Abstract
Background and objectives: Low socioeconomic status (SES) is associated with both development and progression of chronic kidney disease (CKD). The impact of SES on severity of CKD at presentation to a renal service is less well known. This study investigated the relationship between SES and severity of CKD in a retrospective, cross-sectional analysis involving 1657 patients at the Sheffield Kidney Institute (Sheffield, UK).
Design, setting, participants, & measurements: SES was assigned to each patient according to electoral ward of residence by postcode and ranked according to the corresponding British Index of Multiple Deprivation score, which comprises five deprivation quintiles (Q1, least deprived; Q5, most deprived). National Kidney Foundation Kidney Disease Outcomes Quality Initiative classification of CKD was used for stratification and analysis. Binary logistic regression analysis was applied for the association of variables/risk factors with CKD (lower GFR) at presentation.
Results: The age-adjusted prevalence of diagnosed CKD at presentation by area of residence, across the five deprivation quintiles, per million population was Q1 = 1495, Q2 = 3530, Q3 = 3398, Q4 = 3989, and Q5 = 19,599. Logistic regression models showed that living in the lowest SES quintile area (Q5) as compared with the highest SES (Q1) was associated with a greater risk for presenting with a lower estimated GFR, after adjustment for sociodemographic, lifestyle, and clinical variables.
Conclusions: Low SES is related to severity of CKD at presentation. Further studies are needed to examine this issue across the various SES categories in the United Kingdom.
Chronic kidney disease (CKD) is now a widely recognized public health issue of major importance (1). The progressive nature of CKD, the ensuing ESRD, and associated cardiovascular morbidity and mortality are putting a considerable burden on global health care resources (1). Socioeconomic differentials in health are widely recognized; individuals of lower socioeconomic status (SES) have a higher risk for mortality and morbidity compared with those of higher SES (2,3). Low SES is associated with increased risk for CKD from studies mostly based in the US population (4–10) and a few in Europe (11,12). The association of CKD with SES in the various studies could be related to a higher number of socioeconomically disadvantaged individuals living in an area or the effect of the characteristics of the area (13).
A number of reasons warrant this study despite extensive literature on the subject. First, there is still a paucity of data on the relationship between area of residence SES and severity of established CKD at presentation to a renal service in the United Kingdom. Second, the previous works on this topic concentrated mainly on patients with ESRD. Third, differences across regions and countries on the population mix, access and quality of health care, and even lifestyle may have a different impact on the relationship between SES and CKD. The underlying hypothesis in this study, therefore, was whether characteristics of an area of residence, in terms of measures of SES, have an impact on level of kidney function of patients with CKD at presentation. We therefore studied the association between area-level SES and severity of established CKD at presentation to a renal service in the United Kingdom and also examined whether any association is independent of established risk factors for CKD.
Materials and Methods
Patients, Design, and Setting
This was a retrospective, cross-sectional review of 1657 patients who had CKD and were referred to our renal service (Sheffield Kidney Institute [SKI], Sheffield Teaching Hospital NHS Foundation Trust, Sheffield, UK) from January 1, 1995, through December 31, 2005.
Inclusion Criteria
Inclusion criteria were as follows: CKD as defined by estimated GFR (eGFR) <60 ml/min per 1.73 m2 for at least 6 mo duration and not on any form of renal replacement therapy (RRT). The sociodemographic, clinical, and biochemical parameters of each patient were retrieved from the SKI computer database of clinical and laboratory data (and case notes). Patients who presented with acute kidney injury (AKI) and those who were on any form of RRT were excluded; also excluded were those with missing postcodes or those whose postcodes were not valid. The SKI is the only tertiary referral center that provides comprehensive renal services to the North Trent region in England, which has a population base of approximately 1.6 million.
Sociodemographic Parameters
The following demographic parameters were collated and analyzed for each patient: Age, gender, and ethnicity (patients were broadly classified as Caucasians and non-Caucasians). All those categorized as white-British were grouped as Caucasians, and all those defined otherwise (including all ethnic minorities) were termed non-Caucasians. Patients’ postcodes were used in evaluating SES data.
Measures of SES
The UK Index of Multiple Deprivation (IMD) Score 2000 was used to measure residential area deprivation, as an indicator of SES in this study (14). This measure of social deprivation (published by the UK government) provides a composite social deprivation score/scale for each electoral ward in England (14). The IMD combines a number of indicators, chosen to cover a range of economic, social, and housing issues, into a single deprivation score for each small area (ward) in England. Separate indices at ward level are provided for each of the six domains of deprivation: Income, employment, health deprivation and disability, education skills and training, barriers to housing, and services. This allows all wards to be ranked according to how deprived they are relative to each other. This information is then brought together into one overall IMD score, which was initially done in 2000. The indices are used widely to analyze patterns of socioeconomic deprivation and relationships with several health outcomes to identify areas that would benefit from special initiatives or programs of action for health and development (15–20).
From their postcodes, CKD prevalence data at an individual level were georeferenced to an electoral ward of residence. Sixty-four cases with missing postcode data were excluded in the analysis.
Patients were ranked according to their corresponding IMD score by their postcode and corresponding ward of residence, and the ranked data were categorized into quintiles, with quintile 1 (Q1) being the least deprived and quintile 5 (Q5) being the most deprived. Patients were aggregated to their geographic area (electoral ward of residence), and prevalence of CKD was calculated using the total adult population in each ward derived from the UK 2001 National Census (21) as the denominator and then categorized into the corresponding IMD quintiles. The prevalence of CKD per ward was then aggregated according to the corresponding IMD quintiles, with adjustment for age by a direct standardization.
