronchopulmonary dysplasia, respiratory distress syndrome, necrotizing enterocolitis, intraventricular hemorrhage of grade≥III, sepsis, respiratory failure requiring mechanical ventilation, and neonatal death.
NA Not applicable.
Association of intra-amniotic microbial DNA with gestational and neonatal outcomes
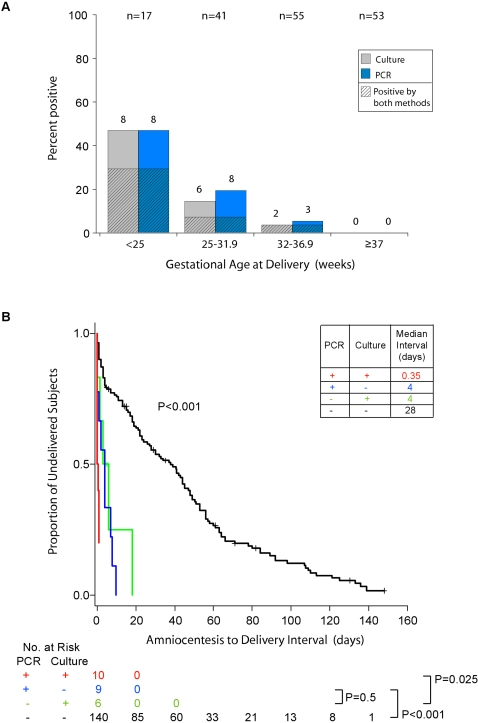
To evaluate the association of microbial DNA with gestational outcomes, results from end-point PCR and culture were correlated with gestational age at delivery. All subjects testing positive by either PCR or culture delivered preterm; this complete separation with respect to test result precluded logistic regression analysis. However, when the study population was stratified according to clinically-relevant gestational-age cut offs, the frequency distribution of positive PCR assays exhibited a clear inverse relationship with gestational age at delivery (Figure 4A). The positive predictive value of PCR for delivery before 37-, 32-, and 25-weeks was 100% for each time point (Table 2).
Figure 4. Gestational Outcomes According to Results of Culture and PCR.
Panel A shows the proportion of subjects from each gestational age cohort who were positive by culture or PCR. The total of number of subjects in each cohort appears across the top of the graph, and the number of positive subjects appears above each bar. Panel B is a Kaplan-Meier analysis of amniocentesis-to-delivery interval according to culture and PCR results. Subjects in whom labor was augmented were censored and are represented by crosses. P values were calculated by means of the Mantel-Haenszel log-rank test. The inset table shows the median interval for each group. For both panels, PCR results refer to end-point PCR for bacteria, fungi and archaea, and culture refers to the use of routine cultivation methods for bacteria (aerobic, anaerobic and genital mycoplasmas) and fungi.
Table 2. Positive Predictive Value (PPV) of Amniotic Fluid PCR and Culture for Pregnancy Outcomes.
| Outcome | Preterm delivery (<37 weeks) | Very preterm delivery (<32 weeks) | Extremely preterm delivery (<25 weeks) | Delivery within one day of amniocentesis |
| Prevalence | 68% (113/166) | 49% (58/118) | 40% (17/43) | 17% (28/166) |
| PPV of PCR | 100% (19/19) | 100% (16/16) | 100% (8/8) | 68% (13/19) |
| PPV of culture | 100% (16/16) | 100% (14/14) | 89% (8/9) | 69% (11/16) |
| PPV of PCR and culture combined | 100% (10/10) | 100% (8/8) | 100% (5/5) | 100% (10/10) |
To evaluate the association of microbial DNA with neonatal outcomes, results from end-point PCR and culture were correlated with a composite of neonatal morbidity and mortality by means of simple and multiple logistic regression. Similar to the outcome of preterm delivery, both PCR and culture exhibited near-complete positive correlation: all but two PCR-positive cases, and all but one culture-positive case were associated with neonatal morbidity and mortality (Supporting Table S2). Although this led to wide confidence limits in the logistic regression models, the magnitude of the odds ratios suggests a significant association for both PCR and culture [adjusted OR 5.2 (95% CI, 0.84 to 32) and 7.1 (95% CI, 0.66 to 76), respectively] (Table 1).
