Abstract
The first step in the anaerobic metabolism of toluene is a highly unusual reaction: the addition of toluene across the double bond of fumarate to produce (R)-benzylsuccinate that is catalyzed by benzylsuccinate synthase. Benzylsuccinate synthase is a member of the glycyl radicalcontaining family of enzymes, and the reaction is initiated by abstraction of a hydrogen atom from the methyl group of toluene. To gain insight into the free energy profile of this reaction, we have measured the kinetic isotope effects on Vmax and Vmax/Km when deuterated toluene is the substrate. At 30 °C the isotope effects are 1.7 ± 0.2 and 2.9 ± 0.1 on Vmax and Vmax/Km, respectively, at 4 °C they increase slightly to2.2 ± 0.2 and 3.1 ± 0.1 respectively. We compare these results with the theoretical isotope effects on Vmax and Vmax/Km that are predicted from the free energy profile for the uncatalyzed reaction, which has previously been computed using density functional theory. The comparison allows us to draw some conclusions on how the enzyme may catalyze this unusual reaction.
Keywords: toluene metabolism, free radicals, enzyme kinetics, Tauera. aromatica, glycyl radical enzyme
Toluene and related aromatic compounds represent an important class of pollutants that are long-lived in the environment and have relatively high water solubility. This is a particular problem in environments where oxygen is scarce, such as ground water supplies. It was initially thought that the only routes to degrade such compounds were through oxidative pathways that require molecular oxygen to functionalize otherwise inert aromatic hydrocarbons. However, the recent discovery in bacteria of anaerobic pathways for the degradation of aromatic compounds has aroused much interest, both for the potential they offer for bio-remediation in situations where oxygen is scarce, and because of the novel chemistry involved (1, 2).
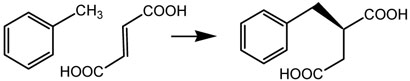
Various denitrifying and sulfate-reducing bacteria such as Thauera aromatica and Desulfobacula toluolica are able to live on toluene as their sole source of carbon and energy under anaerobic conditions (3, 4). The first step in this pathway is the addition of toluene across the double bond of fumarate to produce (R)-benzylsuccinate, as shown in scheme 1, in which hydrogen in the methyl group of toluene is retained in the product (5). This remarkable chemical transformation is catalyzed by benzylsuccinate synthase1 (BSS).
Scheme 1.
Reaction catalyzed by benzylsuccinate synthase
BSS as a member of the growing class of glycyl radical-containing enzymes that includes pyruvate formate-lyase (PFL) and anaerobic ribonucleotide reductase (6-11). EPR studies show that the resting enzyme harbors an organic radical similar to that observed in these enzymes (12-14). Like other glycyl radical enzymes, BSS is extremely oxygen sensitive, and exposure to air results in oxidative cleavage of the α subunit at the site of the presumed glycyl radical (6).
We recently investigated the stereochemistry of hydrogen transfer using deuterated toluene (15). We showed that when fumarate was the co-substrate, deuterium was transferred from toluene to the C-3 pro-(S) position of benzylsuccinate, implying that the addition of toluene to the double bond of fumarate was syn. However, when maleate (which is also recognized by the enzyme (16)) was the co-substrate, the addition of toluene occurs in an anti fashion. These observations provide evidence for the formation of the C-3 radical of benzylsuccinate as an intermediate, in which rotation about the C-2, C-3 bond can occur to relieve the sterically unfavorable cis conformation of the carboxylate groups when maleate is the co-substrate.
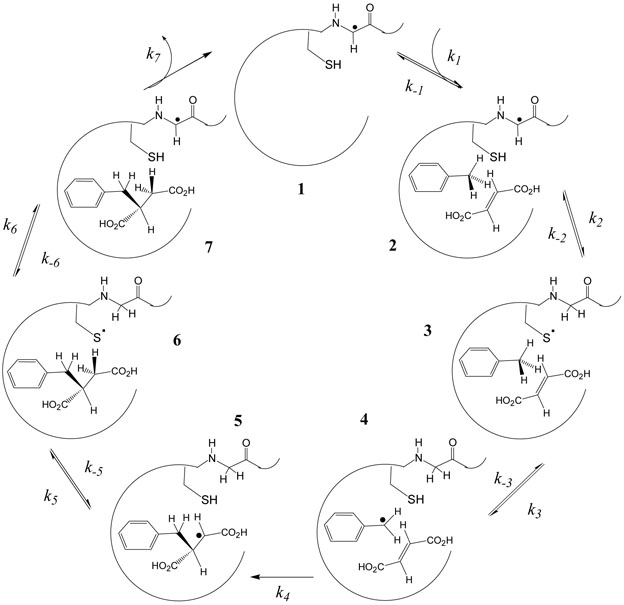
Based on the observations above, the mechanism shown in Figure 1 is proposed. This involves the initial transfer of the glycyl radical to an active site cysteine residue, which then abstracts hydrogen from toluene to generate a benzylic radical. The benzylic radical subsequently undergoes addition to fumarate to generate the C-3 radical of (R)-benzylsuccinate. The abstracted hydrogen is transferred to the product to generate benzylsuccinate and the cysteinyl radical, and lastly the radical is transferred back to glycine.
