Abstract
Aims
N-terminal-pro-B-type-natriuretic-peptide (NT-pro-BNP) concentrations are altered in renal failure. This study examined the effect of baseline and change from baseline NT-pro-BNP on cardiovascular outcome and mortality in haemodialysis patients.
Methods and results
On the basis of the German Diabetes and Dialysis Study, which evaluated atorvastatin in 1255 type 2 diabetes mellitus (T2DM) haemodialysis patients (median follow-up 4 years), the impact of NT-pro-BNP on pre-specified, adjudicated endpoints was investigated: sudden death (SD; n = 160), stroke (n = 99), myocardial infarction (MI; n = 200), cardiovascular events (CVEs: cardiac death, MI, stroke; n = 465), all-cause mortality (n = 612). Patients with baseline NT-pro-BNP ≥9252 pg/mL (fourth quartile) exhibited a more than four-fold risk of stroke [hazard ratio (HR) 4.1; 95% confidence interval (CI) 2.0–8.4] and a more than two-fold risk of SD (HR 2.0; 95% CI 1.2–3.3), CVE (HR 2.0; 95% CI 1.5–2.7), and mortality (HR 2.1; 95% CI 1.6–2.7) compared with patients with baseline NT-pro-BNP ≤ 1433 pg/mL (first quartile). Change in NT-pro-BNP was strongly associated with SD, CVE, and mortality. Doubling of NT-pro-BNP increased the risk of death by 46% (95% CI 1.1–2.0). Neither baseline nor change in NT-pro-BNP was significantly associated with MI.
Conclusion
Increasing NT-pro-BNP is a risk factor for SD, CVE, and mortality in haemodialysis patients with T2DM. Whether NT-pro-BNP-guided treatment improves outcome needs to be evaluated prospectively.
Keywords: NT-pro-BNP, Serial measurement, Cardiovascular events, Mortality, Haemodialysis, Diabetes mellitus
Introduction
Brain natriuretic peptide (BNP) and its counterpart N-terminal-pro-B-type-natriuretic-peptide (NT-pro-BNP) are associated with morbidity and mortality in a wide range of patients.1–4 This has stimulated interest about the role of NT-pro-BNP in renal patients. In chronic kidney disease, NT-pro-BNP was associated with coronary artery disease (CAD) and left ventricular hypertrophy.5 In peritoneal dialysis patients, NT-pro-BNP was related to left ventricular dysfunction, cardiovascular events (CVEs), and mortality.6,7 Finally, in a cohort of 399 haemodialysis patients, NT-pro-BNP was associated with 2 year all-cause death8 and, in a smaller cohort, with 3 year cardiac morbidity and mortality.9
Up to now, the association of baseline NT-pro-BNP with sudden death (SD), stroke, and myocardial infarction (MI) has not been evaluated in renal patients. Furthermore, the effect of a longitudinal change in NT-pro-BNP on outcome has not been investigated before. Therefore, we assessed the effect of baseline and change from baseline NT-pro-BNP on SD, stroke, MI, CVE, and all-cause mortality by performing post hoc analyses of the German Diabetes Dialysis Study [4D-Study (Die Deutsche Diabetes Dialyse Studie)],10 which evaluated atorvastatin in 1255 patients with type 2 diabetes mellitus (T2DM) on maintenance haemodialysis treatment.
Methods
Study design and participants
The 4D-Study methodology has previously been reported.11 Briefly, the 4D-Study was a randomized trial including 1255 patients with T2DM, 18–80 years, and previous duration of haemodialysis of <2 years. Between March 1998 and October 2002, patients were recruited in 178 dialysis units throughout Germany. After a period of 4 weeks, patients were randomly assigned to double-blind treatment with either 20 mg of atorvastatin (n = 619) or placebo (n = 636) once daily. Study visits took place three times before randomization (visit 1–3), at randomization (visit 4), and at four weeks (visit 5) and every six months (visit 6 etc.) after randomization. At each follow-up until March 2004, a blood sample was taken and information was recorded about any suspected endpoint or serious adverse event. The primary study endpoint of the 4D-Study was a composite of death from cardiac causes, MI, and stroke, whichever occurred first. Death from any cause, SD, stroke, and MI (fatal and non-fatal MI) were secondary study endpoints. 4D-Study endpoints were centrally adjudicated by three members of the endpoint committee blinded to study treatment and according to pre-defined criteria.10
For the present analysis, the combined cardiovascular endpoint (cardiac death, MI, stroke), all-cause death, SD, stroke, and MI were all chosen to be separate outcome measures and were based on the primary judgement of the endpoint committee during the 4D-Study.
NT-pro-BNP was measured in blood samples taken at study visit 3 (1 week before randomization) and visit 6 (6 months after randomization). If there was no sample available at visit 6 (n = 20), a sample taken at the following study visit was chosen (visit 7: n = 19; visit 11: n = 1). One of those 20 patients experienced a non-fatal stroke before visit 7 and was therefore censored for analysis.
Laboratory procedures
All laboratory measurements of the 4D-Study including NT-pro-BNP were performed at the Department of Clinical Chemistry, University of Freiburg, Germany. Measurements of NT-pro-BNP were performed by electrochemiluminescence immunoassay on an E 170 analyser (Roche Diagnostics, Mannheim, Germany). Inter-assay coefficients of variance were <5%. Blood samples were taken before the start of dialysis and administration of drugs.