Temporal and dose response associations
To evaluate the association of microbial DNA with timing of delivery, results from end-point PCR and culture were correlated with the interval from amniocentesis to delivery. Kaplan-Meier survival estimates, censored for subjects in whom labor was augmented, and evaluated for differences by means of the Mantel-Haenszel log-rank test, revealed an overall association between a positive test result (PCR and/or culture) and a shortened amniocentesis-to-delivery interval (P<0.001) (Figure 4B). Pairwise comparison of the two cohorts who were positive by one method alone (i.e., P+/C− vs. P−/C+) revealed no difference in the amniocentesis-to-delivery interval of subjects who were positive only by PCR as compared with those who were positive only by culture (P = 0.5), indicating that a positive PCR alone is as significant as a positive culture alone (Figure 4B). However, pairwise comparison of the two cohorts testing positive by culture (i.e., P+/C+ vs. P−/C+) demonstrated a shorter amniocentesis-to-delivery interval for subjects who were positive by both culture and PCR as compared with those who were positive by culture but negative by PCR (P = 0.025, false discovery rate 26/1000) (Figure 4B). These unexpected findings support independent prognostic value for PCR even in the setting of a positive culture. The positive predictive value of PCR or culture for delivery within one day of amniocentesis was approximately 68% for either method alone, and 100% for both methods combined (Table 2). Cox proportional hazards regression, adjusted for nine covariates, estimated the hazard ratio of a positive PCR to be 4.6 (95% CI, 2.2 to 9.5), and of a positive culture to be 6.4 (95% CI, 2.8 to 15) (Supporting Table S3).
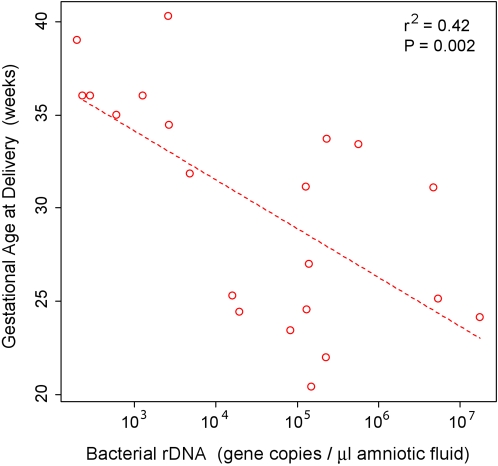
To evaluate dose-response associations, bacterial rDNA concentration was estimated by broad-range real-time PCR, and correlated with gestational age at delivery. Regression modeling revealed a significant correlation (r2 = 0.42; P<0.002) (Figure 5).
Figure 5. Gestational Age at Delivery as a Function of Bacterial rDNA Concentration.
Data are for samples yielding detectable results within the sensitivity and dynamic range of the real-time PCR assay (∼250–1×108 genes/µl amniotic fluid).
Discussion
This study revealed significant associations between the presence and quantity of microbes or their DNA in amniotic fluid, and preterm delivery or other clinically-relevant outcomes. Microbial prevalence, as revealed by means of end-point PCR and culture combined (15%), was 56% higher than that found by culture alone (9.6%), which is the conventional diagnostic approach. PCR detected microbial DNA in culture-negative samples despite the limited sample volumes available for DNA extraction (200 µl per patient), and the fact that PCR was performed 2 to 6 years after sample collection and culturing, which may have limited the number of PCR-positive samples owing to DNA degradation [24]. The prevalence of a positive amniotic fluid culture/PCR was inversely related to gestational age at delivery, reflecting a previously-reported pattern [9]. Importantly, compared to both methods combined, culture exhibited a false negative rate of 27% (3/11) in subjects delivering before 25 weeks (Figure 4A), a cohort with a neonatal mortality rate exceeding 70% in meta-analyses [25]. This underscores the need for improved diagnostic assays in this setting.