A second social deprivation score directly derived from the 2001 census, the UV67 table of household characteristics (21), was also used as an alternative analysis. The UV67 has several dimensions for socioeconomic deprivation. A household can be “deprived” in any of the following dimensions:
Education: No member of the household aged 16 to pensionable age has at least five General Certificate of Secondary Education (grade A through C) or equivalent, and no member of the household aged 16 to 18 is in full-time education.
Health and disability: Any member of the household has general health “not good” in the year before census or has a limiting long-term illness.
Housing: The household's accommodation is either overcrowded (occupancy indicator is −1 or less) or is in a shared dwelling or does not have sole use of bath/shower and toilet or has no central heating.
Clearly, in this study, we did not obtain individual patients’ SES indices or scores but instead relied on the IMD and the UV67 deprivation scores for the area of residence derived from their individual postcodes.
Clinical and Laboratory Parameters
Participants were classified as having hypertension or not on the basis of treatment for hypertension or the values of BP at presentation using the Seventh Joint National Committee for Definition and Classification of Hypertension (JNC 7) (22). Diabetes status was determined on the basis of a recorded history of diabetes or the current use of diabetic medication. Smoking status was categorized as never smoked, former smoker, or current cigarette smoker. Body mass index (BMI) was calculated as weight/height2 (kg/m2).
Biochemical measurements of serum creatinine (SCr) and total cholesterol were obtained using standard methods, at the Chemical Pathology Laboratory of Sheffield Teaching Hospitals NHS Trust using a Beckmann XL autoanalyzer. The coefficient of variation of SCr and total cholesterol at the time of data retrieval for this analysis was 2 to 3 and 3% respectively.
Measures and Definitions
eGFR was used in defining CKD in this study and was estimated from the Modification of Diet in Renal Disease (MDRD) study equation as follows (23):
eGFR (ml/min per 1.73 m2) = 186.3 × SCr−1.154 × age−0.203 × (0.742 for women) × 1.21 (if black)
CKD was defined primarily by the presence of persistent eGFR <60 ml/min per 1.73 m2 for at least 6 mo. This was classified according to the National Kidney Foundation Kidney Disease Outcomes Quality Initiative classification of CKD (NKF-K/DOQI guidelines) (24) as follows: eGFR (ml/min per 1.73 m2) 30 to 59 for stage 3, 15 to 29 for stage 4, and <15 for stage 5. Cases for which it was not possible to ascertain whether decreased kidney function was acute or chronic because of one or two test results of eGFR during the follow-up period were excluded from the study to ensure chronicity. eGFR <60 ml/min per 1.73 m2 was further dichotomized into severe CKD (<30 ml/min per 1.73 m2) and moderate CKD (≥30 ml/min per 1.73 m2) for modeling analysis (25).
Statistical Analyses
Calculations and analyses were performed using the statistical package SPSS 14.0 (SPSS, Chicago, IL). The level of significance was determined as P < 0.05. Continuous data are reported as mean with SD. In case of a skewed distribution, the median and interquartile range are presented. Prevalence is presented as percentages. The prevalence of CKD was adjusted for age by direct standardization. Differences between groups were tested by one-way ANOVA or Kruskal-Wallis test in case of a skewed distribution of a variable. Graded categorical associations were tested with a χ2 test for trend.
To study the impact of low SES (area deprivation) on CKD at presentation, we used a binomial logistic regression analysis, with a binary outcome: Severe CKD (eGFR <30 ml/min per 1.73 m2) and moderate CKD (eGFR ≥30 ml/min per 1.73 m2). The SES was the primary independent variable and was entered categorically according to the IMD quintiles, in the prediction of lower eGFR (severe CKD) at presentation to the renal service. The group in the least deprived IMD quintile (Q1) was used as the reference category.
The following available variables that might explain the association between low SES and CKD in these patients were included in the model, with area deprivation (IMD quintiles) as the primary independent variable, in the prediction of lower eGFR (severe CKD) at presentation: Age, gender (male versus female), ethnicity (Caucasians versus non-Caucasians), BMI, serum cholesterol, smoking (yes/no), diabetes status, (yes/no) and hypertension status (yes/no).
The alternative social deprivation score, the UV67 table of household characteristics, was also used in the analysis as a primary independent variable. We also tested for differences in effects of SES on the outcome by gender, diabetes status (yes/no), and hypertension status (yes/no).
As a further sensitivity analysis, we used logistic models to estimate relative risks (RR) and their 95% confidence intervals (CI) for severe CKD at presentation, adjusted for age and gender where appropriate, across the following strata of the study population: Individuals without diabetes and hypertension, individuals with diabetes, and individuals with hypertension and also across gender and ethnicity categories. ArcGIS 9.1 software (ESRI) was used in the geospatial analysis and mapping of the CKD prevalence data.
Results
General Characteristics
The participants were predominantly male (57.7%) and white (93.1%), with a mean (±SD) age of 58.2 ± 17.1 yr. The overall mean BMI was 23.3 ± 4.4 kg/m2, and mean eGFR was 35.6 ± 23.3 ml/min per 1.73 m2. The overall IMD score was 39.9 (4.7 to 73.4), and the total score for each IMD quintile is shown in Table 1. The characteristics of the study population across the five IMD quintiles are also shown in Table 1. No significant differences in trend for age, gender, ethnicity, smoking, and BMI were found; however, there were significantly more patients with diabetes and hypertension among those within the fifth IMD quintile (most deprived) compared with the lower quintiles (Table 1). There was a decreasing trend in baseline kidney function (eGFR) from the first (least deprived) to the fifth (most deprived) quintiles (Table 1).
Table 1.