Statistical analysis
Baseline patient characteristics are presented according to baseline NT-pro-BNP quartiles. Continuous variables were expressed with mean and standard deviation, and categorical variables were expressed with absolute and relative frequencies. The effect of NT-pro-BNP (baseline and change from baseline) on outcome was assessed by Kaplan–Meier estimates for incidences of the pre-specified endpoints and by relative risks derived from Cox regression analyses, i.e. hazard ratios (HRs) and corresponding 95% confidence intervals. The Cox regression analyses were adjusted for the confounders of gender, age, atorvastatin treatment, phosphate, low-density lipoprotein, haemoglobin, glycated haemoglobin, albumin, smoking, systolic/diastolic blood pressure, body mass index, ultrafiltration volume, duration of dialysis, arterio-venous (AV) fistula, history of stroke or transitory ischaemic attack, CAD (e.g. MI, coronary artery bypass grafting, percutaneous coronary intervention, or angiographically documented CAD), peripheral vascular disease, and congestive heart failure (CHF; predominantly New York Heart Association class II).
In detail, the following analyses were performed. First, the association of baseline NT-pro-BNP with the pre-defined outcome measures of SD, stroke, MI, CVE, and mortality was analysed, both as a continuous variable (logarithmically transformed because values were not normally distributed and the linearity assumption was satisfied for the log-transformed values) and as a categorical variable (baseline NT-pro-BNP quartiles). Second, the association between change in NT-pro-BNP and outcome was evaluated: as a continuous variable (logarithmically transformed ratio of post-baseline/baseline NT-pro-BNP) and as a categorical variable (less than −10%, −10 up to +10%, >10 up to 100%, and >100% change in NT-pro-BNP). Third, since baseline levels might affect this association, additional analyses regarding the effect of the change in NT-pro-BNP on outcome were done in subgroups of patients with baseline NT-pro-BNP values less than or equal to and greater than the median. Finally, we performed subgroup analyses regarding co-morbidity (patients with and without history of CAD, and with and without history of CHF) and determined the association of baseline and change in NT-pro-BNP with total mortality (other outcomes had no adequate power in subgroups). In general, to compare NT-pro-BNP between the atorvastatin and placebo groups, the Wilcoxon rank-sum test was used, whereas the Wilcoxon signed-rank test was applied to compare baseline with post-baseline NT-pro-BNP within groups. All P-values are reported two-sided. In the models assessing the effect of the change in NT-pro-BNP on outcome, patients experiencing an endpoint before the second NT-pro-BNP measurement were used as censored at this time point (22 patients experienced a CVE, eight a stroke, and 14 a non-fatal MI prior to the second measurement). Analyses were done using SAS version 8.2.
Results
Of 1255 patients taking part in the 4D-Study, 1249 had a baseline and 1205 had a post-baseline NT-pro-BNP measurement, with 1203 patients having both. In total, 633 placebo and 616 atorvastatin samples at baseline and 606 placebo and 599 atorvastatin samples after a median of 182 days (interquartile range 177–185 days) were analysed. The mean follow-up-period was 3.96 years (median 4.0 years) on atorvastatin and 3.91 years (median 4.08 years) on placebo. During follow-up, 612 patients died (160 of SD). Furthermore, 465 patients reached the composite cardiovascular endpoint (CVE) (cardiac death, non-fatal MI, stroke), with MI and stroke occurring in 200 and 99 patients, respectively.
Median baseline NT-pro-BNP was high (3361 pg/mL). During follow-up, NT-pro-BNP increased significantly (median increase of 219 pg/mL, P < 0.001). No significant difference between the atorvastatin and placebo group with respect to baseline, post-baseline, and change from baseline NT-pro-BNP was detected. Patients' baseline characteristics are shown in Table 1.
Table 1.