The microbial diversity revealed by end-point PCR and culture combined (18 taxa) was almost twice as rich as that found by culture alone (11 taxa). Among the taxa identified by PCR, but not culture, were phylotypes that would not have been predicted from prior studies. One taxon appeared to be a novel species, and possibly a novel genus. The sequence of this taxon (clone PL036-b24; GenBank accession no. EU932745) clustered nearest the genus Leptotrichia within the phylum Fusobacteria (Figure 2). Two other fastidious members of this phylum went undetected by culture: Leptotrichia amnionii (n = 2) and Sneathia (formerly Leptotrichia) sanguinegens (n = 4). The latter was the third most frequently encountered taxon overall (Figure 2). Both taxa were provided valid species descriptions only recently [26], [27]. Sporadic reports of their detection in peripartum bacteremia [28]–[30], pyosalpinx [31], [32], and in amniotic fluid [27], [33], [34] associated with various clinical syndromes, suggest they possess pathogenic potential. L. amnionii has also been associated with bacterial vaginosis [14], a condition conferring an approximately two-fold risk of preterm delivery [35]–[37]. This study extends support for Leptotrichia sp., Sneathia sp. and related taxa as intra-amniotic pathogens, and suggests that their prevalence in the setting of preterm labor is underestimated. For other bacterial phylotypes detected only by PCR (Delftia acidovorans [Genbank accession no. EU932748], uncultured Bacteroidetes bacterium [Genbank accession no. EU932749], and Neisseria cinerea [Genbank accession no. EU932746]), we found no reports of their previous detection in either human amniotic fluid or maternal-fetal membranes.
A single fungal phylotype, Candida albicans, was detected in two subjects, both of whom were PCR-positive, and one of whom was culture-positive. We detected no members of Archaea, the most recently characterized domain of life [38]. This suggests that if archaea are capable of amniotic cavity invasion, this occurs rarely or involves abundance levels below the detection threshold of our assays.
Prior studies have applied species-specific [10], [11], [39]–[44] or broad-range [33], [34], [45]–[47] PCR to amniotic fluid, within various clinical settings. These early investigations yielded insights but had limitations. Indeed, the broad-range studies involved small sample sizes, and included rDNA sequencing that was limited [33], shallow (one sequence per positive PCR reaction) [34], [46], or absent [45], [47]. These studies also lacked measures of microbial abundance. Cumulatively, however, these important studies demonstrated the sensitivity of PCR in this setting, including the potential for detecting fastidious taxa such as Ureaplasma spp., and revealed associations with certain outcomes.
To overcome limitations of prior studies, we analyzed 166 subjects for microbes belonging to three broad taxonomic groups. Microbial diversity was assessed by sequencing up to 24 clones per subject. Microbial rDNA abundance was measured in parallel. Using this approach, we demonstrated greater microbial diversity than expected from prior studies. Compared to findings from culture, the molecular detection of these diverse microbes exhibited equivalent associations with indices of host inflammation, and with adverse gestational and neonatal outcomes.
Our study does not prove causation. To do so for multifactorial biological phenomena often entails successive investigations that expand knowledge within a coherent framework until sufficient evidence accumulates. Any single framework, however, may have limitations. The postulates of Robert Koch, for example, cannot be fulfilled for uncultivated pathogens [48]. However, an alternative causal framework based on epidemiologic guidelines proposed by Sir Austin Bradford Hill [49] may be particularly appropriate for molecular investigations [16]. Of Hill's nine criteria, our broad-range molecular findings demonstrate statistical support for three: strength of association, temporality and biological gradient. Additionally, our study augments a fourth general criterion of consistency of association (similar findings over multiple investigations using different methodology), and conforms to criteria of coherence, plausibility, analogy and experimentation as they pertain to intra-amniotic infection [8], [50].
Despite this study's progress in defining the microbial diversity associated with preterm delivery, much likely awaits discovery. Many detected phylotypes, such as the uncultivated Bacteroidetes bacterium (GenBank accession no. EU932749) and the previously-uncharacterized Fusobacteria bacterium (GenBank accession no. EU932745), were singletons that have been rarely, if ever, reported from human amniotic fluid. It can thus be inferred, based on ecological approaches for estimating species richness in sampled environments [51], that the amniotic cavity remains a largely unexplored niche.
