Abstract
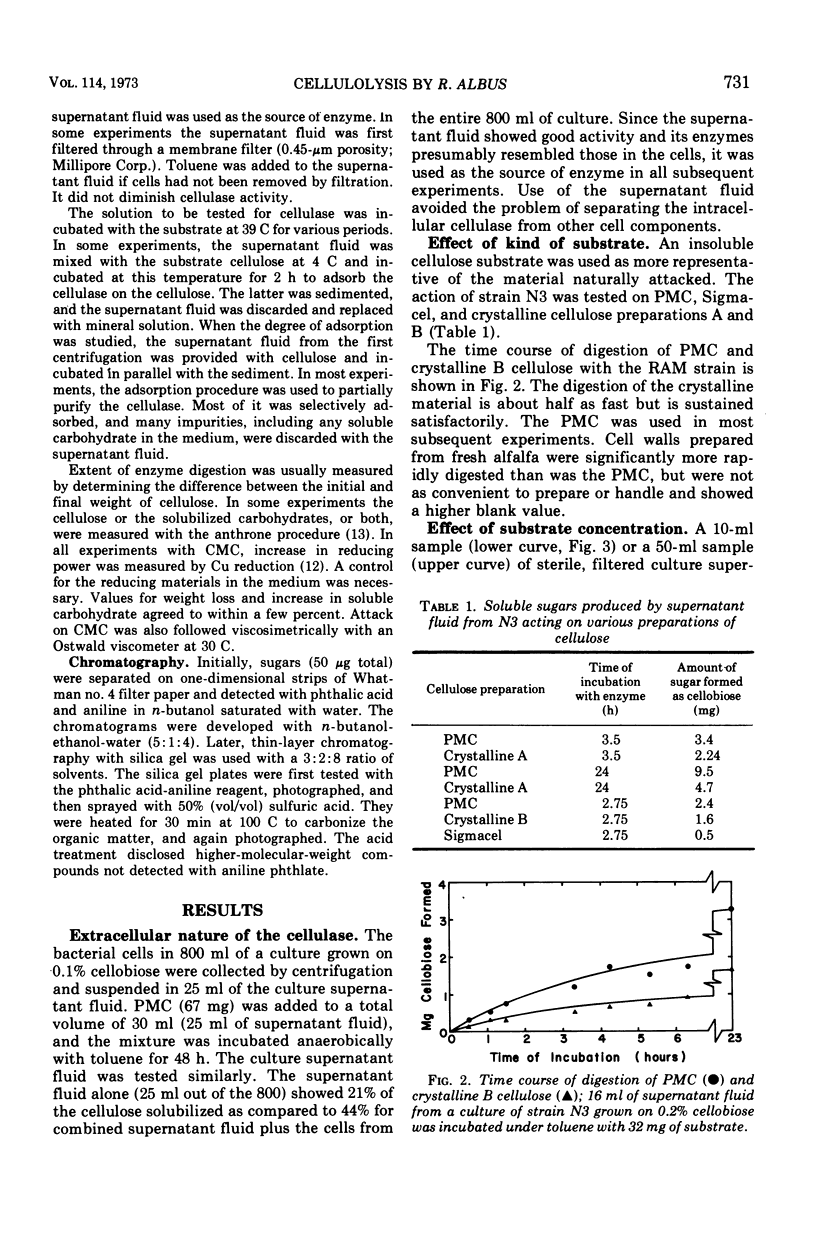
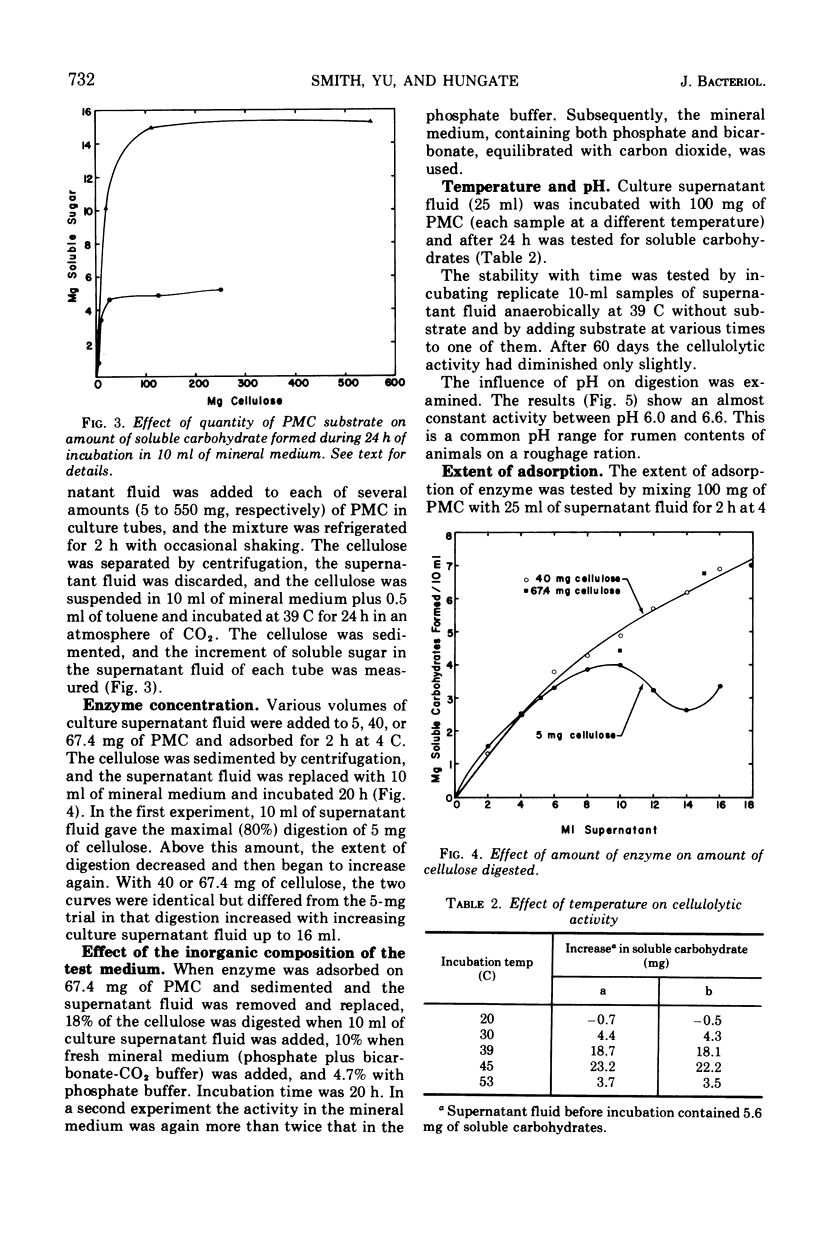
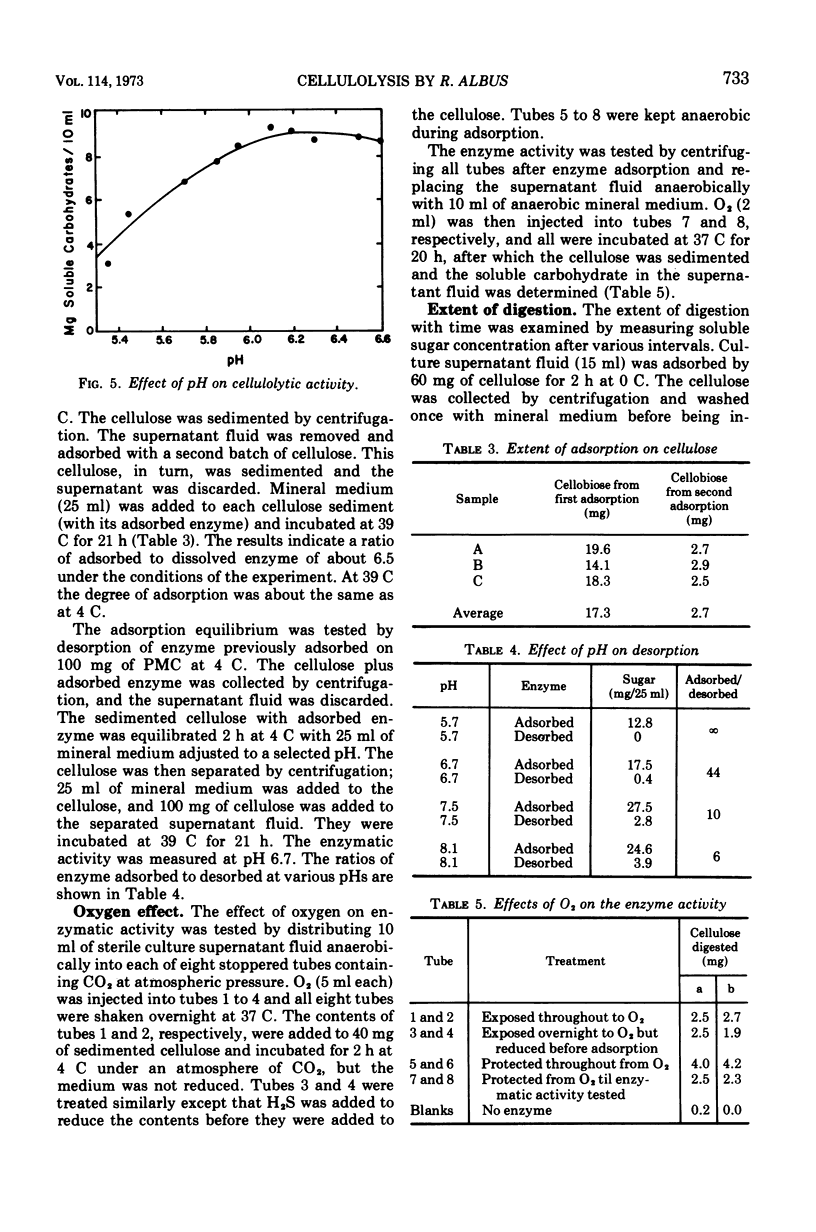
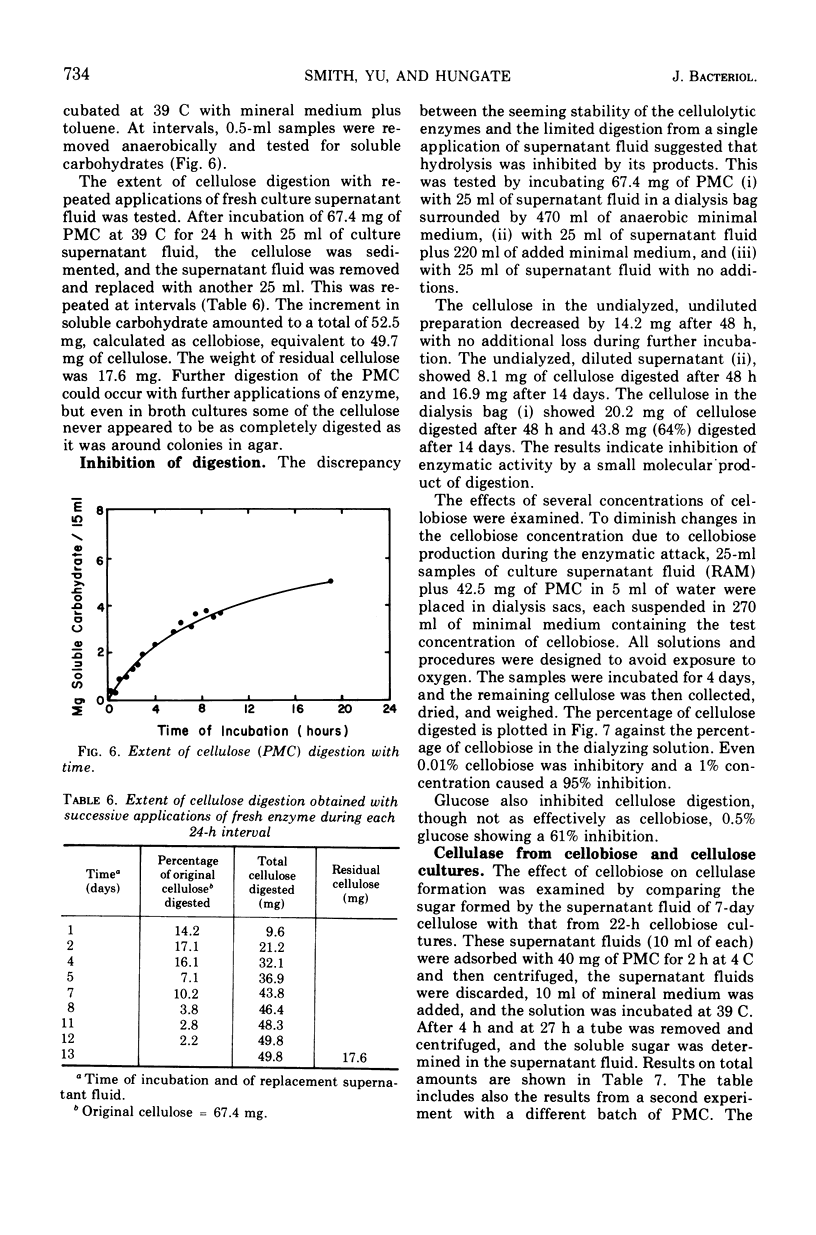
The factors influencing the digestion of pebble-milled cellulose by enzymes were studied by using several strains of Ruminococcus albus including a mutant characterized by a more eccentric location of its colony in the clearing produced by digestion of the cellulose in the thin layer lining the wall of a culture tube. Most of the cellulase is extracellular. As much as 65% of the cellulose could be digested by the cell-free enzymes provided the quantity of cellulose was small. Fresh enzyme was repeatedly administered or the digestion experiment was arranged in a dialysis bag through which