Baseline patient characteristics according to quartiles of baseline N-terminal-pro-B-type-natriuretic-peptide
| Variable | NT-pro-BNP (pg/mL) |
|||
|---|---|---|---|---|
| Quartile 1 ≤1433 (n = 313) | Quartile 2 1434–3361 (n = 312) | Quartile 3 3362–9251 (n = 312) | Quartile 4 ≥9252 (n = 312) | |
| Age, years | 64.8 ± 8.7 | 65.7 ± 8.4 | 66.5 ± 7.6 | 65.8 ± 8.2 |
| Gender male, % (n) | 54 (170) | 55 (171) | 52 (161) | 55 (170) |
| Ever smoking, % (n) | 37 (116) | 42 (130) | 40 (125) | 43 (134) |
| Body mass index, kg/m2 | 28.9 ± 5.2 | 28.2 ± 4.5 | 27.1 ± 4.6 | 26.0 ± 4.4 |
| Systolic blood pressure, mmHg | 143 ± 21 | 144 ± 22 | 147 ± 23 | 149 ± 22 |
| Diastolic blood pressure, mmHg | 75 ± 11 | 75 ± 10 | 76 ± 11 | 77 ± 11 |
| Time receiving dialysis, months | 8.0 ± 6.8 | 8.4 ± 7.2 | 8.2 ± 6.8 | 8.5 ± 6.7 |
| Ultrafiltration volume, kg | 1.97 ± 1.19 | 2.30 ± 1.16 | 2.33 ± 1.18 | 2.42 ± 1.22 |
| Shunt, % (n) | 94 (294) | 95 (297) | 94 (294) | 90 (279) |
| Historya | ||||
| Arrhythmia, % (n) | 11 (33) | 13 (39) | 25 (77) | 27 (85) |
| MI, CABG, PCI, or CHD, % (n) | 21 (67) | 27 (83) | 31 (98) | 38 (117) |
| CHFb, % (n) | 27 (84) | 30 (93) | 37 (115) | 48 (149) |
| Stroke/TIA, % (n) | 18 (57) | 17 (53) | 19 (60) | 17 (52) |
| Peripheral vascular disease, % (n) | 36 (113) | 46 (145) | 41 (128) | 54 (169) |
| Haemoglobin, g/dL | 11.1 ± 1.4 | 10.9 ± 1.2 | 10.9 ± 1.3 | 10.6 ± 1.4 |
| Glycated haemoglobin, % | 6.79 ± 1.25 | 6.67 ± 1.27 | 6.75 ± 1.26 | 6.67 ± 1.26 |
| Phosphate, mg/L | 1.86 ± 0.43 | 1.99 ± 0.52 | 1.95 ± 0.55 | 2.00 ± 0.57 |
| Albumin, g/dL | 3.87 ± 0.27 | 3.84 ± 0.28 | 3.81 ± 0.31 | 3.76 ± 0.33 |
| LDL cholesterol, mg/dL | 127 ± 30 | 125 ± 31 | 126 ± 30 | 125 ± 29 |
Data are given as mean ± standard deviation. To convert haemoglobin values to mmol/L, multiply by 0.6206; to convert values for phosphate to mmol/L, multiply by 0.32; to convert values for LDL cholesterol to mmol/L, multiply by 0.02566. CABG, coronary artery bypass grafting surgery; PCI, percutaneous coronary intervention; CHD, coronary heart disease, documented by coronary angiography; TIA, transitory ischaemic attack.
aTypes of disease and intervention are not mutually exclusive.
bPredominantly New York Heart Association II.
Baseline N-terminal-pro-B-type-natriuretic-peptide and outcome
Absolute risks
Overall, the absolute mortality risks were high and increasing over quartiles of baseline NT-pro-BNP. After 4 years of follow-up, the cumulative incidences of all-cause mortality were 0.323 in the first quartile, 0.427 in the second quartile, 0.545 in the third quartile, and 0.677 in the highest quartile of baseline NT-pro-BNP.
Regarding the change in NT-pro-BNP, mortality risks increased with rising NT-pro-BNP levels. The 4 year cumulative incidences of death were 0.420 in patients with decreasing NT-pro-BNP, 0.424 in patients with stable levels, and 0.481 in patients with an increase of NT-pro-BNP >10 and ≤100%. The highest risk of 0.654 was seen in patients with more than a doubling in NT-pro-BNP levels.
Relative risks
Baseline NT-pro-BNP was significantly associated with the risk of SD, stroke, CVEs, and mortality. Per unit increase in log-transformed baseline NT-pro-BNP, the relative risks of experiencing these endpoints increased, as being reflected by the adjusted HRs: HRSD 1.28 (1.12–1.47), HRstroke 1.39 (1.17–1.65), HRCVE 1.24 (1.15–1.34), HRmortality 1.27 (1.18–1.36). With regard to MI, no association was seen [HRMI 1.08 (0.96–1.22)]. One unit increase in log-transformed NT-pro-BNP was equal to a 2.72-fold increase in absolute values.
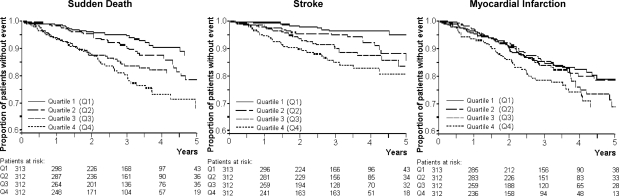
Furthermore, the relation between NT-pro-BNP and all-cause death was independent of baseline co-morbidity, as similar HRs of all-cause death were found in subgroups of patients with and without CHF [HRCHF 1.26 (1.13–1.41); HRnoCHF 1.27 (1.15–1.39)] and with and without CAD [HRCAD 1.26 (1.12–1.42); HRnoCAD 1.28 (1.17–1.40)]. Results of analyses using baseline NT-pro-BNP as a categorical variable are shown in Figure 1 and Table 2.
Figure 1.
Kaplan–Meier estimates for time to sudden death, stroke, and myocardial infarction in subgroups of patients according to quartiles of baseline N-terminal-pro-B-type-natriuretic-peptide (Q1, Quartile 1: N-terminal-pro-B-type-natriuretic-peptide ≤ 1433 pg/mL; Q2, quartile 2: N-terminal-pro-B-type-natriuretic-peptide 1434–3361 pg/mL; Q3, quartile 3: N-terminal-pro-B-type-natriuretic-peptide 3362–9251 pg/mL; Q4, quartile 4: N-terminal-pro-B-type-natriuretic-peptide ≥ 9252 pg/mL).
Table 2.