Figure 1.
Proposed mechanism for benzylsuccinate synthase.
The rapid loss of activity encountered during attempts to purify the enzyme and its extreme oxygen sensitivity (6) has severely hindered efforts to study the enzyme. As a result, virtually nothing is known experimentally regarding the kinetics or energetics of this highly unusual enzyme reaction. However, a density functional theoretical study has evaluated the BSS reaction and computed the relative stabilities of the radical intermediates and activation energies of each step in the putative mechanism (17). The rate-determining step in the reaction is predicted to be addition of the benzyl radical to fumarate, which is effectively irreversible (ΔG = -10.1 kcal/mol). To test this prediction and gain insight into the nature of the rate-determining step in BSS we have measured the deuterium kinetic isotope effects on Vmax and Vmax/Km for the addition of deuterated toluene to fumarate.
MATERIALS AND METHODS
Growth of Thauera Aromatica
T. Aromatica was grown at 30 °C under a N2 atmosphere in the minimal medium described by Coschigano (7), containing 0.5 mM toluene as the carbon source and 5 mM nitrate as an oxidizing agent. The toluene and nitrate concentrations were maintained at their approximate starting concentrations by additional feedings during growth. Cells were grown to an OD600 of 0.7 – 0.9 and harvested under anaerobic conditions by centrifugation at 6,000 g for 30 min in sealed nitrogen-flushed centrifuge tubes. The cell pellet was suspended in degassed buffer containing 10 % glycerol, 0.5 mM of sodium dithionite and the cells pelleted again at 6,000 g for 30 min. The cell pellet was frozen with liquid nitrogen and stored at −80 °C.
Preparation of cell extracts
All experiments were performed in a glove box (Coy Laboratory Products) in a 3% H2/ 97% N2 atmosphere, at 4 °C in the cold room, with oxygen concentration not higher than 5 ppm. Typically, one volume of cell pellet was resuspended in 2.5 volumes of a buffer comprising 40 mM triethanolamine/HCl, 10 mM fumaric acid and 10% glycerol at pH 8.0. The resuspended cells were sonicated at 0 °C with four bursts (13 W × 30 s), with a 1 min rest between each burst to prevent the solution over heating. The lysate was centrifuged at 14, 000 g at 4 °C for 10 min to remove the cell debris and the supernatant containing the soluble proteins was used immediately.
Enzymatic synthesis of the benzylsuccinic acid
Typically 400 – 600 μL of the cell extract was added to 500 μL aqueous toluene solution (5.75 mM) and the reaction mixture was incubated at 4 °C or 30 °C. At various times 200 μL of the reaction solution was removed and the reaction quenched by addition of 50 μl 4 M HCl. The protein residue was removed by centrifugation at 14, 000 g at ambient temperature for 15 min, and the supernatant was further analyzed by reverse phase HPLC.
HPLC analysis of benzylsuccinic acid
Chromatography was performed at room temperature with detection at 254 nm using a Vydac reverse-phase C-18 column (5 μM, 4.6×250). The column was equilibrated in 0.3% formic acid and compounds were eluted with an ascending gradient of CH3CN: 0 – 35 % over 6 mL; 35 – 47 % over 12 mL; and 47 – 100 % over 5 mL at a flow rate of 1 mL/min. Using this gradient, benzylsuccinic acid eluted at 11.8 min. The identity of the benzylsuccinic acid was confirmed by mass spectrometry as described below. The area of the benzylsuccinic acid peak was determined after subtraction of the base line from the t = 0 chromatograph and the amount of benzylsuccinic acid formed determined by reference to standard curves constructed with authentic commercially available benzylsuccinic acid.
Electrospray ionization mass spectroscopy
The deuterium content of benzylsuccinic acid was analyzed by ESI-MS spectroscopy under the negative ion mode. Samples collected after HPLC were directly analyzed by time-of-flight electrospray mass spectrometry (LCT Micromass) under the negative ion mode. The samples were introduced into the spectrometer through an in-line HPLC pump, in a carrier solvent of 90% methanol/10% water at 0.1 mL/min. The desolvation temperature was 150 °C. The spectral data were analyzed using the MassLynx 4.0 software suite. Mass calculated for C11H12O4 (M-): 208.07, found: 207.04; mass calculated for C11H4D8O4 (M-): 216.12, found: 215.10.