This study has some limitations, some of which could affect results from molecular assays disproportionately, thus potentially contributing to a negative PCR in some culture-positive cases. First, DNA may have degraded during the interval from amniocentesis to molecular analysis (range, 2–6 years) [24], rendering it non-amplifiable in some samples. Second, the starting sample volume of 200 µl was probably suboptimal for use with broad-range PCR assays, which are less sensitive than species-specific PCR assays. Third, molecular findings may have been affected by PCR inhibition [52], or by biases in DNA extraction [52], PCR amplification efficiency [52], or PCR primer specificity [53]. Fourth, our approach may have failed to detect phylotypes present in polymicrobial samples at low relative abundance. Last, we did not target viruses or non-fungal eukaryotic microbes.
In conclusion, among patients with spontaneous preterm labor and intact membranes, the amniotic cavity harbors a greater diversity of microbes than previously suspected, including uncultivated, previously-uncharacterized taxa. The strength, temporality and gradient with which detected microbes were associated with clinically relevant outcomes, including preterm delivery, suggest a causal relationship. Despite these insights, the microbial census of the amniotic cavity is unfinished. Taken together, our findings support a contributory role for occult intra-amniotic infection in preterm delivery and its neonatal sequelae, and argue for further large-scale prospective molecular investigations.
Supporting Information
Supporting Materials and Methods
(0.04 MB DOC)
Approach to Amniotic Fluid Analysis. Results reported in this study are shaded either grey (conventional analyses) or blue (molecular analyses). *PCR assays targeted the domain Bacteria, domain Archaea and the fungal division of Eukarya.
(0.28 MB TIF)
Baseline Subject Characteristics According to Results of PCR and Culture of Amniotic Fluid.
(0.04 MB DOC)
Characteristics of Individual Subjects Who Tested Positive by PCR or Culture.
(0.15 MB DOC)
Association of Demographic and Microbiologic Variables with Shortened Amniocentesis-to-Delivery Interval.
(0.03 MB DOC)
Broad-range PCR Assays Used in this Study.
(0.06 MB DOC)
Acknowledgments
We are grateful to study subjects. We thank Nicola Casali, University of California, Berkeley, for thoughtful comments on the manuscript. The authors wish to acknowledge essential contributions of staff of the Perinatology Research Branch and Detroit Medical Center, including Nancy Hauff, Sandy Field, Lorraine Nikita, Vicky Ineson, Evie Russell, Mahbubeh Mahmoudieh, Julie McKinley, Sue Rehel, Shannon Donegan, Linda Bouey, Carolyn Sudz, Sylvia Warren, Shelley Mullen, Gail Bartley, Denise Bayoneto, Judy Kerman, Barbara Steffy, Milagros Kitchen, and Leandra Ga-Pinlac, as well as Doctors Sonia Hassan, Pooja Mittal, Yoram Sorokin, Theodore Jones, and Susan Berman.
Footnotes
Competing Interests: The authors have declared that no competing interests exist.