Risk of all-cause death, combined cardiovascular endpoint, myocardial infarction, sudden death, and stroke by quartiles of baseline N-terminal-pro-B-type-natriuretic-peptide
| NT-pro-BNP (pg/mL) | Quartile 1 ≤1433 (n = 313) | Quartile 2 1434–3361 (n = 312) | Quartile 3 3362–9251 (n = 312) | Quartile 4 ≥9252 (n = 312) |
|---|---|---|---|---|
| All-cause death | ||||
| Number of events during study, n | 98 | 136 | 166 | 212 |
| Adjusted HRa (95% confidence interval) | —b | 1.24 (0.95–1.62), P = 0.116 | 1.65 (1.27–2.13), P < 0.001 | 2.06 (1.60–2.67), P < 0.001 |
| Adjusted HRa (95% confidence interval) with NT-pro-BNP as a continuous variable, 1.27 (1.18–1.36), P < 0.001 | ||||
| Combined cardiovascular endpoint (myocardial infarction, cardiac death, stroke) | ||||
| Number of events during study, n | 79 | 106 | 129 | 151 |
| Adjusted HRa (95% confidence interval) | —b | 1.21 (0.90–1.63), P = 0.206 | 1.67 (1.25–2.23), P < 0.001 | 2.00 (1.49–2.67), P < 0.001 |
| Adjusted HRa (95% confidence interval) with NT-pro-BNP as a continuous variable, 1.24 (1.15–1.34), P < 0.001 | ||||
| Sudden death | ||||
| Number of events during study, n | 23 | 37 | 45 | 55 |
| Adjusted HRa (95% confidence interval) | —b | 1.31 (0.77–2.24), P = 0.319 | 1.74 (1.04–2.93), P = 0.036 | 1.98 (1.18–3.32), P = 0.01 |
| Adjusted HRa (95% confidence interval) with NT-pro-BNP as a continuous variable, 1.28 (1.12–1.47), P < 0.001 | ||||
| Stroke | ||||
| Number of events during study, n | 11 | 22 | 29 | 37 |
| Adjusted HRa (95% confidence interval) | —b | 1.97 (0.94–4.12), P = 0.072 | 2.74 (1.35–5.58), P = 0.005 | 4.13 (2.03–8.39), P < 0.001 |
| Adjusted HRa (95% confidence interval) with NT-pro-BNP as a continuous variable, 1.39 (1.17–1.65), P < 0.001 | ||||
| Myocardial infarction | ||||
| Number of events during study, n | 47 | 48 | 51 | 54 |
| Adjusted HRa (95% confidence interval) | —b | 0.93 (0.62–1.40), P = 0.729 | 1.14 (0.76–1.72), P = 0.525 | 1.23 (0.80–1.88), P = 0.350 |
| Adjusted HRa (95% confidence interval) with NT-pro-BNP as a continuous variable, 1.08 (0.96–1.22), P = 0.219 | ||||
aAdjustment for gender, age, treatment, phosphate, low-density lipoprotein, haemoglobin, glycated haemoglobin, albumin, smoking, systolic/diastolic blood pressure, body mass index, ultrafiltration volume, duration of dialysis, AV fistula, history of stroke or transitory ischaemic attack, CAD (e.g. MI, coronary artery bypass grafting, percutaneous coronary intervention, and angiographically documented CAD), peripheral vascular disease, and CHF.
bThe first quartile was used as reference for each of the others.
Change from baseline N-terminal-pro-B-type-natriuretic-peptide and the association with outcome
With rising NT-pro-BNP (continuous variable), the relative risk of SD, combined CVEs, and all-cause mortality increased significantly, although no significant association with the risk of stroke or MI was detected. These results were independent of baseline NT-pro-BNP, as analyses stratified by baseline levels (less than or equal to and greater than the median) showed a similar pattern. Furthermore, the increasing risk of death with rising NT-pro-BNP was independent of baseline co-morbidity (CHF and CAD).
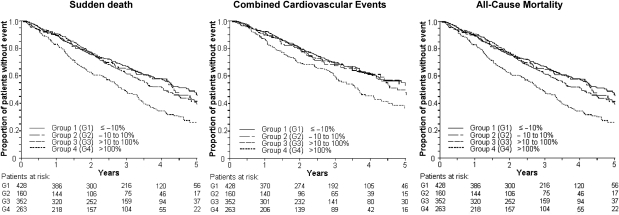
Results of analyses using the change from baseline NT-pro-BNP as a categorical variable are shown in Figure 2 and Table 3. Patients with more than a doubling in NT-pro-BNP had a 51% higher risk of SD, a 37% higher risk of CVEs, and a 46% higher risk of all-cause death than patients with stable levels. Regarding SD, further subgroup analysis showed that patients with low baseline NT-pro-BNP (less than or equal to the median) and a subsequent increase of >100% exhibited a more than three-fold increase in the risk of SD (HR 3.51, 95% CI 1.20–10.27) compared with patients with stable levels. Accordingly, the relative risk of SD was more than halved in patients with baseline NT-pro-BNP above the median and a subsequent decrease >10% (HR 0.42, 95% CI 0.23–0.76) compared with patients with stable NT-pro-BNP.
Figure 2.
Kaplan–Meier estimates for time to sudden death, cardiovascular event (cardiac death, non-fatal myocardial infarction, or stroke), and all-cause mortality in subgroups of patients according to change from baseline N-terminal-pro-B-type-natriuretic-peptide (group 1: less than –10%; group 2: –10 up to +10%; group 3: >10 up to 100%; and group 4: >100%).
Table 3.
