Abstract
The NUP98-HOXD13 (NHD13) fusion gene occurs in patients with myelodysplastic syndrome (MDS) and acute nonlymphocytic leukemia (ANLL). We reported that transgenic mice expressing NHD13 develop MDS, and that more than half of these mice eventually progress to acute leukemia. The latency period suggests a requirement for at least 1 complementary event before leukemic transformation. We conducted a candidate gene search for complementary events focused on genes that are frequently mutated in human myeloid leukemia. We investigated 22 ANLL samples and found a high frequency of Nras and Kras mutations, an absence of Npm1, p53, Runx1, Kit and Flt3 mutations, and a single Cbl mutation. Our findings support a working hypothesis that predicts that ANLL cases have one mutation which inhibits differentiation, and a complementary mutation which enhances proliferation or inhibit apoptosis. In addition, we provide the first evidence for spontaneous collaborating mutations in a genetically engineered mouse model of ANLL.
Introduction
We previously reported a transgenic mouse model for myelodysplastic syndrome (MDS), in which NUP98-HOXD13 (NHD13) mice develop MDS at an early age, and progress to acute leukemia between 4 and 14 months of age.1 This latency period is likely due to a requirement for additional genetic events before leukemic transformation. Many studies have investigated the nature of such secondary events through experimental induction of complementary events, such as retroviral insertional mutagenesis2–4 or ENU-induced mutagenesis.5,6 To our knowledge, no study has investigated the nature of complementary mutations that occur spontaneously. Therefore, we evaluated NHD13 ANLL samples for the presence of mutations commonly seen in patients with ANLL.
Methods
DNA and RNA isolation
All animal experiments were conducted with the approval of the NIH Intramural Animal Care and Use Committee. Peripheral blood complete blood counts were obtained, bone marrow was harvested for cytospins, and paraffin-embedded spleen and liver were stained with hematoxylin and eosin. Routine immunohistochemical stains included F4/80, CD3, B220, and myeloperoxidase (MPO), and ANLL diagnosis was based on the Bethesda proposals for hematopoietic neoplasms in mice.7,8 Effaced spleen tissue from NHD13 mice with acute leukemia was snap frozen on dry ice. DNA and RNA were prepared by standard techniques.
RT-PCR and PCR
Reverse transcription (RT) was performed using Superscript II (Invitrogen, Carlsbad, CA). Genomic- and RT–polymerase chain reaction (PCR) were performed using either Supermix (Invitrogen) or Taq DNA Polymerase (Invitrogen). Primers, thermal cycling profiles, and regions amplified are listed in Tables S1 and S2 (available on the Blood website; see the Supplemental Materials link at the top of the online article). PCR products were purified using Qiagen (Valencia, CA) protocols, and were directly sequenced (Retrogen, San Diego, CA). Sequence chromatograms were manually inspected to detect mutations (Figure S1).
Reference sequences (NCBI accession numbers9) used were as follows: c-Cbl: NM_007619.2; Flt3: NM_010229.2; Kit: NM_021099.2; Kras: NM_021284.4; Nras: NM_010937.2; Npm1: NM_002520.5; Runx1: NM_001111021.1; p53: NM_011640.1.
Transfection
Wild-type and mutant Nras cDNAs were generated by RT-PCR using the Expand Long Template PCR System (Roche, Basel, Switzerland) and primers described in Table S1. Products were cloned into an EF1a expression vector and plasmids containing either wild-type or mutant Nras were used for transfection. Cells were transfected with Lipofectamine 2000 (Invitrogen) and selected with 500 μg/mL G418 (Invitrogen) before withdrawal of IL-3.
Results and discussion
p53 is mutated in approximately 50% of all cancers.10 In ANLL patients however, p53 mutations are less common, occurring in approximately 10% of cases.11 We used an RT-PCR strategy to screen this gene in a cohort of 22 mice with ANLL, and obtained a PCR product from 18 mice. Sequencing of these products detected no mutations (Table 1).
Table 1.