digestion products could diffuse. Cellobiose and, to a lesser extent, glucose inhibited digestion. Pebble-milled filter paper, moist crystalline cellulose from cotton, and dry crystalline cellulose (Sigmacel) were digested, but in decreasing rapidity, respectively. Carboxymethylcellulose was digested more rapidly than pebble-milled cellulose but to approximately the same final extent as judged by Cu reduction values. Cell walls from alfalfa were digested. The enzyme preparation was active over the pH range 6.0 to 6.8 and showed most rapid cellulose digestion at 45 C. Part of the cellulolytic activity was irreversibly destroyed by exposure to oxygen. Much of the enzyme was absorbed on cellulose. The absorption and desorption characteristics, as well as the partial inhibition by oxygen, indicate that multiple enzymes are involved.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Amein M., Leatherwood J. M. Mechanism of cellobiose epimerase. Biochem Biophys Res Commun. 1969 Jul 23;36(2):223–227. doi: 10.1016/0006-291x(69)90318-0. [DOI] [PubMed] [Google Scholar]
- Fusee M. C., Leatherwood J. M. Regulation of cellulase from Ruminococcus. Can J Microbiol. 1972 Mar;18(3):347–353. doi: 10.1139/m72-053. [DOI] [PubMed] [Google Scholar]
- HAMMERSTROM R. A., CLAUS K. D., COGHLAN J. W., McBEE R. H. The constitutive nature of bacterial cellulases. Arch Biochem Biophys. 1955 May;56(1):123–129. doi: 10.1016/0003-9861(55)90341-6. [DOI] [PubMed] [Google Scholar]
- HUNGATE R. E. Microorganisms in the rumen of cattle fed a constant ration. Can J Microbiol. 1957 Mar;3(2):289–311. doi: 10.1139/m57-034. [DOI] [PubMed] [Google Scholar]
- HUNGATE R. E. POLYSACCHARIDE STORAGE AND GROWTH EFFICIENCY IN RUMINOCOCCUS ALBUS. J Bacteriol. 1963 Oct;86:848–854. doi: 10.1128/jb.86.4.848-854.1963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leatherwood J. M. Cellulase from Ruminococcus albus and mixed rumen microorganisms. Appl Microbiol. 1965 Sep;13(5):771–775. doi: 10.1128/am.13.5.771-775.1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- TREVELYAN W. E., HARRISON J. S. Studies on yeast metabolism. I. Fractionation and microdetermination of cell carbohydrates. Biochem J. 1952 Jan;50(3):298–303. doi: 10.1042/bj0500298. [DOI] [PMC free article] [PubMed] [Google Scholar]