Change from baseline N-terminal-pro-B-type-natriuretic-peptide and risk of all-cause death, combined cardiovascular events, myocardial infarction, sudden death, and stroke
| Per cent change from baseline NT-pro-BNP (pg/mL) |
||||
|---|---|---|---|---|
| Decrease (less than −10%) (n = 428) | No change (−10 to 10%) (n = 160) | Increase (>10–100%) (n = 352) | Increase (>100%) (n = 263) | |
| All-cause death | ||||
| Number of events during study, n | 183 | 71 | 163 | 167 |
| Adjusted HRa (95% confidence interval) | 0.90 (0.68–1.19), P = 0.454 | —b | 0.99 (0.74–1.31), P = 0.931 | 1.46 (1.10–1.95), P = 0.009 |
| Adjusted HRa (95% confidence interval) with NT-pro-BNP as a continuous variable, 1.23 (1.14–1.34), P < 0.001 | ||||
| Combined cardiovascular endpoint (myocardial infarction, cardiac death, stroke) | ||||
| Number of events during study, n | 144 | 52 | 117 | 110 |
| Adjusted HRa (95% confidence interval) | 1.00 (0.72–1.38), P = 0.996 | —b | 0.98 (0.70–1.36), P = 0.898 | 1.37 (0.98–1.92), P = 0.068 |
| Adjusted HRa (95% confidence interval) with NT-pro-BNP as a continuous variable, 1.19 (1.07–1.32), P = 0.001 | ||||
| Sudden death | ||||
| Number of events during study, n | 39 | 23 | 37 | 53 |
| Adjusted HRa (95% confidence interval) | 0.61 (0.36–1.02), P = 0.060 | —b | 0.70 (0.42–1.19), P = 0.192 | 1.51 (0.91–2.51), P = 0.109 |
| Adjusted HRa (95% confidence interval) with NT-pro-BNP as a continuous variable, 1.45 (1.25–1.69), P < 0.001 | ||||
| Stroke | ||||
| Number of events during study, n | 32 | 12 | 26 | 18 |
| Adjusted HRa (95% confidence interval) | 0.88 (0.45–1.73), P = 0.706 | —b | 0.91 (0.45–1.83), P = 0.790 | 0.96 (0.45–2.02), P = 0.908 |
| Adjusted HRa (95% confidence interval) with NT-pro-BNP as a continuous variable, 1.10 ( 0.89–1.37), P = 0.391 | ||||
| Myocardial infarction | ||||
| Number of events during study, n | 69 | 19 | 51 | 41 |
| Adjusted HRa (95% confidence interval) | 1.30 (0.78–2.18), P = 0.319 | —b | 1.14 (0.69–1.95), P = 0.630 | 1.35 (0.77–2.36), P = 0.290 |
| Adjusted HRa (95% confidence interval) with NT-pro-BNP as a continuous variable, 1.08 ( 0.92–1.26), P = 0.380 | ||||
aAdjustment for gender, age, treatment, phosphate, low-density lipoprotein, haemoglobin, glycated haemoglobin, albumin, ever smoking, systolic/diastolic blood pressure, body mass index, ultrafiltration volume, duration of dialysis, AV fistula, history of stroke or transitory ischaemic attack, CAD (e.g. MI, coronary artery bypass grafting, percutaneous coronary intervention, and angiographically documented CAD), peripheral vascular disease, and CHF.
bThe group with no change (i.e. a change of −10 to 10%) was used as reference for each of the others.
Discussion
This study investigated the association of two consecutive NT-pro-BNP measurements with the outcome in a large prospective patient cohort with a high incidence of pre-specified and centrally adjudicated endpoints during 4 years of follow-up. In patients with T2DM on haemodialysis, baseline NT-pro-BNP was a risk factor for SD, stroke, CVEs, and all-cause mortality. In addition, the increase in NT-pro-BNP during follow-up was associated with SD, CVEs, and all-cause mortality. Notably, neither baseline nor change from baseline NT-pro-BNP was associated with MI.
Few data exist that specifically focus on NT-pro-BNP and the risk of SD. Only one study in heart failure patients has investigated this association before.12 Against this background, the present analysis adds important new information to current knowledge when demonstrating a clear association between baseline NT-pro-BNP and risk of SD. This might have been due to the high incidence of SD in the present analysis (n = 160) and the long follow-up period of 4 years compared with the former one (n = 44, 1.68 years). Moreover, the follow-up measurement allowed for identifying subgroups of patients at higher and lower risk. In addition to the consistently high risk of patients with increasing NT-pro-BNP, subgroup analysis in patients with high baseline NT-pro-BNP showed a lower adjusted relative risk of SD for those with decreasing NT-pro-BNP (>10% decrease) compared with patients with stable levels. Since SD is common in patients with T2DM on haemodialysis (60% of cardiovascular and 26% of all-cause death) and mortality is high, NT-pro-BNP might be useful in risk stratification when considering therapeutic interventions (e.g. more frequent dialysis, beta-blocker, implantable cardioverter defibrillator).13,14
In contrast to patients with normal renal function, the association between stroke and NT-pro-BNP has not been evaluated in patients with kidney disease.4,15–17 We found that baseline NT-pro-BNP was a risk factor for stroke, although the change in NT-pro-BNP had no significant effect on the occurrence of cerebrovascular events. The association of NT-pro-BNP and stroke was independent of anaemia18 and arrhythmia, but we did not adjust for lesion size, insular involvement, or stroke severity, which were shown to eliminate NT-pro-BNP from a multivariate model in patients with acute stroke.19
Limited data have been reported about NT-pro-BNP and MI in patients with stable vascular disease.20 As Omland et al.,20 we found no association between NT-pro-BNP and MI, and furthermore found that changes in NT-pro-BNP did not affect the risk of MI, independently of baseline NT-pro-BNP levels. This suggests that the association between NT-pro-BNP and mortality as well as combined CVEs was unlikely to be explained by the risk of MI.