Mutations in NHD13 acute leukemias
| Mouse | Strain | Age, mo | Diagnosis | Genes |
|||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| NpmI | p53 | Runx1 | Cbl | Flt3 | Kit | K-ras | N-Ras | ||||
| 63 | C57BL6 | 11 | AML with maturation | GL | GL | GL | GL | GL | GL | GL | GL |
| 251 | C57BL6 | 9 | AML with maturation | GL | GL | ND | GL | GL | GL | GL | codon 12 GGT>GAT (G>D) |
| 254 | C57BL6 | 11 | AML with maturation | GL | GL | GL | GL | GL | GL | GL | GL |
| 1135 | C57BL6 | 12 | AML with maturation | GL | GL | GL | GL | GL | GL | codon 12 GGT>GAT (G>D) | GL |
| 2434 | C57BL6 | 13 | AML with maturation | GL | GL | GL | GL | GL | GL | GL | codon 12 GGT>AGT (G>S) |
| 2437 | C57BL6 | 13 | AML with maturation | GL | ND | GL | GL | GL | GL | GL | GL |
| 2554 | C57BL6 | 12 | AML with maturation | GL | ND | GL | GL | GL | GL | GL | GL |
| 1079 | C57BL6 | 10 | AML with maturation | GL | GL | GL | GL | GL | GL | GL | GL |
| 1080 | C57BL6 | 10 | AML with maturation | GL | ND | GL | 297 bp deletion | GL | GL | GL | GL |
| 1134 | C57BL6 | 10 | AML without maturation | GL | GL | GL | GL | GL | GL | GL | GL |
| 1155 | C57BL6 | 10 | AML without maturation | GL | GL | GL | GL | GL | GL | GL | GL |
| 2413 | C57BL6 | 8 | AML without maturation | GL | GL | GL | GL | GL | GL | GL | GL |
| 2417 | C57BL6 | 9 | AML without maturation | GL | GL | GL | GL | GL | GL | codon 12 GGT>GAT (G>D) | GL |
| 2420 | C57BL6 | 14 | AML without maturation | GL | GL | GL | GL | GL | GL | GL | GL |
| 1892 | C57BL6 | 11 | MPD-like leukemia | GL | GL | GL | GL | GL | GL | GL | codon 12 GGT>GAT (G>D) |
| 2590 | C57BL6 | 5 | MPD-like leukemia | GL | ND | GL | GL | GL | GL | GL | GL |
| 2679 | C57BL6 | 6 | MPD-like leukemia | GL | GL | GL | GL | GL | GL | GL | GL |
| 1017 | FVB | 9 | AMKL | GL | GL | GL | GL | GL | GL | codon 12 GGT>GTT (G>V) | GL |
| 1668 | FVB | 10 | AML with maturation | GL | GL | GL | GL | GL | GL | GL | GL |
| 4875 | FVB | 15 | AML with maturation | GL | GL | GL | GL | GL | GL | GL | GL |
| 1019 | FVB | 14 | AML with maturation | GL | GL | GL | GL | GL | GL | codon 12 GGT>GTT (G>V) | GL |
| 1018 | FVB | 14 | Erythroid leukemia (spleen) | GL | GL | GL | GL | GL | GL | GL | GL |
| 1138 | C57BL6 | 8 | Biphenotypic pre-T LBL/AML | ND | ND | ND | ND | ND | ND | GL | GL |
| 2423 | C57BL6 | 8 | Biphenotypic pre-T LBL/AML | ND | ND | ND | ND | ND | ND | GL | GL |
| 2553 | C57BL6 | 10 | pre-T LBL | ND | ND | ND | ND | ND | ND | GL | GL |
| 2410 | C57BL6 | 11 | pre-T LBL | ND | ND | ND | ND | ND | ND | GL | GL |
| 1075 | FVB | 8 | pre-T LBL | ND | ND | ND | ND | ND | ND | GL | GL |
| 1901 | FVB | 10 | pre-T LBL | ND | ND | ND | ND | ND | ND | GL | codon 61 CAA>AAA (Q>K) |
| 2738 | FVB | 7 | pre-T LBL | ND | ND | ND | ND | ND | ND | GL | GL |
| 2918 | FVB | 6 | pre-T LBL | ND | ND | ND | ND | ND | ND | GL | GL |
| 8013 | FVB | 9 | pre-T LBL | ND | ND | ND | ND | ND | ND | GL | GL |
| 1018 | FVB | 14 | pre-T LBL (thymus) | ND | ND | ND | ND | ND | ND | codon 12 GGT>GAT (G>D) | GL |
MPD-like leukemia indicates myeloproliferative disease-like leukemia; AMKL, acute megakaryocytic leukemia; GL, germline; and ND, not done.
