Abstract
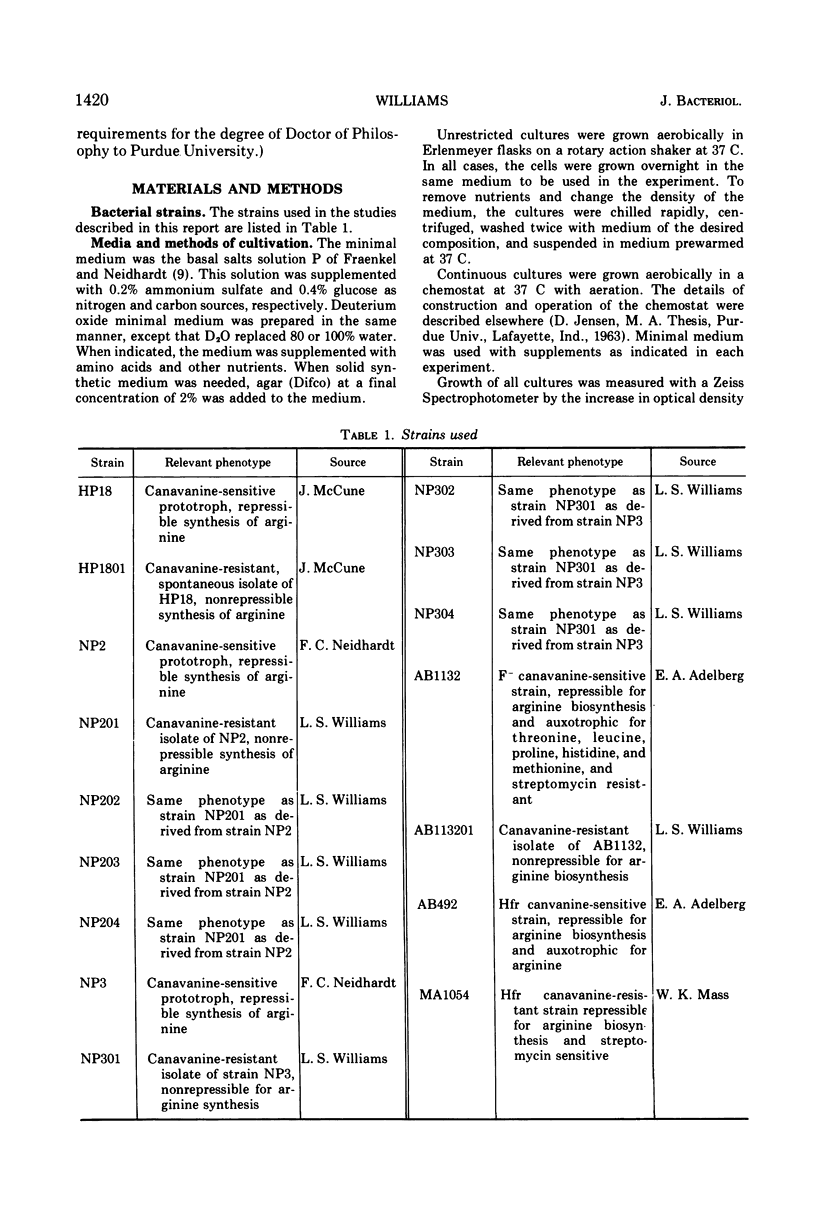
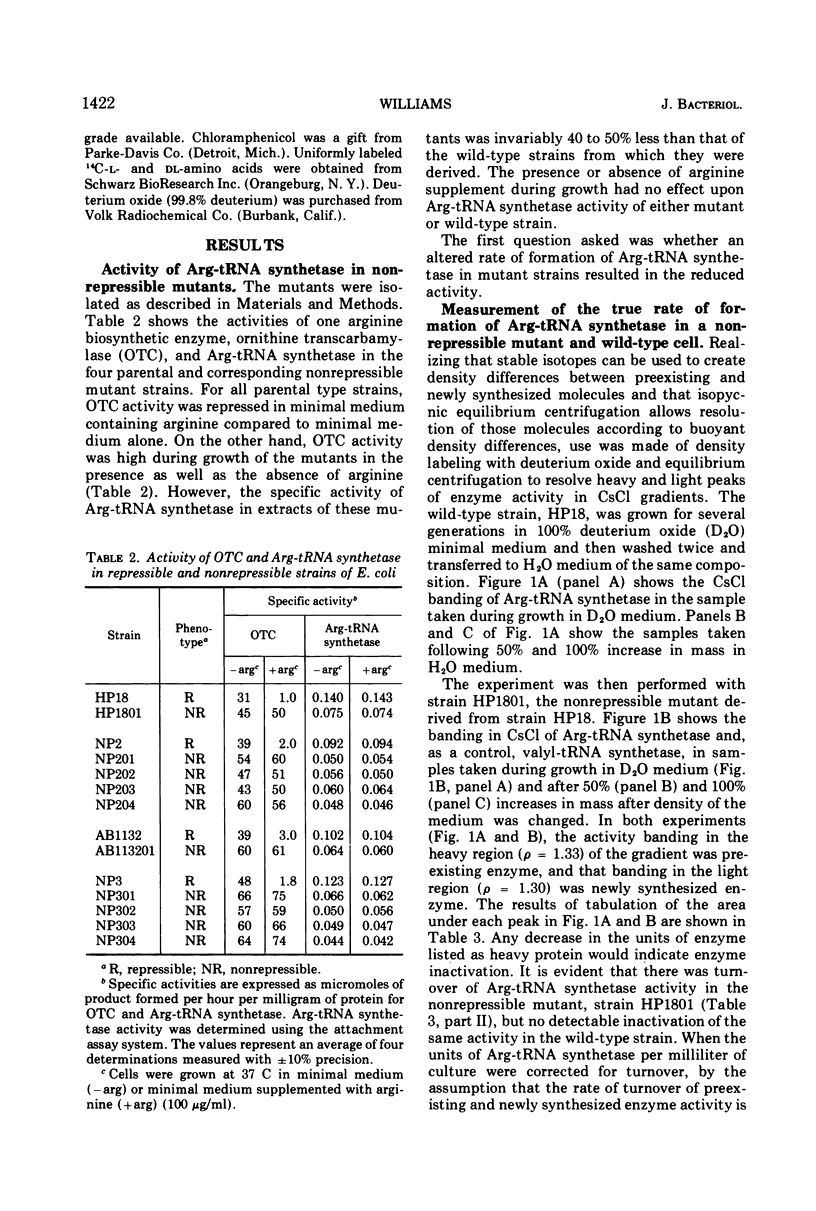
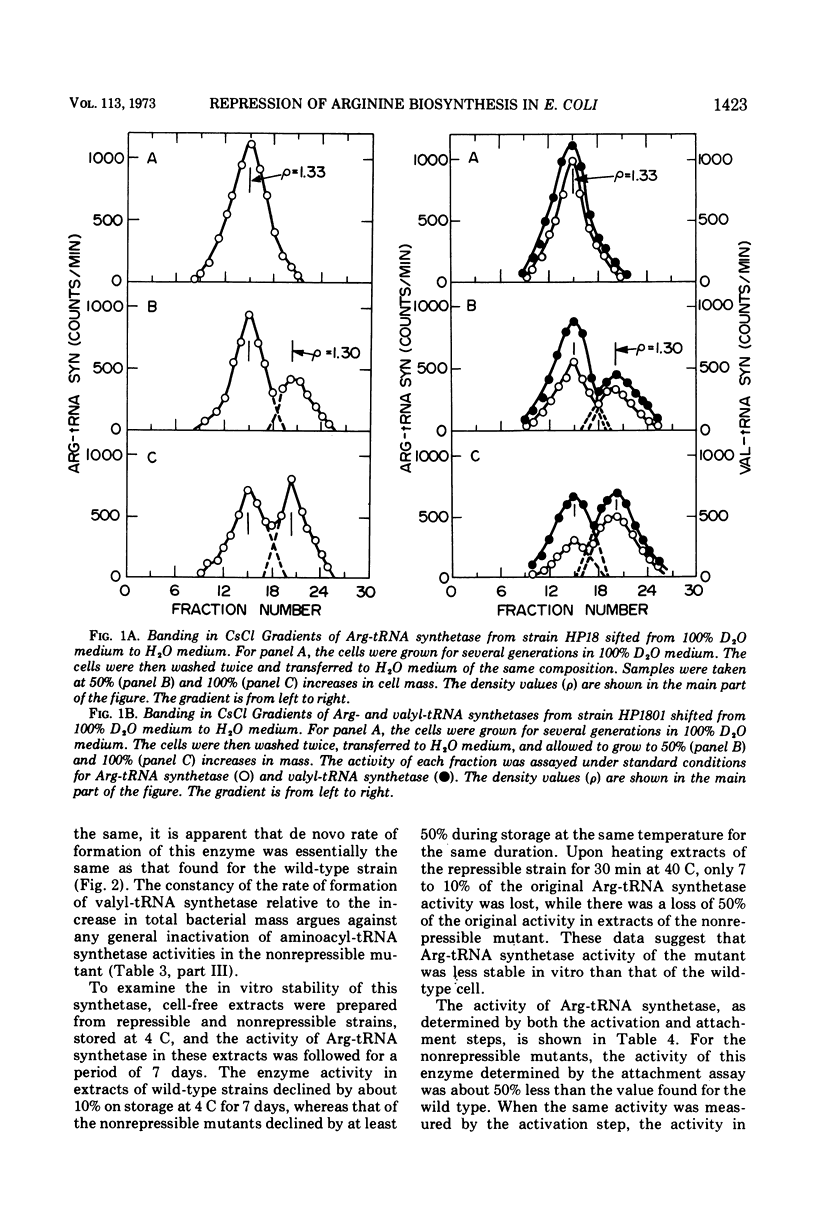
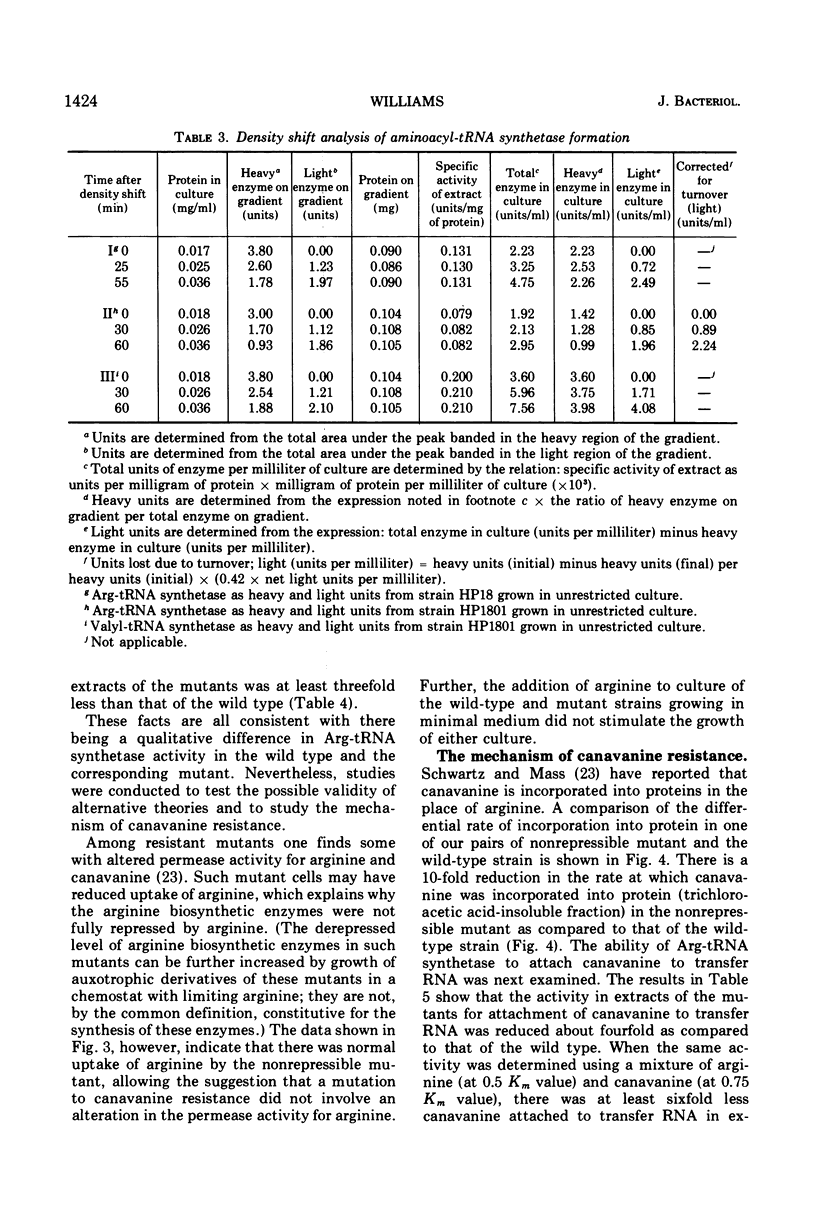
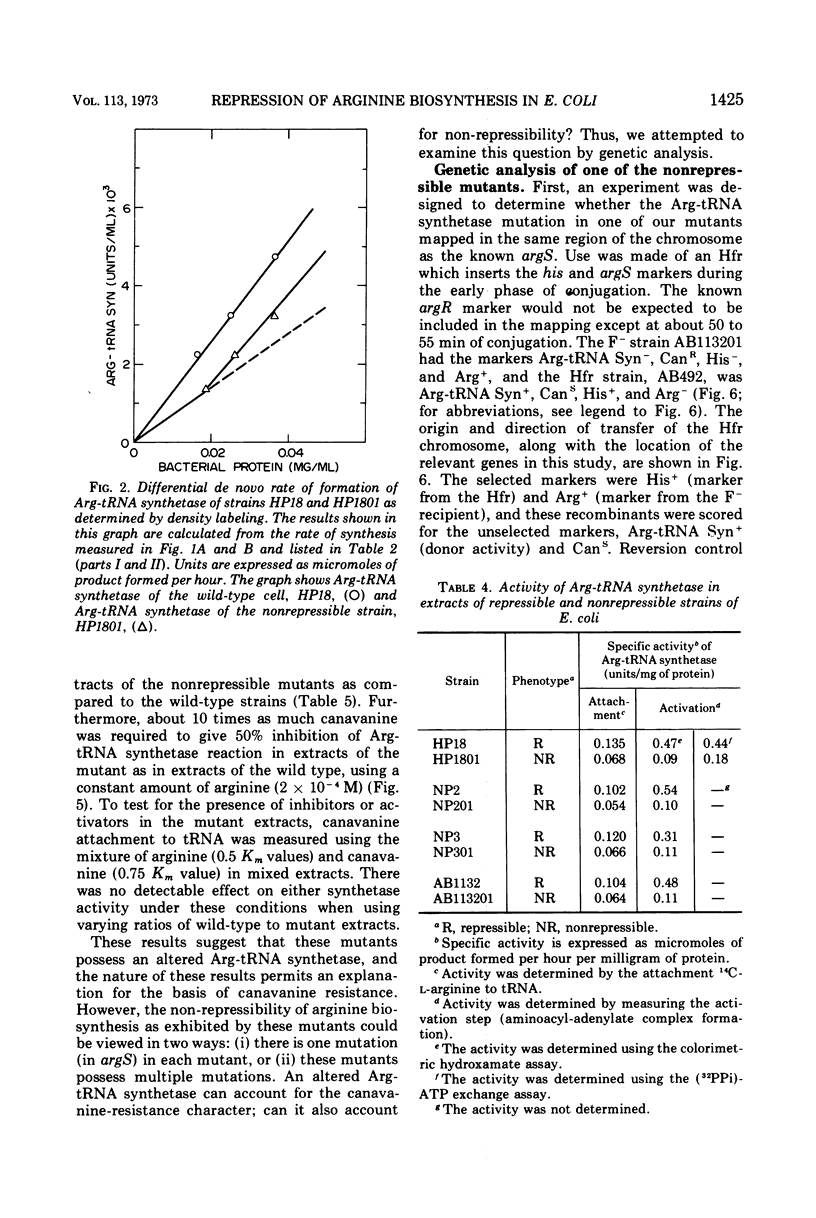
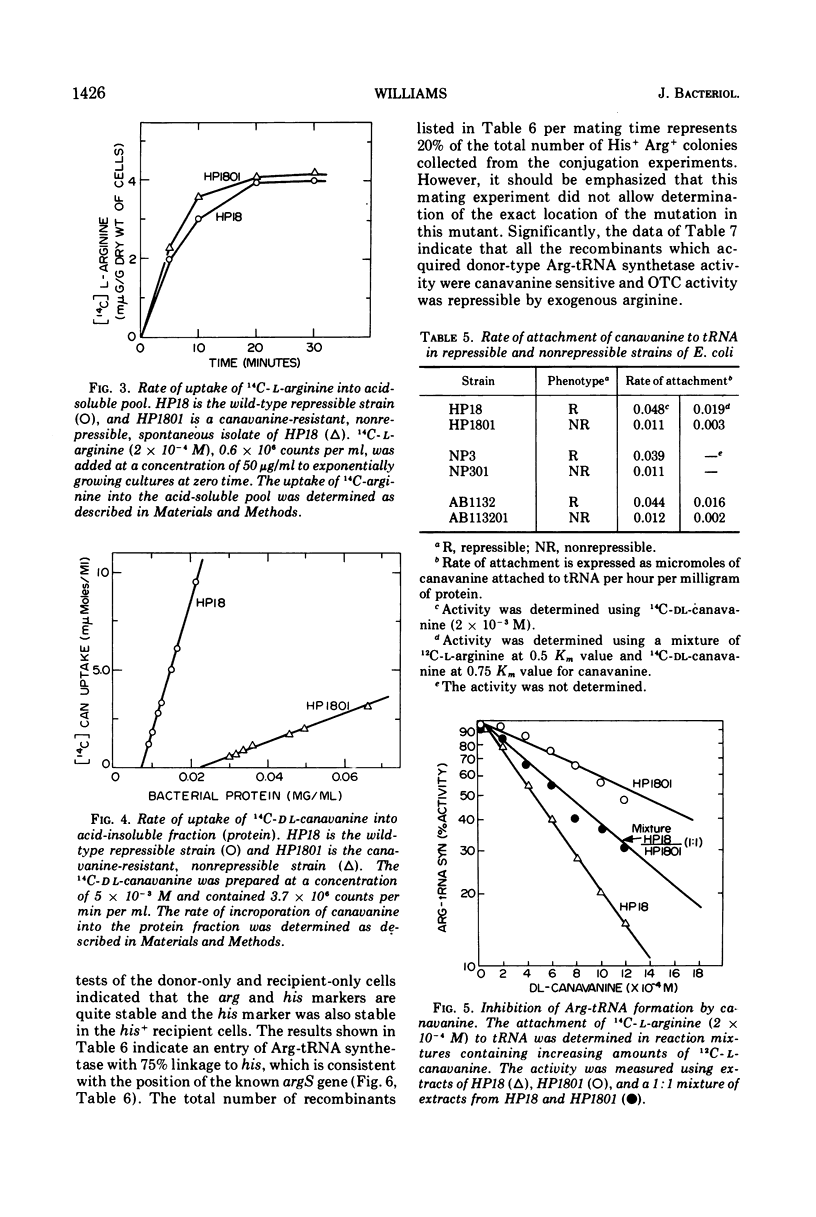
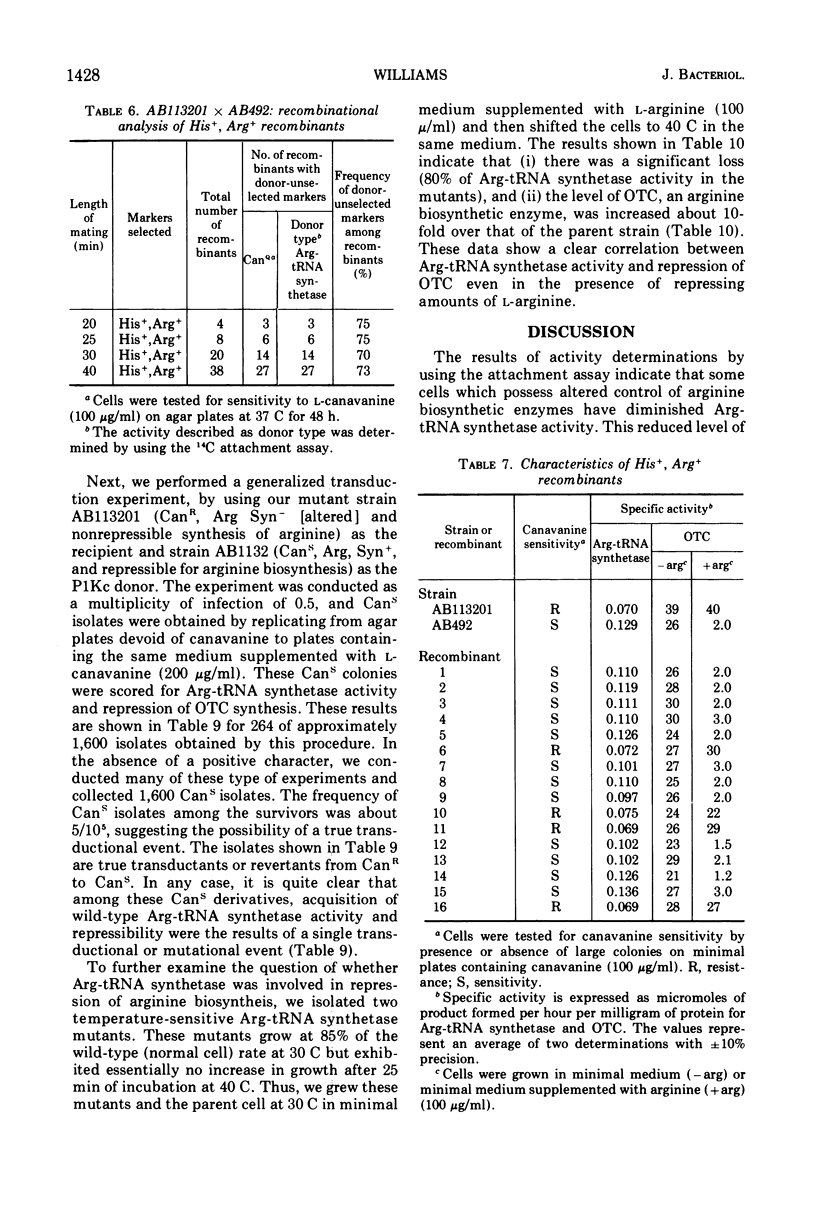