Analysis of deuterated toluene mixtures
To accurately determine the ratio of C7H8 to C7D8 in experiments to measure Vmax/Km isotope effects, aqueous toluene solutions were analyzed by GC/MS spectroscopy using a Finnigan Trace MS gas chromatograph/mass spectrometer.
RESULTS AND DISCUSSION
Isotope effects are a powerful tool to study the chemical mechanisms and energetics of enzyme reactions. For BSS a theoretical study has calculated the free energy profile for the enzyme-catalyzed reaction, providing predictions about the rate-determining step that may be experimentally tested by measuring isotope effects. We therefore decided to measure the deuterium kinetic isotope effect for the BSS-catalyzed reaction when deuterium was present in the methyl group of toluene. In these experiments we have employed cell-free extracts of T. aromatica, because, although the purification of the enzyme has been reported (6), large losses of activity occurred during purification and the purified enzyme was highly unstable. We were not been able to obtain active preparations of the enzyme using the reported purification procedure.
Kinetic analyses are generally performed using highly purified enzymes to guard against the risk of interference due to contaminating enzymes or inhibitors. However, since isotope effects involve purely a comparison between the rates of reaction of labeled and unlabeled substrates they can be reliably measured in crude enzyme preparations provided no other isotopically-sensitive reactions are available to either the substrate or products of the reaction, which could lead to the results being skewed. In the present case these criteria are met, because BSS is the only enzyme for which toluene is a substrate under anaerobic conditions. The product of the reaction, benzylsuccinate, is the substrate for benzylsuccinyl-CoA transferase, which catalyzes the transfer of coenzyme A from succinyl-CoA; in the absence of this reaction there are no other fates for benzylsuccinate. As discussed below, we found no evidence for benzylsuccinyl-CoA transferase activity. This is not surprising because any endogenous succinyl-CoA in the cell-free extract is likely to be rapidly hydrolysed under the slightly basic buffer conditions employed in the reaction.
Enzyme assay
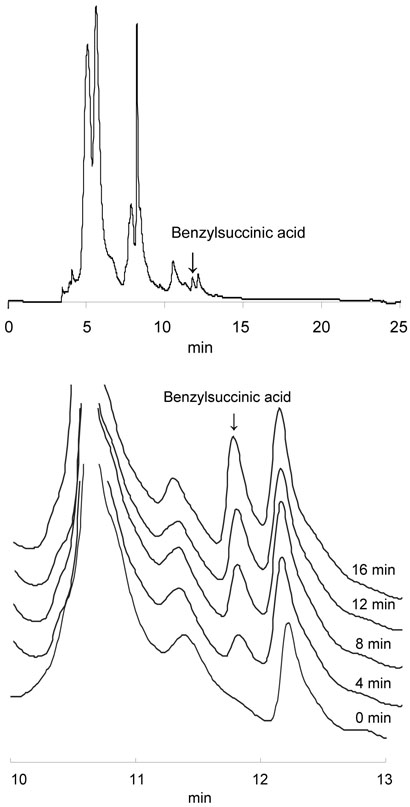
We developed an HPLC-based assay for BSS activity, in place of the radioactivity-based assay used previously (6, 13). This allowed us both to reliably quantify the amount of benzylsuccinate produced, as shown in Figure 2, and subsequently analyze the deuterium content of the benzylsuccinate peak by mass spectrometry. Furthermore, it allowed us to assess whether any additional products were being formed during incubation of the substrates with the cell-free extract. We were unable to detect the formation of any other metabolites – no other peaks appeared or disappeared – during the incubation period.
Figure 2.
HPLC assay of BSS activity in cell-free extracts of T. aromatica. Top Typical HPLC chromatograph with position of the benzylsuccinic acid peak indicated. Bottom Enlargement of the region between 10 and 13 min illustrating the time-dependent increase in benzylsuccinic acid.