By now, follow-up measurements of NT-pro-BNP in chronic vascular disease and their association with various endpoints have not been reported in this form. The increase in NT-pro-BNP from baseline (continuous variable) was associated with an increased risk of combined CVEs and mortality, independent of baseline NT-pro-BNP and—regarding the risk of death—independent of baseline co-morbidity (history of CHF or CAD). In categorized analysis, the increase >100% was associated with a higher risk of outcome compared with patients with stable levels (NT-pro-BNP ± 10% from baseline), whereas smaller increases (<100%) or decreases (>10%) in NT-pro-BNP had no significant effect on outcome.
As other investigators,1–4,7–9 we found baseline NT-pro-BNP to be a risk factor for all-cause mortality. The HR for mortality (HRfourth vs. first quartile 2.06; 95% CI 1.60–2.67) was lower than that observed in peritoneal dialysis patients7 (HR 4.97; 95% CI 1.35–18.28) and patients from the Ludwigshafen Risk and Cardiovascular Health Study without kidney disease3 (HR 3.8; 95% CI 1.99–7.28). This might be due to the high mortality in the present patient cohort. After a follow-up period of 4 years, the cumulative rate of death in patients with NT-pro-BNP ≥400 and <2000 pg/mL was 0.34, and that in patients with NT-pro-BNP >2000 pg/mL was 0.57. In individuals from the Ludwigshafen Risk and Cardiovascular Health Study,3 the respective values were 0.12 and 0.29. As overall mortality was remarkably lower in the latter cohort, the relative risk of dying could be considerably higher. However, further comparisons were impeded by varying categorizations of NT-pro-BNP, follow-up time, etc. As NT-pro-BNP in other studies equally affected cardiovascular morbidity and hypervolaemia, it remains speculative whether optimal adjustment of dry weight might influence prognosis.9,21
Similar to other patients with chronic kidney disease, baseline NT-pro-BNP (median 3361 pg/mL) largely exceeded normal values (<50–334 pg/mL)7–9,22,23 in the present study. According to a previous work, left ventricular wall tension,24 associated haemodynamic factors,5–7,25 cardiac ischaemia,26,27 and reduced renal extraction22,23,28 are possible causative factors. In addition, NT-pro-BNP was shown to depend on volume status and residual diuresis in haemodialysis patients9,21 and to be higher in diabetic patients.29 In the present study, baseline data seem to be well compatible with this. However, the percentage of patients with AV fistula potentially causing a volume overload was lowest in patients with NT-pro-BNP levels in the fourth quartile. This was against expectations since the creation of an AV fistula was shown to produce an elevation of BNP.30,31 In the present study, median NT-pro-BNP increased slightly over time, possibly reflecting a progression of disease. A similar increase is known from patients at high risk of major vascular events from the Heart Protection Study, where simvastatin effectively improved outcome, and patients on simvastatin showed less increase in NT-pro-BNP compared with the placebo group.16 In the present study, baseline, post-baseline, and change in NT-pro-BNP levels between atorvastatin- and placebo-treated patients did not differ significantly.
Our study has certain limitations. It is a post hoc analysis within a selected cohort of German patients with T2DM on haemodialysis. Therefore, the relationship between NT-pro-BNP and risk may not be generalizable. We did not analyse heart failure events because of their low incidence and because they (except for death due to heart failure) have not been pre-specified endpoints. Although preliminary observations in this data set suggest that the concentrations of NT-pro-BNP are independent of the haemodialysis ultrafiltration volume, a more in-depth analysis as to whether intensive haemodialysis would reduce NT-pro-BNP and thus potentially attenuate risk would be desirable.
In summary, NT-pro-BNP is an important risk factor for death and CVEs in patients with T2DM on maintenance haemodialysis treatment. Not only high baseline NT-pro-BNP but also increases in NT-pro-BNP over time are associated with poor long-term clinical outcome. Whether treatment strategies guided by NT-pro-BNP levels will decrease morbidity and mortality needs to be evaluated in future.
Conflict of interest: none declared.
Funding
We thank Roche Diagnostics for providing the reagents for the measurement of NT-pro-BNP. Funding to pay the Open Access publication charges for this article was provided by the Department of Clinical Chemistry, University of Freiburg, Germany.