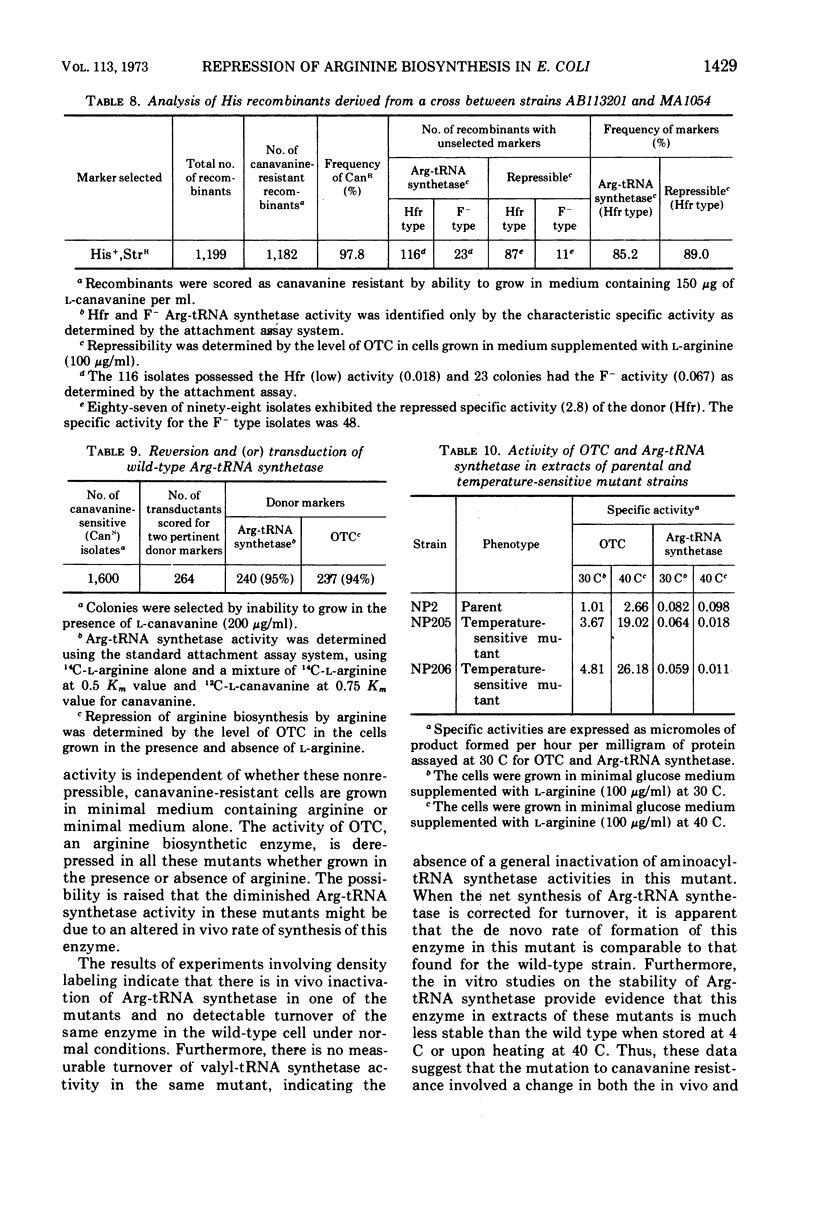
The physiological role of arginyl-transfer ribonucleic acid (Arg-tRNA) synthetase (E.C. 6.1.1.13, arginine: RNA ligase adenosine monophosphate) in repression of arginine biosynthetic enzymes was examined. Mutants with nonrepressible synthesis of arginine biosynthetic enzymes were isolated from various strains of Escherichia coli by resistance to growth inhibition by canavanine, an arginine analogue. These mutants possessed reduced Arg-tRNA synthetase activities which were qualitatively different from the synthetase activity of the wild type. The mutant enzymes exhibited turnover in vivo and were less stable in vitro than the wild type at both 4 C and 40 C; they possessed different affinities for both arginine and canavanine as measured by the three common assay systems for aminoacyl-tRNA synthetases. Furthermore, in one case it was shown that (i) the mutant possesed unaltered uptake of arginine, and (ii) that the mutant possessed diminished ability to incorporate canavanine into proteins and to attach canavanine to tRNA. These observations suggested that the mutation to canavanine resistance involved a structural change in Arg-tRNA synthetase. Likewise, the results of genetic experiments suggested that the mutants differed from the wild-type strain at only one locus, and that this lies in the region of the chromosomes that includes a structural gene for Arg-tRNA synthetase. It appears that Arg-tRNA synthetase may be involved in some way in repression by arginine of its own biosynthetic enzymes.