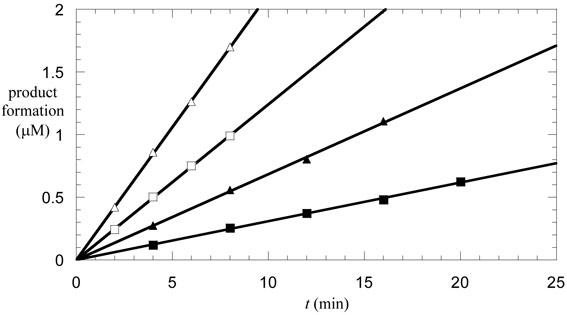
In the assay, the substrate concentrations were typically 2 mM for toluene and 10 mM for fumarate; reducing the concentration of either substrate by half did not change the rate of reaction, so these concentrations may be assumed to be saturating. (The Km for toluene is not known accurately, but is below 100 μM; L. L. unpublished data.) Under the conditions of the assay, the formation of benzylsuccinate was linear with time for up to 20 min, as shown in Figure 3. The activity of the enzyme was measured at both 4 °C and 30 °C. The specific activity of the cell extract at 4 °C was 2.4 nmol/min/mg of protein, whereas at 30 °C the specific activity rose to 7.4 nmol/min/mg. This level of activity is similar to that reported in permeablized cells of Azoracus sp (18), and some 4 orders of magnitude higher than that reported for purified enzyme from T. aromatica (6). As judged by SDS-PAGE, the BSS α-subunit, a conspicuous band of Mr 98,000, comprised about 3.5 % of the total proteins present in the cell free extract, which allows us to estimate kcat for BSS as approximately 0.11 s−1 at 4 °C and 0.35 s−1 at 30 °C.
Figure 3.
Determination of the isotope effect on Vmax for BSS. Initial velocity plots for BSS, reacting with saturating concentrations of toluene. The symbols represent the reactions of unlabelled toluene (triangles) or d8-toluene (squares) at 4 °C (closed symbols) or 30 °C (open symbols).
Deuterium kinetic isotope effects
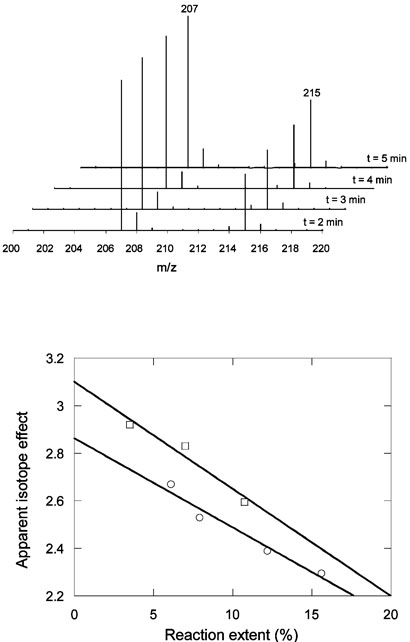
The isotope effects on Vmax/Km were measured in competition experiments in which an equimolar mixture of toluene and d8-toluene was incubated with enzyme-containing cell-free extract for various times. The reaction was quenched, and the amount of benzylsuccinic acid in the sample was determined by HPLC; the deuterium content of the benzylsuccinic acid was subsequently determined by mass spectrometry (Figure 4). In these experiments apparent kinetic isotope effects were measured at relatively low extents of reaction of between 3 and 15 %. Under these conditions the isotopic composition of the starting toluene varies approximately linearly with the extent of reaction. The kinetic isotope effect on D(V/K) was therefore calculated by linear extrapolation of the apparent kinetic isotope effect to zero extent of reaction (Figure 4). D(V/K) = 2.9 ± 0.1 at 30 °C, a value which rises slightly to 3.1 ± 0.1 at 4 °C
Figure 4.
Determination of V/K isotope effect for BSS Top Representative electrospray mass spectra (negative mode) showing the ratio of benzylsuccinate (M-1 = 207) to d8-benzylsuccinate (M-1 = 215) produced by incubation of BSS with a 1:1 mixture of toluene and d8-toluene at 30 °C. Bottom Determination of the isotope effect on Vmax/Km by extrapolation of the apparent isotope effect to zero extent of reaction. Data shown for 4 °C (□)or at 30 °C (○).
The isotope effects on Vmax, were determined by direct comparison of the initial rates at which toluene and d8-toluene were converted to benzylsuccinate (Figure 3). At 30 °C DV = 1.7 ± 0.2, whereas at 4 °C the isotope effect rises to 2.2 ± 0.2. These isotope effects are significantly lower that the corresponding D(V/K) isotope effects, suggesting that although hydrogen abstraction from toluene is a kinetically significant step it is not fully rate-determining. There are two isotopically sensitive steps in the BSS reaction – abstraction of hydrogen from toluene and return of hydrogen to benzylsuccinate – however, it is more likely that the isotope effect is manifested on the former step because the latter step occurs after the addition of the benzylic radical to fumarate, which is expected to be essentially irreversible.