We next searched for mutations of the Npm1 gene, which is mutated in 32% of human ANLLs,12 in 22 ANLL samples from NHD13 mice. These mutations cluster in exon 12 and typically occur as 4 base-pair insertions producing a frame shift. We amplified and sequenced exon 12 of the Npm1 gene; no mutations were detected (Table 1). Runx1 encodes a hematopoietic transcription factor mutated in 11% of ANLL patients.12 We used RT-PCR to screen the NHD13 samples, and obtained a PCR product from 21 mice. No mutations were detected (Table 1).
Flt3 and Kit encode receptor tyrosine kinases,13 that are mutated at frequencies of 24% and 5% respectively in unselected ANLL cases,12 and in 13% and 2% of ANLL cases that evolved from MDS.14 We screened the juxtamembrane and kinase domain regions, where mutations are known to occur (Flt3 exons 14, 15, and 20 and Kit exons 8 and 17), and found no Flt3 or Kit mutations (Table 1). The “2-class” model for myeloid leukemia predicts that one class I (proliferation or survival promoting) and one class II (differentiation blocking) event are required for leukemic transformation.15 This model would predict that NHD13 expression and activating mutations of either Flt3 or Kit would collaborate, and, given the frequent mutations of these genes in ANLL cases, we expected to find mutations of these genes among our mouse cohort. It may be that these genes are not prone to mutations in mice, or, alternatively, they do not complement the NHD13 transgene.
We next investigated the Nras and Kras genes, which encode signaling molecules downstream of receptor tyrosine kinases.16 These genes are mutated in 16% and 4% of ANLL cases, respectively12; the mutations are single nucleotide missense mutations in codons 12, 13, or 61 and result in a constitutively active RAS protein.16 We amplified and sequenced exons 2 and 3 of these genes and identified 3 (14%) Nras and 4 (18%) Kras mutations. All 7 of these ras mutations occurred within codon 12 and resulted in a single amino acid substitution which is predicted to encode a constitutively active NRAS or KRAS protein. Similar to findings in patients with ANLL,17 we noted no clear correlation between leukemic subtype and the presence of NRAS or KRAS mutations. Because of reports that indicate NRAS or KRAS mutations can be found in patients with MDS (most commonly patients with refractory anemia with excess blasts),14 we assayed 12 NHD13 mice with MDS for Nras or Kras mutations. No Nras or Kras mutations were identified in these mice, a statistically significant difference between the MDS and ANLL groups (7/22 vs 0/12; P = .036 by Fisher exact test), suggesting that the ras mutations occurred as leukemia progression events. Finally, because ras mutations have also been associated with pre-T lymphoblastic leukemia/lymphoma (pre-T LBL), we studied 10 NHD13 mice with pre-T LBL and identified one Nras and one Kras mutation (Table 1).
We previously reported the derivation of an IL-3–dependent cell line from embryonic stem cells that expressed an NHD13“knock-in” allele.18 To examine the transforming potential of the ras mutations in the context of NHD13 expression, we cloned a G12D mutant Nras cDNA into an EF1a-driven expression vector. Expression of the mutant Nras conferred IL-3–independent growth, whereas expression of the wild-type Nras had no effect on growth factor dependence (Figure S2). This finding supports the assertion that ras mutations (class I) complement the NHD13 fusion protein (class II).
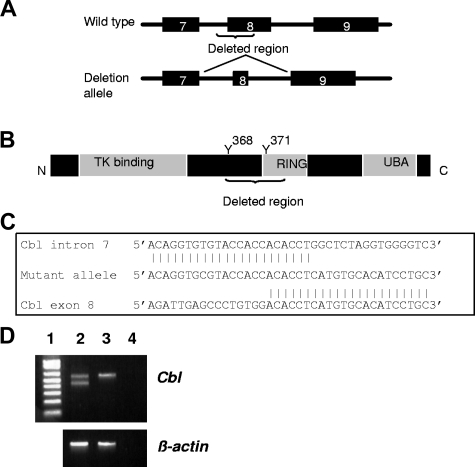
Cbl is a ubiquitin ligase responsible for targeting degradation of receptor tyrosine kinases. Intriguingly, CBL mutations that lead to FLT3 activation have recently been identified in AML patients, clustered around CBL exon 8.19,20 We screened the ANLL samples for mutations of Cbl exon 8, and identified a deletion of 297 bp in one sample that results in loss of the exon 8 splice acceptor. RT-PCR and sequence analysis of the mutant PCR product demonstrated an aberrant Cbl transcript lacking exon 8 (Figure 1), which is similar to findings identified in the MOLM-13 cell line and one ANLL patient.19 This transcript encodes a protein lacking a portion of the RING domain and 2 tyrosine residues which are critical for activation of the CBL protein.21 The transcripts lacking exon 8 are in-frame and may produce a dominant negative protein similar to that produced by the v-Cbl oncogene.19,21 The genomic breakpoint shows a 6 nucleotide microhomology region (Figure 1C) suggesting the deletion was mediated via nonhomologous end-joining.22
Figure 1.