Baseline characteristics of the study population across the social deprivation quintilesa
| Characteristic | Q1(n = 338) | Q2(n = 318) | Q3(n = 322) | Q4(n = 317) | Q5(n = 362) | Pb |
|---|---|---|---|---|---|---|
| IMD score (median [range]) | 7.4 (4.7 to 16.2) | 22.2 (18.1 to 24.2) | 31.9 (28.6 to 37.3) | 49.0 (41.0 to 55.6) | 70.8 (66.9 to 73.4) | |
| Age (yr; mean [SD]) | 58.0 ± 16.8 | 58.7 ± 19.2 | 59.0 ± 18.0 | 58.8 ± 17.1 | 58.7 ± 17.4 | 0.510 |
| Male (n [%]) | 200 (59.2) | 175 (55.0) | 147 (45.7) | 143 (45.1) | 192 (53.0) | 0.450 |
| Caucasian (n [%]) | 310 (91.7) | 293 (92.1) | 300 (93.2) | 302 (95.3) | 338 (93.4) | 0.540 |
| Smoking (n [%]) | 116 (34.3) | 131 (41.2) | 112 (34.8) | 117 (36.9) | 135 (37.3) | 0.430 |
| Hypertension (yes; n [%]) | 166 (49.1) | 171 (53.8) | 181 (56.2) | 201 (63.4) | 247 (68.2) | 0.013 |
| Diabetes (yes; n [%]) | 57 (16.9) | 44 (13.8) | 60 (18.6) | 48 (15.1) | 73 (20.2) | 0.046 |
| BMI (kg/m2; mean [SD]) | 23.4 ± 4.5 | 23.1 ± 3.9 | 23.4 ± 4.4 | 22.8 ± 4.7 | 23.4 ± 4.8 | 0.420 |
| Serum cholesterol (mmol/L; mean [SD]) | 5.4 ± 1.7 | 5.4 ± 1.6 | 5.5 ± 1.7 | 5.5 ± 1.9 | 5.6 ± 1.8 | 0.048 |
| eGFR(ml/min/1.73m2; mean [SD]) | 41.8 ± 30.6 | 33.7 ± 27.0 | 35.0 ± 29.5 | 22.4 ± 20.6 | 20.0 ± 18.9 | 0.010 |
| Age-adjusted prevalence rates of CKD pmp (95% CI) | 1495 (1132 to 1667) | 3530 (2493 to 4887) | 3398 (2162 to 4785) | 3989 (2532 to 5667) | 19,599 (7119 to 25,062) | 0.014 |
BMI, body mass index; CKD, chronic kidney disease; CI, confidence interval; eGFR, estimated GFR; IMD, Index of Multiple Deprivation; Q, social deprivation quintile (Q1 = least deprived, Q5 = most deprived); pmp, per million population.
For trend across quintiles (Q1 through Q5).
Distribution of CKD and Socioeconomic Deprivation Indexes