Comparison of isotope effects predicted by the calculated free energy profile with experimentally determined isotope effects
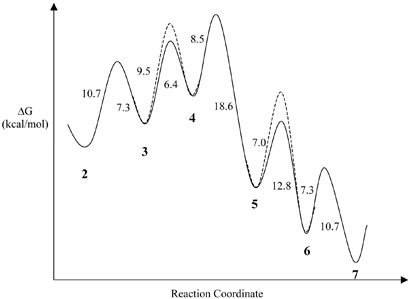
The free energy profile for the BSS-catalyzed reaction has been modeled computationally using density functional theory calculations at the B3LYP level of theory (17). The model included the transfer of hydrogen between the active site cysteine residue and the glycyl radical, but otherwise no information about the protein was assumed because the structure of the enzyme is not known. The calculated free energy profile is shown in Figure 5 and may considered as an approximation to that for the uncatalyzed reaction, assuming that all the reacting species have been brought into a reasonable alignment for the reaction to proceed.
Figure 5.
Free energy profile for the BSS reaction calculated by density functional theory. The data used to construct the figure are taken from reference (17). The heights of the energy barriers (kcal/mol) are indicated by the side of each barrier; the bold numbers refer to the enzyme species shown in Figure 1; the steps subject to a deuterium isotope effect are indicated by dashed lines.
We wished to use our experimentally-determined isotope effects on Vmax and Vmax/Km to compare how the true free energy profile for BSS may differ from the calculated one, and thereby gain insights into how the enzyme effect catalysis in the reaction. This may be achieved as follows: First, the ΔG‡ calculated for each transition state of the reaction mechanism can be used to calculate the corresponding elementary rate constants the mechanism. Second, the expressions for kcat and kcat/Km can be derived in terms of the elementary rate constants for the mechanism, and from these the corresponding expressions for the isotope effects on Vmax and Vmax/Km can also be derived. Third, although we do not know the exact value of the intrinsic isotope effect on C-H cleavage is for the reaction, by making a reasonable guess we can evaluate whether the calculated free energy profile predicts isotope effects on Vmax and Vmax/Km that are in qualitative agreement with those measured experimentally. In particular, we can determine whether the energy profile predicts the isotope effects to be: a) fully suppressed, i.e. DV = D(V/K) = 1; b) fully expressed, i.e. DV = D(V/K) = DVint; c) DV suppressed relative to D(V/K) i.e. DV < D(V/K); or d) in the most unusual case, D(V/K) suppressed relative to D V i.e. DV > D(V/K).
Expressions for kcat and kcat/Km
Expressions for kcat and kcat/Km for the mechanism shown in Figure 1 can be derived using the method of net rate constants (19), among other methods. To simplify matters, we assume, based on the computed free energy profile, that formation of the carbon-carbon bond between benzyl radical and fumarate (k4) is essentially irreversible, in which case subsequent steps in the mechanism can be neglected from the analysis. This is a reasonable assumption because a strong carbon-carbon sigma bond is formed at the expense of a weaker carbon-carbon pi bond. We also assume that neither substrate binding nor product release is rate-limiting, which is also reasonable in view of the isotope effects we have measured. The expressions for kcat and kcat/Km are given by equations 1 and 2 respectively, where the elementary rate constants refer to the steps shown in Figure 1; k-1 in Equation 2 is the off rate for substrate binding.
| Equation 1 |
| Equation 2 |
Expressions for DV and D(V/K)
The expressions for the observed isotope effects on kcat and kcat/Km can be derived by assuming that only k3 and k−3 are altered in the reaction with deuterated toluene. They are obtained by dividing equations 1 and 2 by the equivalent expressions in which k3 and k−3 are replaced with Dk3 and Dk−3 respectively to give equations 3 and 4.
| Equation 3 |
| Equation 4 |
These equations may be rewritten by substituting Dk3 by k3/DVint and Dk−3 by k−3/DVint, where DVint is the intrinsic isotope effect on hydrogen transfer, to give equations 5 and 6; this assumes that any equilibrium isotope effects are small enough to be ignored. These equations describe how the observed isotope effects on Vmax and Vmax/Km change as a function of the elementary rate constants for the various steps in the mechanism.
| Equation 5 |
| Equation 6 |
Comparison of theory and experiment
The elementary rate constants for each step in the BSS reaction were calculated from the computationally determined free energies at 4 °C and at 30 °C using the standard equation derived from basic transition state theory (Equation 7). We made the usual assumption that the transmission coefficient is unity; i.e. no attempt was made to account for the fact that the Arrhenius pre-exponential factors may be different for the individual transition states in the mechanism.
| Equation 7 |
The rate constants are tabulated in Table 1. From these, kcat for the overall reaction was calculated using equation 1 to be 8.4 s−1 at 4 °C and 96 s−1 at 30 °C. This is well within the range of turnover numbers expected for a “typical” enzyme, although significantly higher than the values of about 0.1 s−1 at 4 °C and 0.35 s−1 at 30 °C for kcat that we calculate from our experiments. However, given that the computationally-determined free energy profile includes no explicit knowledge of the protein environment and does not account for substrate binding/product release or protein conformational changes, the numbers agree reasonably well.