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alexander R. R., Calvo J. M., Freundlich M. Mutants of Salmonella typhimurium with an altered leucyl-transfer ribonucleic acid synthetase. J Bacteriol. 1971 Apr;106(1):213–220. doi: 10.1128/jb.106.1.213-220.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- BERG P. Studies on the enzymatic utilization of amino acyladenylates; the formation of adenosine triphosphate. J Biol Chem. 1958 Sep;233(3):601–607. [PubMed] [Google Scholar]
- Celis T. F., Maas W. K. Studies on the mechanism of repression of arginine biosynthesis in Escherichia coli. IV. Further studies on the role of arginine transfer RNA repression of the enzymes of arginine biosynthesis. J Mol Biol. 1971 Nov 28;62(1):179–188. doi: 10.1016/0022-2836(71)90138-0. [DOI] [PubMed] [Google Scholar]
- Chrispeels M. J., Boyd R. F., Williams L. S., Neidhardt F. C. Modification of valyl tRNA synthetase by bacteriophage in Escherichia coli. J Mol Biol. 1968 Feb 14;31(3):463–475. doi: 10.1016/0022-2836(68)90421-x. [DOI] [PubMed] [Google Scholar]
- EIDLIC L., NEIDHARDT F. C. PROTEIN AND NUCLEIC ACID SYNTHESIS IN TWO MUTANTS OF ESCHERICHIA COLI WITH TEMPERATURE-SENSITIVE AMINOACYL RIBONUCLEIC ACID SYNTHETASES. J Bacteriol. 1965 Mar;89:706–711. doi: 10.1128/jb.89.3.706-711.1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- EIDLIC L., NEIDHARDT F. C. ROLE OF VALYL-SRNA SYNTHETASE IN ENZYME REPRESSION. Proc Natl Acad Sci U S A. 1965 Mar;53:539–543. doi: 10.1073/pnas.53.3.539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- FANGMAN W. L., NEIDHARDT F. C. DEMONSTRATION OF AN ALTERED AMINOACYL RIBONUCLEIC ACID SYNTHETASE IN A MUTANT OF ESCHERICHIA COLI. J Biol Chem. 1964 Jun;239:1839–1843. [PubMed] [Google Scholar]
- FRAENKEL D. G., NEIDHARDT F. C. Use of chloramphenicol to study control of RNA synthesis in bacteria. Biochim Biophys Acta. 1961 Oct 14;53:96–110. doi: 10.1016/0006-3002(61)90797-1. [DOI] [PubMed] [Google Scholar]
- Faanes R., Rogers P. Repression of enzymes of arginine biosynthesis by L-canavanine in arginyl-transfer ribonucleic acid synthetase mutants of Escherichia coli. J Bacteriol. 1972 Oct;112(1):102–113. doi: 10.1128/jb.112.1.102-113.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freundlich M. Valyl-Transfer RNA: Role in Repression of the Isoleucine-Valine Enzymes in Escherichia coli. Science. 1967 Aug 18;157(3790):823–825. doi: 10.1126/science.157.3790.823-a. [DOI] [PubMed] [Google Scholar]
- Hirshfield I. N., Horn P. C., Hopwood D. A., Maas W. K., DeDeken R. Studies on the mechanism of repression of arginine biosynthesis in Escherichia coli. 3. Repression of enzymes of arginine biosynthesis in arginyl-tRNA synthetase mutants. J Mol Biol. 1968 Jul 14;35(1):83–93. doi: 10.1016/s0022-2836(68)80038-5. [DOI] [PubMed] [Google Scholar]
- Jacoby G. A., Gorini L. A unitary account of the repression mechanism of arginine biosynthesis in Escherichia coli. I. The genetic evidence. J Mol Biol. 1969 Jan 14;39(1):73–87. doi: 10.1016/0022-2836(69)90334-9. [DOI] [PubMed] [Google Scholar]
- Karlström O., Gorini L. A unitary account of the repression mechanism of arginine biosynthesis in Escherichia coli. II. Application to the physiological evidence. J Mol Biol. 1969 Jan 14;39(1):89–94. doi: 10.1016/0022-2836(69)90335-0. [DOI] [PubMed] [Google Scholar]