References
- 1.Doust JA, Pietrzak E, Dobson A, Glasziou P. How well does B-type natriuretic peptide predict death and cardiac events in patients with heart failure: systematic review. BMJ. 2005;330:625. doi: 10.1136/bmj.330.7492.625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kragelund C, Gronning B, Kober L, Hildebrandt P, Steffensen R. N-terminal pro-B-type natriuretic peptide and long-term mortality in stable coronary heart disease. N Engl J Med. 2005;352:666–675. doi: 10.1056/NEJMoa042330. [DOI] [PubMed] [Google Scholar]
- 3.Marz W, Tiran B, Seelhorst U, Wellnitz B, Bauernsachs J, Winklelmann BR, Boehm BO. N-terminal pro-B-type natriuretic peptide predicts total and cardiovascular mortality in individuals with or without stable coronary artery disease: the Ludwigshafen Risk and Cardiovascular Health Study. Clin Chem. 2007;53:1075–1083. doi: 10.1373/clinchem.2006.075929. [DOI] [PubMed] [Google Scholar]
- 4.Wang TJ, Larson MG, Levy D, Benjamin EJ, Leip EP, Omland T, Wolf PA, Vasan RS. Plasma natriuretic peptide levels and the risk of cardiovascular events and death. N Engl J Med. 2004;350:655–663. doi: 10.1056/NEJMoa031994. [DOI] [PubMed] [Google Scholar]
- 5.deFilippi CR, Fink JC, Nass CM, Chen H, Christenson R. N-terminal pro-B-type natriuretic peptide for predicting coronary disease and left ventricular hypertrophy in asymptomatic CKD not requiring dialysis. Am J Kidney Dis. 2005;46:35–44. doi: 10.1053/j.ajkd.2005.04.007. [DOI] [PubMed] [Google Scholar]
- 6.Lee JA, Kim DH, Yoo SJ, Oh DJ, Yu SH, Kang ET. Association between serum N-terminal pro-brain natriuretic peptide concentration and left ventricular dysfunction and extracellular water in continuous ambulatory peritoneal dialysis patients. Perit Dial Int. 2006;26:360–365. [PubMed] [Google Scholar]
- 7.Wang AY, Lam CW, Yu CM, Wang M, Chan IH, Zhang Y, Lui SF. N-terminal pro-brain natriuretic peptide: an independent risk predictor of cardiovascular congestion, mortality, and adverse cardiovascular outcomes in chronic peritoneal dialysis patients. J Am Soc Nephrol. 2007;18:321–330. doi: 10.1681/ASN.2005121299. [DOI] [PubMed] [Google Scholar]
- 8.Apple FS, Murakami MM, Pearce LA, Herzog CA. Multi-biomarker risk stratification of N-terminal pro-B-type natriuretic peptide, high-sensitivity C-reactive protein, and cardiac troponin T and I in end-stage renal disease for all-cause death. Clin Chem. 2004;50:2279–2285. doi: 10.1373/clinchem.2004.035741. [DOI] [PubMed] [Google Scholar]
- 9.Sommerer C, Beimler J, Schwenger V, Heckele N, Katus HA, Giannitsis E, Zeier M. Cardiac biomarkers and survival in haemodialysis patients. Eur J Clin Invest. 2007;37:350–356. doi: 10.1111/j.1365-2362.2007.01785.x. [DOI] [PubMed] [Google Scholar]
- 10.Wanner C, Krane V, Marz W, Olschewski M, Mann JF, Ruf G, Ritz E. Atorvastatin in patients with type 2 diabetes mellitus undergoing hemodialysis. N Engl J Med. 2005;353:238–248. doi: 10.1056/NEJMoa043545. [DOI] [PubMed] [Google Scholar]
- 11.Wanner C, Krane V, Marz W. Randomized controlled trial on the efficacy and safety of atorvastatin in patients with type 2 diabetes on hemodialysis (4D study): demographic and baseline characteristics. Kidney Blood Press Res. 2004;27:259–266. doi: 10.1159/000080241. [DOI] [PubMed] [Google Scholar]
- 12.Berger R, Huelsman M, Strecker K, Bojic A, Moser P, Stanek B, Pacher R. B-type natriuretic peptide predicts sudden death in patients with chronic heart failure. Circulation. 2002;105:2392–2397. doi: 10.1161/01.cir.0000016642.15031.34. [DOI] [PubMed] [Google Scholar]
- 13.Bleyer AJ, Hartman J, Brannon PC, Reeves-Daniel A, Satko SG, Russell G. Characteristics of sudden death in hemodialysis patients. Kidney Int. 2006;69:2268–2273. doi: 10.1038/sj.ki.5000446. [DOI] [PubMed] [Google Scholar]
- 14.Herzog CA. Cardiac arrest in dialysis patients: approaches to alter an abysmal outcome. Kidney Int Suppl. 2003:S197–S200. doi: 10.1046/j.1523-1755.63.s84.17.x. [DOI] [PubMed] [Google Scholar]
- 15.Bibbins-Domingo K, Gupta R, Na B, Wu AH, Schiller NB, Whooley MA. N-terminal fragment of the prohormone brain-type natriuretic peptide (NT-proBNP), cardiovascular events, and mortality in patients with stable coronary heart disease. JAMA. 2007;297:169–176. doi: 10.1001/jama.297.2.169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Emberson JR, Ng LL, Armitage J, Bowman L, Parish S, Collins R. N-terminal pro-B-type natriuretic peptide, vascular disease risk, and cholesterol reduction among 20,536 patients in the MRC/BHF heart protection study. J Am Coll Cardiol. 2007;49:311–319. doi: 10.1016/j.jacc.2006.08.052. [DOI] [PubMed] [Google Scholar]