Funding: This work was supported, in part, by the Intramural Research Program of the National Institute of Child Health and Human Development, NIH, DHHS, and by NIH grant T32 AI052073-01 (Epidemiology of Emerging Infections and Bioterrorism). DAR is supported by an NIH Directors Pioneer Award (NIH DP1OD000964). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Lawn JE, Cousens S, Zupan J. 4 million neonatal deaths: when? Where? Why? Lancet. 2005;365:891–900. doi: 10.1016/S0140-6736(05)71048-5. [DOI] [PubMed] [Google Scholar]
- 2.Goldenberg RL, Rouse DJ. Prevention of premature birth. N Engl J Med. 1998;339:313–320. doi: 10.1056/NEJM199807303390506. [DOI] [PubMed] [Google Scholar]
- 3.Simhan HN, Caritis SN. Prevention of preterm delivery. N Engl J Med. 2007;357:477–487. doi: 10.1056/NEJMra050435. [DOI] [PubMed] [Google Scholar]
- 4.Committee on Understanding Premature Birth and Assuring Healthy Outcomes. Preterm Birth: Causes, Consequences, and Prevention. Washington D.C.: The National Academy Press; 2006. [Google Scholar]
- 5.Green NS, Damus K, Simpson JL, Iams J, Reece EA, et al. Research agenda for preterm birth: recommendations from the March of Dimes. Am J Obstet Gynecol. 2005;193:626–635. doi: 10.1016/j.ajog.2005.02.106. [DOI] [PubMed] [Google Scholar]
- 6.Bucher HU, Ochsner Y, Fauchere JC. Two years outcome of very pre-term and very low birthweight infants in Switzerland. Swiss Med Wkly. 2003;133:93–99. doi: 10.4414/smw.2003.10099. [DOI] [PubMed] [Google Scholar]
- 7.Goldenberg RL, Hauth JC, Andrews WW. Intrauterine infection and preterm delivery. N Engl J Med. 2000;342:1500–1507. doi: 10.1056/NEJM200005183422007. [DOI] [PubMed] [Google Scholar]
- 8.Romero R, Gomez R, Chaiworapongsa T, Conoscenti G, Kim JC, et al. The role of infection in preterm labour and delivery. Paediatr Perinat Epidemiol. 2001;15(Suppl 2):41–56. doi: 10.1046/j.1365-3016.2001.00007.x. [DOI] [PubMed] [Google Scholar]
- 9.Watts DH, Krohn MA, Hillier SL, Eschenbach DA. The association of occult amniotic fluid infection with gestational age and neonatal outcome among women in preterm labor. Obstet Gynecol. 1992;79:351–357. doi: 10.1097/00006250-199203000-00005. [DOI] [PubMed] [Google Scholar]
- 10.Yoon BH, Romero R, Kim M, Kim EC, Kim T, et al. Clinical implications of detection of Ureaplasma urealyticum in the amniotic cavity with the polymerase chain reaction. Am J Obstet Gynecol. 2000;183:1130–1137. doi: 10.1067/mob.2000.109036. [DOI] [PubMed] [Google Scholar]
- 11.Yoon BH, Romero R, Lim JH, Shim SS, Hong JS, et al. The clinical significance of detecting Ureaplasma urealyticum by the polymerase chain reaction in the amniotic fluid of patients with preterm labor. Am J Obstet Gynecol. 2003;189:919–924. doi: 10.1067/s0002-9378(03)00839-1. [DOI] [PubMed] [Google Scholar]
- 12.Hugenholtz P. Exploring prokaryotic diversity in the genomic era. Genome Biol. 2002;3:REVIEWS0003. doi: 10.1186/gb-2002-3-2-reviews0003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Eckburg PB, Bik EM, Bernstein CN, Purdom E, Dethlefsen L, et al. Diversity of the human intestinal microbial flora. Science. 2005;308:1635–1638. doi: 10.1126/science.1110591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fredricks DN, Fiedler TL, Marrazzo JM. Molecular identification of bacteria associated with bacterial vaginosis. N Engl J Med. 2005;353:1899–1911. doi: 10.1056/NEJMoa043802. [DOI] [PubMed] [Google Scholar]
- 15.Relman DA, Loutit JS, Schmidt TM, Falkow S, Tompkins LS. The agent of bacillary angiomatosis. An approach to the identification of uncultured pathogens. N Engl J Med. 1990;323:1573–1580. doi: 10.1056/NEJM199012063232301. [DOI] [PubMed] [Google Scholar]