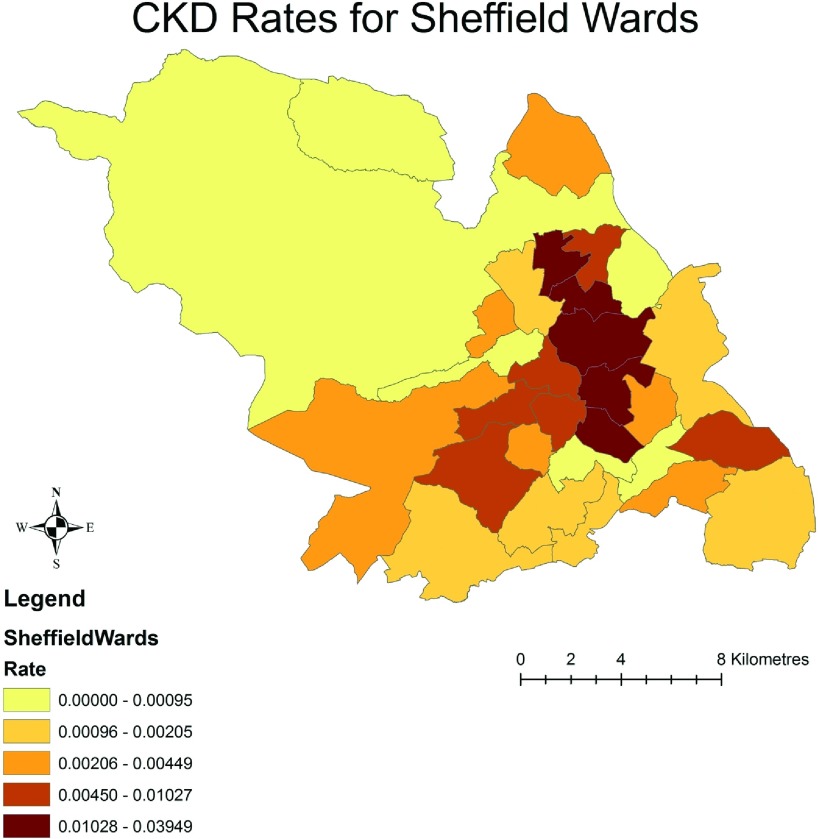
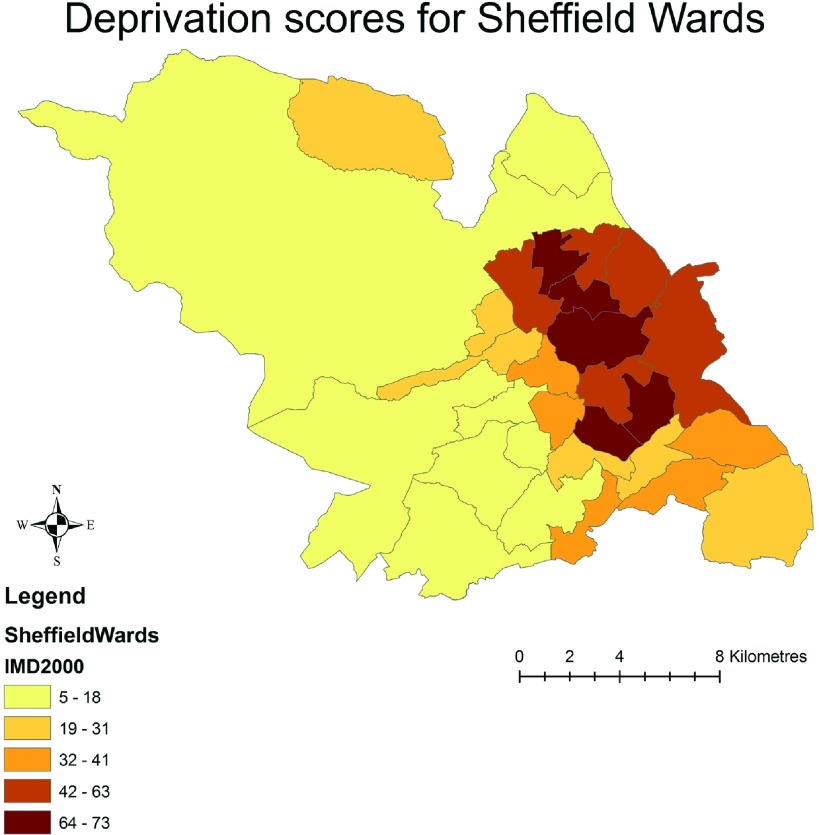
The crude distribution of prevalent CKD for the Sheffield wards stratified by the IMD quintiles is depicted in Figure 1. The darker shaded areas indicate the higher prevalence rate of CKD. The figure illustrates a clear pattern of higher prevalence of CKD within the socioeconomically deprived areas of residence (wards), in comparison with the least deprived areas (Figure 1). Figure 2 illustrates the IMD-derived deprivation scores for the same geographic units of the city. Again, the darker shading indicates the higher socioeconomic deprivation, and a visual inspection reveals that the areas of high deprivation map onto the areas of high CKD prevalence rates as shown in Figure 1.
Figure 1.
Crude distribution of prevalent chronic kidney disease (CKD) for the various wards of Sheffield. The darker shaded areas indicate higher rates of CKD.
Figure 2.
Index of Multiple Deprivation (IMD) scores for the same wards of the city. The darker shading indicates higher socioeconomic deprivation. A visual inspection reveals that the patterns are very similar to the map for CKD rates shown in Figure 1.
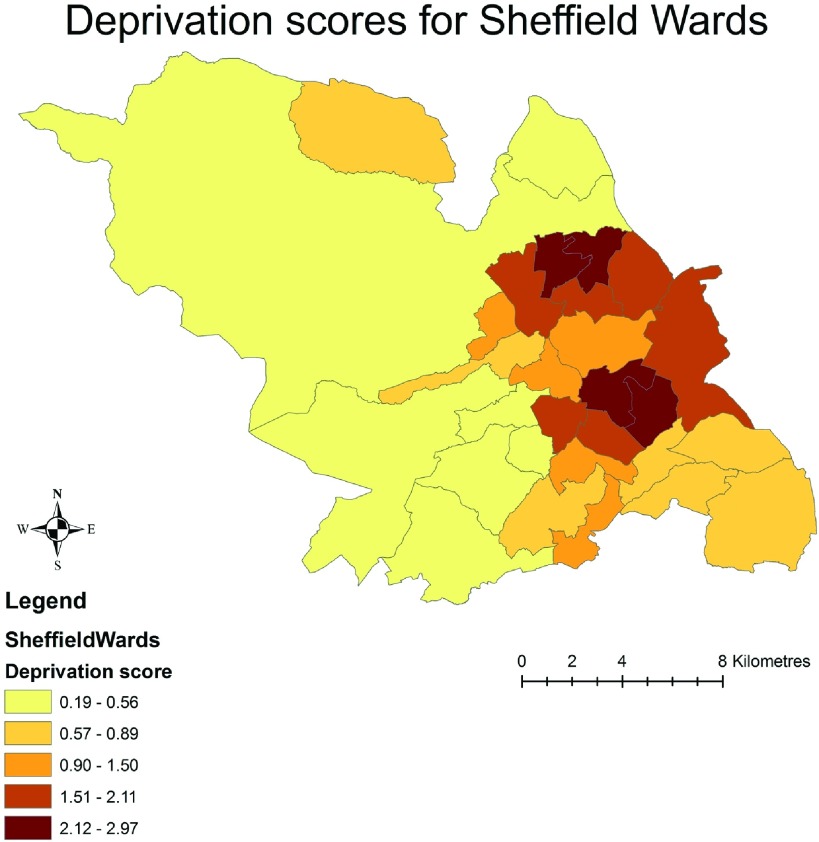
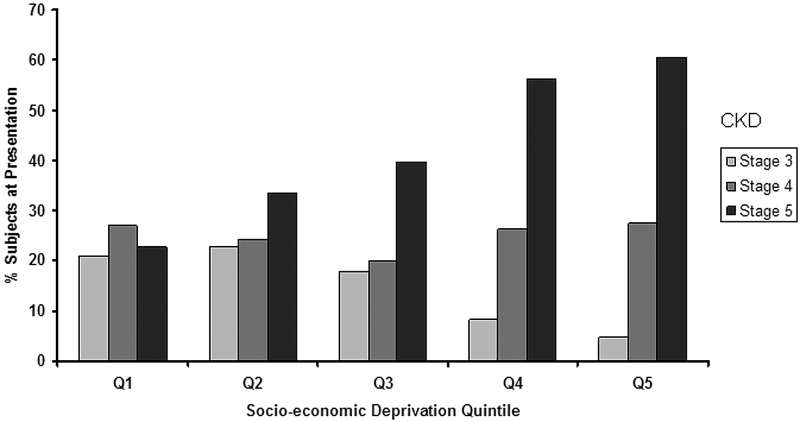
As a sensitivity analysis, an alternative socioeconomic deprivation score, directly derived from the 2001 census, the UV67 table of household characteristics, was used. Using these data, Figure 3 confirmed a similar geographic pattern to the CKD rates represented in Figure 1. Figure 4 shows an increasing trend in the number of patients presenting with advanced CKD (stage 5) to the unit from the least deprived (Q1) to the most deprived category (Q5; P = 0.012 for trend).