Table 1.
Calculated values for elementary rate constants in the BSS-catalyzed reaction at 4 °C and 30 °C.
To compare our experimentally-determined isotope effects with those predicted by the computationally-determined free energy profile, we used equations 5 and 6 with the data in Table 1 to calculate the extent to which intrinsic isotope effect, DVint, is masked by other steps in under Vmax and Vmax/Km conditions. Although equation 6 requires us to assume a value for k−1, the substrate off-rate, if the term k2k3k4 in equation 6 is small enough to be neglected then k−1 will cancel from equation 6. In practice, if k−1 is greater than ~50 s−1 this criterion is met and the value of k−1 does not influence D(V/K)obs. (If k−1 were to be very slow, corresponding to a “sticky” substrate, this would lead to the unusual case in which D(V/K)obs is actually smaller than DVobs, which is the opposite to our experimental finding.)
Our calculations show that at 30 °C DVobs = 0.03 DVint + 0.97 and, assuming k−1 is not limiting, D(V/K)obs = 0.03 DVint+ 0.97, whereas at 4 °C DVobs = 0.02 DVint + 0.98 and D(V/K)obs = 0.02 DVint+ 0.98. Thus the calculated free energy profile predicts that DVobs and D(V/K)obs would not only be almost fully suppressed at both 30 °C and 4 °C, but they would have the same value. In contrast, we find that D(V/K)obs both is significantly larger than DVobs and, although the value of 3 for D(V/K)obs is unlikely to represent the full intrinsic isotope effect, it is probably significantly more than 2 – 3 % of DVint. For example, if the free energy profile was correct, then a value of 3 for D(V/K)obs would imply that DVint would be ~ 65 at 30 °C and ~ 100 at 4 °C!
Such large values are clearly much higher than those predicted for deuterium isotope effects by the semi-classical theory, and would suggest that the calculated free energy profile is incompatible with the experimental data. However, we should note that deuterium isotope effects in the range of 30 - 50 have been measured in several enzymes that catalyze hydrogen atom abstractions from carbon, for example: lipoxygenase, galactose oxidase and B12-dependent isomerases (20-25), in reactions that proceed with a high degree of quantum tunneling. Given that the BSS-catalyzed reaction involves radical chemistry similar to the enzymes mentioned above, the possibility that the intrinsic deuterium isotope effects are indeed extremely large cannot be entirely excluded. However, even allowing for a very large intrinsic isotope effect, the computed free energy profile is unable to account for the experimental observation that D(V/K)obs is significantly larger than DVobs.
To be consistent with our data, the free energy profile must qualitatively reproduce the observations that D(V/K)obs is larger than DVobs and that both isotope effects are to a significant degree suppressed from the intrinsic value. These requirements are not sufficient to uniquely specify the free energy profile, but they do place a number of constraints on the relative heights of the energy barriers.
To account for our data the calculated free energy profile needs to be altered as follows. First, for D(V/K)obs to be larger than DVobs a kinetically significant step in the reaction must occur after the isotopically sensitive step. Therefore k4, or more probably a later step such as product release, must be either of a similar magnitude or slower than either k3 or k2. Secondly, k3, which is the isotopically sensitive step, must be either of a similar magnitude to, or slower than, k2, otherwise this step is not kinetically significant. Lastly, k2 must of a similar magnitude to k−2 because if k2 >> k−2 (i.e. by more than a factor of 10) then D(V/K)obs will tend become smaller than DVobs, corresponding to a “sticky” substrate. A free energy profile that qualitatively reproduces our experimental observations is illustrated in Figure 5.