- 16.Fredericks DN, Relman DA. Sequence-based identification of microbial pathogens: a reconsideration of Koch's postulates. Clin Microbiol Rev. 1996;9:18–33. doi: 10.1128/cmr.9.1.18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gibbs RS, Blanco JD, St Clair PJ, Castaneda YS. Quantitative bacteriology of amniotic fluid from women with clinical intraamniotic infection at term. J Infect Dis. 1982;145:1–8. doi: 10.1093/infdis/145.1.1. [DOI] [PubMed] [Google Scholar]
- 18.Redline RW, Faye-Petersen O, Heller D, Qureshi F, Savell V, et al. Amniotic infection syndrome: nosology and reproducibility of placental reaction patterns. Pediatr Dev Pathol. 2003;6:435–448. doi: 10.1007/s10024-003-7070-y. [DOI] [PubMed] [Google Scholar]
- 19.Romero R, Quintero R, Nores J, Avila C, Mazor M, et al. Amniotic fluid white blood cell count: a rapid and simple test to diagnose microbial invasion of the amniotic cavity and predict preterm delivery. Am J Obstet Gynecol. 1991;165:821–830. doi: 10.1016/0002-9378(91)90423-o. [DOI] [PubMed] [Google Scholar]
- 20.Yoon BH, Romero R, Kim CJ, Jun JK, Gomez R, et al. Amniotic fluid interleukin-6: a sensitive test for antenatal diagnosis of acute inflammatory lesions of preterm placenta and prediction of perinatal morbidity. Am J Obstet Gynecol. 1995;172:960–970. doi: 10.1016/0002-9378(95)90028-4. [DOI] [PubMed] [Google Scholar]
- 21.Romero R, Jimenez C, Lohda AK, Nores J, Hanaoka S, et al. Amniotic fluid glucose concentration: a rapid and simple method for the detection of intraamniotic infection in preterm labor. Am J Obstet Gynecol. 1990;163:968–974. doi: 10.1016/0002-9378(90)91106-m. [DOI] [PubMed] [Google Scholar]
- 22.Goldenberg RL, Andrews WW, Hauth JC. Markers of preterm birth. Prenat Neonat Med. 1998;3:43–46. [Google Scholar]
- 23.Hillier SL, Martius J, Krohn M, Kiviat N, Holmes KK, et al. A case-control study of chorioamnionic infection and histologic chorioamnionitis in prematurity. N Engl J Med. 1988;319:972–978. doi: 10.1056/NEJM198810133191503. [DOI] [PubMed] [Google Scholar]
- 24.Katsoulis J, Heitz-Mayfield LJ, Weibel M, Hirschi R, Lang NP, et al. Impact of sample storage on detection of periodontal bacteria. Oral Microbiol Immunol. 2005;20:128–130. doi: 10.1111/j.1399-302X.2004.00200.x. [DOI] [PubMed] [Google Scholar]
- 25.Lorenz JM, Wooliever DE, Jetton JR, Paneth N. A quantitative review of mortality and developmental disability in extremely premature newborns. Arch Pediatr Adolesc Med. 1998;152:425–435. doi: 10.1001/archpedi.152.5.425. [DOI] [PubMed] [Google Scholar]
- 26.Collins MD, Hoyles L, Tornqvist E, von Essen R, Falsen E. Characterization of some strains from human clinical sources which resemble “Leptotrichia sanguinegens”: description of Sneathia sanguinegens sp. nov., gen. nov. Syst Appl Microbiol. 2001;24:358–361. doi: 10.1078/0723-2020-00047. [DOI] [PubMed] [Google Scholar]
- 27.Shukla SK, Meier PR, Mitchell PD, Frank DN, Reed KD. Leptotrichia amnionii sp. nov., a novel bacterium isolated from the amniotic fluid of a woman after intrauterine fetal demise. J Clin Microbiol. 2002;40:3346–3349. doi: 10.1128/JCM.40.9.3346-3349.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.De Martino SJ, Mahoudeau I, Brettes JP, Piemont Y, Monteil H, et al. Peripartum bacteremias due to Leptotrichia amnionii and Sneathia sanguinegens, rare causes of fever during and after delivery. J Clin Microbiol. 2004;42:5940–5943. doi: 10.1128/JCM.42.12.5940-5943.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hanff PA, Rosol-Donoghue JA, Spiegel CA, Wilson KH, Moore LH. Leptotrichia sanguinegens sp. nov., a new agent of postpartum and neonatal bacteremia. Clin Infect Dis. 1995;20(Suppl 2):S237–239. doi: 10.1093/clinids/20.supplement_2.s237. [DOI] [PubMed] [Google Scholar]