Cbl deletion mutant. (A) Exons 7-9 of the wild-type and mutant Cbl gene. Deletion of the exon 8 splice acceptor causes exon 8 to be spliced of the transcript. (B) CBL protein structure showing known functional domains. Y indicates tyrosine residue that is phosphorylated to activate CBL function. (C) Nucleotide sequence at breakpoint, demonstrating 6 base pair microhomology. (D) Expression of the mutant allele. Lane 1, 1 kb ladder, lane 2, cbl mutant, lane 3, wild-type control, lane 4, no template. Note the lower PCR product in lane 2 corresponding to the mutant allele.
Although we cannot exclude the possibility that the NHD13 transgene alone was sufficient to initiate leukemia, the results regarding Nras, Kras, Cbl, Runx1 and Npm1 are consistent with the current “2-class” model for leukemogenesis. Of note, although ras mutations have been studied for decades, and there are numerous reports of mutagen-induced ras mutations in mice, there have been no reports of spontaneous ras mutations in murine leukemia. The absence of Kit and Flt3 mutations was surprising in the context of the aforementioned 2-class hypothesis, and it will be of interest to learn whether these mutations were not identified in NHD13 mice because they do not complement NHD13, or because they simply did not occur. The results described herein reinforce the prevailing 2-class model of leukemogenic events, and also have implications for the nature of NHD13-mediated leukemogenesis.
Supplementary Material
Acknowledgments
We are grateful to our colleagues Ying-Wei Lin, Helge Hartung, Yangjo Chung, and Chul Won Choi for significant discussion and technical assistance.
This research was supported by the Intramural Research Program of the NIH and NCI.
Footnotes
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Authorship
Contribution: C.S. designed and performed research, analyzed data, and wrote the first draft of the manuscript; L.Y.L. and S.B. designed and performed research and analyzed data; and P.D.A. designed research, analyzed data, and wrote the final draft of the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Peter D. Aplan, NIH/NCI/CCR/Genetics Branch, Navy 8, Room 5101, 8901 Wisconsin Avenue, Bethesda, MD 20889-5105; e-mail: aplanp@mail.nih.gov.
References
- 1.Lin YW, Slape C, Zhang Z, Aplan PD. NUP98-HOXD13 transgenic mice develop a highly penetrant, severe myelodysplastic syndrome that progresses to acute leukemia. Blood. 2005;106:287–295. doi: 10.1182/blood-2004-12-4794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Slape C, Hartung H, Lin YW, Bies J, Wolff L, Aplan PD. Retroviral insertional mutagenesis identifies genes that collaborate with NUP98-HOXD13 during leukemic transformation. Cancer Res. 2007;67:5148–5155. doi: 10.1158/0008-5472.CAN-07-0075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Johansson FK, Brodd J, Eklof C, et al. Identification of candidate cancer-causing genes in mouse brain tumors by retroviral tagging. Proc Natl Acad Sci U S A. 2004;101:11334–11337. doi: 10.1073/pnas.0402716101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Castilla LH, Perrat P, Martinez NJ, et al. Identification of genes that synergize with Cbfb-MYH11 in the pathogenesis of acute myeloid leukemia. Proc Natl Acad Sci U S A. 2004;101:4924–4929. doi: 10.1073/pnas.0400930101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Higuchi M, O'Brien D, Kumaravelu P, Lenny N, Yeoh EJ, Downing JR. Expression of a conditional AML1-ETO oncogene bypasses embryonic lethality and establishes a murine model of human t(8;21) acute myeloid leukemia. Cancer Cell. 2002;1:63–74. doi: 10.1016/s1535-6108(02)00016-8. [DOI] [PubMed] [Google Scholar]