Figure 3.
Alternate deprivation score (UV67) for the wards of Sheffield, with a similar spatial pattern for the CKD prevalence rates as shown in Figure 1.
Figure 4.
Distribution of patients (%) with various stages of prevalent CKD (stages 3 through 5) across the five IMD quintiles (Q1 through Q5). There is an increasing trend of advanced CKD at presentation (stage 5; ▪) from the least (Q1) to the highest (Q5) IMD quintile (P = 0.012 for trend).
Age-adjusted prevalence of CKD (pmp) by IMD quintiles is shown in Table 1. Patients within the highest IMD quintile (more deprived) also had the highest age-adjusted prevalence rate for CKD as compared with the patients in the lower quintiles (less deprived; P = 0.014 for trend).
Risk for CKD and Socioeconomic Deprivation Index
Tables 2 through 4 show the RR (95% CI) for severe CKD (lower eGFR) at presentation by IMD quintiles, shown across gender (male versus female), ethnicity (Caucasian/non-Caucasian), and diabetes and hypertension categories of the study population. In both genders, patients in the most deprived quintiles had a greater risk for CKD compared with the patients from less deprived categories (Table 2). Among Caucasian patients, there was a greater risk from the patients in the most deprived IMD quintiles (Table 3). In the non-Caucasian patients, those in the fifth IMD quintile carried the greatest risk (RR 9.33; 95% CI 1.03 to 84.20), a risk higher than the corresponding category in the Caucasian population (Table 3). Analysis by stratification into groups of patients with diabetes and hypertension, and without diabetes and hypertension showed an increased risk for severe CKD (lower eGFR) at presentation in the patients within the most deprived IMD quintiles (4 and 5), as compared with patients within the lower quintiles in each of these groups (Table 4).
Table 2.
Adjusted RR for Severe CKD (eGFR <30 ml/min per 1.73 m2) at presentation by gender
| IMD Quintile | RR (95% CI) | P |
|---|---|---|
| Male | ||
| 1 (least deprived) | 1 | |
| 2 | 1.08 (0.70 to 1.64) | 0.720 |
| 3 | 0.82 (0.53 to 1.26) | 0.350 |
| 4 | 2.29 (1.39 to 3.75) | 0.001 |
| 5 (most deprived) | 6.69 (3.70 to 12.04) | 0.001 |
| Female | ||
| 1 (least deprived) | 1 | |
| 2 | 2.23 (1.37 to 3.61) | 0.001 |
| 3 | 1.61 (1.02 to 2.54) | 0.037 |
| 4 | 5.99 (3.51 to 10.23) | 0.001 |
| 5 (most deprived) | 9.76 (5.34 to 17.82) | 0.001 |
Table 4.
Adjusted RR for severe CKD (eGFR <30 ml/min per 1.73 m2) at presentation by clinical status
| IMD Quintile | RR (95% CI) | P |
|---|---|---|
| Diabetic | ||
| 1 (least deprived) | 1.00 | |
| 2 | 2.26 (0.94 to 5.41) | 0.070 |
| 3 | 1.05 (0.50 to 2.22) | 0.880 |
| 4 | 4.30 (1.63 to 11.36) | 0.003 |
| 5 (most deprived) | 5.75 (2.32 to 14.29) | 0.001 |
| Hypertensive | ||
| 1 (least deprived) | 1.00 | |
| 2 | 2.12 (1.35 to 3.33) | 0.001 |
| 3 | 1.47 (0.95 to 2.26) | 0.080 |
| 4 | 3.92 (2.44 to 6.27) | 0.001 |
| 5 (most deprived) | 9.05 (5.30 to 15.45) | 0.001 |
| Nondiabetic and nonhypertensive | ||
| 1 (least deprived) | 1.00 | |
| 2 | 1.27 (0.90 to 1.78) | 0.160 |
| 3 | 1.10 (0.78 to 1.54) | 0.560 |
| 4 | 3.51 (2.35 to 5.23) | 0.001 |
| 5 (most deprived) | 7.83 (4.89 to 12.54) | 0.001 |
Table 3.
Adjusted RR for severe CKD (eGFR <30 ml/min per 1.73 m2) at presentation by ethnicity
| IMD Quintile | RR (95% CI) | P |
|---|---|---|
| Caucasian | ||
| 1 (least deprived) | 1.00 | |
| 2 | 1.50 (1.78 to 2.80) | 0.014 |
| 3 | 1.13 (0.82 to 1.56) | 0.420 |
| 4 | 3.56 (2.47 to 5.14) | 0.001 |
| 5 (most deprived) | 7.70 (5.07 to 11.90) | 0.001 |
| Non-Caucasian | ||
| 1 (least deprived) | 1.00 | |
| 2 | 1.21 (0.30 to 4.76) | 0.780 |
| 3 | 0.77 (0.20 to 3.01) | 0.710 |
| 4 | 7.50 (0.10 to 88.82) | 0.900 |
| 5 (most deprived) | 9.33 (1.03 to 84.20) | 0.047 |
Multivariate Regression Analysis
The results of the stepwise multivariate logistic regression modeling analysis are shown in Table 5. Univariately, increasing socioeconomic deprivation (across quintiles) was associated with increasing odds for severe CKD (eGFR <30 ml/min per 1.73 m2) at presentation (model 1). After adjustment for age, gender, and ethnicity, this association remained significant for the higher quintiles of social deprivation (third through fifth; model 2). The trend persisted after correction for other clinical and biochemical variables associated with the risk for severe CKD, only for the most deprived IMD quintile (fifth). The most deprived category (fifth quintile) as compared with the least deprived (first quintile), was associated with a greater risk for presenting with a lower eGFR (<30 ml/min per 1.73 m2; odds ratio 4.36; 95% CI 1.09 to 17.38), even when the influence of the various population's characteristics was taken into account in the various strata of the multivariate model (model 3). Other independent determinants in the final model were diabetes and hypertension (Table 5).