- 30.Thilesen CM, Nicolaidis M, Lokebo JE, Falsen E, Jorde AT, et al. Leptotrichia amnionii, an Emerging Pathogen of the Female Urogenital Tract. J Clin Microbiol. 2007;45:2344–2347. doi: 10.1128/JCM.00167-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gundi VA, Desbriere R, La Scola B. Leptotrichia amnionii and the female reproductive tract. Emerg Infect Dis. 2004;10:2056–2057. doi: 10.3201/eid1011.031019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hebb JK, Cohen CR, Astete SG, Bukusi EA, Totten PA. Detection of novel organisms associated with salpingitis, by use of 16S rDNA polymerase chain reaction. J Infect Dis. 2004;190:2109–2120. doi: 10.1086/425929. [DOI] [PubMed] [Google Scholar]
- 33.Gardella C, Riley DE, Hitti J, Agnew K, Krieger JN, et al. Identification and sequencing of bacterial rDNAs in culture-negative amniotic fluid from women in premature labor. Am J Perinatol. 2004;21:319–323. doi: 10.1055/s-2004-831884. [DOI] [PubMed] [Google Scholar]
- 34.Miralles R, Hodge R, McParland PC, Field DJ, Bell SC, et al. Relationship between antenatal inflammation and antenatal infection identified by detection of microbial genes by polymerase chain reaction. Pediatr Res. 2005;57:570–577. doi: 10.1203/01.PDR.0000155944.48195.97. [DOI] [PubMed] [Google Scholar]
- 35.Hillier SL. The complexity of microbial diversity in bacterial vaginosis. N Engl J Med. 2005;353:1886–1887. doi: 10.1056/NEJMp058191. [DOI] [PubMed] [Google Scholar]
- 36.Hillier SL, Nugent RP, Eschenbach DA, Krohn MA, Gibbs RS, et al. Association between bacterial vaginosis and preterm delivery of a low-birth-weight infant. The Vaginal Infections and Prematurity Study Group. N Engl J Med. 1995;333:1737–1742. doi: 10.1056/NEJM199512283332604. [DOI] [PubMed] [Google Scholar]
- 37.Leitich H, Bodner-Adler B, Brunbauer M, Kaider A, Egarter C, et al. Bacterial vaginosis as a risk factor for preterm delivery: a meta-analysis. Am J Obstet Gynecol. 2003;189:139–147. doi: 10.1067/mob.2003.339. [DOI] [PubMed] [Google Scholar]
- 38.Woese CR, Kandler O, Wheelis ML. Towards a natural system of organisms: proposal for the domains Archaea, Bacteria, and Eucarya. Proc Natl Acad Sci U S A. 1990;87:4576–4579. doi: 10.1073/pnas.87.12.4576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Blanchard A, Hentschel J, Duffy L, Baldus K, Cassell GH. Detection of Ureaplasma urealyticum by polymerase chain reaction in the urogenital tract of adults, in amniotic fluid, and in the respiratory tract of newborns. Clin Infect Dis. 1993;17(Suppl 1):S148–153. doi: 10.1093/clinids/17.supplement_1.s148. [DOI] [PubMed] [Google Scholar]
- 40.Gerber S, Vial Y, Hohlfeld P, Witkin SS. Detection of Ureaplasma urealyticum in second-trimester amniotic fluid by polymerase chain reaction correlates with subsequent preterm labor and delivery. J Infect Dis. 2003;187:518–521. doi: 10.1086/368205. [DOI] [PubMed] [Google Scholar]
- 41.Leon R, Silva N, Ovalle A, Chaparro A, Ahumada A, et al. Detection of Porphyromonas gingivalis in the amniotic fluid in pregnant women with a diagnosis of threatened premature labor. J Periodontol. 2007;78:1249–1255. doi: 10.1902/jop.2007.060368. [DOI] [PubMed] [Google Scholar]