- LENNOX E. S. Transduction of linked genetic characters of the host by bacteriophage P1. Virology. 1955 Jul;1(2):190–206. doi: 10.1016/0042-6822(55)90016-7. [DOI] [PubMed] [Google Scholar]
- LOVELESS A., HOWARTH S. Mutation of bacteria at high levels of survival by ethyl methane sulphonate. Nature. 1959 Dec 5;184:1780–1782. doi: 10.1038/1841780a0. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Mosteller R. D., Yanofsky C. Evidence that tryptophanyl transfer ribonucleic acid is not the corepressor of the tryptophan operon of Escherichia coli. J Bacteriol. 1971 Jan;105(1):268–275. doi: 10.1128/jb.105.1.268-275.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nazario M. The accumulation of argininosuccinate in Neurospora crassa. II. Inhibition of arginyl-tRNA synthesis by argininosuccinate. Biochim Biophys Acta. 1967 Aug 22;145(1):146–152. doi: 10.1016/0005-2787(67)90663-6. [DOI] [PubMed] [Google Scholar]
- Roth J. R., Antón D. N., Hartman P. E. Histidine regulatory mutants in Salmonella typhimurium. I. Isolation and general properties. J Mol Biol. 1966 Dec 28;22(2):305–323. doi: 10.1016/0022-2836(66)90134-3. [DOI] [PubMed] [Google Scholar]
- Rouget P., Chapeville F. Leucyl-tRNA synthetase. Two forms of the enzyme: relation between structural and catalytic properties. Eur J Biochem. 1971 Dec 10;23(3):459–467. doi: 10.1111/j.1432-1033.1971.tb01641.x. [DOI] [PubMed] [Google Scholar]
- SCHLESINGER S., MAGASANIK B. EFFECT OF ALPHA-METHYLHISTIDINE ON THE CONTROL OF HISTIDINE SYNTHESIS. J Mol Biol. 1964 Sep;9:670–682. doi: 10.1016/s0022-2836(64)80174-1. [DOI] [PubMed] [Google Scholar]
- SCHWARTZ J. H., MAAS W. K. Analysis of the inhibition of growth produced by canavanine in Escherichia coli. J Bacteriol. 1960 Jun;79:794–799. doi: 10.1128/jb.79.6.794-799.1960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silbert D. F., Fink G. R., Ames B. N. Histidine regulatory mutants in Salmonella typhimurium 3. A class of regulatory mutants deficient in tRNA for histidine. J Mol Biol. 1966 Dec 28;22(2):335–347. doi: 10.1016/0022-2836(66)90136-7. [DOI] [PubMed] [Google Scholar]
- Szentirmai A., Szentirmai M., Umbarger H. E. Isoleucine and valine metabolism of Escherichia coli. XV. Biochemical properties of mutants resistant to thiaisoleucine. J Bacteriol. 1968 May;95(5):1672–1679. doi: 10.1128/jb.95.5.1672-1679.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor A. L. Current linkage map of Escherichia coli. Bacteriol Rev. 1970 Jun;34(2):155–175. doi: 10.1128/br.34.2.155-175.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- VON EHRENSTEIN G., LIPMANN F. Experiments on hemoglobin biosynthesis. Proc Natl Acad Sci U S A. 1961 Jul 15;47:941–950. doi: 10.1073/pnas.47.7.941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams L. S., Neidhardt F. C. Synthesis and inactivation of aminoacyl-transfer RNA synthetases during growth of Escherichia coli. J Mol Biol. 1969 Aug 14;43(3):529–550. doi: 10.1016/0022-2836(69)90357-x. [DOI] [PubMed] [Google Scholar]
- Yem D. W., Williams L. S. Inhibition of arginyl-transfer ribonucleic acid synthetase activity of Escherichia coli by arginine biosynthetic precursors. J Bacteriol. 1971 Aug;107(2):589–591. doi: 10.1128/jb.107.2.589-591.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]