- 6.Castilla LH, Garrett L, Adya N, et al. The fusion gene Cbfb-MYH11 blocks myeloid differentiation and predisposes mice to acute myelomonocytic leukaemia. Nat Genet. 1999;23:144–146. doi: 10.1038/13776. [DOI] [PubMed] [Google Scholar]
- 7.Kogan SC, Ward JM, Anver MR, et al. Bethesda proposals for classification of nonlymphoid hematopoietic neoplasms in mice. Blood. 2002;100:238–245. doi: 10.1182/blood.v100.1.238. [DOI] [PubMed] [Google Scholar]
- 8.Morse HC, 3rd, Anver MR, Fredrickson TN, et al. Bethesda proposals for classification of lymphoid neoplasms in mice. Blood. 2002;100:246–258. doi: 10.1182/blood.v100.1.246. [DOI] [PubMed] [Google Scholar]
- 9.National Center for Biotechnology Information. NCBI database. [Accessed May 7. 2008]; http://www.ncbi.nlm.nih.gov/
- 10.Hollstein M, Sidransky D, Vogelstein B, Harris CC. p53 mutations in human cancers. Science. 1991;253:49–53. doi: 10.1126/science.1905840. [DOI] [PubMed] [Google Scholar]
- 11.Fenaux P, Preudhomme C, Quiquandon I, et al. Mutations of the P53 gene in acute myeloid leukaemia. Br J Haematol. 1992;80:178–183. doi: 10.1111/j.1365-2141.1992.tb08897.x. [DOI] [PubMed] [Google Scholar]
- 12.Forbes S, Clements J, Dawson E, et al. Cosmic 2005. Br J Cancer. 2006;94:318–322. doi: 10.1038/sj.bjc.6602928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Reilly JT. Receptor tyrosine kinases in normal and malignant haematopoiesis. Blood Rev. 2003;17:241–248. doi: 10.1016/s0268-960x(03)00024-9. [DOI] [PubMed] [Google Scholar]
- 14.Bacher U, Haferlach T, Kern W, Haferlach C, Schnittger S. A comparative study of molecular mutations in 381 patients with myelodysplastic syndrome and in 4130 patients with acute myeloid leukemia. Haematologica. 2007;92:744–752. doi: 10.3324/haematol.10869. [DOI] [PubMed] [Google Scholar]
- 15.Gilliland DG, Tallman MS. Focus on acute leukemias. Cancer Cell. 2002;1:417–420. doi: 10.1016/s1535-6108(02)00081-8. [DOI] [PubMed] [Google Scholar]
- 16.Schubbert S, Shannon K, Bollag G. Hyperactive Ras in developmental disorders and cancer. Nat Rev Cancer. 2007;7:295–308. doi: 10.1038/nrc2109. [DOI] [PubMed] [Google Scholar]
- 17.Bacher U, Haferlach T, Schoch C, Kern W, Schnittger S. Implications of NRAS mutations in AML: a study of 2502 patients. Blood. 2006;107:3847–3853. doi: 10.1182/blood-2005-08-3522. [DOI] [PubMed] [Google Scholar]
- 18.Slape C, Chung YJ, Soloway PD, Tessarollo L, Aplan PD. Mouse embryonic stem cells that express a NUP98-HOXD13 fusion protein are impaired in their ability to differentiate and can be complemented by BCR-ABL. Leukemia. 2007;21:1239–1248. doi: 10.1038/sj.leu.2404648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Caligiuri MA, Briesewitz R, Yu J, et al. Novel c-CBL and CBL-b ubiquitin ligase mutations in human acute myeloid leukemia. Blood. 2007;110:1022–1024. doi: 10.1182/blood-2006-12-061176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sargin B, Choudhary C, Crosetto N, et al. Flt3-dependent transformation by inactivating c-Cbl mutations in AML. Blood. 2007;110:1004–1012. doi: 10.1182/blood-2007-01-066076. [DOI] [PubMed] [Google Scholar]
- 21.Levkowitz G, Waterman H, Ettenberg SA, et al. Ubiquitin ligase activity and tyrosine phosphorylation underlie suppression of growth factor signaling by c-Cbl/Sli-1. Mol Cell. 1999;4:1029–1040. doi: 10.1016/s1097-2765(00)80231-2. [DOI] [PubMed] [Google Scholar]
- 22.Corneo B, Wendland RL, Deriano L, et al. Rag mutations reveal robust alternative end joining. Nature. 2007;449:483–486. doi: 10.1038/nature06168. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.