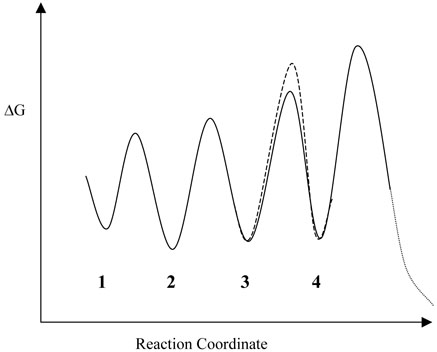
In physical terms, the main difference between the experimentally deduced free energy profile (Figure 5) and the calculated one (Figure 6) is that starting glycyl radical and the intermediate cysteinyl and benzylic radicals appear to be much closer in energy on the enzyme than on paper. The relative heights of the energy barriers between the intermediates are the same in the two free energy profiles. It may be that the calculations reproduce the energies of the radicals poorly, but we think this is unlikely as in general DFT produces reliable results for the ground state energies of small molecules. More interestingly, this comparison suggests that the enzyme plays a role in stabilizing the cysteinyl and benzylic radicals relative to the glycyl radical, or, equally, it could destabilize the glycyl radical. This may be an important and general mechanism for controlling the reactivity of radicals in this class of enzymes. This would represent another example of a generally observed principle of enzymic catalysis: that enzymes act to shift the internal equilibrium constants for the conversion of bound reaction intermediates towards unity. Indeed, enzymes that utilize free radicals in catalysis appear to be particularly adept at stabilizing free radical species in their active sites (9).
Figure 6.
Qualitative free energy profile for the initial steps in the BSS-catalyzed reaction that is consistent with the experimentally observed isotope effects on Vmax and Vmax/Km. The numbers refer to the enzyme species shown in Figure 1; the step subject to a deuterium isotope effect is indicated by dashed lines.
ACKNOWLEDGMENT
We thank Dr. Peter Coschigano (Ohio University) for advice on the culture of T. aromatica and Christel Fox and Dr. Mou-Chi Cheng for assistance.
Footnotes
This research was supported by a grant from the National Institutes of Health, GM 59227 to E.N.G.M.
The abbreviations used are: BSS, benzylsuccinate synthase; D(V/K), the deuterium kinetic isotope effect on Vmax/Km; DV, the deuterium kinetic isotope effect on Vmax.
REFERENCES
- 1.Spormann AM, Widdel F. Metabolism of alkylbenzenes, alkanes, and other hydrocarbons in anaerobic bacteria. Biodegradation. 2000;11:85–105. doi: 10.1023/a:1011122631799. [DOI] [PubMed] [Google Scholar]
- 2.Heider J, Spormann AM, Beller HR, Widdel F. Anaerobic bacterial metabolism of hydrocarbons. Fems Microbiol. Rev. 1998;22:459–473. [Google Scholar]
- 3.Biegert T, Fuchs G, Heider F. Evidence that anaerobic oxidation of toluene in the denitrifying bacterium Thauera aromatica is initiated by formation of benzylsuccinate from toluene and fumarate. Eur. J. Biochem. 1996;238:661–668. doi: 10.1111/j.1432-1033.1996.0661w.x. [DOI] [PubMed] [Google Scholar]
- 4.Rabus R, Heider J. Initial reactions of anaerobic metabolism of alkylbenzenes in denitrifying and sulfate reducing bacteria. Arch. Microbiol. 1998;170:377–384. [Google Scholar]
- 5.Beller HR, Spormann AM. Analysis of the novel benzylsuccinate synthase reaction for anaerobic toluene activation based on structural studies of the product. J. Bacteriol. 1998;180:5454–5457. doi: 10.1128/jb.180.20.5454-5457.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Leuthner B, Leutwein C, Schulz H, Horth P, Haehnel W, Schiltz E, Schagger H, Heider J. Biochemical and genetic characterization of benzylsuccinate synthase from Thauera aromatica: a new glycyl radical enzyme catalysing the first step in anaerobic toluene metabolism. Mol. Microbiol. 1998;28:615–628. doi: 10.1046/j.1365-2958.1998.00826.x. [DOI] [PubMed] [Google Scholar]
- 7.Coschigano PW. Transcriptional analysis of the tutE tutFDGH gene cluster from Thauera aromatica strain T1. Appl. Environ. Microbiol. 2000;66:1147–1151. doi: 10.1128/aem.66.3.1147-1151.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Achong GR, Rodriguez AM, Spormann AM. Benzylsuccinate synthase of Azoarcus sp strain T: Cloning, sequencing, transcriptional organization, and its role in anaerobic toluene and m-xylene mineralization. J. Bacteriol. 2001;183:6763–6770. doi: 10.1128/JB.183.23.6763-6770.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Frey PA. Radical mechanisms of enzymatic catalysis. Ann. Rev. Biochem. 2001;70:121–148. doi: 10.1146/annurev.biochem.70.1.121. [DOI] [PubMed] [Google Scholar]