Table 5.
Results of multivariate logistic regression analysis, relating CKD (lower eGFR) with Area Deprivation as the primary independent variable under investigation
| Model | Variable | OR (95% CI) |
|---|---|---|
| 1 | IMD deprivation quintile | |
| 1 (least deprived) | 1.00 (reference) | |
| 2 | 1.43 (1.01 to 2.00)a | |
| 3 | 1.45 (1.30 to 1.67)a | |
| 4 | 3.00 (2.00 to 4.71)a | |
| 5 (most deprived) | 11.90 (8.50 to 16.70)a | |
| 2 | IMD deprivation quintile | |
| 1 (least deprived) | 1.00 (reference) | |
| 2 | 1.39 (0.98 to 1.95) | |
| 3 | 1.46 (1.30 to 1.68)a | |
| 4 | 3.16 (2.01 to 4.90)a | |
| 5 (most deprived) | 12.60 (8.90 to 17.80)a | |
| Age (per year) | 1.00 (0.99 to 1.01) | |
| Gender (male versus female) | 1.65 (1.50 to 1.84) | |
| Race (Caucasian/non-Caucasian) | 1.01 (0.99 to 1.03) | |
| 3 | IMD deprivation quintile | |
| 1 (least deprived) | 1.00 (reference) | |
| 2 | 0.51 (0.07 to 3.28) | |
| 3 | 0.23 (0.10 to 2.68) | |
| 4 | 0.99 (0.11 to 8.63) | |
| 5 (most deprived) | 4.36 (1.09 to 17.38)a | |
| Age (per year) | 1.04 (0.97 to 1.04) | |
| Gender (male versus female) | 3.27 (0.30 to 37.40) | |
| Ethnicity (Caucasian/non-Caucasian) | 0.69 (0.36 to 1.31) | |
| BMI (per kg/m2) | 1.02 (0.87 to 1.90) | |
| Serum cholesterol (mmol/L) | 0.82 (0.58 to 1.15) | |
| Smoking (yes/no) | 1.24 (0.37 to 4.18) | |
| Diabetes (yes/no) | 1.87 (1.19 to 4.00)a | |
| Hypertension (yes/no) | 1.75 (1.19 to 2.91)a |
P < 0.01.
Discussion
This study found a greater risk for severe CKD at presentation to a UK renal service in patients who lived in areas with low SES. There was a similar trend in this relationship between SES and severe CKD across various demographic and clinical categories of the population studied. Control for the risk factors associated with CKD did not cancel completely the observed relationship between low SES (Area deprivation) and severe CKD. A previous study in UK also found the incidence of diagnosed/referred CKD to be greater in the more deprived areas, with poorer prognosis and decreased survival (11). A similar association has also been shown in national surveys in the US, with the relationship being more prevalent among the African-American groups (4–10). The relatively small size of the non-Caucasian population in our study makes any comment on the impact of racial differences difficult to ascertain.
In our study, the association observed between SES and the greater risk for presenting with advanced CKD remained even after controlling for some of the classical CKD risk factors recognized in the literature (24–26), although we cannot rule out the influences by residual confounding of other, unmeasured risk factors for CKD in this study. Of note, SES on its own does not affect kidney function or link directly with onset of CKD, but the associated biologic exposures would explain fully or in part its relationship with CKD. There are several ways in which low SES (area deprivation) could influence CKD distribution in the community. These could be demographic (age, gender, and ethnic variation influences) clinical (hypertension, diabetes, and hyperlipidemia), behavioral (diet, obesity, smoking, alcohol, and recreational drug use), or the differences in health care delivery system (access, referral pattern, and awareness among primary health care providers) (27).
It might be that there is a higher preponderance of elderly living in the more deprived areas of Sheffield, and increasing age is an established risk factor for CKD; however, age does not fully explain the relationship seen in this study in our multivariate model, and there was no significant difference in the age distribution across the population in the various IMD quintiles. Similarly, there is a higher prevalence in deprived areas in the United Kingdom of chronic diseases such as hypertension and diabetes known to be leading causes of CKD (28–30). Although in our study, hypertension and diabetes accounted for some of the risk for presentation with advanced CKD, these factors did not fully explain the relationship between socioeconomic deprivation and a higher burden of CKD in the population studied.
Of the lifestyle factors, obesity is a public health concern globally and is shown to be highly prevalent in socioeconomically deprived areas of the population in the United Kingdom (30). Obesity is also known to cluster with several risk factors and markers suggested to be causally related with CKD and an independent predictor of CKD in the population (31). We therefore investigated whether BMI could influence the relationship between social deprivation and severe CKD at presentation, but the relationship remained after adjustment for BMI in the various logistic regression models. Areas of residence with low SES have also been shown to have higher proportions of smokers, also a risk factor for CKD (32); however, smoking behavior in this study did not explain the relationship seen between area-level deprivation and severe CKD at presentation. Overall, the classical CKD risk factors did not explain fully the relationship between area-level low SES and advanced CKD in this study.