- 42.Oyarzun E, Yamamoto M, Kato S, Gomez R, Lizama L, et al. Specific detection of 16 micro-organisms in amniotic fluid by polymerase chain reaction and its correlation with preterm delivery occurrence. Am J Obstet Gynecol. 1998;179:1115–1119. doi: 10.1016/s0002-9378(98)70115-2. [DOI] [PubMed] [Google Scholar]
- 43.Perni SC, Vardhana S, Korneeva I, Tuttle SL, Paraskevas LR, et al. Mycoplasma hominis and Ureaplasma urealyticum in midtrimester amniotic fluid: association with amniotic fluid cytokine levels and pregnancy outcome. Am J Obstet Gynecol. 2004;191:1382–1386. doi: 10.1016/j.ajog.2004.05.070. [DOI] [PubMed] [Google Scholar]
- 44.Yi J, Yoon BH, Kim EC. Detection and biovar discrimination of Ureaplasma urealyticum by real-time PCR. Mol Cell Probes. 2005;19:255–260. doi: 10.1016/j.mcp.2005.04.002. [DOI] [PubMed] [Google Scholar]
- 45.Hitti J, Riley DE, Krohn MA, Hillier SL, Agnew KJ, et al. Broad-spectrum bacterial rDNA polymerase chain reaction assay for detecting amniotic fluid infection among women in premature labor. Clin Infect Dis. 1997;24:1228–1232. doi: 10.1086/513669. [DOI] [PubMed] [Google Scholar]
- 46.Jalava J, Mantymaa ML, Ekblad U, Toivanen P, Skurnik M, et al. Bacterial 16S rDNA polymerase chain reaction in the detection of intra-amniotic infection. Br J Obstet Gynaecol. 1996;103:664–669. doi: 10.1111/j.1471-0528.1996.tb09835.x. [DOI] [PubMed] [Google Scholar]
- 47.Markenson GR, Martin RK, Tillotson-Criss M, Foley KS, Stewart RS, Jr, et al. The use of the polymerase chain reaction to detect bacteria in amniotic fluid in pregnancies complicated by preterm labor. Am J Obstet Gynecol. 1997;177:1471–1477. doi: 10.1016/s0002-9378(97)70093-0. [DOI] [PubMed] [Google Scholar]
- 48.Rivers TM. Viruses and Koch's Postulates. J Bacteriol. 1937;33:1–12. doi: 10.1128/jb.33.1.1-12.1937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Hill AB. The Environment and Disease: Association or Causation? Proc R Soc Med. 1965;58:295–300. [PMC free article] [PubMed] [Google Scholar]
- 50.Gravett MG, Adams KM, Sadowsky DW, Grosvenor AR, Witkin SS, et al. Immunomodulators plus antibiotics delay preterm delivery after experimental intraamniotic infection in a nonhuman primate model. Am J Obstet Gynecol. 2007;197:518 e511–518. doi: 10.1016/j.ajog.2007.03.064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Hughes JB, Hellmann JJ, Ricketts TH, Bohannan BJ. Counting the uncountable: statistical approaches to estimating microbial diversity. Appl Environ Microbiol. 2001;67:4399–4406. doi: 10.1128/AEM.67.10.4399-4406.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.von Wintzingerode F, Gobel UB, Stackebrandt E. Determination of microbial diversity in environmental samples: pitfalls of PCR-based rRNA analysis. FEMS Microbiol Rev. 1997;21:213–229. doi: 10.1111/j.1574-6976.1997.tb00351.x. [DOI] [PubMed] [Google Scholar]
- 53.Baker GC, Smith JJ, Cowan DA. Review and re-analysis of domain-specific 16S primers. J Microbiol Methods. 2003;55:541–555. doi: 10.1016/j.mimet.2003.08.009. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supporting Materials and Methods
(0.04 MB DOC)
Approach to Amniotic Fluid Analysis. Results reported in this study are shaded either grey (conventional analyses) or blue (molecular analyses). *PCR assays targeted the domain Bacteria, domain Archaea and the fungal division of Eukarya.
(0.28 MB TIF)
Baseline Subject Characteristics According to Results of PCR and Culture of Amniotic Fluid.
(0.04 MB DOC)
Characteristics of Individual Subjects Who Tested Positive by PCR or Culture.
(0.15 MB DOC)
Association of Demographic and Microbiologic Variables with Shortened Amniocentesis-to-Delivery Interval.
(0.03 MB DOC)
Broad-range PCR Assays Used in this Study.
(0.06 MB DOC)