- 10.Marsh ENG, Huhta MS, Patwardhan A. S-adenosylmethionine radical enzymes. Bioorganic Chem. 2004;32:326–340. doi: 10.1016/j.bioorg.2004.06.001. [DOI] [PubMed] [Google Scholar]
- 11.Selmer T, Pierik AJ, Heider J. New glycyl radical enzymes catalysing key. metabolic steps in anaerobic bacteria. Biol. Chem. 2005;386:981–988. doi: 10.1515/BC.2005.114. [DOI] [PubMed] [Google Scholar]
- 12.Duboc-Toia C, Hassan AK, Mulliez E, Ollagnier-de Choudens S, Fontecave M, Leutwein C, Heider J. Very high-field EPR study of glycyl radical enzymes. J. Am. Chem. Soc. 2003;125:38–39. doi: 10.1021/ja026690j. [DOI] [PubMed] [Google Scholar]
- 13.Verfurth K, Pierik AJ, Leutwein C, Zorn S, Heider J. Substrate specificities and electron paramagnetic resonance properties of benzylsuccinate synthases in anaerobic toluene and m-xylene metabolism. Arch. Microbiol. 2004;181:155–162. doi: 10.1007/s00203-003-0642-4. [DOI] [PubMed] [Google Scholar]
- 14.Krieger CJ, Roseboom W, Albracht SPJ, Spormann AM. A stable organic free radical in anaerobic benzylsuccinate synthase of Azoarcus sp strain T. J. Biol. Chem. 2001;276:12924–12927. doi: 10.1074/jbc.M009453200. [DOI] [PubMed] [Google Scholar]
- 15.Qiao C, Marsh ENG. Mechanism of benzylsuccinate synthase: Stereochemistry of toluene addition to fumarate and maleate. J. Am. Chem. Soc. 2005;127:8608–8609. doi: 10.1021/ja051972f. [DOI] [PubMed] [Google Scholar]
- 16.Beller HR, Spormann AM. Substrate range of benzylsuccinate synthase from Azoarcus sp strain T. Fems Microbiol. Lett. 1999;178:147–153. doi: 10.1111/j.1574-6968.1999.tb13771.x. [DOI] [PubMed] [Google Scholar]
- 17.Himo F. Catalytic mechanism of benzylsuccinate synthase, a theoretical study. J. Phys. Chem. B. 2002;106:7688–7692. [Google Scholar]
- 18.Beller HR, Spormann AM. Anaerobic activation of toluene and o-xylene by addition to fumarate in denitrifying strain T. J. Bacteriol. 1997;179:670–676. doi: 10.1128/jb.179.3.670-676.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Cleland WW. Partition analysis and the concept of net rate constants as tools in enzyme kinetics. Biochemistry. 1975;14:3220–3224. doi: 10.1021/bi00685a029. [DOI] [PubMed] [Google Scholar]
- 20.Rickert KW, Klinman JP. Nature of hydrogen transfer in soybean lipoxygenase 1: Separation of primary and secondary isotope effects. Biochemistry. 1999;38:12218–12228. doi: 10.1021/bi990834y. [DOI] [PubMed] [Google Scholar]
- 21.Whittaker MM, Ballou DP, Whittaker JW. Kinetic isotope effects as probes of the mechanism of galactose oxidase. Biochemistry. 1998;37:8426–8436. doi: 10.1021/bi980328t. [DOI] [PubMed] [Google Scholar]
- 22.Marsh ENG, Ballou DP. Coupling of cobalt-carbon bond homolysis and hydrogen atom abstraction in adenosylcobalamin-dependent glutamate mutase. Biochemistry. 1998;37:11864–72. doi: 10.1021/bi980512e. [DOI] [PubMed] [Google Scholar]
- 23.Cheng MC, Marsh ENG. Isotope effects for deuterium transfer between substrate and coenzyme in adenosylcobalamin-dependent glutamate mutase. Biochemistry. 2005;44:2686–2691. doi: 10.1021/bi047662b. [DOI] [PubMed] [Google Scholar]
- 24.Chowdhury S, Banerjee R. Evidence for quantum mechanical tunneling in the coupled cobalt-carbon bond homolysis-substrate radical generation reaction catalyzed by methylmalonyl-CoA mutase. J. Am. Chem. Soc. 2000;122:5417–5418. [Google Scholar]
- 25.Bandarian V, Reed GH. Isotope effects in the transient phases of the reaction catalyzed by ethanolamine ammonia-lyase: Determination of the number of exchangeable hydrogens in the enzyme-cofactor complex. Biochemistry. 2000;39:12069–12075. doi: 10.1021/bi001014k. [DOI] [PubMed] [Google Scholar]