- 17.Kistorp C, Raymond I, Pedersen F, Gustafsson F, Faber J, Hildebrandt P. N-terminal pro-brain natriuretic peptide, C-reactive protein, and urinary albumin levels as predictors of mortality and cardiovascular events in older adults. JAMA. 2005;293:1609–1616. doi: 10.1001/jama.293.13.1609. [DOI] [PubMed] [Google Scholar]
- 18.Nybo M, Kristensen SR, Mickley H, Jensen JK. The influence of anaemia on stroke prognosis and its relation to N-terminal pro-brain natriuretic peptide. Eur J Neurol. 2007;14:477–482. doi: 10.1111/j.1468-1331.2006.01591.x. [DOI] [PubMed] [Google Scholar]
- 19.Etgen T, Baum H, Sander K, Sander D. Cardiac troponins and N-terminal pro-brain natriuretic peptide in acute ischemic stroke do not relate to clinical prognosis. Stroke. 2005;36:270–275. doi: 10.1161/01.STR.0000151364.19066.a1. [DOI] [PubMed] [Google Scholar]
- 20.Omland T, Sabatine MS, Jablonski KA, Rice MM, Hsia J, Wergeland R, Landaas S, Rouleau JL, Domanski MJ, Hall C, Pfeffer MA, Braunwald E. Prognostic value of B-type natriuretic peptides in patients with stable coronary artery disease: the PEACE Trial. J Am Coll Cardiol. 2007;50:205–214. doi: 10.1016/j.jacc.2007.03.038. [DOI] [PubMed] [Google Scholar]
- 21.van de Pol AC, Frenken LA, Moret K, Baumgarten R, van der Sande FM, Beerenhout CM, Kooman JP, Leunissen KM. An evaluation of blood volume changes during ultrafiltration pulses and natriuretic peptides in the assessment of dry weight in hemodialysis patients. Hemodial Int. 2007;11:51–61. doi: 10.1111/j.1542-4758.2007.00154.x. [DOI] [PubMed] [Google Scholar]
- 22.Khan IA, Fink J, Nass C, Chen H, Christenson R, deFilippi CR. N-terminal pro-B-type natriuretic peptide and B-type natriuretic peptide for identifying coronary artery disease and left ventricular hypertrophy in ambulatory chronic kidney disease patients. Am J Cardiol. 2006;97:1530–1534. doi: 10.1016/j.amjcard.2005.11.090. [DOI] [PubMed] [Google Scholar]
- 23.Vickery S, Price CP, John RI, Abbas NA, Webb MC, Kempson ME, Lamb EJ. B-type natriuretic peptide (BNP) and amino-terminal proBNP in patients with CKD: relationship to renal function and left ventricular hypertrophy. Am J Kidney Dis. 2005;46:610–620. doi: 10.1053/j.ajkd.2005.06.017. [DOI] [PubMed] [Google Scholar]
- 24.Jessup M, Brozena S. Heart failure. N Engl J Med. 2003;348:2007–2018. doi: 10.1056/NEJMra021498. [DOI] [PubMed] [Google Scholar]
- 25.Ellinor PT, Low AF, Patton KK, Shea MA, MacRae CA. Discordant atrial natriuretic peptide and brain natriuretic peptide levels in lone atrial fibrillation. J Am Coll Cardiol. 2005;45:82–86. doi: 10.1016/j.jacc.2004.09.045. [DOI] [PubMed] [Google Scholar]
- 26.Goetze JP, Christoffersen C, Perko M, Arendrup H, Rehfeld JF, Kastrup J, Nielsen LBI. Increased cardiac BNP expression associated with myocardial ischemia. FASEB J. 2003;17:1105–1107. doi: 10.1096/fj.02-0796fje. [DOI] [PubMed] [Google Scholar]
- 27.Sakai H, Tsutamoto T, Ishikawa C, Tanaka T, Fujii M, Yamamoto T, Takashima H, Horie M. Direct comparison of brain natriuretic peptide (BNP) and N-terminal pro-BNP secretion and extent of coronary artery stenosis in patients with stable coronary artery disease. Circ J. 2007;71:499–505. doi: 10.1253/circj.71.499. [DOI] [PubMed] [Google Scholar]
- 28.Goetze JP, Jensen G, Moller S, Bendtsen F, Rehfeld JF, Henriksen JH. BNP and N-terminal proBNP are both extracted in the normal kidney. Eur J Clin Invest. 2006;36:8–15. doi: 10.1111/j.1365-2362.2006.01594.x. [DOI] [PubMed] [Google Scholar]
- 29.Pfister R, Tan D, Thekkanal J, Hellmich M, Erdmann E, Schneider CA. NT-pro-BNP measured at discharge predicts outcome in multimorbid diabetic inpatients with a broad spectrum of cardiovascular disease. Acta Diabetol. 2007;44:91–97. doi: 10.1007/s00592-007-0248-4. [DOI] [PubMed] [Google Scholar]
- 30.De Stefano LM, Matsubara LS, Matsubara BB. Myocardial dysfunction with increased ventricular compliance in volume overload hypertrophy. Eur J Heart Fail. 2006;8:784–789. doi: 10.1016/j.ejheart.2006.02.005. [DOI] [PubMed] [Google Scholar]
- 31.Iwashima Y, Horio T, Takami Y, Inenga T, Nishikimi T, Takishita S, Kawano Y. Effects of the creation of arteriovenous fistula for hemodialysis on cardiac function and natriuretic peptide levels in CRF. Am J Kidney Dis. 2002;40:974–982. doi: 10.1053/ajkd.2002.36329. [DOI] [PubMed] [Google Scholar]
References
The above article uses a new reference style being piloted by the EHJ that shall soon be used for all articles.