Of our limitations, the IMD and the UV67 scores used as proxies for area-level SES in the United Kingdom cover aspects of income, housing, and access to services including health care but are not direct measures of the quality of health care delivery in the communities (14,21). Second, unmeasured environmental, social, behavioral, and genetic factors were beyond the scope of these SES scoring systems (24,25).
Further limitations include the absence of information on duration of residence; therefore, whether the association between area deprivation (low SES) and CKD is a reflection of environmental exposure time is unknown. On this note, changes in SES as a result of reverse causation over a lifetime cannot be ruled out completely. Moreover, we lack information about individual SES on the basis of occupational class, income, and level of education. Thus, the ecologic fallacy here must be noted, in that data at one geographic scale should be used at other scales with caution. The IMD score indicates that an area has a particular level of deprivation, but it is not necessarily the case that specific individuals living in that area are similarly deprived.
We also lack data on low birth weight and achieved educational level shown to be associated with area deprivation (27). Other limitations included the retrospective nature of the study and that the population studied might not be fully representative of the overall population. Moreover, given the cross-sectional nature of the analysis, causation cannot be established; however, that our findings are consistent with other published reports suggests that biases are less likely (4–12).
Similarly, the impact that an earlier diagnosis among the less deprived category of the population would lead to a selection bias is another potential limitation; however, there was a balanced representation of study subjects across all of the IMD quintiles. Moreover, detection bias, if any, would pick up more patients with high SES, thereby reducing the strength of the observed relationship. Of note, access to health care in the United Kingdom is free to all permanent residents, thus minimizing these potential biases.
What implications arise from these findings? There are growing concerns worldwide about the rising epidemic of ESRD and the high burden of CKD. Such concerns can be linked to the increasing costs of the various forms of RRT and adverse outcomes of CKD. These adverse outcomes vary from ESRD, increasing cardiovascular morbidity and mortality, and premature mortality. Such negative health outcomes can be prevented or at least delayed through early detection and prevention programs. The early detection and prevention of CKD require a good understanding of the burden and risk factors/markers that are associated with CKD in the community (1,24,25).
This study provides information on the relationship between area of residence deprivation (low SES) and severity of CKD at presentation to a renal service in the United Kingdom. Namely, that the prevalence of advanced stages of CKD at the time of presentation to renal service is higher from the areas with low SES, as defined by either the IMD or UV67 measure of area socioeconomic deprivation.
Conclusions
This study has demonstrated an association between low SES (area deprivation) and a lower eGFR at presentation to the renal service, independent of some of the classical CKD risk factors. The specific components of area of residence level of SES that influence kidney function and thus CKD are still unclear; however, the relationship suggests that environmental and socioeconomic factors need to be emphasized when developing modalities for CKD prevention in the community.
Disclosures
None.
Acknowledgments
A.K.B. was an International Society of Nephrology Fellow at the Sheffield Kidney Institute (University of Sheffield, Sheffield, UK) and acknowledges the support of the International Society of Nephrology in his research and clinical training. We also acknowledge the Sheffield Area Kidney Patient Association and the Sheffield Kidney Research Foundation for supporting Research in the Sheffield Kidney Institute.
Published online ahead of print. Publication date available at www.cjasn.org.
References
- 1.El Nahas AM, Bello AK: Chronic kidney disease: The global challenge. Lancet 365 :331 –340,2005 [DOI] [PubMed] [Google Scholar]
- 2.Acheson D: Independent Inquiry into Inequalities in Health Report, London, Stationery Office,1999
- 3.Adler NE, Ostrove JM: Socioeconomic status and health: What we know and what we don't. Ann N Y Acad Sci 896 :3 –15,1999 [DOI] [PubMed] [Google Scholar]
- 4.Byrne C, Nedelman J, Luke RG: Race, socioeconomic status, and the development of end-stage renal disease. Am J Kidney Dis 23 :16 –22,1994 [DOI] [PubMed] [Google Scholar]
- 5.Young EW, Mauger EA, Jiang KH, Port FK, Wolfe RA: Socioeconomic status and end-stage renal disease in the United States. Kidney Int 45 :907 –911,1994 [DOI] [PubMed] [Google Scholar]
- 6.Perneger TV, Whelton PK, Klag MJ: Race and end-stage renal disease: Socio-economic status and access to health care as mediating factors. Arch Intern Med 155 :1201 –1208,1995 [PubMed] [Google Scholar]
- 7.Klag MJ, Whelton PK, Randall BL, Neaton JD, Brancati FL, Stamler J: End-stage renal disease in African-American and white men, 16-year MRFIT findings. JAMA 277 :1293 –1298,1997 [PubMed] [Google Scholar]
- 8.Rostand SG: US minority groups and endstage renal disease: A disproportionate share. Am J Kidney Dis 14 :411 –413,1992 [DOI] [PubMed] [Google Scholar]
- 9.Krop JS, Coresh J, Chambless LE, Shahar E, Watson RL, Szklo M, Brancati FL: A community-based study of explanatory factors for the excess risk of early renal function decline in blacks vs whites with diabetes. Arch Intern Med 159 :1777 –1783,1999 [DOI] [PubMed] [Google Scholar]
- 10.Brancati FL, Whittle JC, Whelton PK, Seidler AJ, Klag MJ: The excess incidence of diabetic endstage renal disease among blacks: A population-based study of potential explanatory factors. JAMA 268 :3079 –3084,1992 [PubMed] [Google Scholar]
- 11.Drey N, Roderick P, Mullee M, Rogerson M: A population-based study of the incidence and outcomes of diagnosed chronic kidney disease. Am J Kidney Dis 42 :677 –684,2003 [DOI] [PubMed] [Google Scholar]
- 12.Fored CM, Ejerblad E, Fryzek JP, Lambe M, Lindblad P, Nyren O, Elinder CG: Socio-economic status and chronic renal failure: A population-based case-control study in Sweden. Nephrol Dial Transplant 18 :82 –88,2003 [DOI] [PubMed] [Google Scholar]
- 13.Macintyre S, Maciver S, Sooman A: Area, class and health: Should we be focusing on places or people? J Social Policy 22 :213 –234,1993 [Google Scholar]
- 14.DETR: Indices of Deprivation 2000, London, Department of the Environment, Transport and the Regions,2000. . Available at: http://www.urban.odpm.gov.uk/imd. Accessed March 26, 2006
- 15.Lang IA, Llewellyn DJ, Langa KM, Wallace RB, Huppert FA, Melzer D: Neighborhood deprivation, individual socioeconomic status, and cognitive function in older people: Analyses from the English Longitudinal Study of Ageing. J Am Geriatr Soc 56 :191 –198,2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ashworth M, Seed P, Armstrong D, Durbaba S, Jones R: The relationship between social deprivation and the quality of primary care: A national survey using indicators from the UK Quality and Outcomes Framework. Br J Gen Pract 57 :441 –448,2007 [PMC free article] [PubMed] [Google Scholar]
- 17.Horton TC, Dias JJ, Burke FD: Social deprivation and hand injury. J Hand Surg Eur 32 :256 –261,2007 [DOI] [PubMed] [Google Scholar]
- 18.Jordan H, Roderick P, Martin D: The Index of Multiple Deprivation 2000 and accessibility effects on health. J Epidemiol Community Health 58 :250 –257,2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Strong M, Maheswaran R, Radford J: Socioeconomic deprivation, coronary heart disease prevalence and quality of care: a practice-level analysis in Rotherham using data from the new UK general practitioner Quality and Outcomes Framework. J Public Health 28 :39 –42,2006 [DOI] [PubMed] [Google Scholar]
- 20.Maheswaran R, Elliott P, Strachan DP: Socioeconomic deprivation, ethnicity, and stroke mortality in Greater London and south east England. J Epidemiol Community Health 51 :127 –131,1997 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.UV67: Classification of Household Deprivation. Available at: http://www.statistics.gov.uk/census2001/pdfs/univariate_tables_final.pdf. Accessed March 10,2007
- 22.Chobanian AV, Bakris GL, Black HR, Cushman WC, Green LA, Izzo JL Jr, Jones DW, Materson BJ, Oparil S, Wright JT Jr, Roccella EJ: The Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure: The JNC 7 report. JAMA 289 :2560 –2572,2003 [DOI] [PubMed] [Google Scholar]
- 23.Levey AS, Bosch JP, Lewis JB, Greene T, Rogers N, Roth D: A more accurate method to estimate glomerular filtration rate from serum creatinine: A new prediction equation. Modification of Diet in Renal Disease Study Group. Ann Intern Med 130 :461 –470,1999 [DOI] [PubMed] [Google Scholar]
- 24.K/DOQI clinical practice guidelines for chronic kidney disease, evaluation classification and stratification. Kidney Disease Outcomes Quality Initiative. Am J Kidney Dis 39 [Suppl 1]:S1 –S246,2002 [PubMed] [Google Scholar]
- 25.Levey AS, Atkins R, Coresh J, Cohen EP, Collins AJ, Eckardt KU, Nahas ME, Jaber BL, Jadoul M, Levin A, Powe NR, Rossert J, Wheeler DC, Lameire N, Eknoyan G: Chronic kidney disease as a global public health problem: Approaches and initiatives—A position statement from Kidney Disease Improving Global Outcomes. Kidney Int 72 :247 –259,2007 [DOI] [PubMed] [Google Scholar]
- 26.Haroun MK, Jaar BG, Hoffman SC, Comstack GW, Klag MJ, Coresh J: Risk factors for chronic kidney disease: A prospective study of 23,534 men and women in Washington County, Maryland. J Am Soc Nephrol 14 :2934 –2941,2003 [DOI] [PubMed] [Google Scholar]
- 27.Shoham DA, Vupputuri S, Kshirsagar AV: Chronic kidney disease and life course socioeconomic status: A review. Adv Chronic Kidney Dis 12 :56 –63,2005 [DOI] [PubMed] [Google Scholar]
- 28.Colhoun HM, Hemingway H, Poulter NR: Socio-economic status and blood pressure: An overview analysis. J Hum Hypertens 12 :91 –110,1998 [DOI] [PubMed] [Google Scholar]
- 29.Connolly V, Unwin N, Sherriff P, Bilous R, Kelly W: Diabetes prevalence and socio-economic status: A population based study showing increased prevalence of type 2 diabetes mellitus in deprived areas. J Epidemiol Community Health 54 :173 –177,2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Evans JM, Newton RW, Ruta DA, MacDonald TM, Morris AD: Socio-economic status, obesity and prevalence of type 1 and type 2 diabetes mellitus. Diabet Med 17 :478 –480,2000 [PubMed] [Google Scholar]
- 31.Gelber RP, Kurth T, Kausz AT, Manson JE, Buring JE, Levey AS, Gaziano JM: Association between body mass index and CKD in apparently healthy men. Am J Kidney Dis 46 :871 –880,2005 [DOI] [PubMed] [Google Scholar]
- 32.Shohaimi S, Luben R, Wareham N, Day N, Bingham S, Welch A, Oakes S, Khaw KT: Residential area deprivation predicts smoking habit independently of individual educational level and occupational social class: A cross sectional study in the Norfolk cohort of the European Investigation into Cancer (EPIC-Norfolk). J Epidemiol Community Health 57 :270 –276,2003 [DOI] [PMC free article] [PubMed] [Google Scholar]